Abstract
Biodiesel is regarded as the most promising renewable biofuel which can satisfy the worldwide energy demand through its robust net energy output with less impact on environment and also an effective alternative or supplement for existing diesel fuel for powering Compression Ignition engines. This present study aims to compare the performance, combustion and emission characteristics of beef tallow biodiesel with its dominant fatty acid esters and their influence on these characteristics. Ethyl stearate, ethyl palmitate and ethyl oleate were identified as dominant fatty acid esters available in a composition of 14.78, 37.36 and 25.17%, respectively. Two set of samples were prepared for testing purpose and were classified as (i) blend samples and (ii) ester samples, with plain diesel as base fuel. The maximum cylinder pressure was found to be in the range between 67 and 71 bar for all the samples and heat release rate was found to be highest for B20 blend. Slight decrease in efficiency along with marginal increase in fuel consumption was a result of long chained fatty acid esters (stearate and oleate esters) whereas increase in CO2 emission and decrease in NOX emission were because of higher oxygen content, cetane number and degree of unsaturation in the ester molecule.
Keywords
Introduction
Increase in demand for energy to satisfy the need for modernization of world has resulted in the depletion of fossil fuel reserve along with increased global temperature and anthropogenic climate change (Schirone and Pellitteri, 2017; Senthil et al., 2016). This present global scenario has risked the inventory of fossil fuel resources and has pushed the planet to rely on renewable energy resources. Biodiesel is one such promising renewable biofuel which is known for its environmental friendliness, self-sustainability, low toxicity, high biodegradability and improvised combustibility along with reduced emission of hydrocarbons, CO, SO2, polycyclic aromatic HC, nitric polycyclic aromatic HC and particulate matter (PM) (Dhiraj et al., 2012; Srinivasan and Jambulingam, 2018a). This carbon neutral fuel is predominantly known for its high oxygen content (Jaichandar and Annamalai, 2011) with zero aromatics and no sulphur content (Atabani et al., 2012; Hassan and Kalam, 2013).
Biodiesel is the common name for any fatty acid alkyl ester produced by transesterifying the fatty acids in triglycerides with short chained alcohol in the presence of catalyst under the influence of continuous heating and stirring (Demirbas, 2008).The sources of triglyceride can be from any organic biomass with good fat content like vegetable and inedible seed oils (Issariyakul and Dalai, 2014; Srinivasan et al., 2017), animal fats (Adewale et al., 2015), waste cooking oil (Phan and Phan, 2008; Yildizhan et al., 2017) and even high fatty greases (Tran et al., 2018). In recent times, leather fleshing wastes (Duraisamy et al., 2016) and animal slaughter house wastes (Srinivasan et al., 2018b) have been also used as raw feedstock for producing biodiesel with lower cost of production (Atabani et al., 2012) and reduced potential threats to humans and environments (Sanli et al., 2014). The major technical challenge associated with these waste animal fats is high free fatty acid content which must be reduced below 1% for an effective transesterification reaction (Srinivasan and Jambulingam, 2018a; Canoira et al., 2008) and can be achieved by either undergoing esterification or glycerolysis reaction as a pre-treatment technique. Numerous homogeneous catalysts like hydroxides of sodium and potassium, methonates and ethonates of sodium and potassium; heterogeneous catalysts like metal oxides, metal zeolites (Chouhan and Sarma, 2011); biocatalysts like enzymes, immobilized lipases and coordinated compound catalysts like ionic liquids have been used for increasing the rate of the transesterification reaction and also the yield of biodiesel. In general, the reaction rate and biodiesel yield are administered by the influencing parameters (optimum molar ratio, calculated catalyst concentration) and operating parameters (reaction time, stirring speed and reaction temperature) of the transesterification reaction, and numerous studies have been carried out on these parameters in terms of optimization and detailed reaction kinetics to achieve the maximum yield. Accordingly, Srinivasan et al. (2018c) optimized the transesterification reaction for waste animal fat by stating 1:3 (tallow:methanol) and 0.5% as optimum molar ratio and catalyst concentration, respectively, whereas Tashtoush et al. (2004) correlated the interaction between the reaction time and temperature of transesterification reaction and was found to be 50°C for time duration of 120 min. In addition, supercritical transesterification (Demirbas, 2009), ultrasonic, microwave-assisted transesterification reaction (O¨zçimen and Yu¨cel, 2011) have been proven to be sustainable methods for improvising the reaction rate and biodiesel yield with higher performance and efficiency.
Even though biodiesel possess superior properties it cannot be directly used in an engine because of its higher viscosity, corrosive activity on cylinder surface, rubber tubings and linings, and carbon deposition and soot formation (Jayed et al., 2011). However, these drawbacks were overcome by blending biodiesel with plain diesel which improvised the performance, combustion and emission (PCE) characteristics with little modification in engine. The necessity for blend was justified in the work carried out by Lapuerta et al. (2008) when a pressure drop was noticed in filter upon using 100% animal biodiesel but was rectified when blended with soybean oil above 50% blend ratios. Apart from plain diesel, biodiesel can be blended with alcohol like methanol (Zhang et al., 2017), ethanol (Kwanchareon et al., 2007), butanol (Yilmaz et al., 2014) as oxygenated fuel with reduced emissions or can even be combusted with biogas (Ramesha et al., 2015), acetylene (Valmiki and Sanjaykumar, 2016), hydrogen gas (Çalık, 2018) and even hydroxyl gas (Uludamar et al., 2017) as inducted fuel in dual fuel mode CI engine for improvising the overall efficiency. Even though these dual fuel modes enhanced the performance and combustion characteristics of biodiesel and had a good control over soot formation, it had least impact in terms of reducing NOX emissions (Singh et al., 2018).
Numerous studies related to the PCE characteristics of oil- and fat-based biodiesel in CI engine have been carried out to understand its viability as a successful replacement or supplement for existing petro diesel. Based on the preliminary study conducted by Demirbas (2007a), it was found that the calorific value of biodiesel was found to be 39–41 MJ/kg which was marginally inferior to gasoline (46 MJ/kg), petro diesel (43 MJ/kg) and petroleum (42 MJ/kg), but superior than coal (32–37 MJ/kg). Selvam and Vadivel (2012) carried out an experimental study on engine characteristics of methyl esters from beef tallow blended with diesel and concluded that for animal fat-based biodiesel blends, there was a slight decrease in brake thermal efficiency and increase in specific fuel consumption when compared with neat diesel fuel because of higher viscosity rates. However, the efficiency rates can be improvised by adding additives to the biodiesel and their influence in improvising the PCE characteristics of biodiesel was summarized in the work carried out by Ali et al. (2013), where natural and synthetic antioxidants, metal-based fuel additives improvised the overall PCE characteristics of biodiesel and resulted in higher efficiency and reduced emission rate in diesel engine. Even though there is only a marginal difference in performance and combustion characteristics between ordinary diesel and biodiesel blend, a drastic variation can be noted in emission characteristics between these fuels. To study these variations, Selvam and Vadivel (2012) compared the emission characteristics between beef tallow biodiesel and plain diesel and noticed that there was a drastic reduction in the concentration of CO, unburned hydrocarbon and smoke density but marginal increase in NOX emission rate. Supporting this, Özgünay et al. (2007) provided an evidence of reduced hydrocarbon content and PMs upon combusting animal fat biodiesel whereas work by Barrios et al. (2014) concluded that animal fat biodiesel blended with plain diesel above 40% resulted in smaller quantity of particle emission than compared to vegetable oil. The size and concentration of PM were also found to be reduced for biodiesel samples than compared to plain diesel upon using in CI engine (Lapuerta et al., 2008). Surprisingly, engine testing carried out by varying different injection pressures for various biodiesel blend samples also resulted in the reduction of CO and HC emissions for lower blends like B10 and B20; however, reduction in NOX emissions was dependent on the blend % (Dharmadhikari et al., 2012).
From these studies, it was clearly evident that waste animal fat can be used as a potential feedstock for production of biodiesel with reduced cost of production and increased sustainability. By means of minor modifications in engine and alteration in the fuel properties by blending or introducing additives, higher efficiency rates with reduced emission can be achieved for this eco-friendly fuel. Although copious amount of studies have been carried out to study the PCE characteristics of biodiesel and their blends in CI engine, only few works have been carried out to understand the influence of fatty acid ester during the combustion reaction. In relevant to this, a brief study was carried to evaluate and compare the PCE characteristics between methyl decanoate and animal fat biodiesel theoretically and experimentally (Szybist et al., 2007). It has been conclusively shown that properties and PCE characteristics of fatty acid esters have a very good impact on the properties and PCE characteristics of biodiesel itself and also suggested that NOX concentration can be reduced by modifying the oleic acid in the triglyceride genetically. However, no previous study has investigated the influence of corresponding dominant fatty acid esters present in biodiesel with respect to their PCE characteristics in CI engine. Significantly, this study would offer important insights into the influence of carbon chain length, cetane number (CN), C/H ratio, degree of unsaturation and molecular structure in PCE characteristics giving an opportunity for an efficient and controlled combustion reaction.
The main purpose of this study is to develop an understanding of PCE characteristics of biodiesel produced from waste beef tallow rendered from leather fleshing waste and animal slaughter houses and compare with PCE characteristics of their dominant fatty acid esters, namely ethyl stearate, ethyl palmitate and ethyl oleate to understand their influence during the combustion of beef tallow biodiesel in CI engine.
Materials and methodology
Collection of raw materials
The leather fleshing wastes and meat processing wastes required for the experiment were collected from tanneries and animal slaughter houses in and around Chennai and Vellore. The collected waste samples were preserved in refrigeration to avoid oxidation (rancidity) and infection. Standard ester samples like ethyl oleate, ethyl stearate and ethyl palmitate were purchased from Sigma Aldrich Chemicals Company along with other chemicals required for production of biodiesel.
Production of beef tallow biodiesel
The production of biodiesel began with rendering of beef tallow using autoclave extraction and was refined by degumming process using orthophosphoric acid to remove phospholipids. The refined tallow was transesterified with calculated amount of ethanol with optimum catalyst concentration under continuous heating and stirring. The end product was separated into biodiesel and glycerol and was refined through water washing and drying.
GC–MS analysis of beef tallow biodiesel
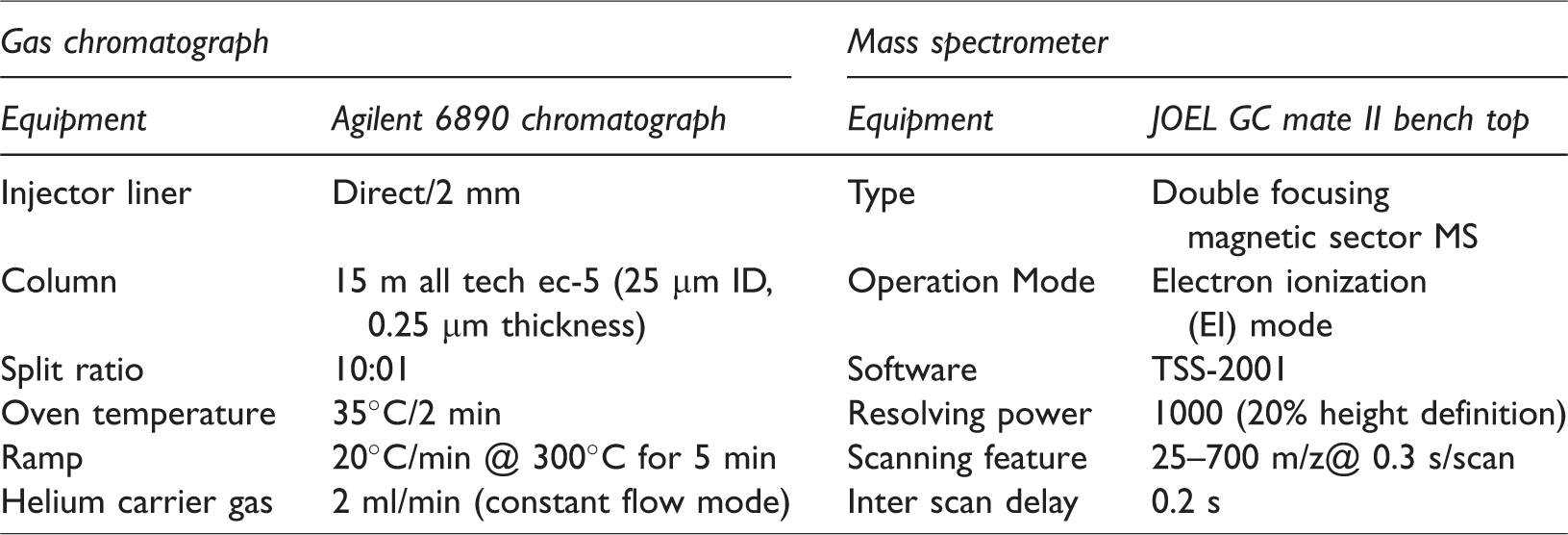
GC–MS analysis was carried out on synthesized tallow biodiesel to quantify the fatty acid esters present in it. The sample was prepared and analysed as per the standard norms. The technical specification of gas chromatograph and mass spectrometer are listed in Table 1.
Technical specification of gas chromatograph and mass spectrometer.
Physico-chemical properties of beef tallow biodiesel
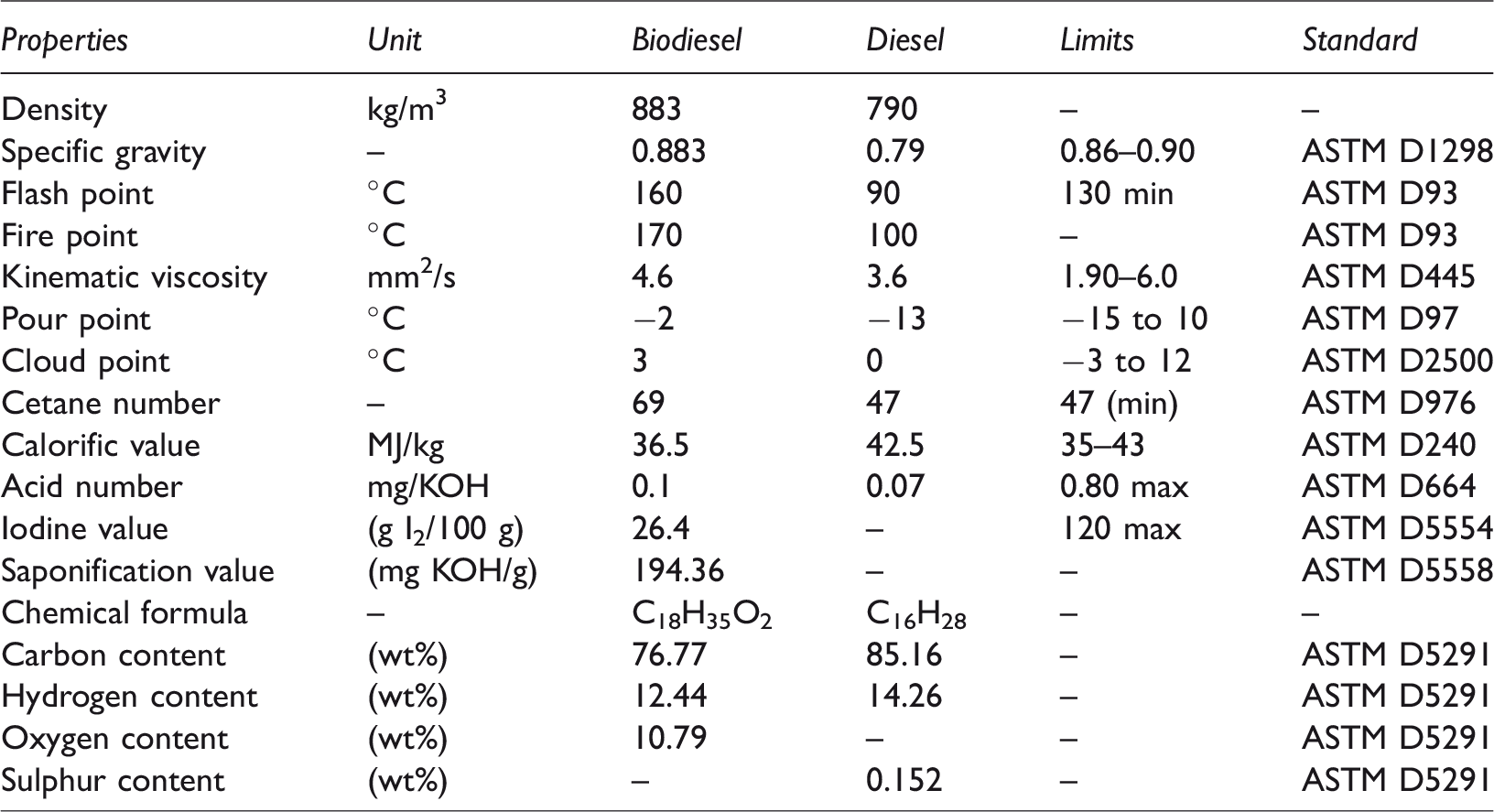
The physico-chemical properties of beef tallow biodiesel were determined using ASTM standards and were compared with plain diesel. All the values were found to be in acceptable range and could be regarded as a successful alternate or supplement for plain diesel. Table 2 summarizes the physico-chemical properties of beef tallow biodiesel.
Physico-chemical properties of beef tallow biodiesel.
Preparation of test samples
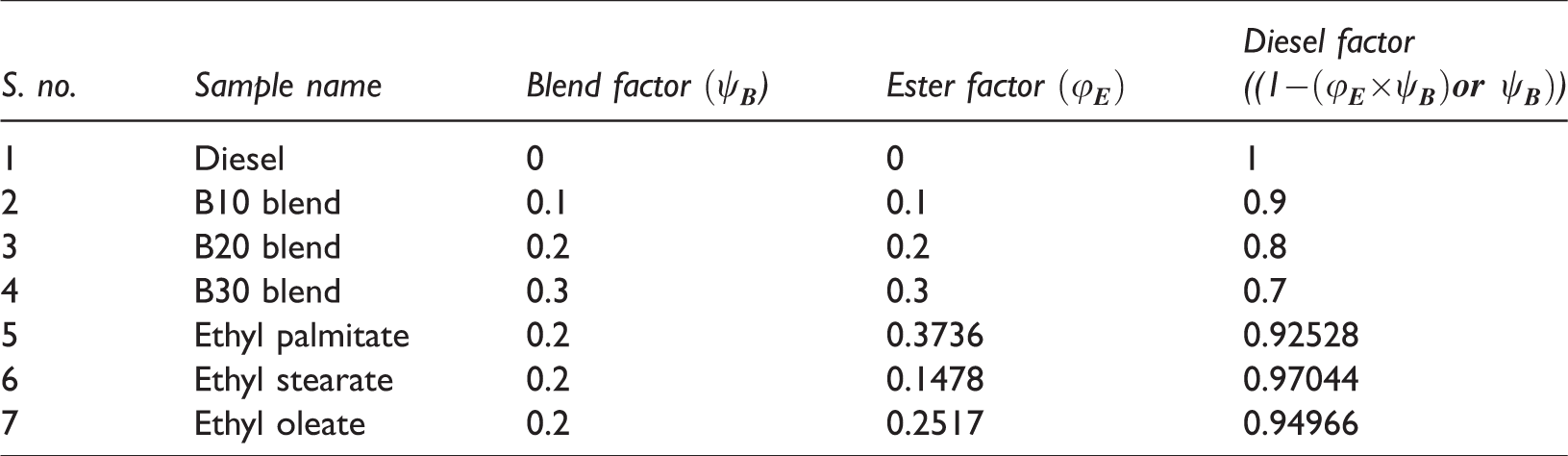
Two different set of samples, namely: (i) blend samples and (ii) ester samples were prepared for studying the behaviour of beef tallow biodiesel at various blend ratios and influence of their dominant fatty acid esters in compression ignition engine, respectively. The blend samples consist of three different blends, namely B10, B20 and B30, which were prepared by varying biodiesel blends as 10, 20 and 30%. Volume of sample required for preparing desired blend is calculated using equations (1) and (2). The ester sample consists of three different samples, namely oleate blend, stearate blend and palmitate blend, prepared by blending ethyl oleate, ethyl stearate and ethyl palmitate separately with ordinary diesel. The volume of sample required for preparing the desired blend was calculated using equations (3) and (4)
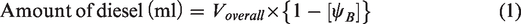
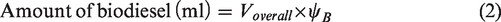
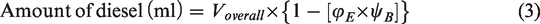
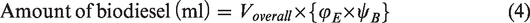
Engine testing of fuel samples
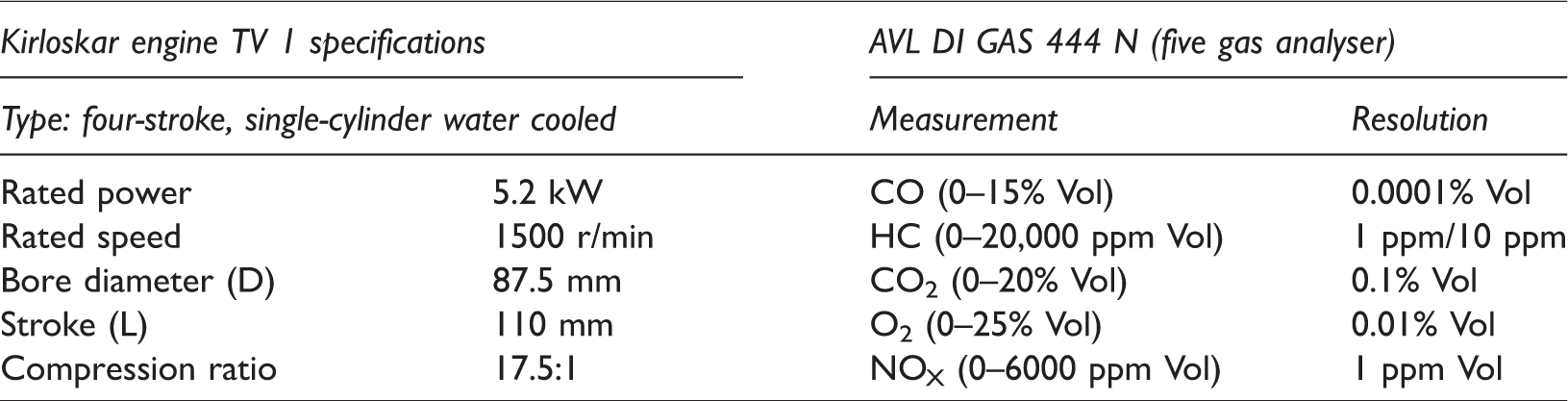
The PCE characteristics of prepared samples were analysed by testing it in a four-stroke, single-cylinder diesel engine. Various combustion parameters like maximum cylinder pressure, cumulative heat release rate, exhaust temperature; performance characteristics like specific fuel consumption, brake and indicated thermal efficiency; and emission characteristics like CO, CO2, O2 and NOX emission were analysed for all the prepared samples to evaluate the quality of biodiesel and also study the influence of fatty acid esters during combustion. Table 3 consolidates the technical specifications of Kirloskar Engine TV1 and AVL DI GAS 444 N (five gas analyser).
Technical specification of Kirloskar engine TV1 and AVL DI GAS 444 N.
Results and discussions
Optimized production of beef tallow biodiesel
The collected leather fleshing wastes and meat processing wastes were autoclaved at 120°C, 15 bar pressure to separate the fatty contents from other non-fatty residues. The rendered tallow was degummed by adding 1 ml of orthophosphoric acid to remove phospholipids present in it. Ethanol-based transesterification was carried out on the refined tallow, where the molar ratio was maintained as 1:6 (ethanol:beef tallow) and KOH as base catalyst in a concentration of 0.5%. The reaction was carried out at 60°C for 2 h under continuous stirring at 350 r/min in a hot plate magnetic stirrer. The reacted mixture was separated as biodiesel and glycerol, which was later refined by progressive water wash followed by drying to remove moisture content. Figure 1 depicts the detailed process flow chart of biodiesel production from waste beef tallow.
Quantification of beef tallow biodiesel
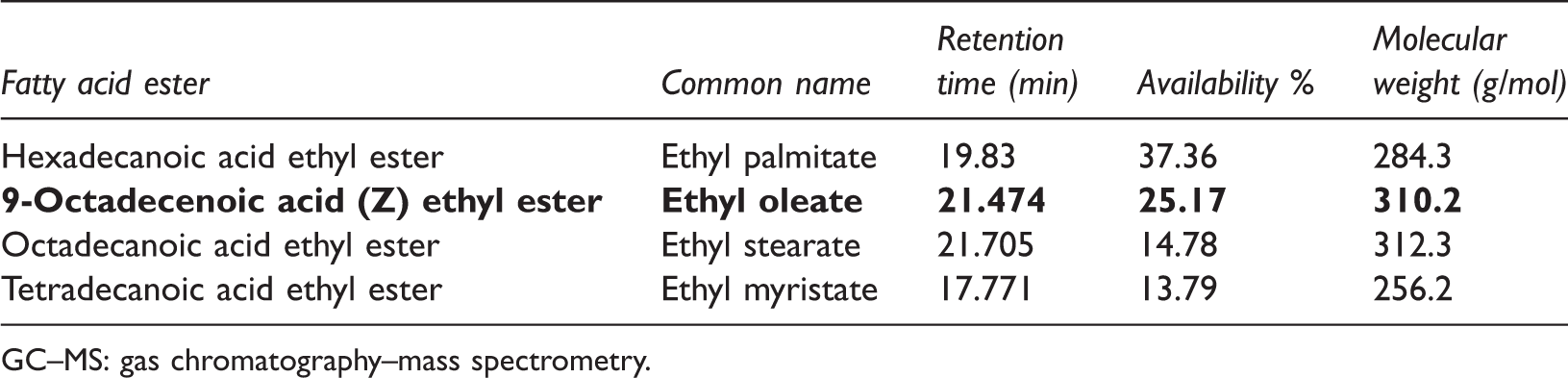
Based on the Gas Chromatographic analysis, ethyl palmitate, ethyl oleate and ethyl stearate were identified as the dominant fatty acid esters present in the beef tallow biodiesel sample based upon the area % occupied by their corresponding peaks in the spectra. The availability % along with their retention time and calculated molecular weight are tabulated in Table 4. These values of availability % were used for calculating the volume of blend required for preparing sample. Figure 2 shows the GC spectra of waste beef tallow biodiesel.
GC Spectral data of waste beef tallow biodiesel.
GC–MS: gas chromatography–mass spectrometry.
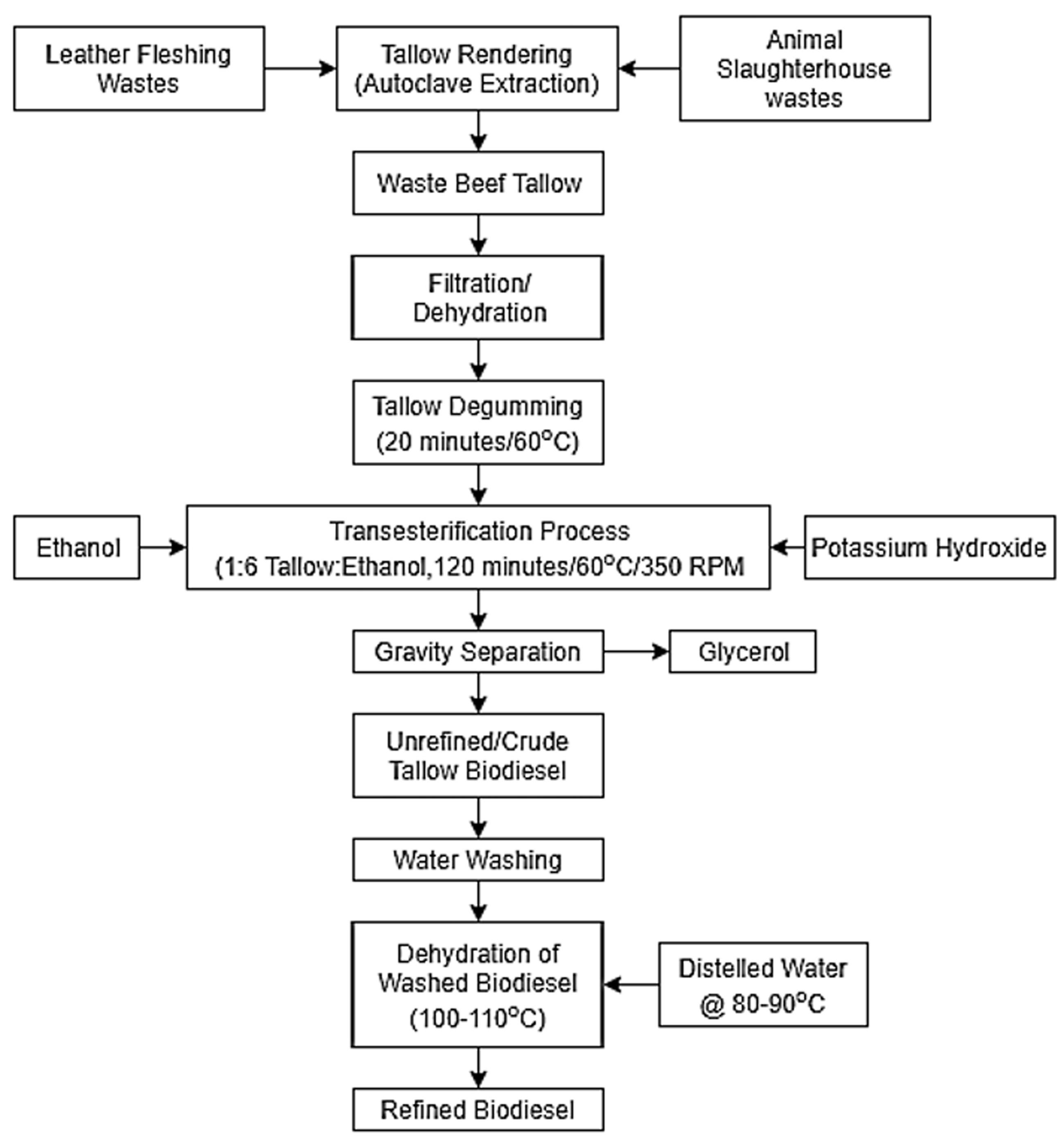
Process flowchart for production of beef tallow biodiesel.
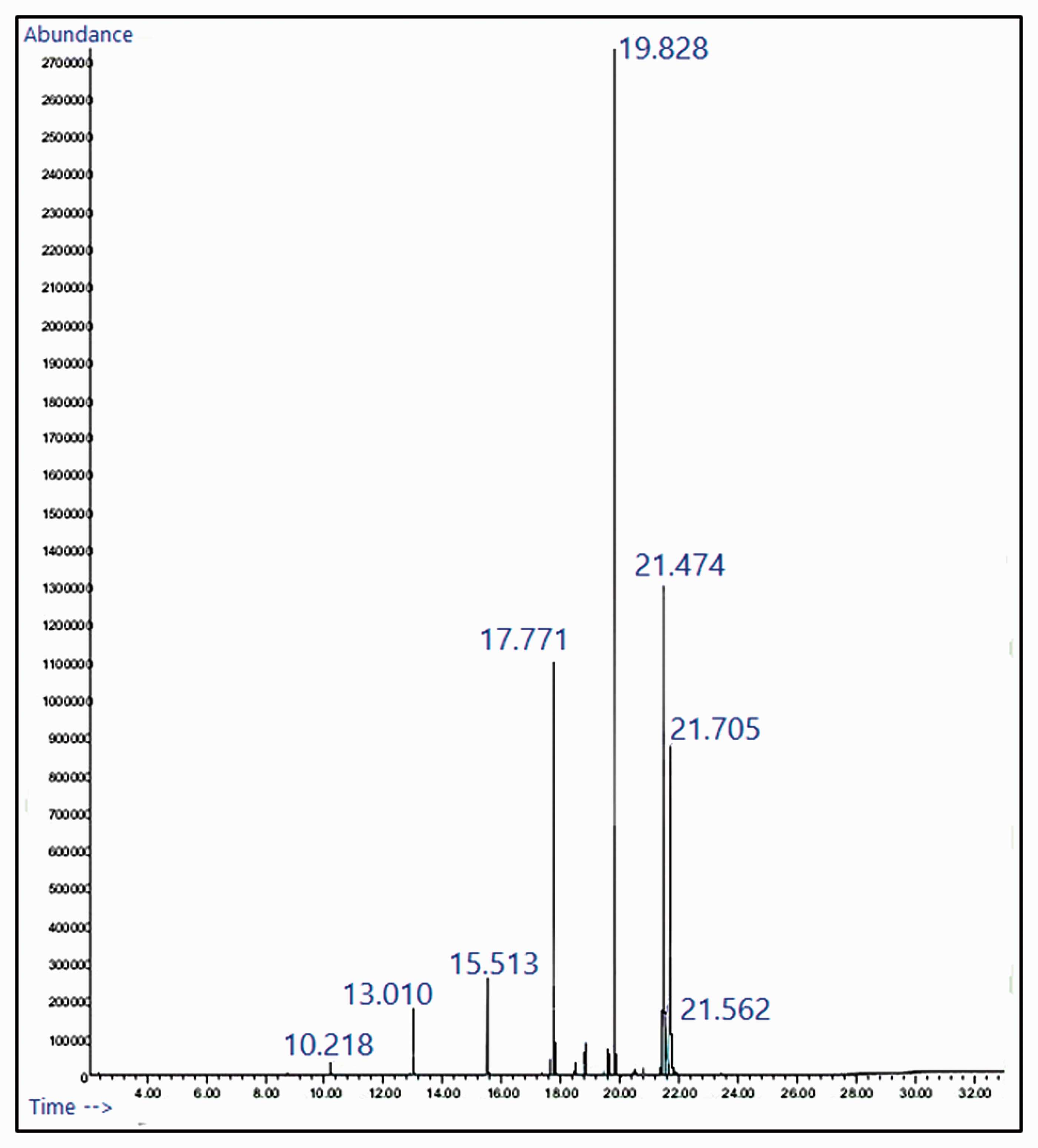
GC spectra of beef tallow biodiesel.
Sample preparation
The volume of fuel samples was dependent on blend factor (
Blend factor and ester factor for various blend and ester samples.
Combustion chemistry
Stoichiometric air-to-fuel ratio
Stoichiometric air-to-fuel ratio is the definite amount of air required to combust 1 mol of fuel completely. Assuming the combustion products as result of oxidation on carbon, hydrogen and oxygen molecules in the fuel, the equation for complete combustion can be written as shown below (Bharathwaaj et al., 2017)
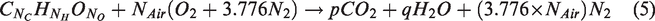
Upon balancing equation (5), the balance of carbon, hydrogen, oxygen and nitrogen can be used to calculate the amount of air (in terms of moles) required to combust 1 mol of biofuel completely by substituting in equation (6)
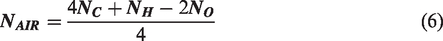
The molecular formula of biodiesel from rendered waste beef tallow was formulated using the number of carbon, hydrogen and oxygen molecule calculated using the GC spectra data and was found to be C18H35O2. On substituting the number of carbon, hydrogen and oxygen molecules as 18, 35 and 2, respectively, in equation (6), the amount of air (in moles) required for combusting 1 mole of biodiesel was calculated as 25.75. Based on calculated Nair, the stoichiometric air-to-fuel ratio was found to be 12.54:1. However, 1.5 times the calculated amount of stoichiometric air must be supplied to the engine for biodiesel to undergo complete combustion thereby making the actual stoichiometric ratio as 18.9:1.
Higher heating value (HHV) and CN
HHV is the calculated amount of heat liberated by unit mass of biodiesel during complete combustion initiated at 25°C and reverting back the temperatures of combustion products to 25°C accounting for latent heat of vaporization of water. Thermophysical properties like HHV and CN can be computed by employing the saponification number (SN) and iodine number (IN) of a biodiesel determined using the preliminary results from GC spectra (Hariram and Bharathwaaj, 2016)


Equations (7) and (8) denote the empirical formula for calculating the IN and SN, respectively, based upon the fatty acid esters determined using the GC spectra (Bharathwaaj et al., 2017) and can be calculated by substituting the number of double bonds available in individual fatty acid molecule, its corresponding availability and molecular weight determined using GC spectra in the above equations. The IN and SN of the biodiesel produced from rendered waste beef tallow were found to be 22.2 and 197.49, respectively
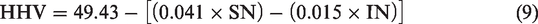
Upon substituting the determined IN and SN value in equation (9) (Bharathwaaj et al., 2017), the HHV was calculated as 41.7 MJ/kg which was lower than compared to plain diesel (45.5 MJ/kg) by 9.11% as biodiesel has reduced hydrocarbon and zero sulphur content which does not contribute much to its HHV. However, in real time applications, biodiesel’s affinity towards moisture content and higher water retaining capacity also tends to reduce its HHV further more than the theoretical value (Demirbas, 2007b).
CN describes the quality of fuel relative to cetane upon ignition and is inversely proportional to ignition delay in fuel and time duration between initiation of injection and first noticeable pressure rise due to combustion. Like HHV, CN can be determined using IN and SN of the tallow biodiesel using the GC spectra and employing these values in equation (10) (Bharathwaaj et al., 2017). Upon calculating, the CN of beef tallow biodiesel was found to be 72 and was found to be 46.8% greater than the plain diesel
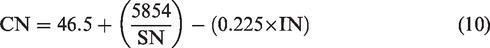
Combustion characteristics
Cylinder pressure
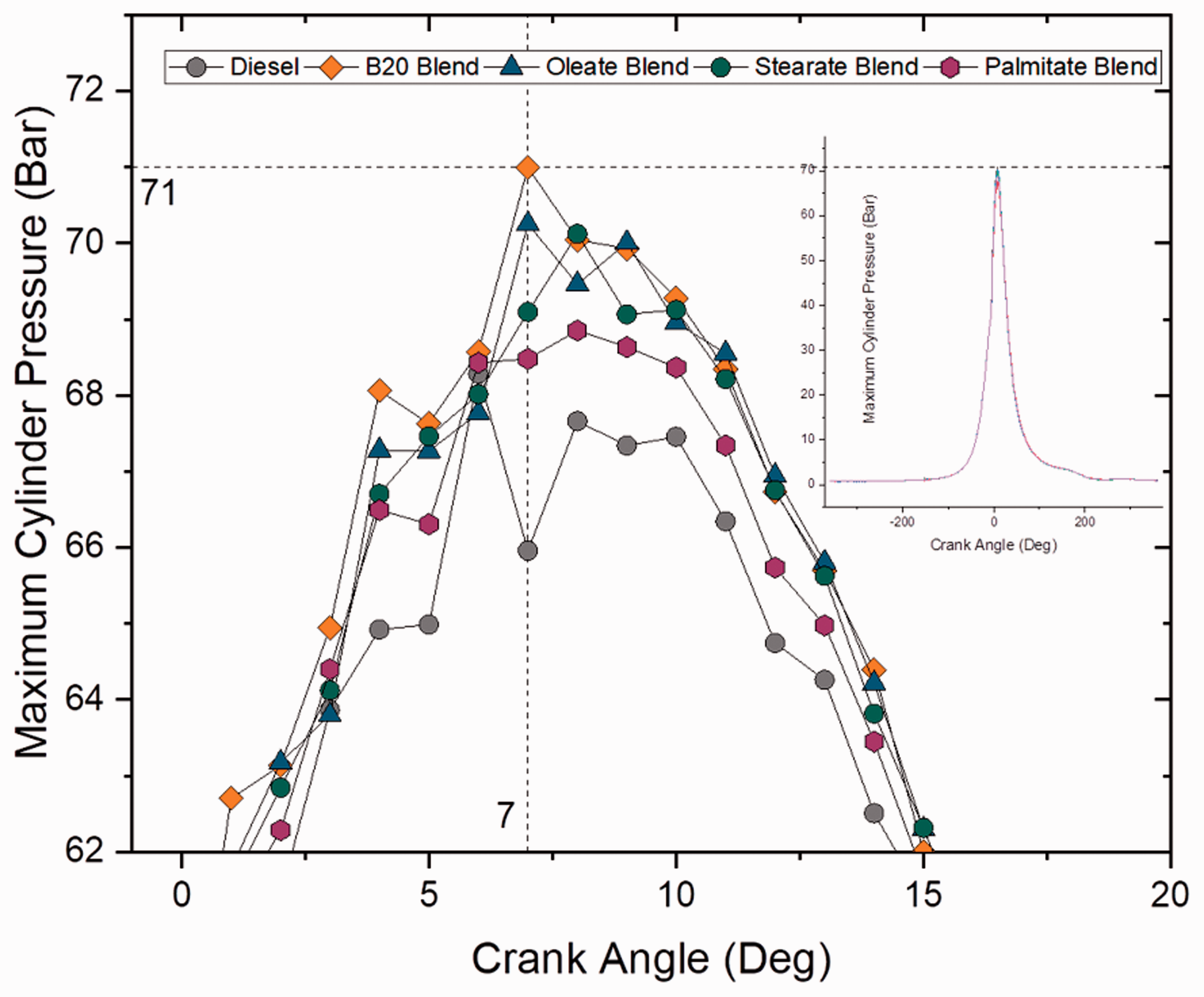
Cylinder pressure defines the fuel’s ability to undergo homogeneous mixing with air and undergo combustion. Figures 3 and 4 show the variation in cylinder pressure at different crank angles for blend samples and ester samples at full load (100% load) conditions. The maximum cylinder pressure for all the blends was in a range between 67 and 71 bar and was found to be maximum shortly after TDC. Maximum cylinder pressure was noted as 71 bar for B20 sample and was 6% greater than the pressure achieved from diesel sample whereas only a 4.4% rise in maximum pressure was noted for other samples. Similarity in cylinder pressure for diesel and biodiesel blends was because of uniform range in heating value and viscosity. However, blend samples followed by ester samples had more cylinder pressure than compared to plain diesel owing to higher oxygen content in it. Furthermore, no incidents of knocking activity were noticed throughout the test period for all samples, as no pulsation on pressure was noticed.
Maximum cylinder pressure for blend samples at 100% load. Maximum cylinder pressure for ester samples at 100% load.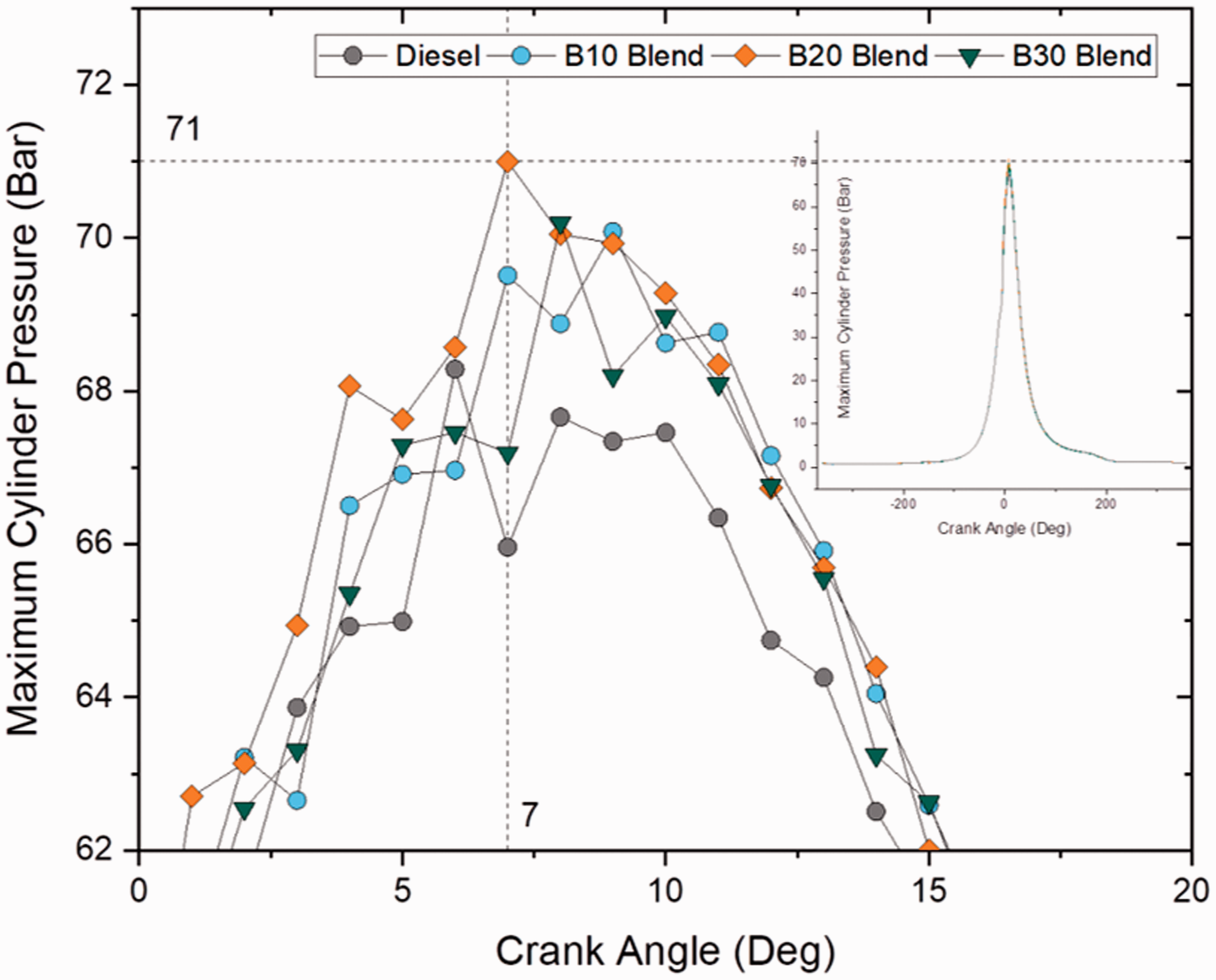
Heat release rate and cumulative heat release rate
Heat release rate pattern of a fuel provides relevant information regarding the initiation of combustion and distribution of heat during the combustion stroke (Xue, 2013). Heat release rate can be calculated from recorded cylinder pressures by employing first law of thermodynamics applicable to closed part of engine cycle (Heywood, 1998)
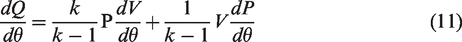
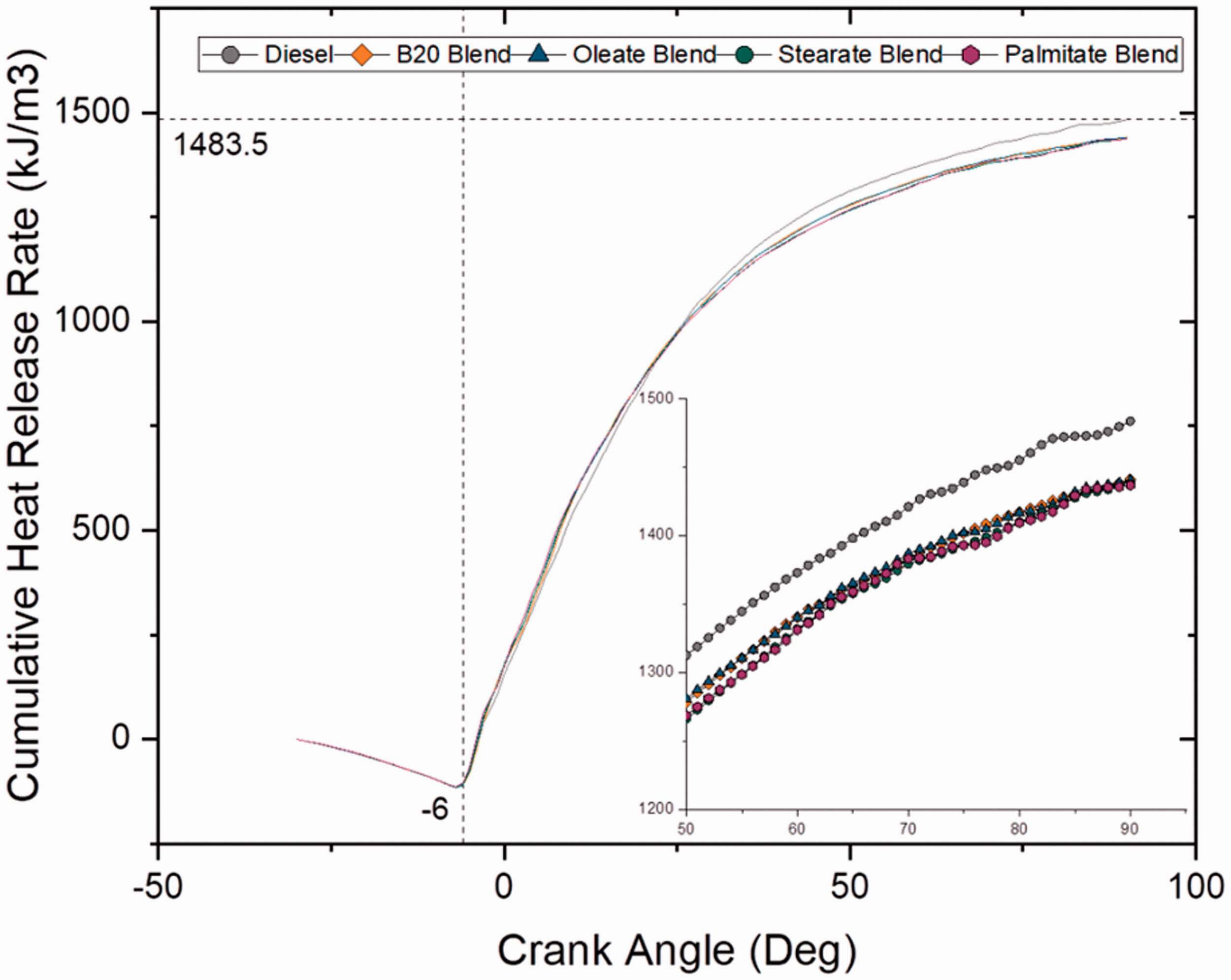
Figures 5 and 6 show cumulative heat release rate at different crank angles for varying blends and different fatty acid ester blends at full load (100% load) conditions. It can be noted that the cumulative heat release rate was highest for plain diesel among all other blends and was calculated as 1470 kJ/m3. This was because of high volatility of diesel sample than compared to blend and ester samples that improvised its overall heat release rate. Even though blend and ester samples had lower heat release rate than diesel sample, CHRR increased with increase in blend ratio for blend samples and increase in carbon chain length for ester samples because of higher heat liberation due to increase in C/H ratio.
Cumulative heat release rate for blend samples at 100% load. Cumulative heat release rate for ester samples at 100% load.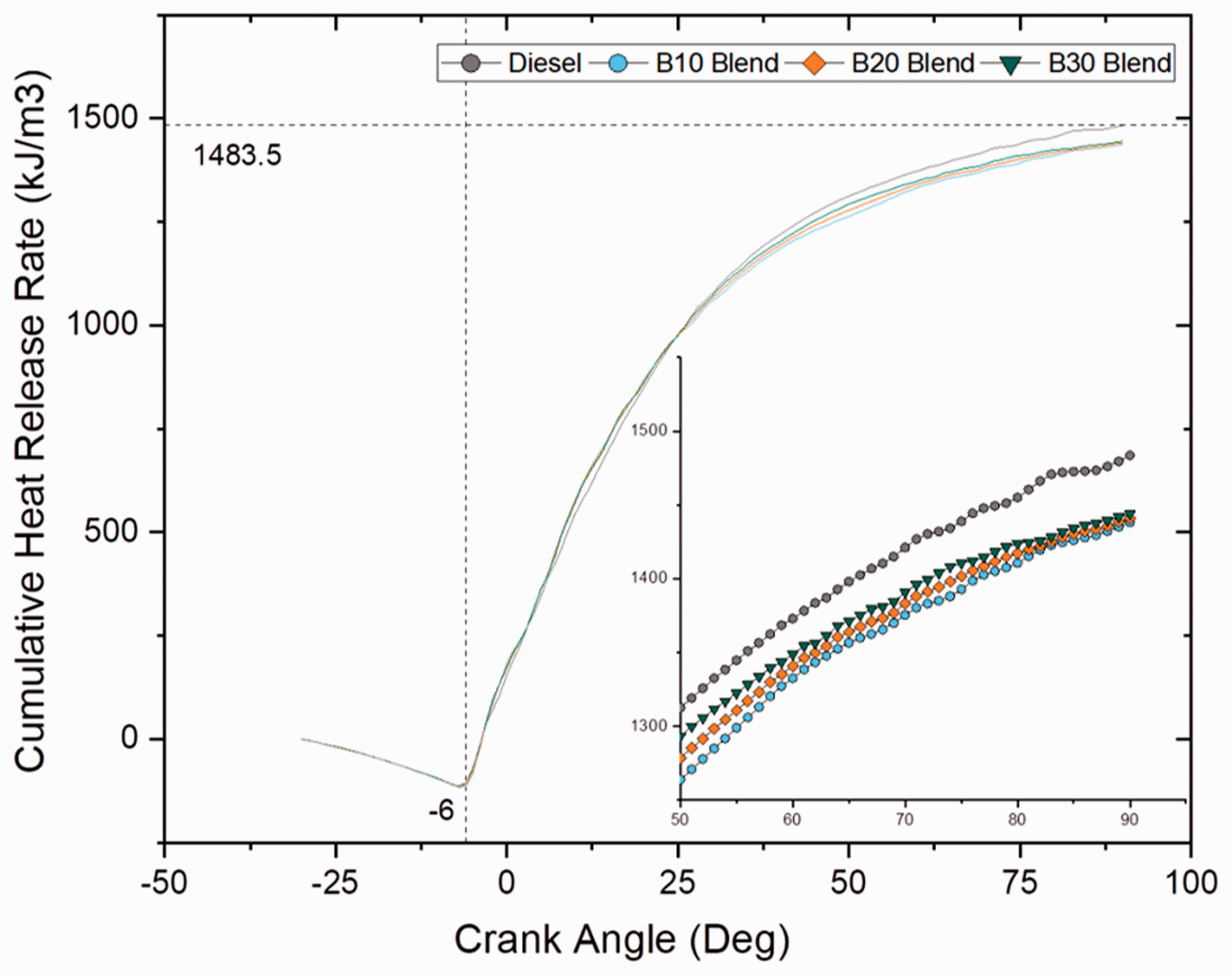
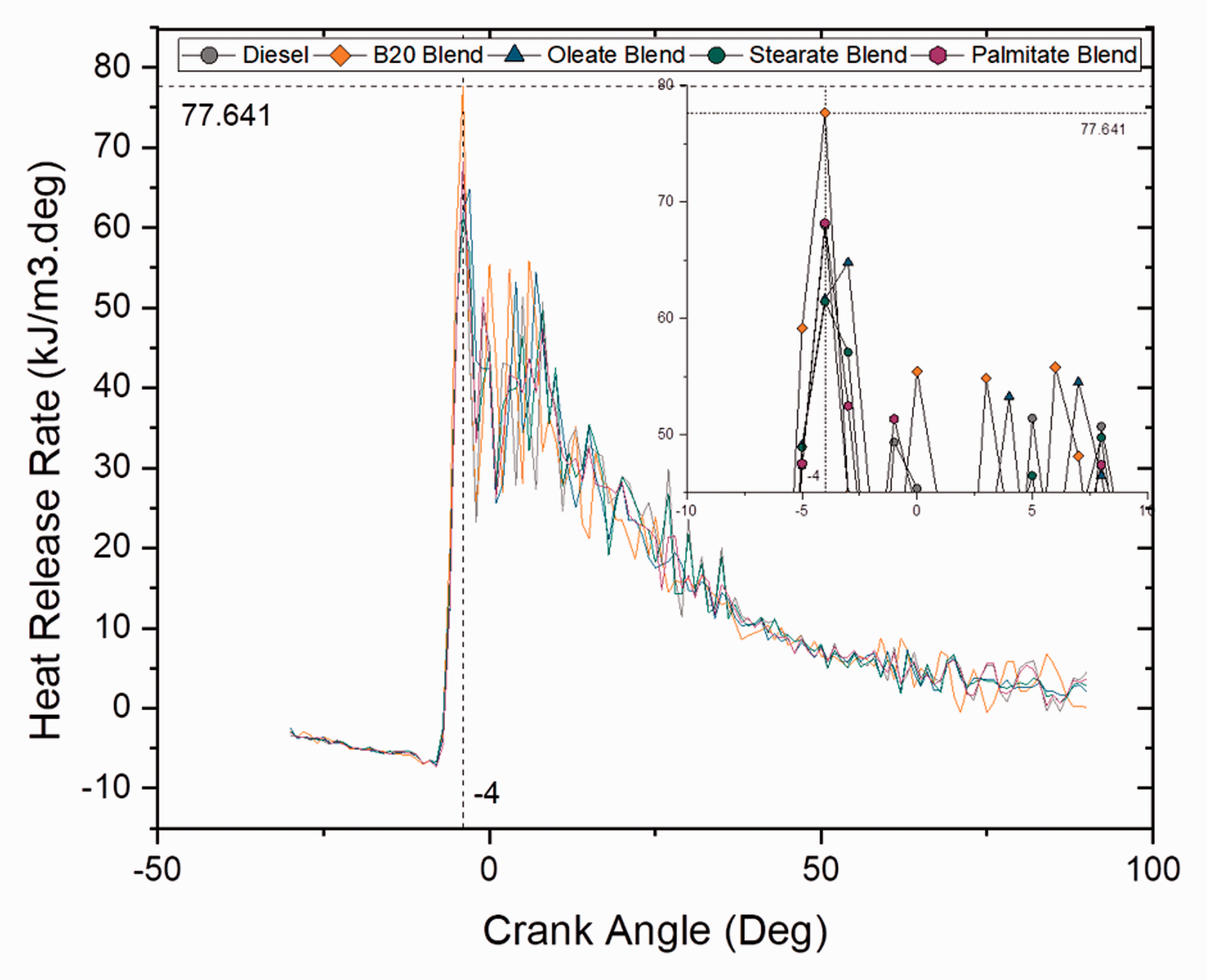
Figures 7 and 8 depict the heat release rate per degree at different crank angles for blend and ester samples at full load (100% load) conditions. Another important finding was that biodiesel (B20) blend had a very good heat release rate than compared to ordinary diesel and ester samples with a heat rate of 77 kJ/m3 deg and was found to be 13.2% higher than diesel sample. This result suggested that higher heat release rate was a result of higher oxygen content and CN. From heat release rate and cumulative heat release pattern, it was noticed that biodiesel blends and ester samples had shorter combustion duration; however, it increased with increase in load condition (Anand et al., 2010; Kumar et al., 2003).
Heat release rate for blend samples at 100% load. Heat release rate for ester samples at 100% load.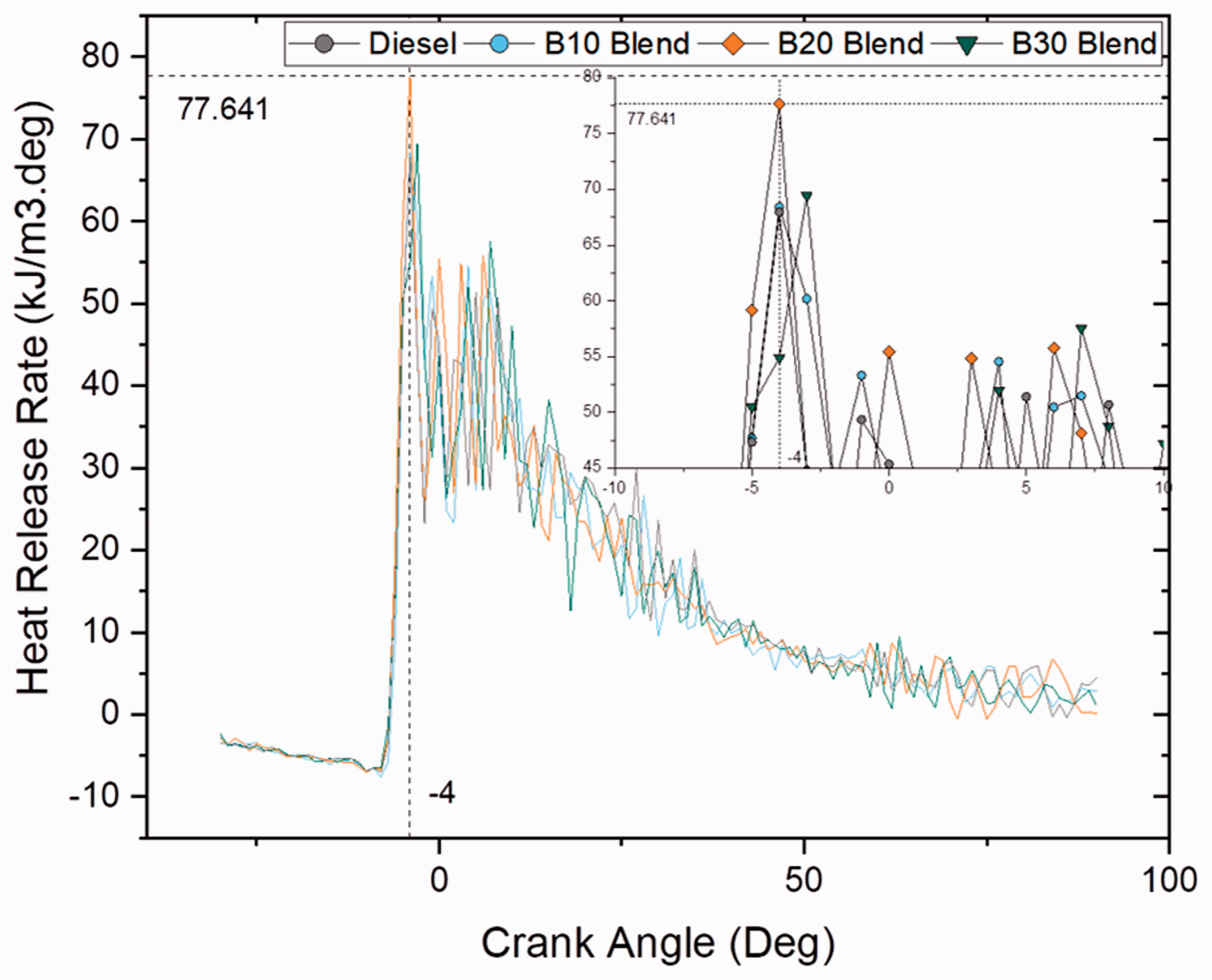
Exhaust gas temperatures
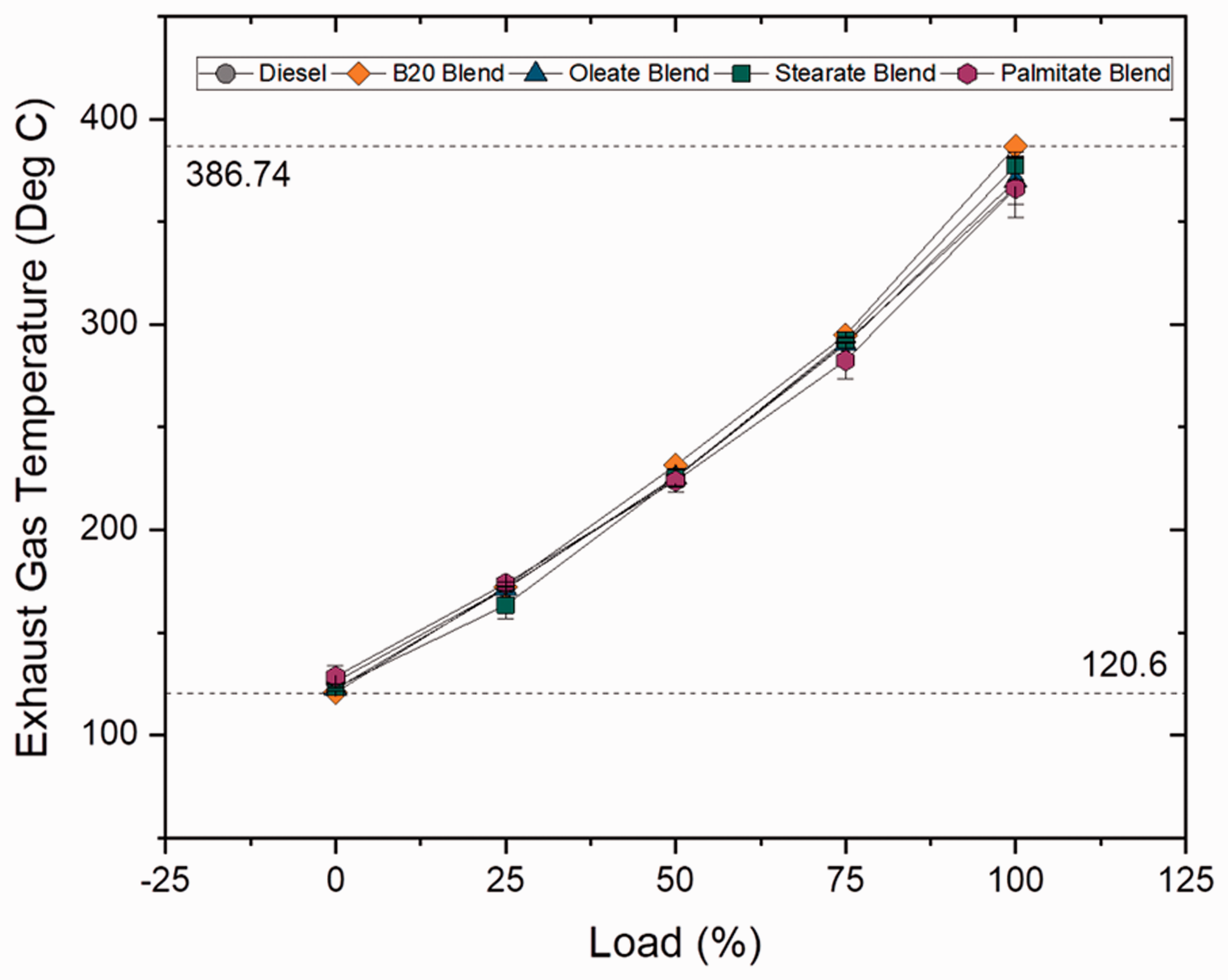
Exhaust gas temperature describes about the engine’s progress in terms of combustion. For delayed combustion start followed by longer combustion duration, exhaust gas temperature tends to be high. The NOX formation is directly influenced by high exhaust gas temperatures and decreases with reduction temperature. Figures 9 and 10 show exhaust gas temperatures at varying loads for blend and ester samples. In case of blend samples, it was concluded that biodiesel blends had higher exhaust gas temperature and increased by 1.14, 5.43 and 8.5% for B10, B20, B30 blends, respectively, when compared to diesel sample owing to reduced premixing time due to ascending CN at higher blends (Abu-Hamdeh and Alnefaie, 2015). In case of ester samples, oleate and stearate blend displayed only a marginal difference in temperature from biodiesel (B20) blend by 0.9 and 2.84%, respectively. It is clearly evident that higher exhaust gas temperature for biodiesel sample is because of higher CN and abundant availability of oxygen content in the sample.
Exhaust gas temperature for blend samples at various loads. Exhaust gas temperature for ester samples at various loads.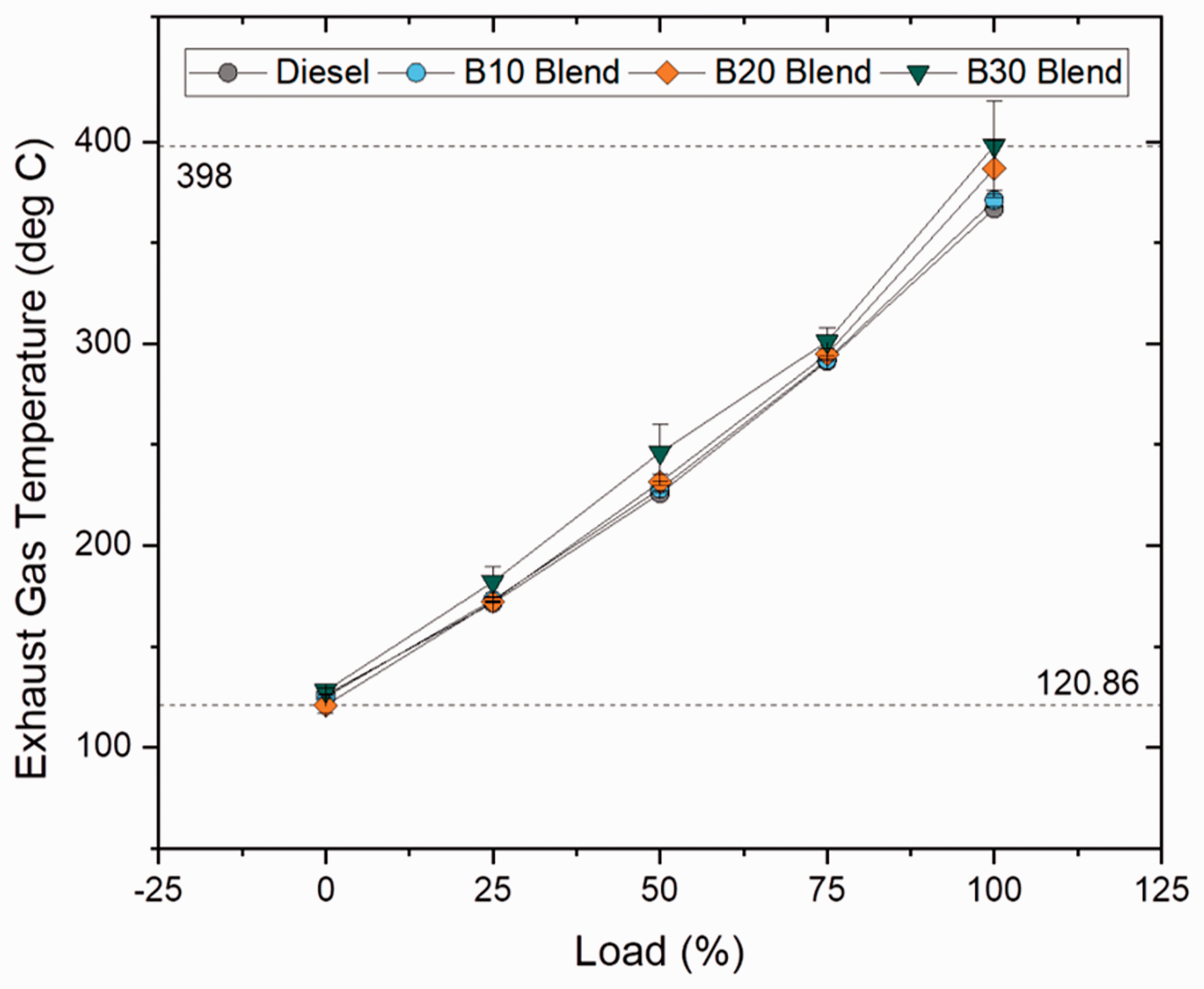
Performance characteristics
Specific fuel consumption
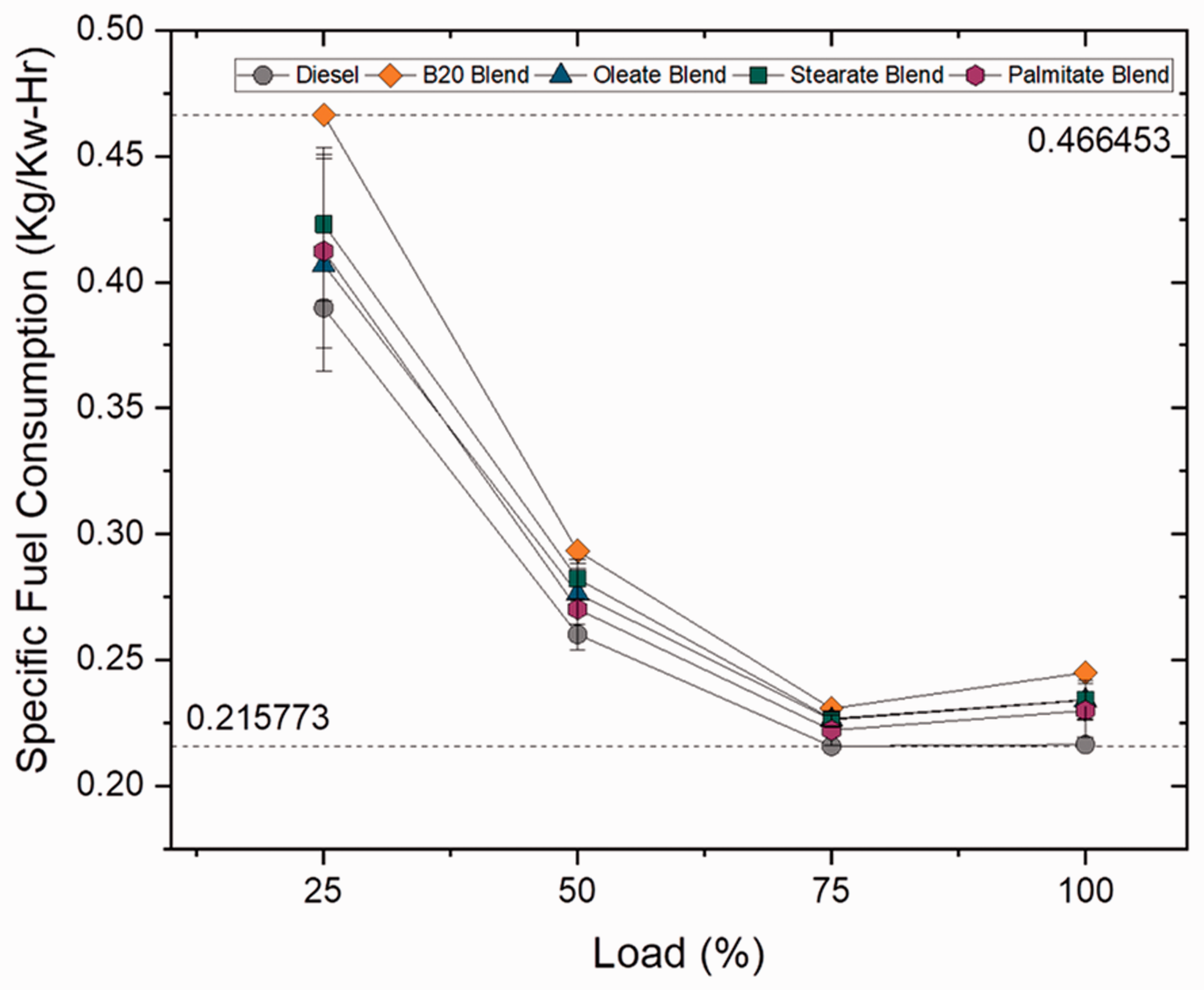
Specific fuel consumption describes the amount of fuel required for deriving 1 unit of power and increases with increase in load condition because of high brake power at higher load conditions (Patil and Patil, 2018). Figures 11 and 12 illustrate the specific fuel consumption at varying loads for blend and ester samples. For blend samples, specific fuel consumption increased progressively with increasing blend ratios as 8.20, 13.22 and 21.26% for B10, B20 and B30 blends, respectively, because of higher density and viscosity range than compared to plain diesel (Kumar et al., 2015). In case of ester samples, stearate and oleate blends displayed a marginal difference in consumption rate by 4.42 and 4.46% from biodiesel (B20) blend owing to their high viscosity range because of increased carbon chain length when compared to palmitate blend (shorter carbon chained ester). However, higher SFC rate for biodiesel (B20) blend was deeply influenced by the presence of stearate and oleate molecules in it. Hence, this proves that the rate of fuel consumption is directly related to length of carbon chain present in fatty acid ester molecule.
Specific fuel consumption for blend samples at various loads. Specific fuel consumption for ester samples at various loads.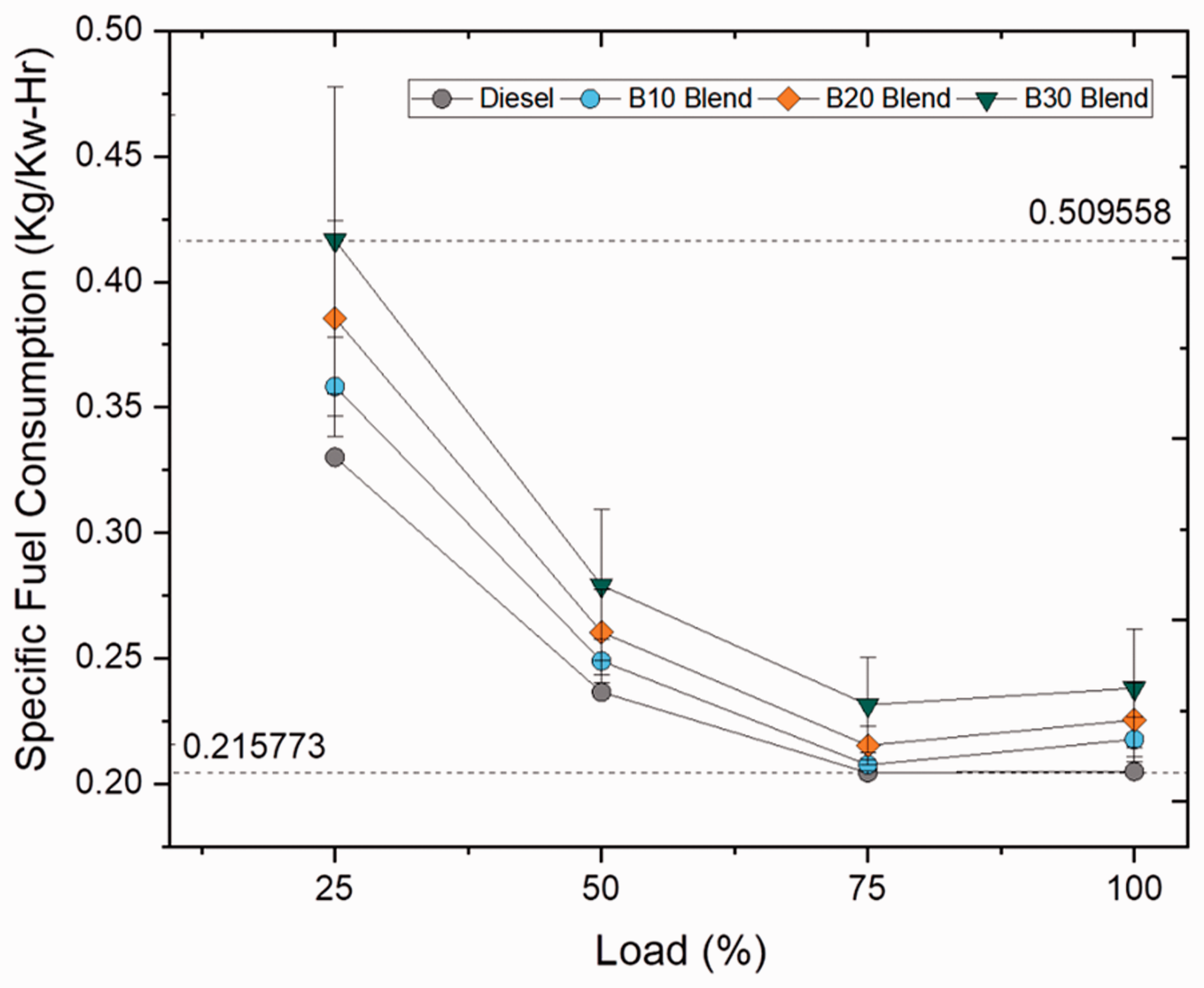
Brake thermal efficiency
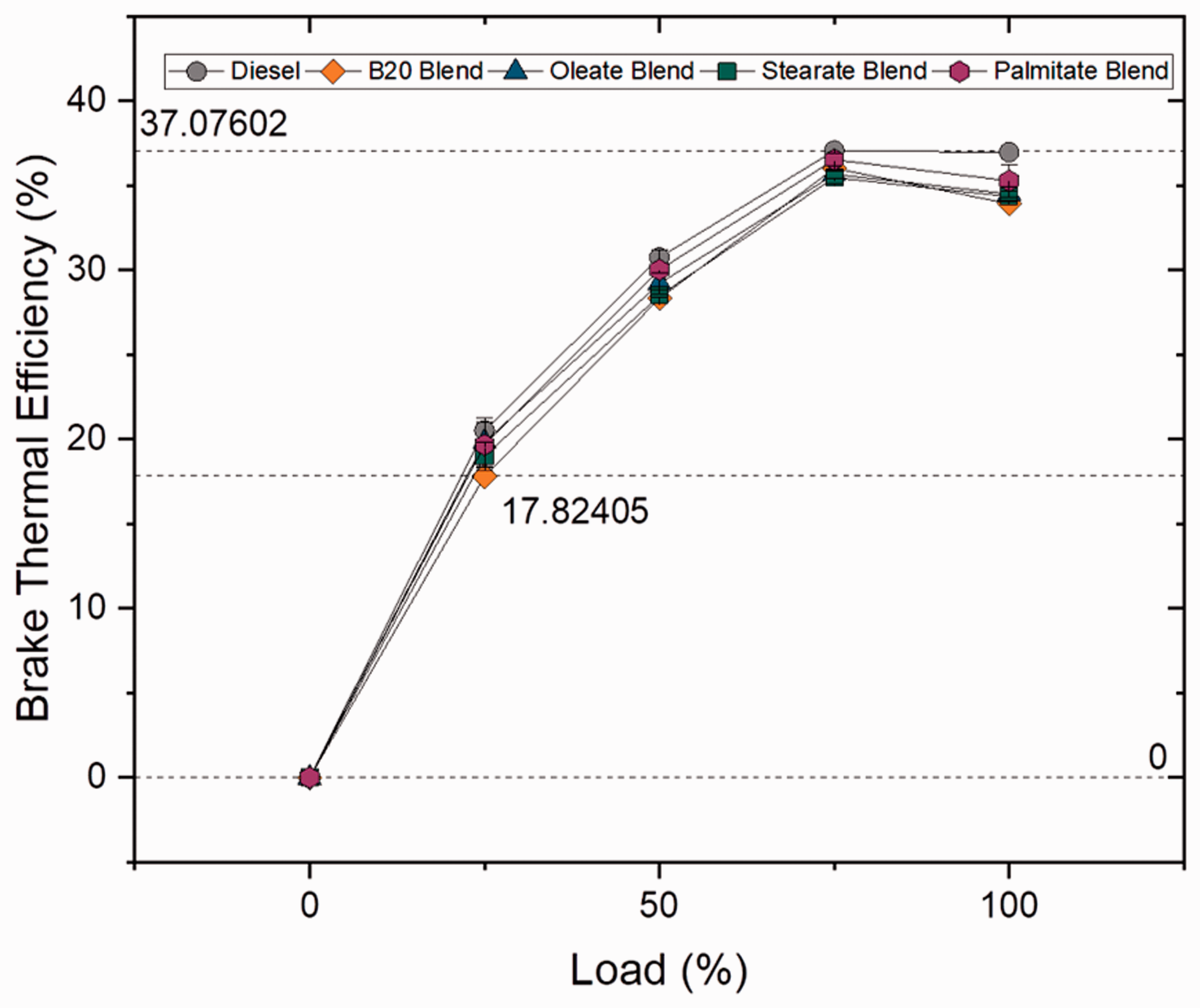
Brake thermal efficiency is defined as the ratio between the brake power to fuel power, where fuel power is the product of mass flow rate and calorific value of the fuel used, and used for evaluating the engine’s ability to convert the thermal energy from the fuel into mechanical energy. BTE increased for all the samples as load increased because of increasing power to be delivered at higher loads (Sekhar et al., 2018). Figures 13 and 14 illustrate the brake thermal efficiency at varying loads for blend and ester samples. For blend samples, it was clearly evident that the BTE was higher for plain diesel and decreased with increase in blend ratio by 5.8, 8.21 and 12.58% for B10, B20 and B30 blends when compared to plain diesel. The brake thermal efficiency reduced for biodiesel samples owing to its higher viscosity and lower calorific value (Prabu, 2018) whereas increased for plain diesel because of its lower SFC rate and superior calorific value (Khiari et al., 2016). Similarly, the BTE was found to be superior for palmitate blend (3.98%) followed by other ester blends (1.68 and 1.24% for oleate and stearate blend, respectively) than compared to biodiesel blend owing to lower specific fuel consumption rate. In spite of high viscosity of biodiesel blends, higher BTEs were achieved at higher loads because of complete combustion of fuel with the help of affluent oxygen content present in biodiesel sample (Debbarma and Misra, 2017). Furthermore, variation in BTE can be understood by studying the combustion duration, phasing and maximum heat release rates (Duraisamy et al., 2016).
Brake thermal efficiency for blend samples at various loads. Brake thermal efficiency for ester samples at various loads.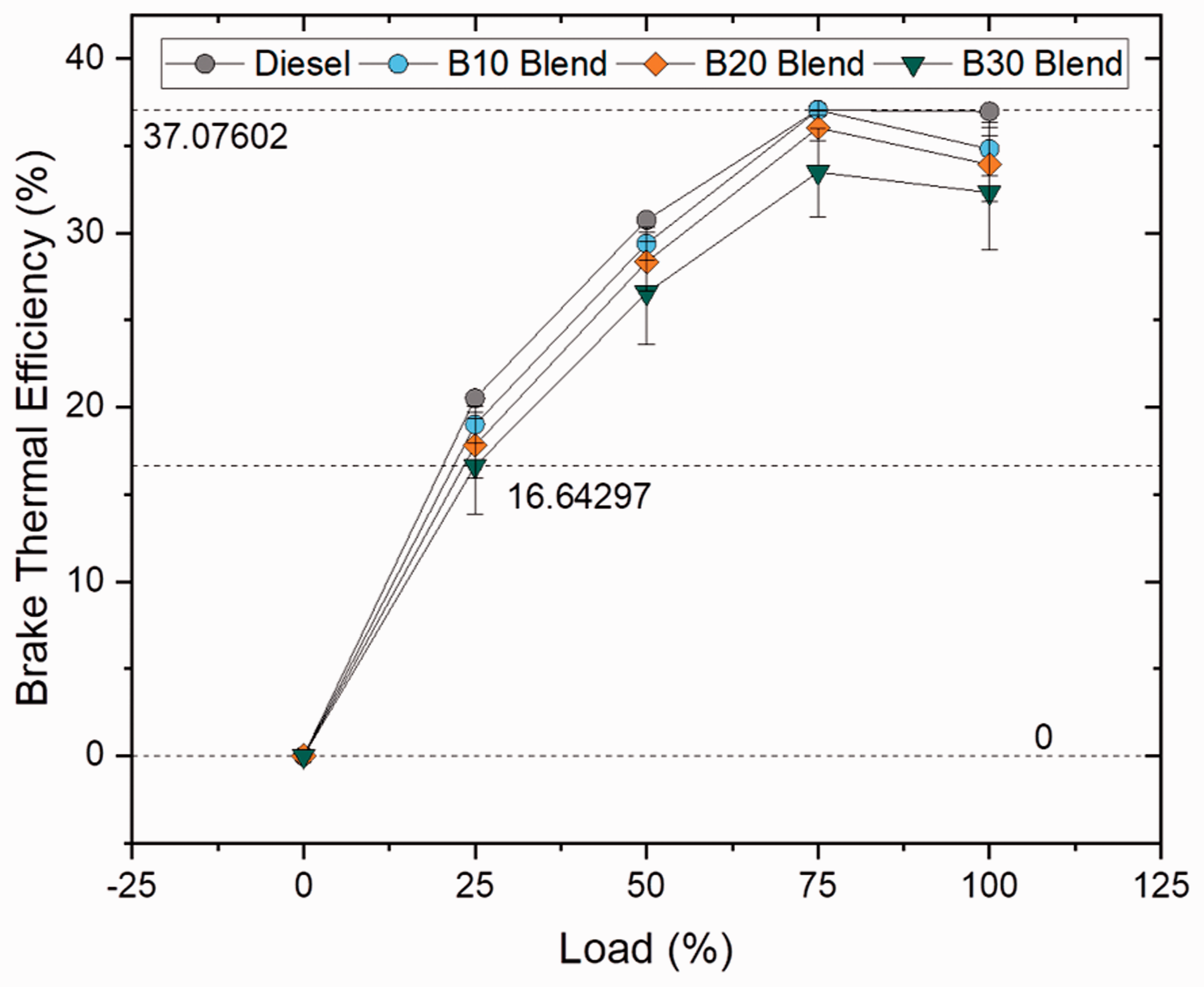
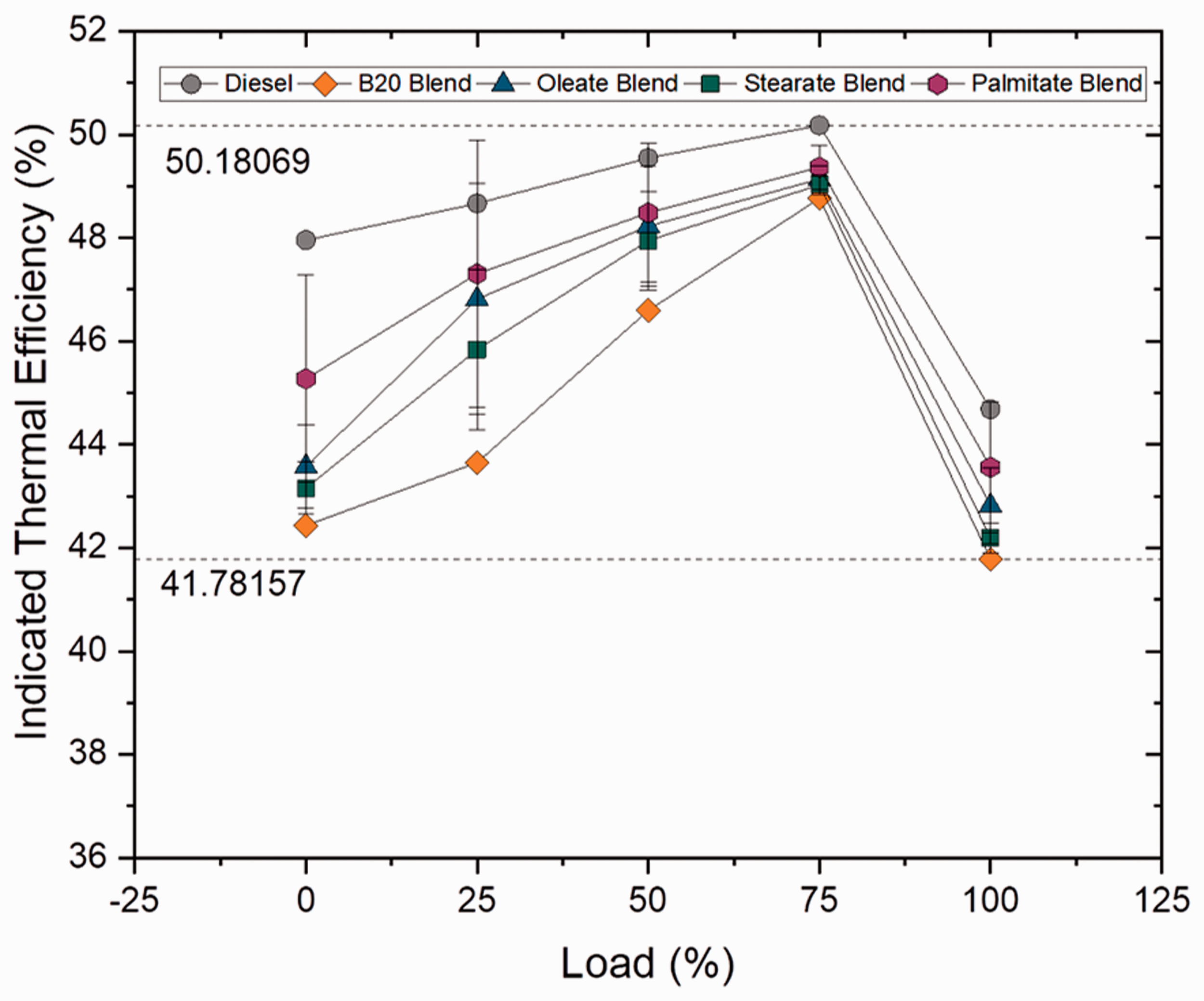
Indicated thermal efficiency
Indicated thermal efficiency is defined as the ratio between the indicated power to fuel power, where fuel power is the product of mass flow rate and calorific value of the fuel used. For both blend and ester samples, indicated thermal efficiency increased gradually up to 75% load condition and decreased as load condition increased further up to 100%. For blend samples, the ITE decreased with increase in blend ratio by 4.8, 6.49 and 8.85% for B10, B20 and B30 samples, respectively, because of higher viscosity range of the samples which led to reduced atomization, vaporization and combustion. Even though heat release rate increased with increase in carbon chain length, the increased viscosity of the samples leads to poor ITE. In case of ester samples, the ITE was found to be 2.48 and 0.97% lower for oleate and stearate blends when compared to biodiesel (B20) blend whereas only a marginal deviation was noticed in between diesel and palmitate sample(4.26%). Similarly, the ITE was found to be lower for biodiesel blend because of higher viscosity and this lower ITE was because of influence of oleate and stearate molecules in it. Figures 15 and 16 illustrate the indicated thermal efficiency at varying loads for blend and ester samples.
Indicated thermal efficiency for blend samples at various loads. Indicated thermal efficiency for ester samples at various loads.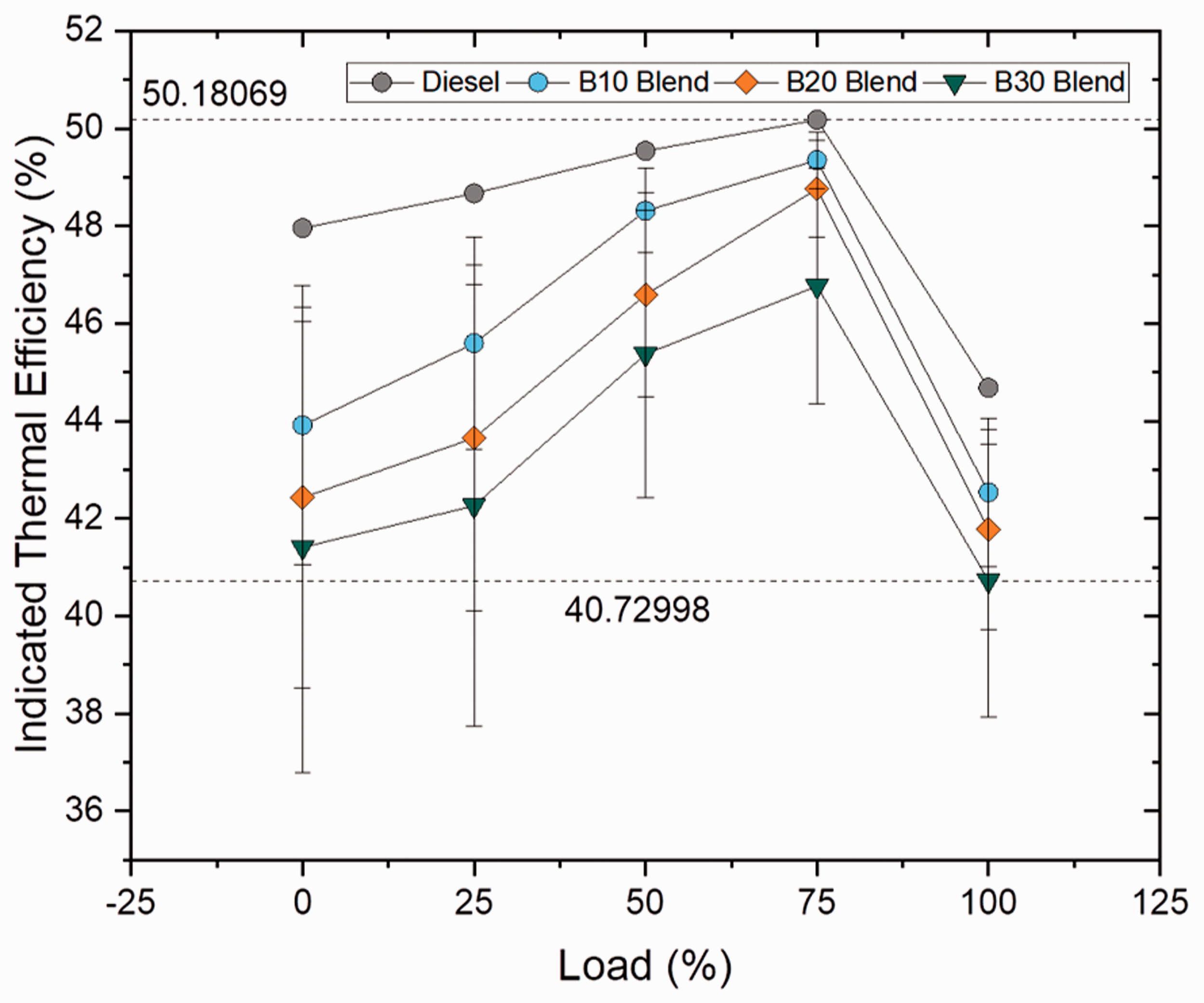
Emission characteristics
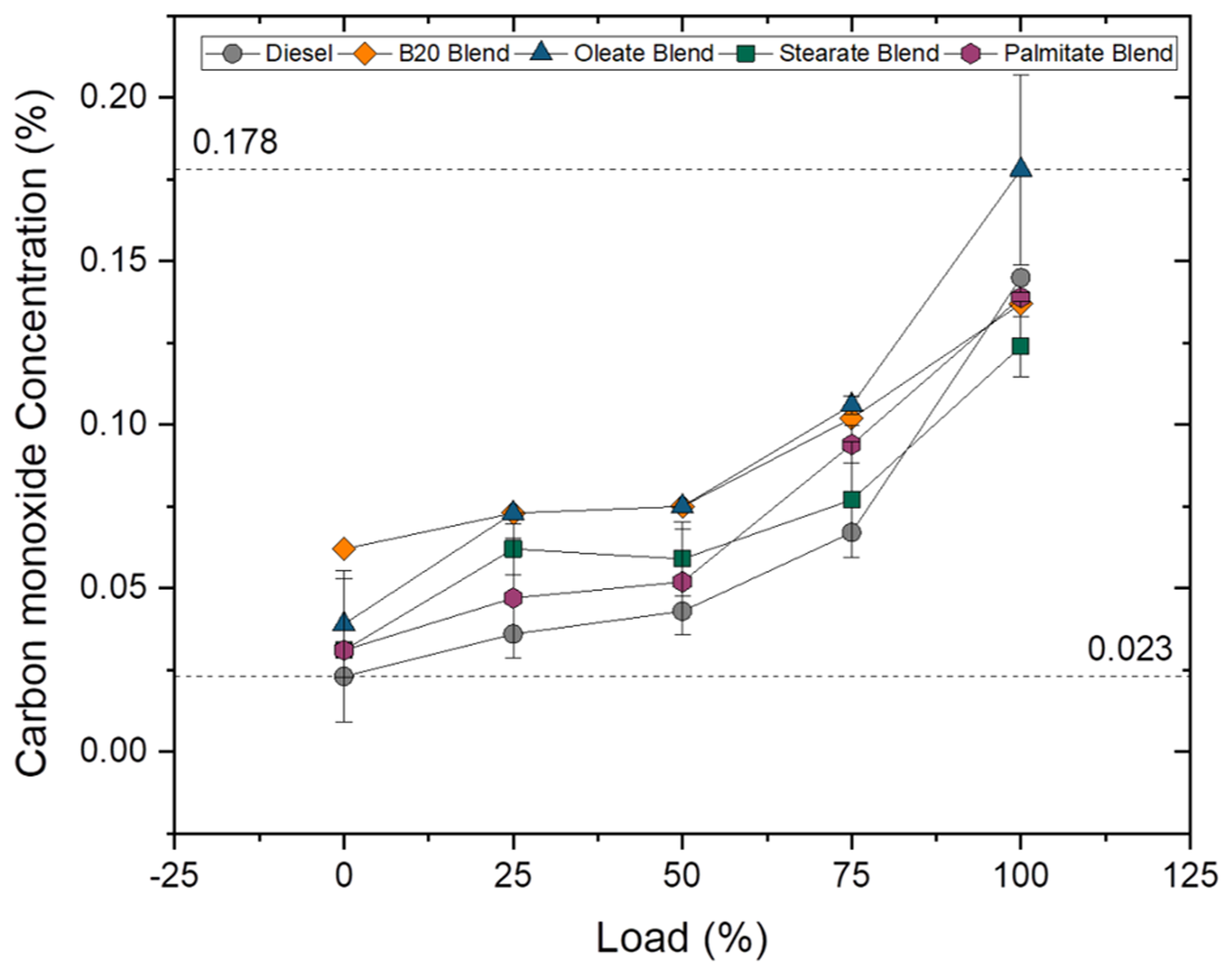
Carbon monoxide (CO) emissions
CO is formed as a result of incomplete combustion and is widely influenced by C/H ratio, degree of unsaturation, type of fuel used and cylinder temperature. The CO emissions for B10 blend and oleate blend were 7.17 and 22.75% higher than plain diesel, but was found to be lesser by 5.83 and 16% for B20 and B30 blends in case of blend samples and 4.31 and 16.93% for palmitate and stearate blends in case of ester samples. In addition to that, the CO emission was found to be 1.46%, 29.92% higher for palmitate and oleate blend and 10.48% lower for stearate blend when compared to biodiesel blend. It was clearly evident that the concentration of CO emission reduced with increase in blend ratio because of high availability of oxygen content. The abundant availability of oxygen at higher blends drastically reduced the CO emission. On the contrary, the CO emission was found to be higher in oleate blend and biodiesel blend because of unsaturated carbon bond (C = C) in both fatty acid ester molecules (Chukwuezie et al., 2017). However, CO emission increased for all samples at 100% load owing to rich fuel consumption to meet the higher power demand. Figures 17 and 18 illustrate the concentration of CO emission at varying loads for blend and ester samples.
CO concentration for blend samples at various loads. CO concentration for ester samples at various loads.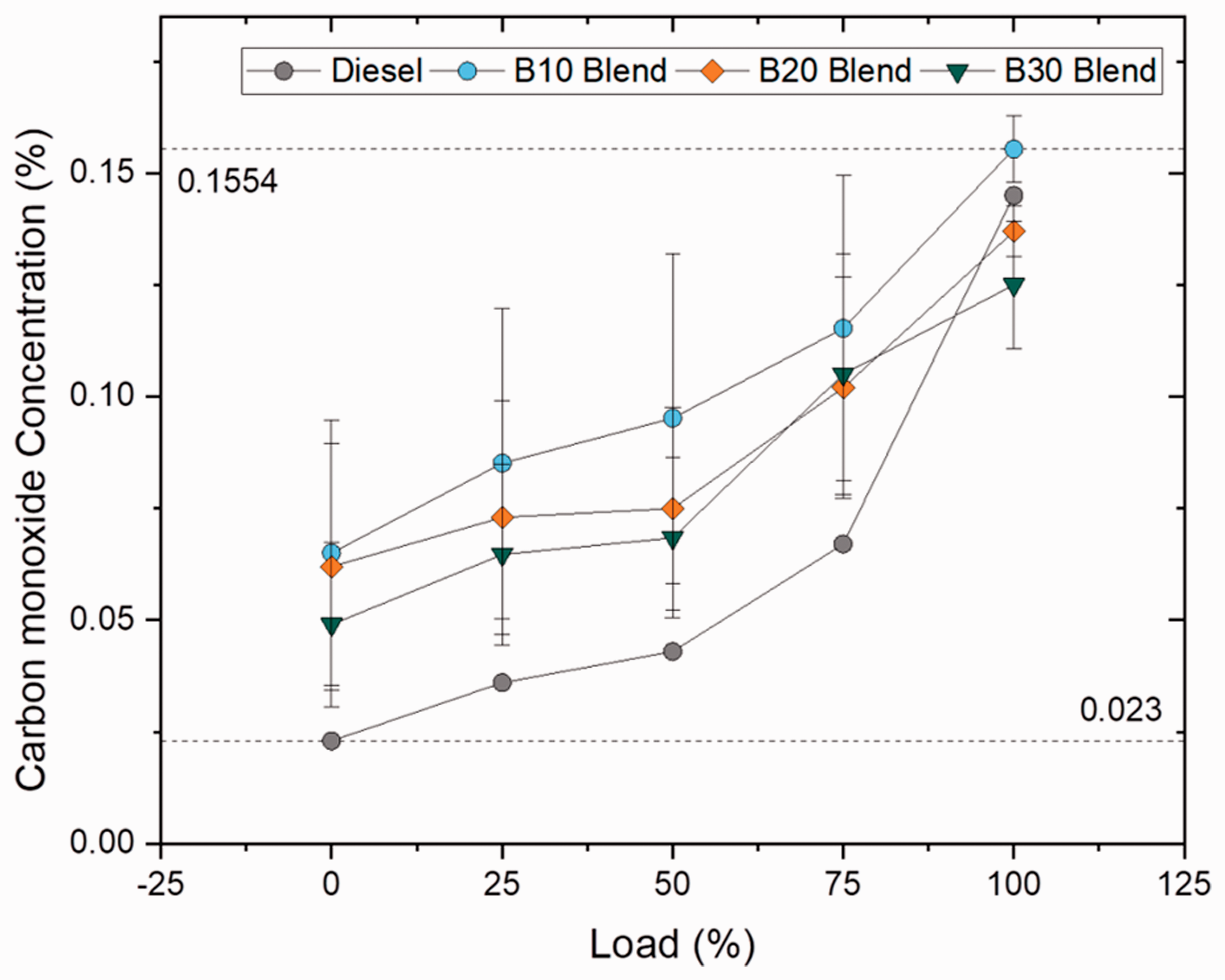
Carbon dioxide (CO2) emissions
CO2 is the primary product formed during combustion of hydrocarbons and is also influenced by C/H ratio, degree of unsaturation, type of fuel used and cylinder temperature. The CO2 emission increased by 4.16, 13 and 21.53% for B10, B20 and B30 blends for blend samples whereas in case of ester samples, a marginal rise by 1.6% was noted for palmitate blend and drastic rise was noticed for stearate and oleate blends which increased by 9.81 and 10.87%. In addition, the higher concentration of CO2 emission for higher blends was a result of higher oxygen content and viscosity rate which provides time for complete combustion during the power stroke (Abdul Malik et al., 2017). The CO2 emission was found to be lesser by 1.92, 2.91 and 11.23% for oleate, stearate and palmitate blends when compared to biodiesel blend. Meanwhile, increased carbon length (C18 and C18:1) resulted in increased CO2 emission for stearate and oleate blends, whose influence was noticed in biodiesel blend. Nonetheless, increase in CO2 emission reduced the concentration of toxic CO emissions. Figures 19 and 20 illustrate concentration of CO2 emission at varying loads for blend and ester samples.
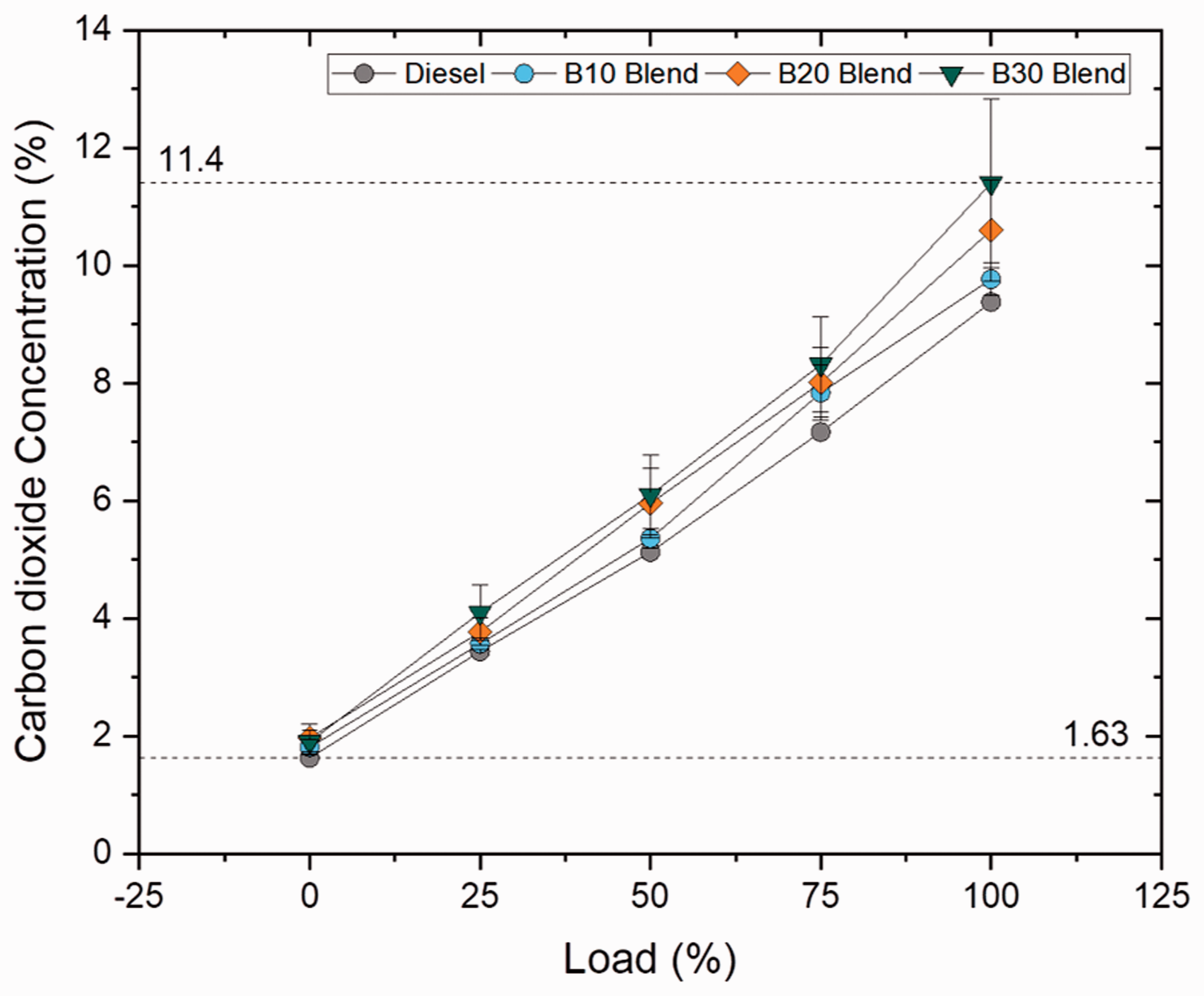
CO2 concentration for blend samples at various loads.
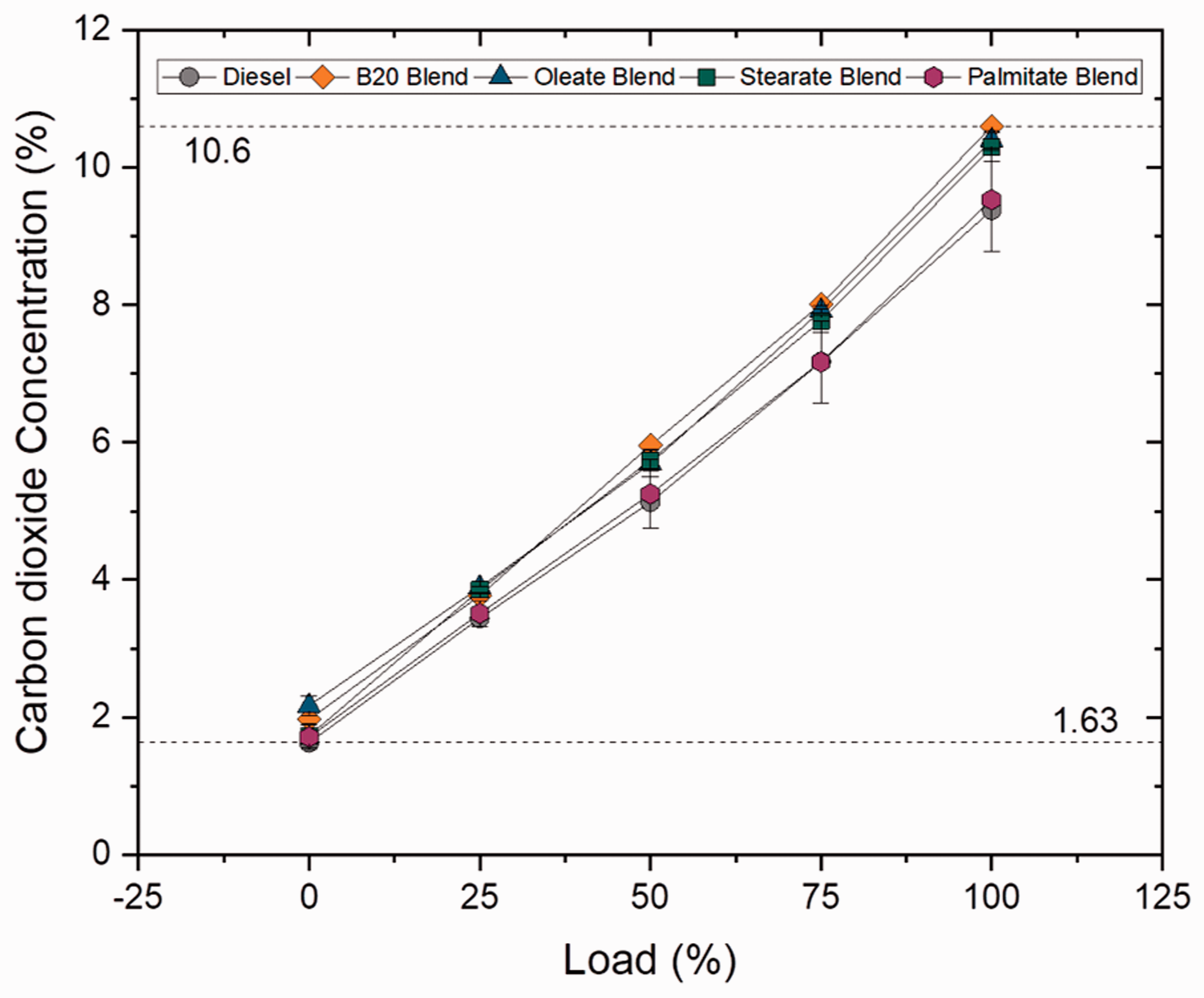
CO2 concentration for ester samples at various loads.
Nitrogen oxide (NOX) emissions
The NOX concentration is administered by peak engine temperature and increases with increase in higher CN, high oxygen content and longer residence time. NOX emission for blend samples reduced by 16.87, 17.88 and 3.17% for B10, B20 and B30 blends when compared to plain diesel and increased with increase in blend ratio because of higher viscosity and oxygen content; however, B20 has reduced NOX emission because of shortened ignition delay and less time for air–fuel mixing which resulted in low NOX emission. The NOX emission was found to be lower for ester samples, by 12.5% for oleate sample and 20% lesser for other ester samples when compared to plain diesel because of reduced combustion heat release rate and higher viscosity of ester samples. These findings suggest that NOX emission was found to be lesser than compared to biodiesel blend by 2.44%, 3.16% for stearate and palmitate blend and increased by 4.78% for oleate blend. It is a common phenomenon that unsaturation in biodiesel causes higher NOX rate; in contrast, lower concentration of NOX was recorded in B20 and ester blends because of lower concentration of unsaturation in the sample. Figures 21 and 22 illustrate the concentration of NOX emission at varying loads for blend and ester samples.
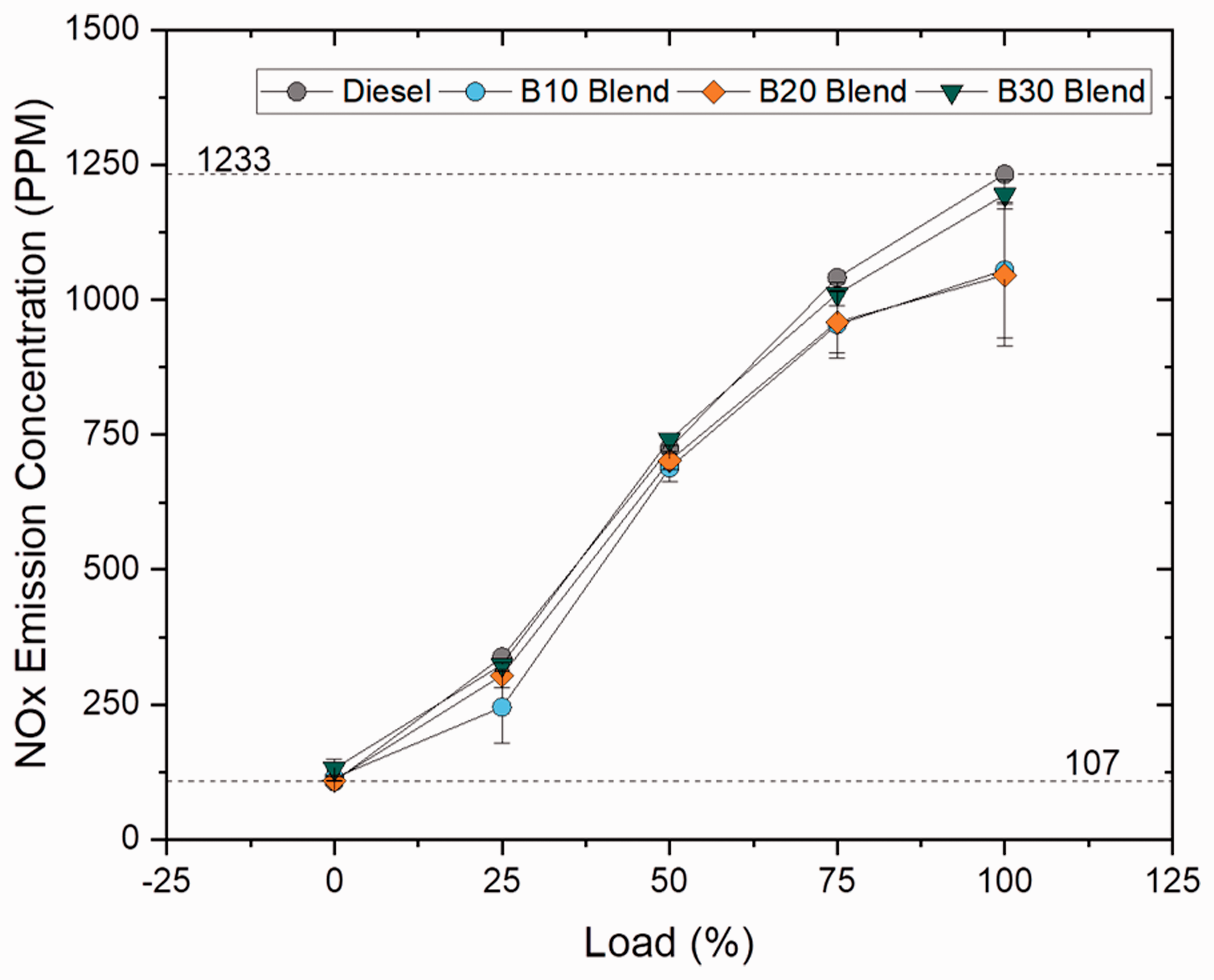
NOX concentration for blend samples at various loads.
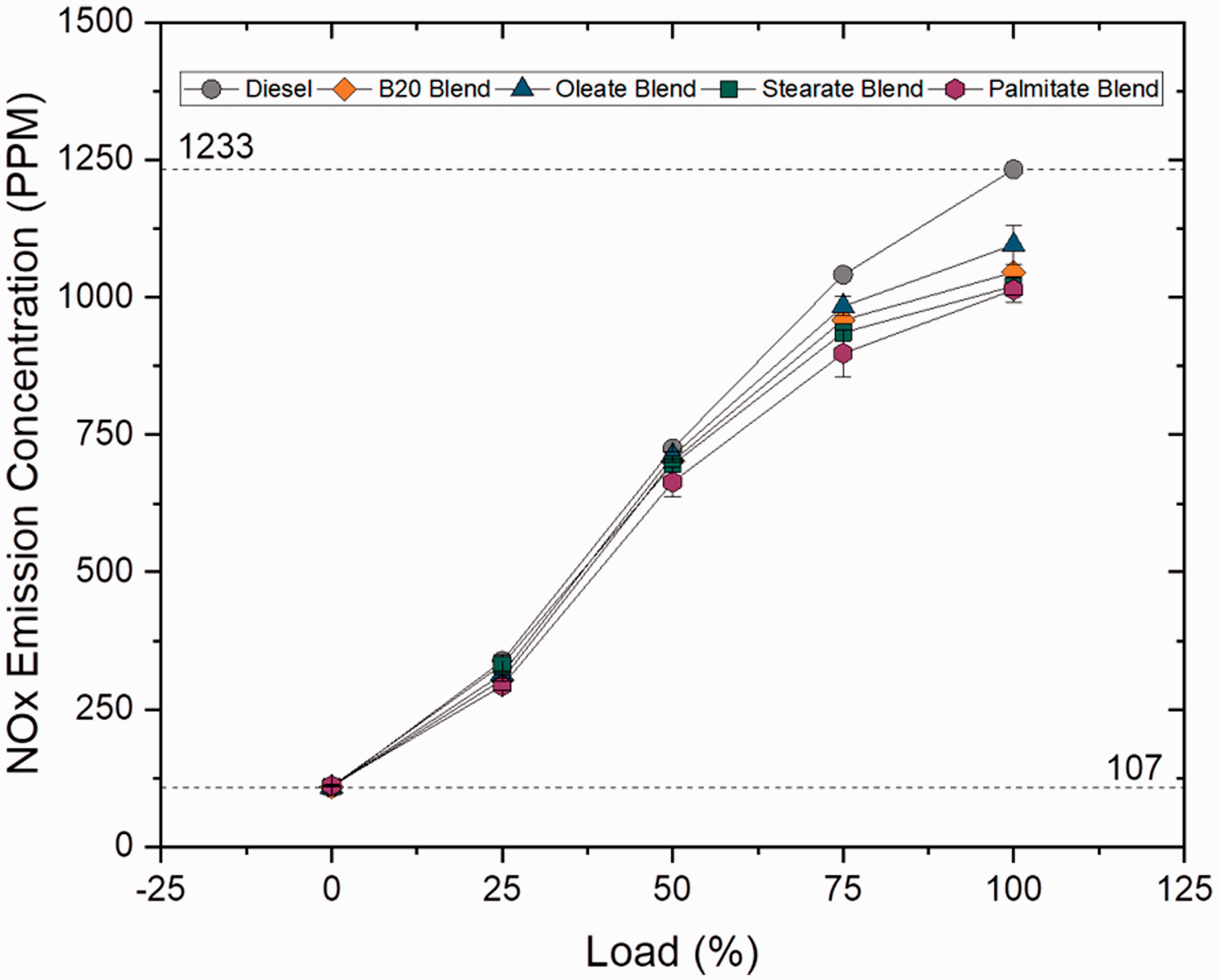
NOX concentration for ester samples at various loads.
Conclusions
Thus, the comparative study related to the PCE characteristics between biodiesel from beef tallow and their dominant fatty acids esters has been carried out and their influence in the combustion process has been analysed. Following were the conclusions drawn from this experimental work.
Ethyl palmitate, ethyl oleate and ethyl stearate were identified as the dominant fatty acid esters in the biodiesel sample produced from waste beef tallow. The maximum cylinder pressure and heat release rate were result of higher CN and high oxygen content which were imparted by all the three fatty acid ester molecules individually. Increase in exhaust gas temperature, specific fuel consumption and reduction in thermal efficiencies were influenced by oleate and stearate esters owing to their longer carbon chain length and higher viscosity rate. Similarly, the CO2 concentration increased because of longer carbon chained stearate and oleate molecules; however, the CO concentration was influenced by the unsaturated double bond in the oleate molecule and NOX concentration was reduced as a result of influence of palmitate molecule.
It was clearly evident that PCE characteristics of biodiesel were deeply influenced by the dominant fatty acid esters by means of C/H ratio, degree of unsaturation, individual CN and carbon chain length. This work provides key evidence that PCE characteristics and physico-chemical properties of any biodiesel are deeply influenced by its dominant fatty acid esters. In conclusion, this work also paves a path for engineering a biofuel as per the requirement and can help in developing a high calorific fuel with reduced/zero emission thereby leading the world towards sustainable development.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
