Abstract
The interactions between supercritical CO2 and coal and their effects on changes in the coal pore structure and organic groups play a critical role in the CO2 geological storage-enhanced coalbed methane recovery. To investigate the effects of supercritical CO2 on organic groups in coals of different ranks and its mechanisms under different temperature and pressure conditions, CO2 sequestration processes in bituminous coals and high-rank coals were replicated using a high-pressure reactor. Four coal samples of different ranks were exposed to supercritical CO2 and water under three temperatures and pressures for 240 h. Fourier transform infrared spectroscopy was used to provide semiquantitative ratios and Fourier transform infrared spectra of coal samples before and after the supercritical CO2–H2O treatment. The results show that interactions between supercritical CO2 and coal were controlled by the coal macromolecular structure, and semianthracite is the inflection point of interaction characteristics for coal samples of different ranks. Bituminous coal, including high- and low-volatility bituminous coal, has a low degree of condensation of its aromatic structure, and its aromatic nuclei can facilitate addition reactions. Swellings primarily break cross-links between aromatic nuclei in the same aromatic layer. These characteristics favor the polymerization addition of aliphatic side chains of aromatic nuclei, causing an increase in the degree of condensation of the aromatic structures in bituminous coal. High-rank coals including semianthracite and anthracite have a high degree of condensation of their aromatic structures, and the aromatic nuclei favor substitution reactions. Swellings primarily break cross-links connecting different aromatic layers, and bond dissociation reactions and sulfuration reactions are more significant for high-rank coal. These characteristics cause a decrease in the degree of condensation of the aromatic structure in high-rank coal. Temperature and pressure have a great impact on interactions between supercritical CO2 and coal and are controlled by the reaction types of the organic groups. With the increase in experimental temperature and pressure, the changes in the organic group content can be classified as the descending type, the rising type, the lower opening parabola type, and the upper opening parabola type. 45.0°C and 10 MPa is the inflection point of the changes in the organic group content. Descending- and rising-type changes favor addition, bond dissociation, and sulfuration reactions, which are endothermic. The reaction rate of supercritical CO2 and the organic groups increases, and the effects caused by temperature and pressure decrease as the temperature and pressure increase. Lower opening parabola- and upper opening parabola-type changes favor substitution, oxidation, and addition polymerization reactions, which are exothermic. These changes were significantly affected by a variety of reactions and were suppressed by high temperature and pressure. When the temperature is ≤45.0°C and the pressure is ≤10 MPa, supercritical CO2 has remarkable effects on alkyl and hydroxy groups and has a stronger effect on bituminous coal. When the temperature is >45.0°C and the pressure is >10 MPa, supercritical CO2 has remarkable effects on oxygen- and sulfur-containing groups and has a greater effect on high-rank coals.
Introduction
Due to the competitive adsorption advantage, CO2 sequestration in a coal seam can effectively displace CH4, which has become a new method for the improvement of coalbed methane (CBM) development and the reduction of CO2 emissions (Bergen et al., 2011). CO2 geological storage-enhanced coalbed methane recovery (CO2-ECBM) is an attractive technology for reducing greenhouse gas emissions and exploiting new energy sources, and it has attracted much attention from many researchers (Rodosta et al., 2011). At the reservoir temperature and pressure (burial depth >800 m), CO2 is likely to be present in a supercritical state (i.e. Tc = 31.06°C, Pc = 7.38 MPa) (Massarotto et al., 2010). The effect of supercritical CO2 (ScCO2) on the transformation of the organic composition and inorganic minerals of coal may lead to physical and chemical structural changes in coal, which lead in turn to changes in permeability and the adsorption capacity of the coal, which is particularly critical for the effectiveness of CO2-ECBM (Day et al., 2008; Kiyama et al., 2011; Lin et al., 2008; Siemons and Busch, 2007).
A ScCO2–H2O system affects coal in two different ways: (1) CO2 can dissolve in water and form an acidic solution containing H2CO3. The acidic solution can leach out inorganic minerals in coal, e.g. calcite, dolomite, magnesite, etc., which can change the pore–fracture structure and the connectivity of coal (Dawson et al., 2015; Liu et al., 2015, 2010; Massarotto et al., 2010; Wang et al., 2016a, 2016b). Recent studies of interactions between a ScCO2–H2O system and coal have focused on that aspect. (2) ScCO2 has good diffusivity and dissolving capacity that can dissolve some of the organic groups on the inner surface of the pores and fractures of coal during the migration process within them, which changes the physical and chemical structure of the coal (Kolak and Burruss, 2006; Mazumder et al., 2006; Zhang et al., 2017). The interactions between ScCO2 and coal organic groups are complex, and their effects are relative to the solubility of a compressed gas and affected by temperature, pressure, pore and fracture widths, coal rank, etc. (Jonathan et al., 2015; Kolak and Burruss, 2006; Pasquali et al., 2008; Stahl et al., 1978).
The ScCO2 extraction rate of coal organic groups slightly increased as the temperature and pressure increased, but the characteristics of the changes are different for coals of different ranks (Kolak and Burruss, 2006; Pasquali et al., 2008; Stahl et al., 1978). In a low-pressure range (7–10 MPa), ScCO2 can extract fewer polar hydrocarbons and lipids, e.g. alkanes and polycyclic aromatic hydrocarbons of lower molecular weight, esters, ethers, lactones, epoxy compounds, etc. (Stahl et al., 1978). The effects of ScCO2 on polar functional groups are weak (Massarotto et al., 2010). In a high-pressure range (>10 MPa), ScCO2 can extract some aliphatic hydrocarbons, hydroxyl groups, and other oxygen-containing groups and cause changes in the coal macromolecular structure and the destruction of the microcrystalline structure (Gathitu et al., 2009; Jonathan et al., 2015; Mirzaeian and Hall, 2006; Okolo et al., 2015). ScCO2 extraction rates of lignite/brown coals, bituminous coals, and anthracites increased at first but subsequently decreased and peaked when the coal carbon content was approximately 86% (Bertier et al., 2006; Liu and Smirnov, 2009; Mirzaeian et al., 2010; Wang et al., 2010).
Interactions between ScCO2 and coal proceed by two kinds of mechanisms. (1) ScCO2 adsorption and diffusion in coal can cause swelling. Swelling decreases interactions among coal macromolecular chains and extends coal macromolecular chains. Low-rank coals swell more readily, and temperature has a weak effect (Day et al., 2008; Gathitu et al., 2009; Siemons and Busch, 2007). (2) ScCO2 can weaken intermolecular forces in coal, causing the dissociation of low molecular weight compounds from the coal macromolecular structure (Tsotsis et al., 2004). Water can leach these low molecular weight compounds from the coal matrix, which may change the pore–fracture structure and rearrange coal physical structure (Tsotsis et al., 2004). The interactions between ScCO2 and low molecular weight compounds are affected by temperature and pressure. Other researchers have suggested that ScCO2 is a nonpolar solvent whose polarity does not change with pressure. Therefore, ScCO2 has a limited effect on low molecular weight compounds in coal, which are mainly polar compounds (Massarotto et al., 2010).
Scientists generally agree that the interactions between ScCO2 and coal organic groups can loosen the surface of coal particles and lead to coal macromolecular rearrangements and obvious plastification, which can increase the porosity and permeability of coal (Day et al., 2008; Siemons and Busch, 2007; Tsotsis et al., 2004; Wang et al., 2010). Meanwhile, different specific surface areas (both of the absolute content and the percentage composition) of pores with different widths decrease, which enhances the desorption ability of a coal (André et al., 2007; Dutka et al., 2012, 2013; Hao et al., 2013). Other scientists have suggested that ScCO2 can only break subagents in coal structure units but are unable to break the space frames of coal formed by chemical cross-links (Kutchko et al., 2013; Mastalerz et al., 2010). Therefore, ScCO2 can slightly change the pore size distribution in coal. Meanwhile, a consensus also exists that the interactions between ScCO2 and coal organic groups can effectively improve the adsorption capacity, specific surface area, and porosity of coal (Kutchko et al., 2013; Mastalerz et al., 2010). In conclusion, the effects of the interactions between ScCO2 and organic groups on coal porosity, permeability, and adsorption capacity and their important roles in CO2-ECBM have been generally recognized. However, the effects of ScCO2 on the changes in coal organic groups and their mechanisms are still controversial. A poor understanding of interactions between a ScCO2–H2O system and organic groups limits our understanding of CO2-ECBM and hinders engineering exploration.
In the present study, simulations of CO2 sequestration processes in coal at different temperatures and pressures combined with Fourier transform infrared spectroscopy (FTIR) were employed to describe the characteristics of the changes in the semiquantitative ratios and the absorbance peaks in FTIR spectra. Interaction mechanisms between a ScCO2–H2O system and organic groups are also discussed for bituminous coals and high-rank coals. This study aims to provide a better understanding of the theoretical effectiveness of CO2-ECBM, which can guide the optimization of CO2-ECBM engineering explorations.
Materials and methodology
Samples
Four groups of Chinese coal samples of different ranks were systematically collected: high-volatility bituminous coal from the Yangzhuang Mine, low-volatility bituminous coal from the Xinyuan Mine, semianthracite from the Yuwu Mine, and anthracite from the Sihe Mine, and these were, respectively, designated Coal #1 to Coal #4 (Table 1). Coal #1 was collected from coal seam #9 in the Taiyuan formation in the Bohaiwan basin. The Bohaiwan basin is rich in unconventional gas resources, including CBM and shale gas. Coals #2 to #4 were collected from coal seam #3 of the Shanxi formation in the southern Qinshui basin. The southern Qinshui basin is the most active region for CBM exploration in China, and it is the only CO2-ECBM demonstration study area in China, which presents favorable conditions for research (Liu et al., 2016, 2017, 2015). The collection, retention, and preparation of the coal samples were conducted according to the relevant standard GB/T 19222-2003 in China (Zhong et al., 2003) and the international standard ISO 7404-2:1985. To prevent these coal samples from further oxidizing, coal samples collected from the coal mine working faces were wrapped in absorbent paper and hermetically sealed in plastic bags at 5°C. The key properties of these coals are shown in Table 1.
Properties of the coals used.
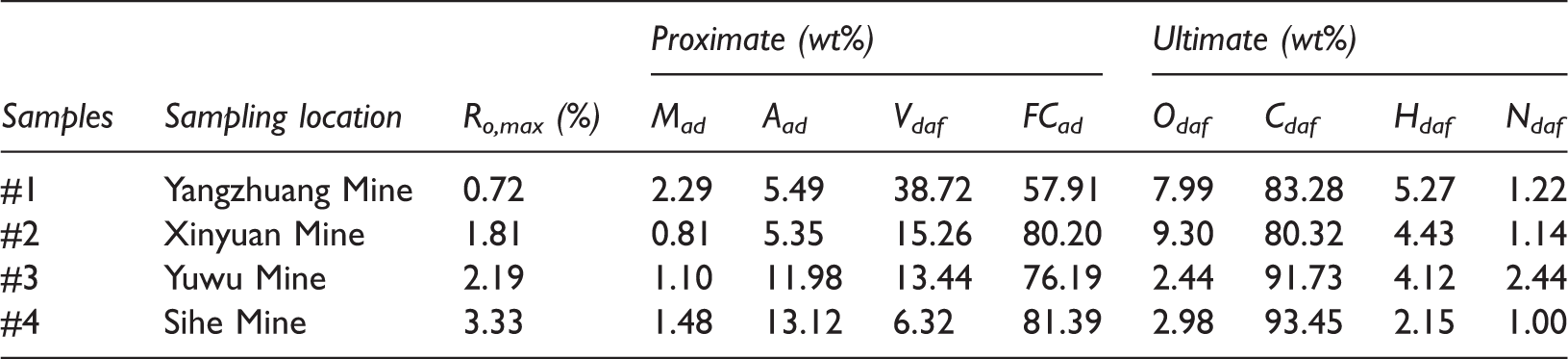
Aad: ash yield, air-drying base; Cad: content of carbon, dry ash-free basis; FCad: fixed carbon content, air-drying base; Had: content of hydrogen, dry ash-free basis; Mad: moisture, air-drying base; Nad: content of nitrogen, dry ash-free basis; Odaf: content of oxygen, dry ash-free basis; Vdaf: volatile matter, dry ash-free basis; wt%: weight percent.
ScCO2–H2O treatment
High-pressure reactor
The interactions between ScCO2–H2O and coal were carried out in a high-pressure reactor (Figure 1). The reactor can simulate the conditions for CO2 sequestration in a deep coal seam, which are a maximum of 35 MPa and 200°C. The key component of this apparatus is a high-pressure tubular stainless steel sample cell, which is 100 mm in diameter and 200 mm in height. A coal sample was held in a 60–80 mesh (0.20–0.25 mm) stainless steel tube in the sample cell, which allowed the water and CO2 to flow through the coal sample.
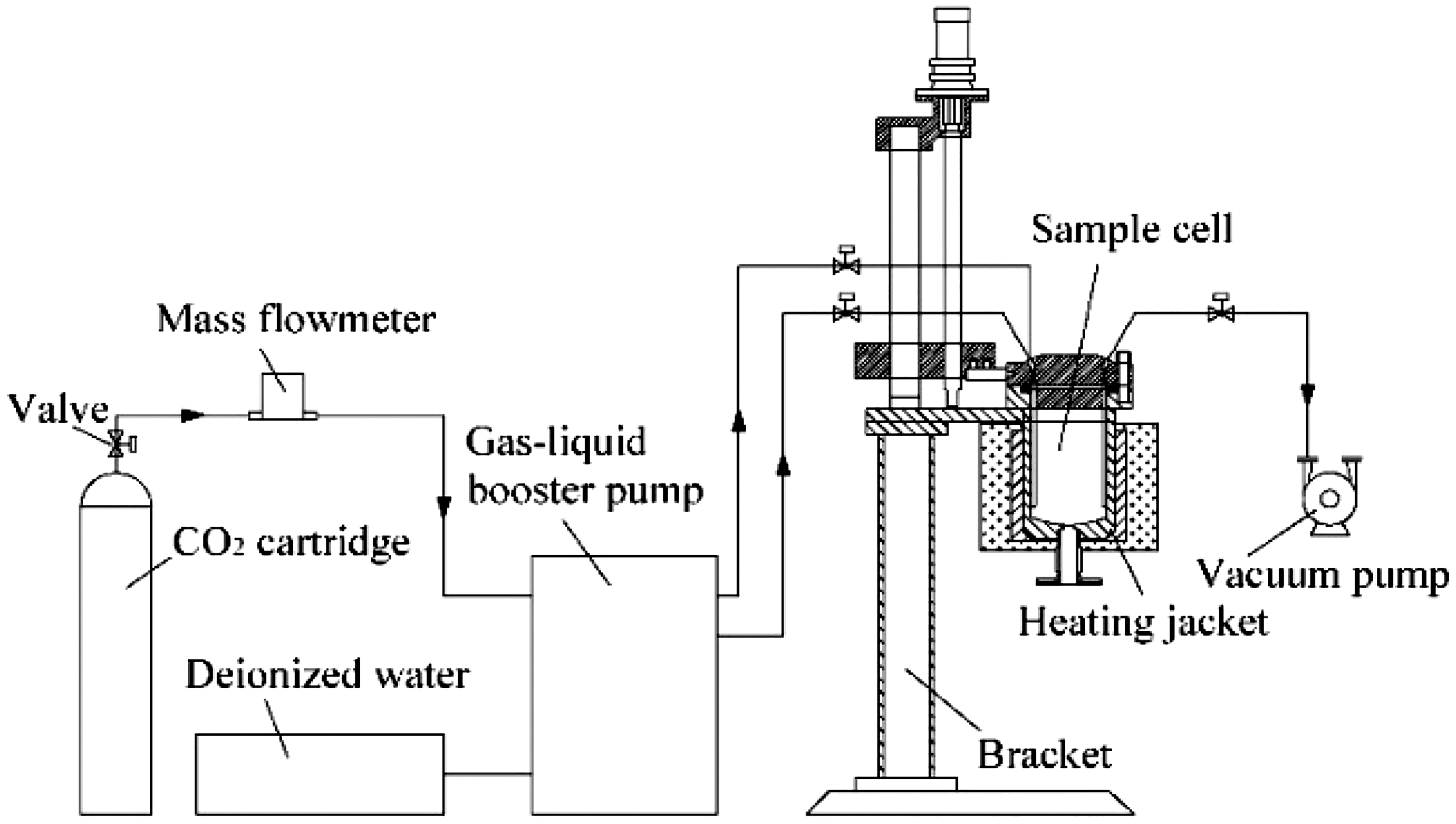
Sketch of the high-pressure reactor.
Coal grain size
In principle, it is better to choose coal with a large particle size for an experimental simulation of in situ conditions because it captures the actual conditions more realistically. In contrast, for laboratory experiments, using coal with a large particle size will result in an inconveniently long reaction time. Therefore, the coal samples used for ScCO2–H2O treatment were coal particles sieved into 4–8 mm grain sizes. Because the volume of the sample cell was approximately 1500 ml, a 200 g coal sample was used for ScCO2–H2O treatment.
Temperatures, pressures, and time in the experiment
The simulation experiments were performed at three burial depths: 1000, 1500, and, 2000 m. Temperatures and pressures of the three burial depths were calculated from the geothermal gradient and the pressure gradient at the sampling location. The ancient sedimentary environment of the Shanxi formation in the Southern Qinshui basin and the Taiyuan formation in the Bohaiwan basin are both believed to be an epicontinental sea setting and belong to the Permo-Carboniferous system of the north China old craton. Their underground constant temperature zones have the same temperature and depth. The temperature and depth of underground constant temperature zones, average geothermal gradient, and average pressure gradient are approximately 9°C, 20 m, 3.54°C/100 m, and 1.0 MPa/100 m (for a burial depth <2500 m), respectively. The temperatures and pressures at the three burial depths according to the average geothermal and pressure gradients are shown in Table 2. These parameters indicate that the CO2 was in a supercritical state during the simulation.
Temperatures and pressures for the simulation experiments.
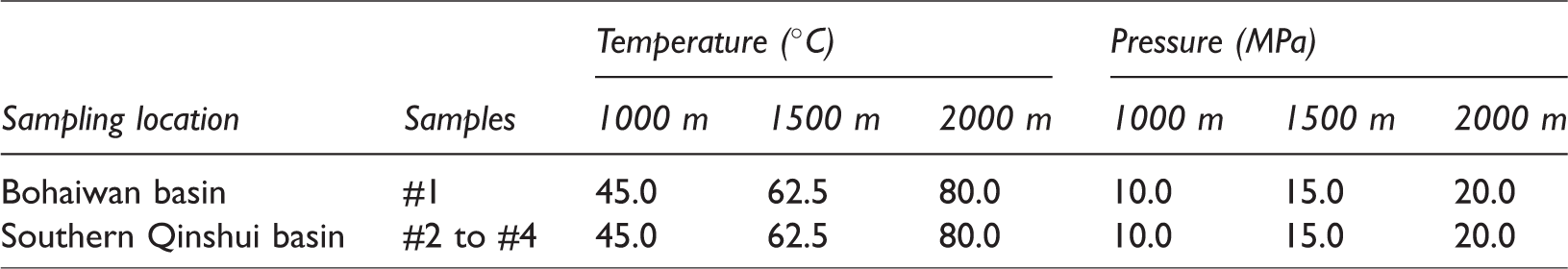
To ensure that an experiment reached reaction equilibrium, an experimental duration of 240 h (10 days) was chosen. Figure 2 shows that the temperatures and pressures in the reactor were very stable over the course of the simulations, giving a pressure of ±0.78 MPa from a predetermined pressure and a temperature of ±0.8°C from a predetermined temperature.
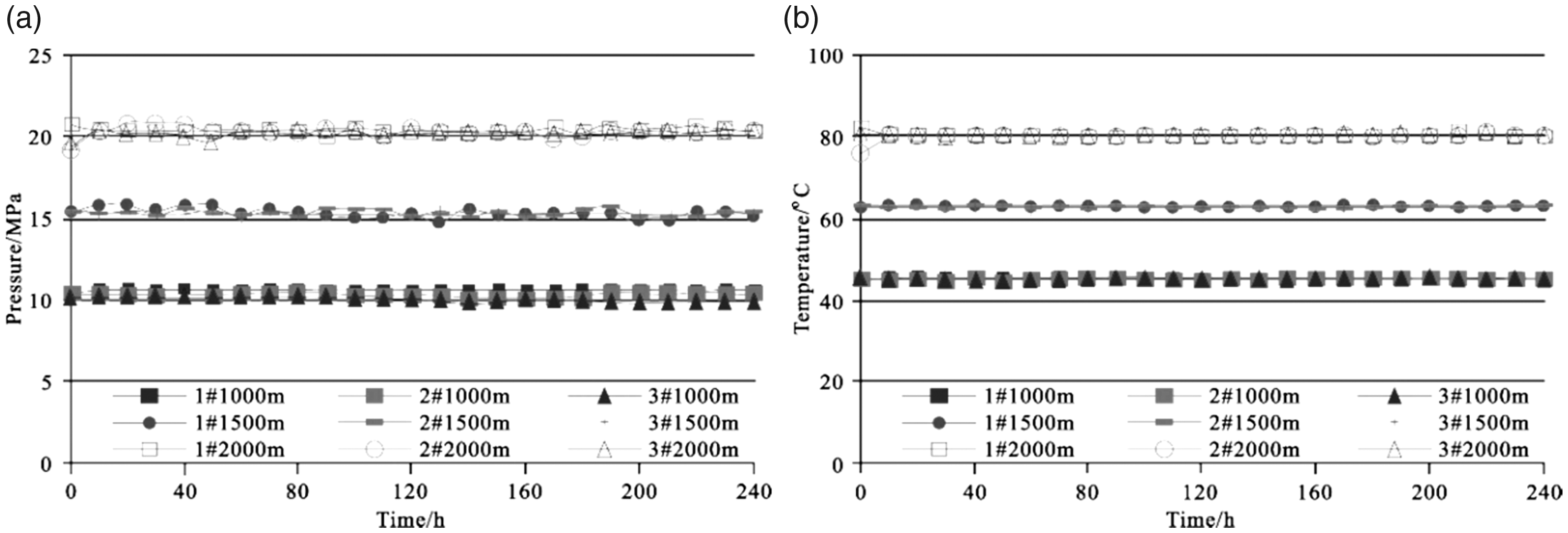
Experimental conditions of the ScCO2–H2O treatment ((a) pressure; (b) temperature).
ScCO2–H2O treatment process
The basic ScCO2–H2O treatment process is as follows.
After the prepared coal samples were placed in the sample cell (Figure 1), it was sealed and then preheated to a predetermined temperature. The sample cell was first evacuated three times, and then 300 ml deionized water was pumped into the sample cell at a flow rate of 5 ml min−1, ensuring that deionized water ran over the coal sample. Gas was pumped into the sample cell to a predetermined pressure. The temperature and pressure were monitored during the experiment to ensure system stability. After 240 h, the system pressure was reduced to atmospheric pressure, and the coal sample was removed from the sample cell and immediately vacuum dried at 50°C for 24 h.
Determination of organic group types and contents
FTIR spectroscopy
FTIR spectroscopy is a common technique for determining coal chemical structure parameters and accurately displays the type and semiquantitative ratio of coal functional groups (Chen et al., 2012, 2015; Mastalerz et al., 2012; Wang et al., 2016b).
FTIR was carried out with a VERTEX 80v FTIR spectrometer produced by Bruker, Germany. To obtain accurate infrared spectra, the coal samples used in the experiment were no larger than 200 mesh (76 µm) and were vacuum dried at 50°C for 24 h. The coal samples were prepared by the KBr disc technique. Pulverized coal was evenly mixed with KBr in a proportion of 1:180 (the sum of the weights was 0.1 g). A KBr disc (0.1 g) was used as a blank background. The scanned area was 400–4000 cm−1 with a scanning resolution of 4 cm−1. The coal samples were scanned 32 times.
Types and ratios of the major organic groups
The types and ratios of the major organic groups of coal were acquired by a semiquantitative analysis of the infrared spectra of the coal molecules. The primary features of the analysis are as follows.
According to the infrared spectra of the four coals, polynomial fitting could be used in the baseline correction. Then, the absorbance peaks could be preliminarily analyzed via the second derivative using the OMNIC software package. By comparing the absorption frequency attributes of the coal molecules with the coal molecular structure, the major organic groups of the coal could be determined. Overlapping peaks could be separated by the peak resolution function of the OMNIC software. Then, the ratios of the major organic groups were analyzed semiqualitatively by calculating the peak areas of the various organic groups. The semiquantitative analysis of the infrared spectra was based on the Lambert–Beer law (equation (1))
where A(v) is the absorbance, K(v) is the absorbance coefficient, b is the thickness of a coal sample in centimeter, c is the concentration of a coal sample in mol l−1, and T(v) is the percent transmittance.
Results and discussion
Semiquantitative ratios of the organic groups after the ScCO2–H2O treatment
There are five coal organic group categories—aromatic groups (aromatic nucleus), alkyl groups, hydroxy groups, oxygen-containing groups, and sulfur-containing groups (Figure 3). The four coals mostly contained hydroxy groups, oxygen-containing groups, and CHx (2.27–36.20, 8.53–28.28, and 17.41–51.49%, respectively), followed by Ar groups (aromatic groups) (5.52–15.84%), with sulfur-containing groups as the fewest (7.50–32.35%) (Figure 3). The semiquantitative ratios of the organic groups of the four coals are greatly different because of differences in the materials that formed the coal. After the ScCO2–H2O treatment, distinct changes in semiquantitative ratios of the organic groups were evident. As the temperature and pressure increased, the changes became significantly regular.
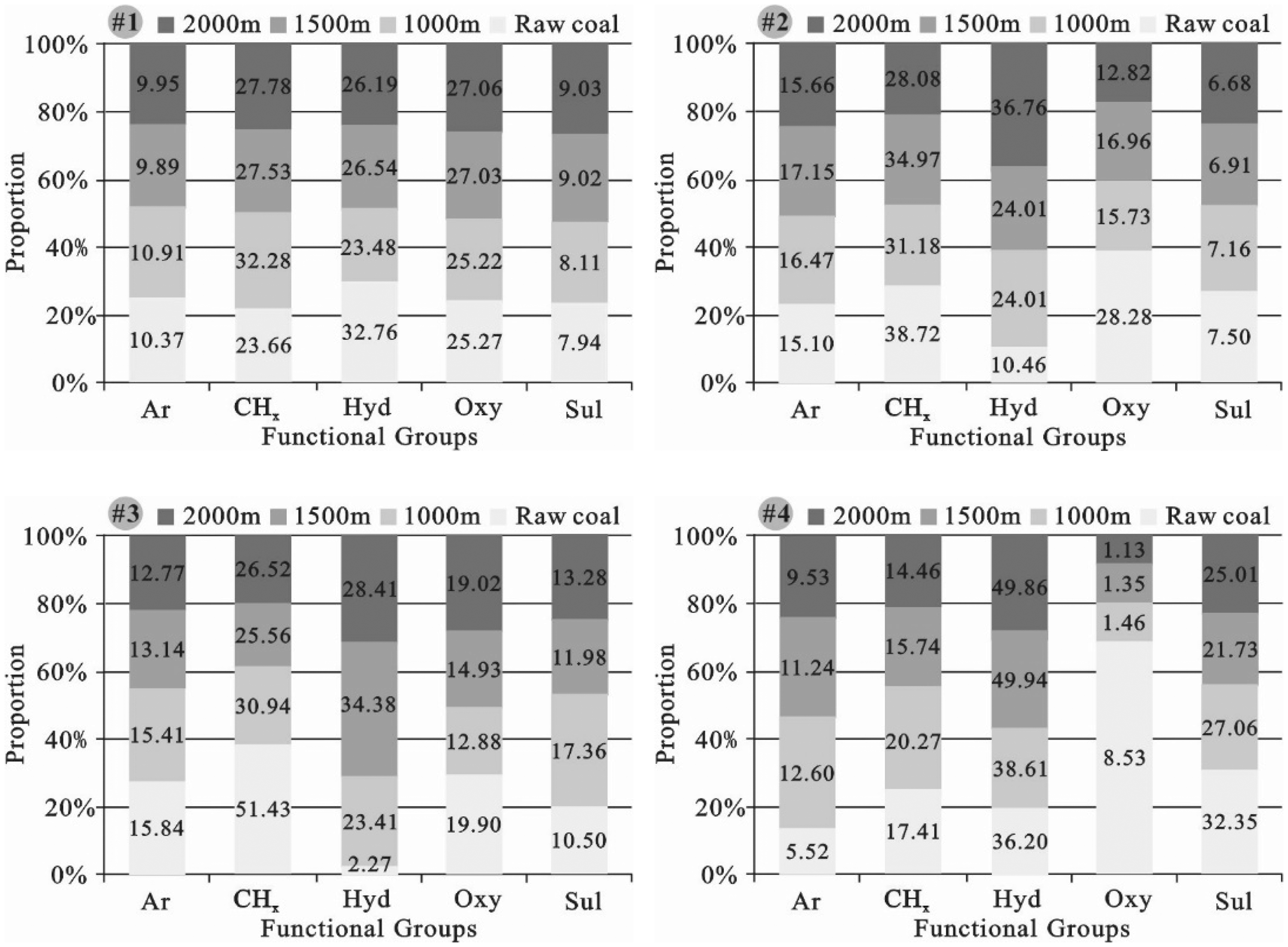
Semiquantitative ratios of five categories of coal organic groups before and after the ScCO2–H2O treatment. Note: Ar, aromatic nucleus; –CHx, alkyl groups, e.g. methyls, methylenes, and methines; Hyd, hydrogen bonding groups, e.g. free hydroxyls, self-association hydrogen bonds, and hydroxyls; Oxy, oxygen-containing groups, e.g. carboxyls, carbonyls, etc.; Sul, sulfur-containing groups, e.g. mercapto group and disulfide bond.
As the coal rank increased, the Ar semiquantitative ratios of Coals #1 to #3 (10.37, 15.10, and 15.84%, respectively) increased. After the ScCO2–H2O treatment, the Ar semiquantitative ratios in Coals #1 and #3 declined slightly compared to raw coal, and it slightly increased in Coals #2 and #4 (Figures 3 and 4).
There are two types of CHx in coal samples—aromatic CHx (Ar–CHx) and aliphatic CHx (CnH2n+1–CHx). The semiquantitative ratios of Ar–CHx (13.47–47.49%) in coal samples are much larger than CnH2n+1–CHx (1.05–10.19%) (Figure 4), which means that the changes in the semiquantitative ratios of CHx are primarily determined by Ar–CHx. The CnH2n+1–CHx semiquantitative ratios gradually decreased (10.19, 7.66, 4.00, and 1.05%, respectively) as Ro,max increased. As the temperature and pressure increased, the semiquantitative ratios of Ar–CHx in Coals #2 to #4 showed a decreasing tendency, but in Coal #1, they showed a significant increase compared to raw coal (Figure 4). Differently than for Ar–CHx, as the temperature and pressure increased, the CnH2n+1–CHx semiquantitative ratio in Coals #1 to #3 decreased, but it slightly increased in Coal #4 (Figure 4).

Semiquantitative ratios of coal organic groups before and after the ScCO2–H2O treatment. Note: Ar, aromatic nucleus; Ar–CHx, aromatic CHx; CnH2n+1–CHx, aliphatic CHx; ·OH, free hydroxyls; OH–O, self-association hydrogen bonds in ethers; –OH, hydroxyls in alcohols, phenols, and ethers; –COOH, carboxyls in fatty alcohols, aldehydes, and ketones; –CO–N, amides in aliphatic compounds; –C–O–, oxygen bonds in aliphatic ethers, phenols, and aromatic oxides; R–C=O, carbonyls in aldehydes, ketones, and esters; Ar–C=O, carbonyls in anhydride; RC=OR′, quinonyls in aromatic ketones; –S–S–, disulfide bonds; –SH, mercapto groups.
Hydrogen bonding plays a vital role in the establishment of the skeletal structure of coal macromolecules (Wang et al., 2016b). The coals contained three types of hydroxy group—OH, an OH–O bond (ether), and –OH groups. The four coals mostly contained –OH, with extremely low semiquantitative ratios of ·OH and OH–O (Figure 4). Therefore, the changes in the hydroxy group semiquantitative ratios are primarily determined by –OH. Coal #1 and #4 had the highest semiquantitative ratios of –OH of 30.76 and 30.26%, respectively, and Coal #3 had the lowest semiquantitative ratio of –OH of 0.20% (Figure 4). Compared to raw coal, the hydroxy group semiquantitative ratios in Coals #2 to #4 significantly increased, but they decreased in Coal #1 (Figure 3). As the temperature and pressure increased, the semiquantitative ratios of ·OH in Coals #2 to #4 decreased, but –OH greatly increased in Coals #2 to #4 (Figure 4). Compared to raw coal, the semiquantitative ratios of ·OH slightly increased in Coal #1, but –OH decreased (Figure 4). OH–O in Coals #2 and #3 was not detected in raw coal, and the semiquantitative ratios of the four coals all increased compared to raw coal.
The four coals mostly contained –C–O– groups at an extremely low semiquantitative ratio (generally <3.00%) of other oxygen-containing groups, e.g. –COOH groups, –CO–N groups, R–C = O groups, Ar–C = O groups, and RC = OR′ groups (Figure 4). The changes in the oxygen-containing group semiquantitative ratios are primarily determined by –C–O–. Some oxygen-containing groups were not detected, e.g. –CO–N in Coals #1 and #3; R–C = O in Coals #1, #3, and #4; and RC = OR′ in Coals #1, #2, and #4 (Figure 4). The semiquantitative ratios of different oxygen-containing groups in Coal #4 are all extremely low, generally <2.00%. As Ro,max increases, the semiquantitative ratios of –C–O– gradually decreased for the four coals (20.90, 24.09, 14.29, and 5.71%, respectively). As the temperature and pressure increased, the oxygen-containing group semiquantitative ratios in Coals #2 to #4 significantly decreased, but they increased in Coal #1 compared to raw coal (Figure 3). The –C–O– semiquantitative ratio in Coals #2 to #4 significantly decreased as the temperature and pressure increased, but –COOH slightly increased (Figure 4). The –C–O– semiquantitative ratio in Coal #1 slightly increased, but –COOH decreased (Figure 4). The semiquantitative ratios of Ar–C = O, –CO–N, R–C = O, and RC = OR′ are low in raw coal, decreased after the ScCO2–H2O treatment, and approached or reached zero at temperatures greater than 45.0°C and pressures greater than 10 MPa, except for RC = OR′ in Coal #3 (Figure 4).
The coals primarily contained two types of sulfur-containing groups: –S–S– and –SH. The –S–S– semiquantitative ratios (4.80–19.20%) are slightly higher than –SH (2.33–13.15%) (Figure 4). The –S–S– semiquantitative ratios and –SH in Coal #4 are extremely high, which are next to –OH (30.62%). After the ScCO2–H2O treatment, the sulfur-containing group semiquantitative ratios in Coals #1 and #3 slightly increased compared to raw coal, but slightly declined in Coals #2 and #4 (Figure 3). As the temperature and pressure increased, the –S–S– semiquantitative ratio in Coals #1 and #3 slightly increased (Figure 4). On the contrary, the –S–S– semiquantitative ratio in Coals #2 and #4 continuously declined (Figure 4). After the ScCO2–H2O treatment, the –SH semiquantitative ratio in Coals #1, #2, and #4 slightly decreased (Figure 4), but slightly increased in Coal #3 (Figure 4).
In addition, the fluctuating amplitudes of semiquantitative ratios of the organic groups compared to raw coal decreased at temperatures greater than 45.0°C and pressures greater than 10 MPa, indicating that the interactions between ScCO2 and organic groups were attenuated.
FTIR spectra of the organic groups after the ScCO2–H2O treatment
Absorbance peak of the aromatic structure
As Ro,max increased, the changes in the absorbance peak intensities of Ar ranged from a significant decrease for Coal #1 (1600.63 cm−1) and a slight decrease for Coal #2 (1596.77 cm−1) to a slight increase for Coal #3 (1598.70 cm−1) and a significant increase for Coal #4 (1598.70 cm−1) (Figure 5). Semianthracite (#3) is the inflection point of the changes in the absorbance peak intensities, indicating that coalification affects the reactions of ScCO2 with Ar. The changes in the absorbance peak intensities of Ar for Coals #1 and #2 are different for their semiquantitative ratios, which means that the changes in their semiquantitative ratios were caused by the changes in the total organic group content. In addition, the peak positions in the spectrum of Coal #2 moved to a higher frequency after the ScCO2–H2O treatment because of inductive effects of oxygen-containing and hydroxy groups.
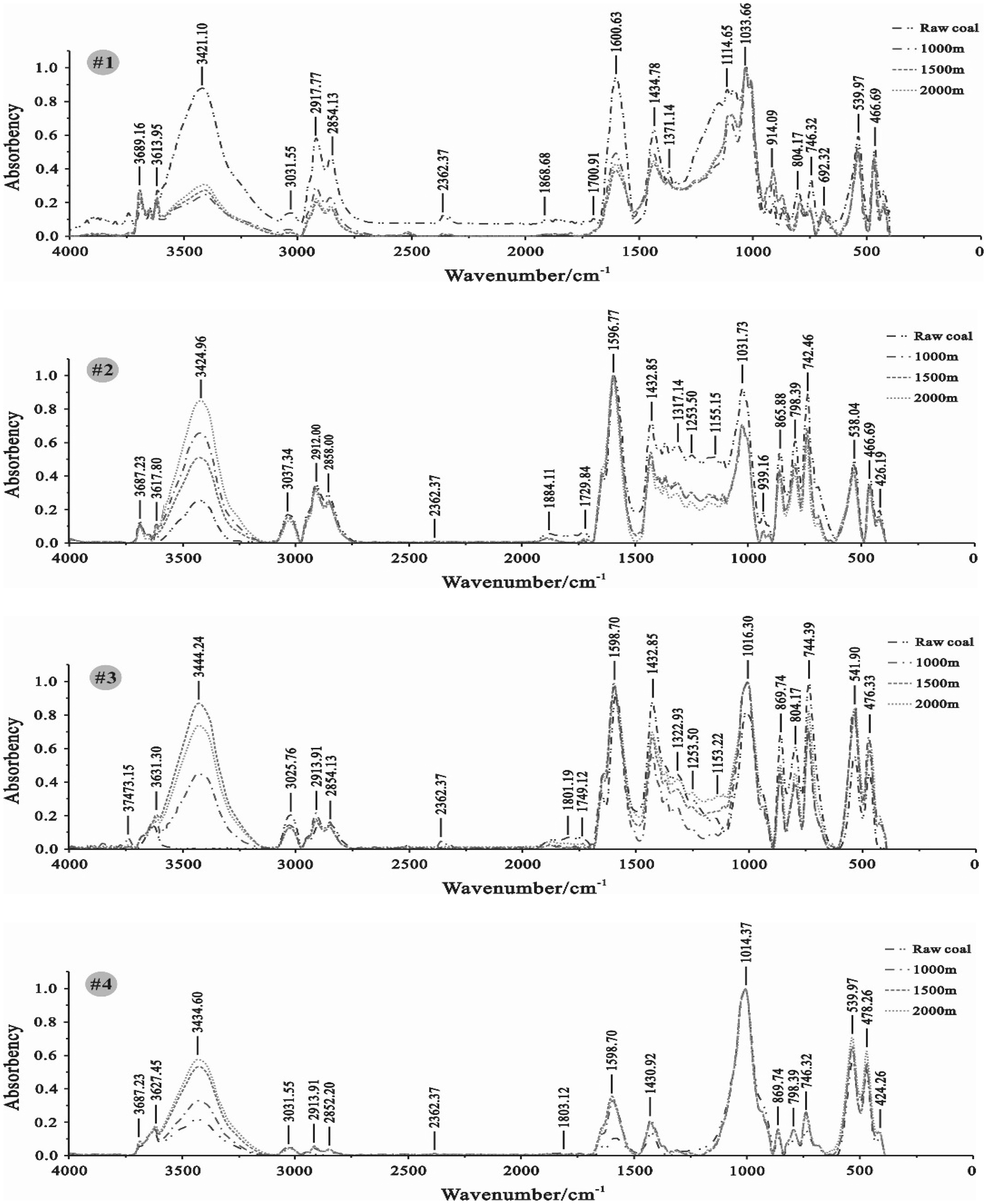
Graph of FTIR spectra of coal samples.
The Ar–CHx of the four coals mostly comprises –CH2 (747–743 cm−1) and –CH3 (1379–1373, 1460–1439 cm−1), with weak absorbance peaks of –CH (3060–3032 cm−1) (Figure 5). The absorbance peak intensity of –CH2 for Coals #2 to #4 was slightly larger than –CH3, and –CH3 had the highest absorbance peak intensity compared to –CH2 and –CH in Coal #1. Ar–CHx mostly undergoes 1,2-substitution (770–735 cm−1), following by 1,3-substitution (810–750 cm−1) and 1,4-substitution (860–800 cm−1) (Figure 5). Some single substitutions (710–690 cm−1) were also evident in Coal #1 (Figure 5).
After the ScCO2–H2O treatment, the absorbance peak intensity of Ar–CHx in Coals #1 to #3 significantly decreased, indicating that the Ar–CHx content was lower (Figure 5). The changes in the absorbance peak intensities of Ar–CHx are weak at temperatures greater than 45.0°C and pressures greater than 10 MPa. Furthermore, Coal #1 had the largest decreasing amplitude for a 1,2-substitution (746.32 cm−1) and a 1,3-substitution (804.17 cm−1) (Figure 5). The Ar–CHx content decrease indicates that the 4H (750 ± 10 cm−1), 3H (810 ± 10 cm−1), and 1H (875 ± 10 cm−1) content in Ar increased by different amounts, and bond dissociation reactions and 1,2-substitution, 1,3-substitution, and 1,4-substitution addition reactions occurred as a result of the ScCO2–H2O treatment.
The absorption band at approximately 3060–3032 cm−1 is the stretching vibration –CH absorption peak, which can be considered as measurement of the adjacent aromatic hydrogens per ring (Wang et al., 2016b). The –CH absorbance peak intensity for Coals #1 to #3 significantly decreased (Figure 5), confirming the increase in adjacent aromatic hydrogens per ring and occurrence of bond dissociation reactions. The absorption band at approximately 747–743 cm−1 is the rocking vibration –CH2 absorption peak, whose intensity is proportional to the number of continuously linked –CH2 groups in a molecular chain and can indicate the length of the methylene chains in coal (Wang et al., 2016b). The length of the methylene chains of Coals #1 to #3 decreased (Figure 5), also indicating that bond dissociation reactions were strong during ScCO2–H2O treatment and were weakly affected by temperature and pressure (Figure 5). The absorbance peak intensity for single substitutions (692.32 cm−1) and 1,4-substitutions (863.95 cm−1) in Coal #1 greatly increased at temperatures greater than 45.0°C and pressures greater than 10 MPa (Figure 5). Some substituent groups in some 1,2- and 1,3-substitutions were reduced to H. Therefore, an increase in single substitution content also confirmed the occurrence of bond dissociation reactions. The para-position on the benzene ring experiences less steric hindrance, so it is easily substituted. Therefore, the increase in 1,4-substitutions indicates that bond dissociation reactions, addition reactions, and electrophilic substitution reactions were synchronized.
The Ar–CHx absorbance peak intensity in Coal #4 significantly increased, weakly affected by the temperature and pressure (Figure 5). The number of substituent groups increased after the ScCO2–H2O treatment, but the adjacent aromatic hydrogens per ring in Coal #4 were lower. That means that Coal #4 undergoes more substitution reactions during ScCO2–H2O treatment. The symmetrical and symmetrical variable-angle absorbance peak of –CH3 (1430.92 cm−1), the rocking vibration absorbance peak of –CH2 (746.32 cm−1), and the stretching vibration absorbance peak of –CH2 (3031.55 cm−1) increased in intensity, which confirmed that substitution reactions had occurred and that branched chains had increased.
Aliphatic hydrocarbon structure absorbance peak
Two absorbance peaks of CnH2n+1–CHx are present in the coal samples, the symmetrical stretching vibration (2858–2847 cm−1) and the asymmetrical stretching vibration (2935–2918 cm−1) of –CH2 (Figure 5). CnH2n+1–CHx in coal samples is mostly –CH2, indicating that coal samples mostly contain long linear and alicyclic structures with fewer branched chains. As Ro,max increased, the changes in the absorbance peak intensities of CnH2n+1–CHx varied from a significant decrease for Coal #1 (2917.77 and 2854.13 cm−1) and a slight decrease for Coal #2 (2912.00 and 2858.00 cm−1) to a slight increase for Coal #3 (2913.91 and 2854.13 cm−1) and a significant increase for Coal #4 (2913.91 and 2852.20 cm−1) (Figure 5). Semianthracite (#3) is the inflection point of the changes in the absorbance peak intensities. The semiquantitative ratios behave in a similar fashion—the changes in the absorbance peak intensities of CnH2n+1–CHx are weak at temperatures greater than 45.0°C and pressures greater than 10 MPa. Therefore, the changes in the CnH2n+1–CHx semiquantitative ratios were caused by the changes in total organic group content, and coalification had a critical role in reactions between ScCO2 and CnH2n+1–CHx.
Hydroxy group absorbance peaks
The ·OH (3684–3625 cm−1) and OH–O (3624–3610 cm−1) absorption bands are not obvious, but superimposed, e.g. in raw Coal #3 and #4 (Figure 5). The changes in the ·OH absorbance peak intensity are consistent with the semiquantitative ratios—Coal #1 was slightly increased, and Coals #2 to #4 were decreased (Figure 5). The absorbance peak intensity of OH–O had a slight increase (Figure 5), indicating that a trace amount of OH–O was produced during the ScCO2–H2O treatment.
The –OH groups in the four coals are mostly –OH (alcohol) (3550–3200 cm−1), with a low content of –OH (phenol) (940–900 cm−1) (Figure 5). The –OH absorption bands of Coals #3 and #4 are not obvious but are superimposed over the absorption bands of silicate minerals (1033 or 1010 cm−1) (Figure 5). The changes in the –OH (alcohol) absorbance peak intensities are consistent with the semiquantitative ratios in that absorbance peak intensities showed large variations. The –OH (phenol) absorbance peak intensity of Coal #1 had a clear increase compared to raw coal (Figure 5), indicating that a small quantity of –OH (phenol) was produced during the ScCO2–H2O treatment. The –OH (phenol) absorbance peak intensity of Coals #2 to #4 slightly decreased (Figure 5), which shows that the changes in –OH content were caused by changes in alcohol –OH groups.
Oxygen-containing groups absorbance peaks
Coal #1 mostly contains –C–O– (aliphatic ether) (1125–1110 cm−1) groups, with a low –C–O– (phenol) (1300–1150 cm−1) and –C–O– (aromatic oxide) (1351–1260 cm−1) content, and the –C–O– (aliphatic ether) and –C–O– (aromatic oxide) contents are slightly greater than the –C–O– (phenol) content (Figure 5). Coal #3 mostly contains –C–O– (aromatic oxide), and the –C–O– content in Coal #4 is low (Figure 5). The –C–O– (aliphatic ether) content is slightly greater than the –C–O– (phenol) and –C–O– (aromatic oxide) contents, and the–C–O– absorption bands were superimposed over those of carbonate minerals (Figure 5). ScCO2–H2O treatment significantly decreased the absorbance peak intensity and the –C–O– content in the coal samples, but temperature and pressure had little effect (Figure 5). The changes in the –C–O– semiquantitative ratio of Coal #1 were caused by the changes in the total organic group content. The –C–O– of Coal #3 is unusual in that at temperatures greater than 45.0°C and pressures greater than 10 MPa, the –C–O– (aliphatic ether) (1153.22 cm−1) and –C–O– (phenol) (1116.58 cm−1) absorbance peak intensities increased as the –C–O– (aromatic oxide) decreased (Figure 5).
The coal samples mostly contain –COOH (fatty alcohol) (2780–2350 cm−1) and no –COOH (aldehyde and ketone) (1720–1690 cm−1) (Figure 5). Only Coals #1 and #3 had obvious –COOH (fatty alcohol) absorption peaks at approximately 2362.37 cm−1, which are not obvious in Coals #2 and #4 (Figure 5). Overall, the ScCO2–H2O treatment slightly increased the –COOH (fatty alcohol) absorbance peak intensities in Coals #2 to #4 but decreased them in Coal #1.
The Ar–C = O stretching vibration absorption band is usually split into two absorption bands at 1860–1800 and 1800–1750 cm−1 because of the coupling of stretching vibrations of the two C = O groups. ScCO2–H2O treatment significantly decreased the Ar–C = O absorbance peak intensity consistently with changes in the semiquantitative ratios (Figure 5). The Ar–C = O stretching vibration absorption band of Coals #1 to #3 moved to a higher frequency (1868.68, 1884.11, and 1874.47 cm−1, respectively), indicating that Coals #1 to #3 mostly contain open chain aliphatic and cyclic aliphatic anhydrides. In addition, the Ar–C = O absorbance peak of Coal #4 is not obvious because the Ar–C = O content of Coal #4 is low (Figure 5).
The –CO–N (1650–1640 cm−1) absorbance peaks were usually superimposed over the Ar (1635–1595 cm−1) and RC = OR′ (1690–1650 cm−1) peaks and were not obvious (Figure 5) due to the conjugative effect between Ar and carbonyl groups. No –CO–N absorbance peaks were seen in Coals #1 and #3, and they disappeared in Coals #2 and #4 after the ScCO2–H2O treatment (Figure 5). The R–C = O absorbance peaks (1736–1722 cm−1) were also not obvious. The only R–C = O absorbance peaks were found in raw Coal #2 and disappeared after the ScCO2–H2O treatment (Figure 5). The only RC = OR′ absorbance peaks were found in Coal #3, and their intensity decreased after the ScCO2–H2O treatment (Figure 5). The changes in the –CO–N, R–C = O, and RC = OR′ absorbance peak intensities after ScCO2–H2O treatment were consistent with their semiquantitative ratios.
Sulfur-containing group absorbance peaks
The changes in the –S–S– content and its absorbance peak intensity (466–478 cm−1) were closely linked to coalification but little affected by temperature and pressure (Figure 5). After ScCO2–H2O treatment, absorbance peak intensity in the high- and low-volatility bituminous coals (Coals #1 and #2) decreased, and the decrease in amplitude decreased with the increase in coalification (Coal #1 > Coal #2) (Figure 5). The absorbance peak intensity of the high-rank coals (Coal #3 and #4) was greater, and the increase in amplitude decreased with the increase in coalification (Coal #3 > Coal #4) (Figure 5). The absorbance peak intensity was highest for semianthracite. The changes in the –SH content and absorbance peak intensity (540 cm−1) are identical to –S–S– and are closely associated with coalification (Figure 5).
In addition, the changes in the –S–S– semiquantitative ratios of Coal #1 and the –SH semiquantitative ratio of Coal #4 were caused by a change in the total organic group content.
Chemical reactions between a ScCO2–H2O system and organic groups
The effects of swelling on Ar, CHx, and hydroxy groups
Solvated electrons in ScCO2 can dissolve some coal and cause the coal to swell (Dawson et al., 2015; Gathitu et al., 2009). Swelling decreased the interactions among the coal macromolecules, causing the extension and reorientation of the coal macromolecules, rupture of the hydrogen bonds, and rearrangement of coal chemical structure (Dawson et al., 2015; Gathitu et al., 2009). Swelling is accompanied by bond dissociation reactions, polyaddition reactions, and hydrogen bond interactions and is a general expression of above-mentioned chemical reactions.
The ratio between the peak areas of CHx with absorption bands of 700–900 cm−1 and the Ar (Ar–CHx/Ar) stretching vibration is a measurement of the degree of condensation of Ar (Chen et al., 2012). The degree of condensation of Ar Coals #1 to #4 progressively increased, which is in agreement with our knowledge of coal ranks (Table 3). After the ScCO2–H2O treatment, the degree of condensation of Ar Coals #2 to #4 decreased greatly, and the decrease in amplitude increased with the coal rank (Table 3). Therefore, the condensation degree of Ar in coal with a lower coal rank (#3) is higher than that with higher coal rank (#4). The degree of condensation of Ar in Coal #1 increased as the temperature and pressure increased (Table 3), indicating that the decrease in the degree of cross-linking caused by swelling is more pronounced in a high-rank coal.
Degree of condensation of coals before and after ScCO2–H2O treatment.
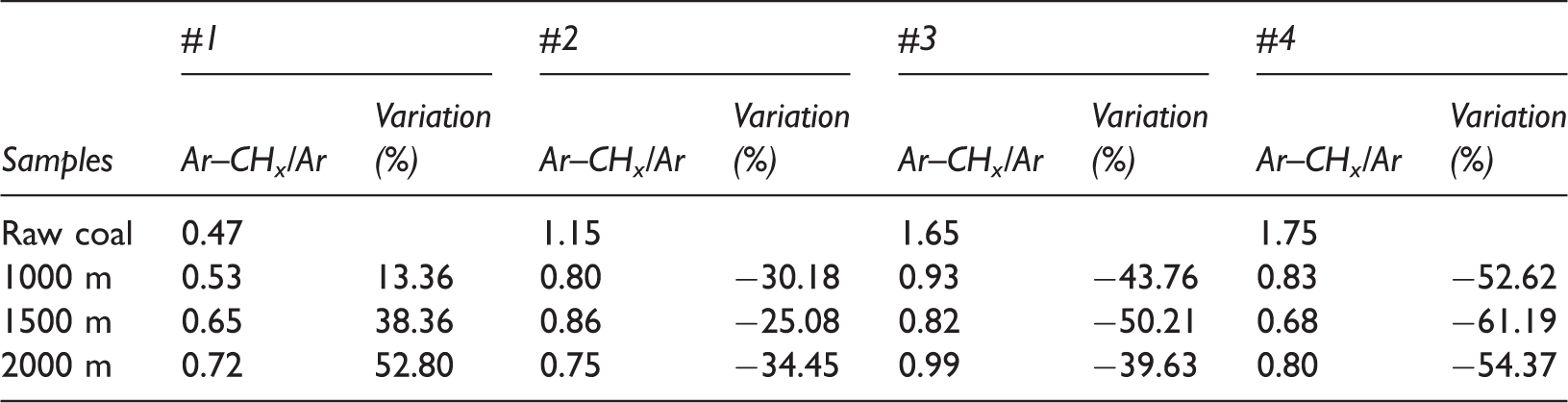
Note: “−” means decrease.
The short-range order of the macromolecular structure of a high-rank coal depends on the directional arrangement of the aromatic layers between the coal macromolecules (Mathews and Chaffee, 2012; Mathews et al., 2011). Macromolecules of a high-rank coal in the same aromatic layer are cross-linked by carbon–carbon bonds (CAr–CAr), and hydrogen bonds and Van Der Waals forces almost disappear, instead of reactions among unfixed π bonds whose structure and chemical properties are stable and have almost no swelling (Mathews and Chaffee, 2012; Mathews et al., 2011). Macromolecules of high-rank coal in different aromatic layers are cross-linked by methyne, ether, thioether, methylene ether, and methyne thioether bonds, etc. (Mathews and Chaffee, 2012; Mathews et al., 2011). Swelling caused by ScCO2 primarily breaks cross-links connecting different aromatic layers, decreasing the forces acting among the aromatic layers and the degree of cross-linking of the macromolecules. The effect becomes stronger as the coal rank increases. Cross-link breakage can produce hydrogen free radicals (·H). ·H can be substituted by CHx, causing Ar–CHx and CnH2n+1–CHx, to increase or it can be changed into –OH, causing an increase in alcoholic hydroxyl groups. Cross-link breakage among aromatic layers and the formation of –OH increase the asymmetry and polarity of the hydrogen bonds (hydroxyl groups are hydrogen bonding groups) and of CAr–CAr. The common effects of polar group conjugations, hydrogen bond interactions, and symmetric interactions of coal macromolecules increase the absorbance peak intensity of Ar.
The short-range order of the macromolecular structure of bituminous coal depends on the directional arrangement of the aromatic layers in the coal macromolecules (Castro-Marcano et al., 2012; Grzybek et al., 2002). The aromatic structure of bituminous coal is mainly polycyclic aromatic hydrocarbons with rings arranged in a straight line, and the aromatic layers are weakly cross-linked (Castro-Marcano et al., 2012; Grzybek et al., 2002). Swelling primarily breaks cross-links in the same aromatic layer. Some branched chains and organic groups with low bond energy were broken by swelling, causing the rearrangement of the coal chemical structure, increased directionality in the aromatic layers, and finally the formation of CAr–CAr cross-links (equation (2)). The formation of CAr–CAr cross-links is also known as a polyaddition reaction. Polyaddition reactions can further increase the degree of condensation and degree of cross-linking of the aromatic structure, and it becomes weaker as the coal rank increases. The breakage of strong polar cross-links and the increase in the symmetry of CAr–CAr concomitantly increase the absorbance peak intensity of Ar
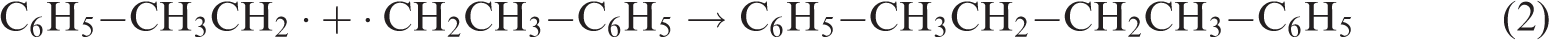
The other effects of swelling caused by ScCO2 on organic groups are effects on the CHx and hydroxy group contents and absorbance peak intensities. Cross-link breakage, e.g. of methyne, ether bonds, and methylene ether bonds, is an important reason for the decrease in the CHx, –OH, and –C–O– contents. These cross-links are primarily found in bituminous coal. Therefore, the decrease in their contents is more pronounced in Coals #1 and #2. Cross-link breakage, produces a large number of ·H, which changes the associations between the aliphatic chains and the organic groups. More –OH exist in coal macromolecules in the form of OH–O, which is an important reason for the increase in the OH–O content and absorbance peak intensity. Meanwhile, the increase in the OH–O contents and absorbance peak intensity confirmed the occurrence of swelling. Some ·H also changed to ·OH via the capture of O. ·OH can combine with Ar or CHx to form –OH. In Coal #1, some ·H combined with Ar to form –OH (phenol). However, the polyaddition reaction inhibited the reaction of ·H and CHx, causing a decrease in –OH (alcohol) and an increase in ·OH. In Coals #2 to #4, the combination of ·H with Ar is difficult because the polycyclic aromatic structure and the breakage of cross-links due to swelling decrease –OH (phenol) groups. However, bond dissociation and hydrolysis reactions increased the –OH (alcohol) content in Coals #2 to #4.
The effects of endothermic reactions on organic groups
Addition reactions.
There are three types of addition reactions between a ScCO2–H2O system and coal organic groups—chlorination, conjugated addition, and nucleophilic addition. Chlorination and conjugated addition meanly affect Ar content and absorbance peak intensity, and nucleophilic addition acts on hydroxy group and oxygen-containing groups.
Cl is a common element in coal (Xu et al., 2016). X-ray photoelectron spectroscopy shows that a large amount of Cl is present in coal, and the Cl content decreases with an increase in coalification (Table 4). The acid environment caused by the dissolution of ScCO2 in water can dissolve Cl in inorganic chlorides. These Cl can participate in addition reactions with Ar under heating (<100°C) and pressurized conditions, and such reactions are termed chlorination (Limantseva et al., 2008). ScCO2 and water are both ionizing solvents that can accelerate chlorination (Arash et al., 2016).
Results of X-ray photoelectron spectroscopy (XPS) of coal samples (Source gun type: Al K Alpha).
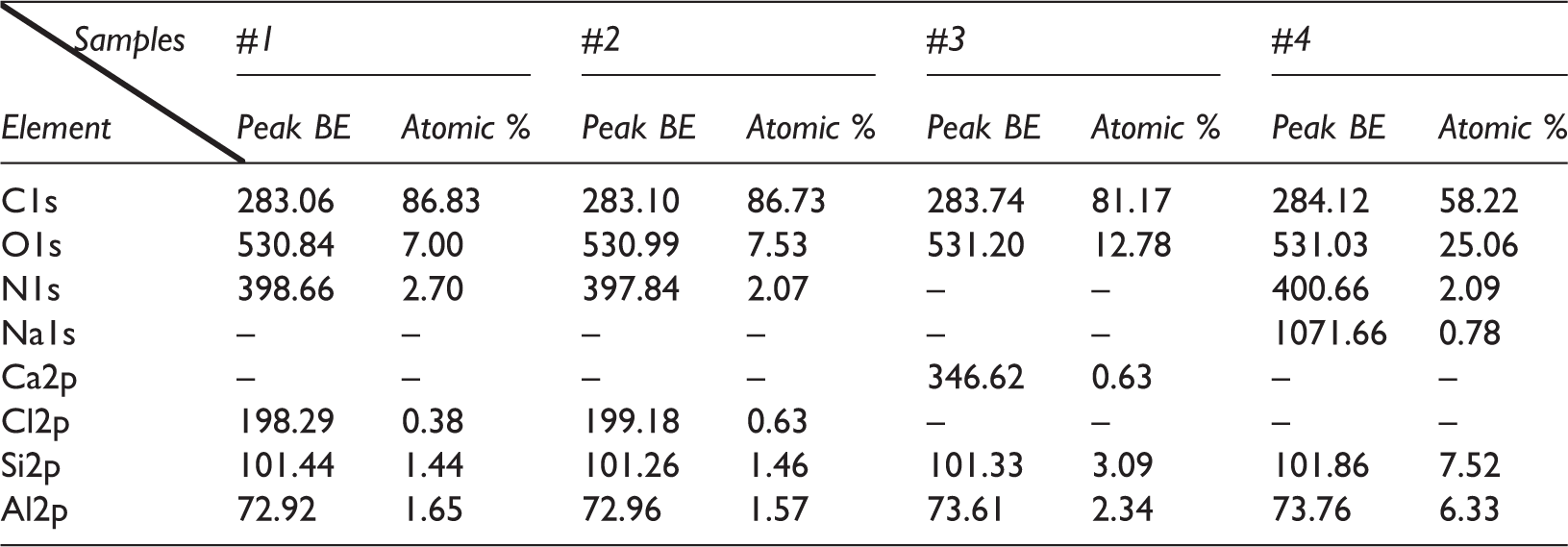
Peak BE: peak binding energy (eV).
In the early phase of chlorination, H on Ar groups can be substituted by Cl, releasing HCl (equation (3))

In the later phases, after H is significantly decreased, the addition reaction occurs (equation (4))

Chlorination destroys carbon–carbon double bonds (C = C) in Ar, causing the decrease in the Ar content and absorbance peak intensity in Coals #1 and #2.
The coal aromatic structure consists of Ar with a different degree of condensation and contains a small amount of hydroaromatic rings, aziridine, and sulfur heterocyclic rings (Castro-Marcano et al., 2012; Grzybek et al., 2002; Mathews and Chaffee, 2012; Mathews et al., 2011). No clear conclusions are possible about the coal macromolecular structure. Studies of coal macromolecular structure are focused on bituminous coal, and research on anthracite is lacking. The best knowledge at present is that the number of condensed aromatic rings in coal is associated with coalification and the carbon content (Castro-Marcano et al., 2012; Grzybek et al., 2002; Mathews and Chaffee, 2012; Mathews et al., 2011). As coalification increases, the number of condensed rings increases, and the oxygen-containing group content decreases. The number of condensed rings in bituminous coal with a carbon content between 70 and 83% is approximately 2–3, and the aromatic structure is mainly benzene, naphthalene, and phenanthrene. The number of condensed rings of bituminous coal with a carbon content between 83 and 90% increases to 3–5, and the aromatic structure is mainly phenanthrene, anthracene, and pyrene. The number of condensed rings of a high-rank coal with a carbon content greater than 90% is greatly increased because of the graphite structure. For a high-rank coal with a carbon content greater than 93%, the number of condensed rings spikes to more than 30 (Castro-Marcano et al., 2012; Grzybek et al., 2002; Mathews and Chaffee, 2012; Mathews et al., 2011).
Coal #1 is a high-volatility bituminous coal with a carbon content of 83.28% (Table 1). Its aromatic structure mostly contains benzene, naphthalene, and phenanthrene. Naphthalene and phenanthrene are polycyclic aromatic hydrocarbons with rings arranged in a straight line. Their resonance energy decreases as the number of rings increases, and they are more reactive than benzene. Coal #2 is low-volatility bituminous coal, with a carbon content of 80.32% (Table 1). Its aromatic structure mostly contains phenanthrene, anthracene, and pyrene. Pyrene has a condensed multibenzene ring structure such that its chemical stability is similar to benzene. The pyrene in Coal #2 makes chlorination more difficult. Coals #3 and #4 are semianthracite and anthracite with carbon contents of greater than 90% (Table 1). The chemical properties of Coals #3 and #4 are stable, and chlorination is difficult. Therefore, Coal #1 is easily chlorinated, greatly increasing its Ar content. Chlorination can also occur in Coal #2, but the increase in the Ar content is less. Little chlorination occurs in Coals #3 and #4, so the Ar content is stable.
ScCO2 and water are both polar solvents that can promote the conjugated addition of Ar. Solvated electrons can participate in conjugated addition with 1,3-dienes in Ar and produce free anions. The free anion captures a proton and forms a free radical. The free radical then acquires a solvated electron and becomes an anion. The anion is an alkali that can capture a proton to form a cycloolefin. Conjugated addition can also destroy C = C bonds in Ar. Its response intensity in different rank coals is similar to chlorination.
Furthermore, swelling and addition reactions together drove the changes in the absorbance peak intensities of Ar and had opposite effects on Ar. Coal #1 mostly underwent addition reactions so that the absorbance peak intensity of Ar decreased. The effects of addition reactions are slightly stronger than swelling in Coal #2, so the absorbance peak intensity of Ar slightly decreased. The effects of swelling are slightly stronger than addition reactions in Coal #3, so the absorbance peak intensity of Ar slightly increased. Coal #4 had the most swelling, so the absorbance peak intensity of Ar increased.
Water is a nucleophilic reagent and can participate in nucleophilic addition with C = O in R–C = O, Ar–C = O, and RC = OR′ groups under acidic conditions (equation (5)). Nucleophilic addition can form –OH groups and decrease the R–C = O, Ar–C = O, and RC = OR′ contents and absorbance peak intensities
2. Bond dissociation reactions.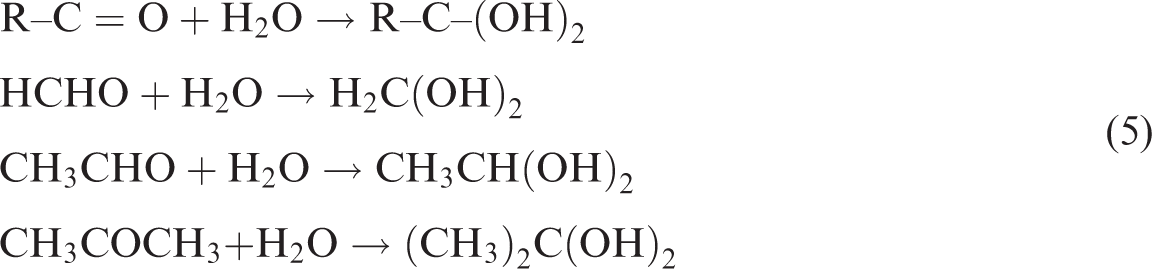
Bond dissociation reactions are important reasons for the changes of CHx. Under acidic conditions caused by ScCO2 and heating conditions, chemical bonds of CHx can have hemolysis and form free radical (·C), which is known as bond dissociation reaction of CHx (equation (6)) (Liang et al., 2014; Wang et al., 2016b). According to the bond dissociation energy, the longer the molecular chains of alkane, the smaller the bond dissociation energy of ·C is. Smaller bond dissociation energy makes ·C instability that can easily be broken. Larger macromolecules are more easily broken in the middle of the molecular chain
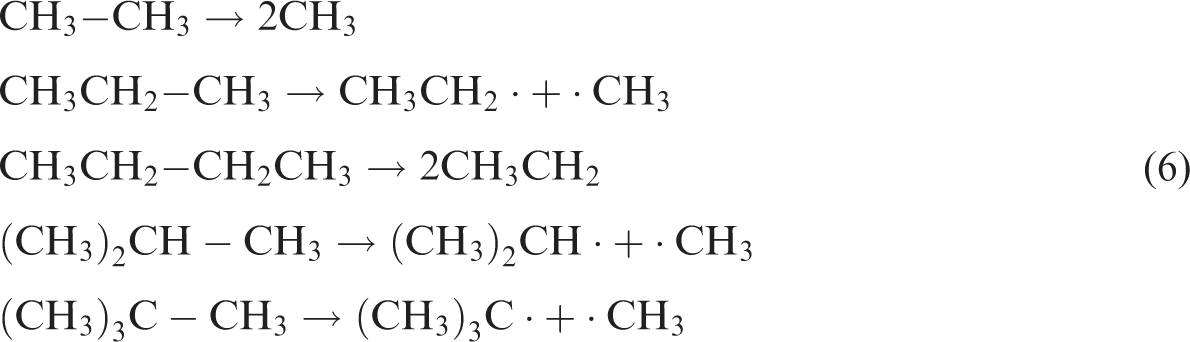
In addition, C = C of Ar also have small bond dissociation energy, particularly polycyclic aromatic hydrocarbons with rings arranged in a straight line, e.g. naphthalene and anthracene, that their aromatic structures can be easily destroyed. That is, one of the reasons for the decrease in Ar contents in Coals #1 and #2.
3. Sulfuration reaction.
Organic sulfur is the main heteroatom in coal and is in the form of a cross-linked structure and heterocyclic ring that are difficult to dissolve and remove (Lei et al., 1994; Sun et al., 2009). Research shows that with the increase in coalification, inorganic sulfur can translate into organic sulfur, making the total sulfur content increase constantly (Lei et al., 1994; Sun et al., 2009). That is, with the increase in coalification, the content of organic sulfur tends to rise, and the sulfur-containing group semiquantitative ratios are anomalously high. Meanwhile, with the increase in coalification, thioether (–S–S–) and mercaptan (–SH) change into thiophene sulfur—the main organic sulfur in high-rank coal.
Under acidic conditions caused by ScCO2 and heating conditions, sulfide minerals in coal, e.g. pyrite, can be dissolved to form H2S. H2S can have direct reactions with organic matter of coal to form organic sulfides, e.g. mercaptan. Mercaptan and thioether can change into each other such that –SH creates –S–S–, and –S–S– breaks into –SH (equation (7)). The cross-linking reaction between S2− and organic matter of coal is known as a sulfuration reaction. A sulfuration reaction can connect one or more S to the coal macromolecules to form cross-links. That is why the sulfur-containing group contents in Coals #3 and #4 increased

In addition, –S–S– and –SH in Coals #1 and #2 are primarily cross-links in coal macromolecules, which can be broken by swelling, making the decrease in sulfur-containing group contents. Moreover, –S–S– and –SH are no longer the main cross-links in Coals #3 and #4 that were weakly affected by swelling and primarily affected by sulfuration reaction.
The effects of exothermic reaction on organic groups
Substitution reaction.
There are two types of substitution reactions between a ScCO2–H2O system and coal organic groups—electrophilic substitution and hydrolysis reaction. They act on CHx and oxygen-containing groups, respectively.
H on Ar can be easily substituted by other organic groups, and the acidic conditions caused by ScCO2 promote the substitution reaction making the increase in substituent, e.g. CHx (Liang et al., 2014; Wang et al., 2016b)
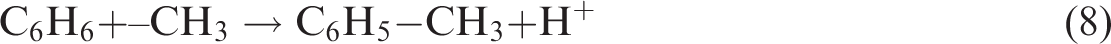
According to the effects of swelling and endothermic reactions, the substitution reaction is weak because of the symmetrical cross-linked structure of bituminous coal (Coals #1 and #2), together with swelling and bond dissociation reaction, causing the decrease in contents of Ar–CHx and CnH2n+1–CHx. The substitution reaction is strong because of the polar cross-linked structure of high-rank coal (Coal #3 and #4), causing the increase in contents of Ar–CHx and CnH2n+1–CHx.
Moreover, the changes of Ar–CHx and CnH2n+1–CHx are the results of swelling, bond dissociation reaction, and substitution reaction. Swelling and bond dissociation reaction decrease CHx contents, while substitution reaction increases CHx contents.
There were more substituent groups and branched chains of aliphatic hydrocarbons in bituminous coal (Coals #1 and #2), which are easily broken. Therefore, swelling and bond dissociation reaction are stronger than substitution reaction, causing the decrease in CHx contents. ·C produced by bond dissociation reaction is so unstable that free radical reaction will continue until the free radicals disappear. The free radical reaction can combine two ·C together that shorten the aliphatic hydrocarbons and increase the degree of condensation of the coal. At the high-rank coal stage (Coals #1 and #2), aliphatic series, aliphatic functional groups, and side chains of Ar have decreased, causing the swelling and bond dissociation reaction of CHx to become weaker. The large number of ·H produced by swelling promoted the substitution reaction, making the substitution reaction stronger than swelling and bond dissociation reaction. The dominance of substitution reaction increased CHx content. In a word, semianthracite (Coal #3) is the inflection point of interaction types for CHx. For Coal #3, CnH2n+1–CHx are more stable than in Ar. Bond dissociation reaction mainly continued to Ar–CHx, while substitution reaction primarily affected CnH2n+1–CHx, causing the decrease in Ar–CHx and increase in CnH2n+1–CHx. These have been proved by the increase in absorbance peak intensity of –C–O– (aliphatic ether) and the decrease in absorbance peak intensity of –C–O– (aromatic ether) in Coal #3.
Hydrolysis reaction is the important reason for the decrease in contents and absorbance peak intensity of oxygen-containing groups. Hydrolysis reaction is actually nucleophilic substitution reaction. Oxygen bonds in –CO–N and –C–O– are unstable and can be easily hydrolyzed under acid conditions and heating conditions (Wang et al., 2016b). The products of the hydrolysis reactions of different groups have some differences
Hydrolysis reaction of amide:
Hydrolysis reaction of –C–O–:
Under the acidity and heating conditions, the hydrolysis reaction of –CO–N and –C–O– caused the decrease in –CO–N and –C–O– content. Oxygen bonds in –CO–N and –C–O– are inactive. Therefore, the reactivity of –CO–N and –C–O– is weak, and the decreasing percentages of their contents are small.
2. Oxidizing reaction.
R–C = O, Ar–C = O, and RC = OR′ are easily oxidized and form –COOH. The reaction conditions of R–C = O, Ar–C = O, and RC = OR′ oxidization are moderate. These oxidizations maybe caused by oxidation reagents in coal or reactive solvents, e.g. Cr, H2O2, and Br. Their essences are nucleophilic additions of –OH and –COOH. These oxidizations may also be caused by oxygen during the experimental process, which is auto-oxidation
Oxidizing reaction:
Auto-oxidation reaction:
The oxidizing reaction of C = O bonds is an important reason for the increase in –COOH content.
The effects of temperatures and pressures on reactions between a ScCO2–H2O system and organic groups
Under ScCO2–H2O treatment, as the temperature and pressure increased, the changes in the organic group content can be classified into four types: descending type, rising type, lower opening parabola type, and upper opening parabola type.
Descending type
The typical organic groups of descending type include oxygen-containing groups with low contents (e.g. R–C = O, Ar–C = O, and RC = OR′), Ar, CnH2n+1–CHx, –S–S–, and –SH in Coals #1 and #2 and ·OH in Coals #2 to #4. With the increase in temperature and pressure, contents and absorbance peak intensity of descending type decrease, indicating that interactions between ScCO2 and organic groups enhanced. When temperatures are >45.0°C and pressures are >10 MPa, their decrease in amplitude significantly decreases, which means that the effects of temperature and pressure on reactions between ScCO2 and organic groups are attenuated.
Under the supercritical state, the dissolving capacity and reaction capacity of ScCO2 to organic groups slightly change with the changes in temperature and pressure. The increase in the organic group content is primarily affected by the reaction capacity and reaction type of the organic groups. Descending type gives priority to endothermic reaction. With the increase in temperature, the chemical properties are more reactive, and the reaction capacity and reaction rates of the organic groups increase. Furthermore, reaction types of the organic groups can be determined. R–C = O, Ar–C = O, and RC = OR′ mostly contained nucleophilic addition. Ar in Coals #1 and #2 primarily had chlorination or conjugated addition (including CAr–CAr cross-linking). CnH2n+1–CHx, –S–S–, and –SH in Coals #1 and #2 and ·OH in Coals #2 to #4 mostly contained bond dissociation reactions caused by swelling.
Rising type
The typical organic groups of rising type include ROR′–OH; –OH in Coals #2 to #4; and Ar, –S–S, and –SH in Coals #3 and #4. With the increase in temperature and pressure, contents and absorbance peak intensity of descending type increase, indicating that interactions between ScCO2 and organic groups enhanced. Additionally, when temperatures are >45.0°C and pressures are >10 MPa, the effects of temperature and pressure on reactions between ScCO2 and organic groups are attenuated.
Rising type also gives priority to endothermic reaction. Furthermore, ROR′–OH and –OH in Coals #2 to #4 mostly contained nucleophilic addition and bond dissociation reactions caused by swelling. Ar in Coals #3 and #4 primarily had a bond dissociation reaction, and –S–S and –SH in Coals #3 and #4 mostly contained sulfuration reaction.
Lower opening parabola type
The typical organic groups of lower opening parabola type include CnH2n+1–CHx in Coals #3 and #4, Ar–CHx in Coal #4, and –COOH in Coals #2 to #4. With the increase in temperature and pressure, the contents and absorbance peak intensity of lower opening parabola type increase first and then decrease when temperatures are >45.0°C and pressures are >10 MPa. After the ScCO2–H2O treatment, the organic group contents are higher than raw coal. With the increase in temperature and pressure, the reaction capacity of ScCO2 decreases.
The lower opening parabola type gives priority to exothermic reaction. High temperature has an inhibiting effect on the exothermic reaction. Moreover, CnH2n+1–CHx in Coals #3 and #4 and Ar–CHx in Coal #4 were primarily formed by substitution reaction of CHx, and –COOH in Coals #2 to #4 mostly contained oxidation reaction.
Upper opening parabola type
The typical organic groups of upper opening parabola type include –C–O–, Ar–CHx in Coals #1 to #3, and –OH and –COOH in Coal #1. With the increase in temperature and pressure, contents and absorbance peak intensity of upper opening parabola type decrease first, and then increase when temperatures are >45.0°C and pressures are >10 MPa. After the ScCO2–H2O treatment, the organic group contents are above raw coal. With the increase in temperature and pressure, reaction capacity of ScCO2 decrease.
Upper opening parabola type also gives priority to exothermic reaction and affected by a variety of chemical reactions. Ar–CHx in Coals #1 to #3 affected by substitution reaction and bond dissociation reaction, and –C–O–, –OH, and –COOH were affected by nucleophilic addition, swelling, bond dissociation reaction caused by swelling, and polyaddition reaction (exothermic reaction). With the increase in temperature and pressure, substitution reaction and polyaddition reaction attenuate, and bond dissociation reaction enhances, causing the organic group contents decrease first and then increase.
In conclusion, 45.0°C and 10 MPa is the inflection point of the changes in the organic group content. When temperatures are >45.0°C and pressures are >10 MPa, the reaction capacity of the organic groups increases, but the effects of temperature and pressure on reactions attenuate, or the reaction capacity decreases. The effects of temperature and pressure on reactions between ScCO2 and organic groups are controlled by reaction types of the organic groups. When temperatures are ≤45.0°C and pressures are ≤10 MPa, ScCO2 has remarkable effects on CHx and hydroxy groups and has a better effect on bituminous coal than high-rank coal. When the temperatures are >45.0°C and pressures are >10 MPa, ScCO2 has remarkable effects on oxygen-containing groups and sulfur-containing groups and has a greater effect on high-rank coal. In this sense, the burial depth of ScCO2 to bituminous coal should not be too deep, and the burial depth of ScCO2 to high-rank coal can be up to 2000 m.
The effects of coalification on reactions between a ScCO2–H2O system and organic groups
According to “Chemical reactions between a ScCO2–H2O system and organic groups”, coal coalification has great effects on ScCO2–H2O treatment. Coal coalification determines coal macromolecular structure, including characteristics of the aromatic structures, categories of main organic groups, and essences of cross-links. With the increase in coalification, coal macromolecules show obvious differences, causing the differences in reaction types and reaction results between ScCO2–H2O and different ranks of coals. For instance, the differences in characteristics of the aromatic structures cause Ar in bituminous coal to give priority to addition reaction and to substitution reaction in high-rank coal, resulting in the decrease in Ar contents in bituminous coal and the increase in absorbance peak intensity in high-rank coal. The differences in categories of main organic groups of coal, especially side chains of aliphatic series, cause side chains of aliphatic series in bituminous coal to have an intense polyaddition reaction and those in high-rank coal to have an obvious bond dissociation reaction, resulting in the increase in the degree of condensation of Ar and decrease in CHx contents in bituminous coal and the decrease in the degree of condensation of Ar and increase in CHx contents in high-rank coal. The differences in cross-links cause cross-link breakage in the same aromatic layer in bituminous coal and breakage of cross-links connecting different aromatic layers in high-rank coal, resulting in the increase in the degree of condensation of Ar and decrease in contents of oxygen-containing groups and sulfur-containing groups in bituminous coal and the decrease in the degree of condensation of Ar and increase in the contents of oxygen-containing groups and sulfur-containing groups in high-rank coal.
In conclusion, semianthracite is the inflection point of reactions between ScCO2–H2O and organic groups in coal samples of different ranks. Semianthracite corresponds to the “third saltation” of coalification. In this phase, oxygen-containing functional groups and side chains in coal almost disappear, and the number of condensed rings rapidly increases. Furthermore, coal macromolecules tend to be ordered. Semianthracite is a demarcation point from a quantitative change to a qualitative change in the coal macromolecules, making it an important transition in ScCO2–H2O treatment.
Conclusions
ScCO2–H2O treatments of four coal samples of different ranks were carried out at the three temperatures and pressures, which are 45.0°C and 10 MPa, 62.5°C and 15 MPa, and 80.0°C and 20 MPa. FTIR spectroscopy provides semiquantitative ratios and FTIR spectra of coal samples before and after the ScCO2–H2O treatment. Then, the changes in the organic groups and effects of temperature and pressure and coalification on reactions between ScCO2–H2O and organic groups were discussed in terms of organic chemistry. The following conclusions can be drawn from this study.
The changes in the organic group content are the result of a variety of reactions and are affected by temperature and pressure and coalification. Reaction types between ScCO2 and coal were controlled by coal macromolecular structure. Semianthracite is the inflection point of reaction types for coal samples of different ranks. The effects of temperature and pressure on interactions between ScCO2 and coal were controlled by the reaction types of the organic groups. With the increase in temperature and pressure, the changes in the organic group content can be classified into four types, which are descending type, rising type, lower opening parabola type, and upper opening parabola type. 45.0°C and 10 MPa is the inflection point of the changes in the organic group content. Bituminous coal including high- and low-volatility bituminous coal has low degree of condensation of the aromatic structure, and its Ar can facilitate addition reactions. The swellings primarily break cross-links between Ar in the same aromatic layer. These characteristics promote the polymerization addition of aliphatic side chains of Ar, causing the increase in the degree of condensation of the aromatic structure and the decrease in contents of CHx, oxygen-containing groups, and sulfur-containing groups in bituminous coal. High-rank coal including semianthracite and anthracite has a high degree of condensation of the aromatic structure, and its Ar gives priority to substitution reactions. The swellings primarily break cross-links connecting different aromatic layers. Bond dissociation reaction and sulfuration reaction are more significant for high-rank coal. These characteristics cause the decrease in the degree of condensation of the aromatic structure and the increase in contents of CHx and sulfur-containing groups in high-rank coal. Descending type and rising type favor addition reaction, bond dissociation reaction, and sulfuration reaction, which are endothermic reactions. The reaction rate of ScCO2 and organic groups increases as the temperature and pressure increase. Lower opening parabola type and upper opening parabola type favor substitution reaction, oxidation reaction, and addition polymerization, which are exothermic reactions. They were significantly affected by a variety of reactions and were suppressed by high temperature and pressure. Thus, when the temperatures are ≤45.0°C and pressures are ≤10 MPa, ScCO2 has remarkable effects on CHx and hydroxy groups and has a better effect on bituminous coal. When the temperatures are >45.0°C and pressures are >10 MPa, ScCO2 has remarkable effects on oxygen-containing groups and sulfur-containing groups and has a greater effect on high-rank coal. These results indicate that the burial depth of ScCO2 to bituminous coal should not be too deep, and the burial depth of ScCO2 to high-rank coal can be up to 2000 m.
Footnotes
Acknowledgements
We would like to thank a number of research students from China University of Mining and Technology for their assistance in the coal sampling and some experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Nos. 41330638, 41402135, and 41727801), the Natural Science Foundation of Jiangsu Province (No. BK20171188), the Scientific Research Foundation of Key Laboratory of Coal-based CO2 Capture and Geological Storage, Jiangsu Province (China University of Mining and Technology) (Nos. 2015A04 and 2016B03), the State Scholarship Fund of China Scholarship Council (CSC) (No. 201706425014), Geological survey project of China Geological Survey (No. 12120115008101), and the Science Foundation of Wuhan Institute of Technology (No. K201726).
