Abstract
To study the geochemical characteristics of the No. 6 Coal of the Guanzigou Mine, the contents of major and trace elements were determined by X-ray fluorescence spectrometry and inductively coupled plasma-mass spectrometry. Results showed that the contents of SiO2, Al2O3, TiO2, Li, Ga, In, and Pb in the No. 6 Coal are higher than the common contents of the average values in Chinese coal, whereas those of other elements, such as Co, Ni, Rb, Cs, and Ba, are lower. The SiO2/Al2O3 ratio (1.22) is close to the ratio in pure kaolinite (1.18) because of the lower proportion of quartz or boehmite in the coal. In the vertical section, all major and most trace elements in the samples are enriched near the parting and floor. The contents of Al2O3, Li, and Ga have reached their minimum mining grade, supporting their recovery as by-products. The high content of Pb should be paid attention to during coal mining and utilizing. The geochemical associations of the elements were assessed according to correlation and cluster analyses. It was found that Al2O3, SiO2, Li, Na2O, Bi, MgO, K2O, Rb, Cs, and Ga are associated with clay minerals. Cd, Pb, TiO2, and Cu are associated with unidentified traces of sulfide minerals or organic matter. In, Tl, MnO, Co, Std, Fe2O3, and CaO are associated with sulfides, carbonates, or organic matter. P2O5, Sr, Be, Ba, Ni, and U are associated with gorceixite or goyazite. These results not only provide fundamental information on coal seam formation, but also favor the comprehensive utilization of the coal and the assessment of environmental and health-related effects of the coal during mining and utilizing processes.
Introduction
Coal represents a major proportion of China’s energy consumption and will continue to do so for a long time. Studying the elemental content of coal can provide information on coal seam formation, enable the comprehensive utilization of the valuable elements in coal (Lin et al., 2013; Lv et al., 2015; Qin et al., 2015a; Zhao et al., 2017), and reveal the presence of toxic elements that may be harmful to the environment and human health (Fu et al., 2013; Swaine, 2000; Swaine and Goodarzi, 1995; Zhao et al., 2014).
Coal with high Al content (Al2O3 > 40% in coal ash) has been identified in some Chinese coals. This type of coal has been named as “high-Al coal” and commonly occurs in the southern region of the Yinshan Oldland, China (Qi et al., 2006). The “high-Al coal” contains many valuable elements (Sun et al., 2016). Coals from Jungar Coalfield belong to the “high-Al coal.”
The Jungar Coalfield in the Ordos Basin is an important coal enrichment and production area. Coal from the central and northern parts of the Jungar Coalfield has been the subject of extensive studies (Dai et al., 2006; Sun et al., 2012a, 2016). Dai et al. (2006) discovered a superlarge gallium ore deposit in Heigaigou Mine of this coalfield, and Sun et al. (2012a, 2013a) found a large lithium deposit in Guanbanwusu Mine of this coalfield. The southern, central, and northern mining areas all belong to the same coalfield and have the same geological background. However, due to differences in local sedimentary environments, the geochemical characteristics of the elements vary (Sun et al., 2010; Xu et al., 2011). There have been no studies on the trace elements of coal in the Guanzigou Mine of the Jungar Coalfield. It is essential to understand the geochemical characteristics of elements in the entire basin. Accordingly, the purpose of this paper is to study the origin, distribution, and mechanism of enrichment of the elements in the coal in the Guanzigou Mine.
Geological setting
The geological setting of the Jungar Coalfield has been described previously (Dai et al., 2006; Liu et al., 1991; Sun et al., 2012a, 2013a; Zhang and Wang, 2009). Geologically, the Guanzigou Mine is located in the southern Jungar Coalfield (Figure 1), adjacent to the Haerwusu Mine, and covers 17.1 km2 and contains 0.46 Gt of coal reserves.
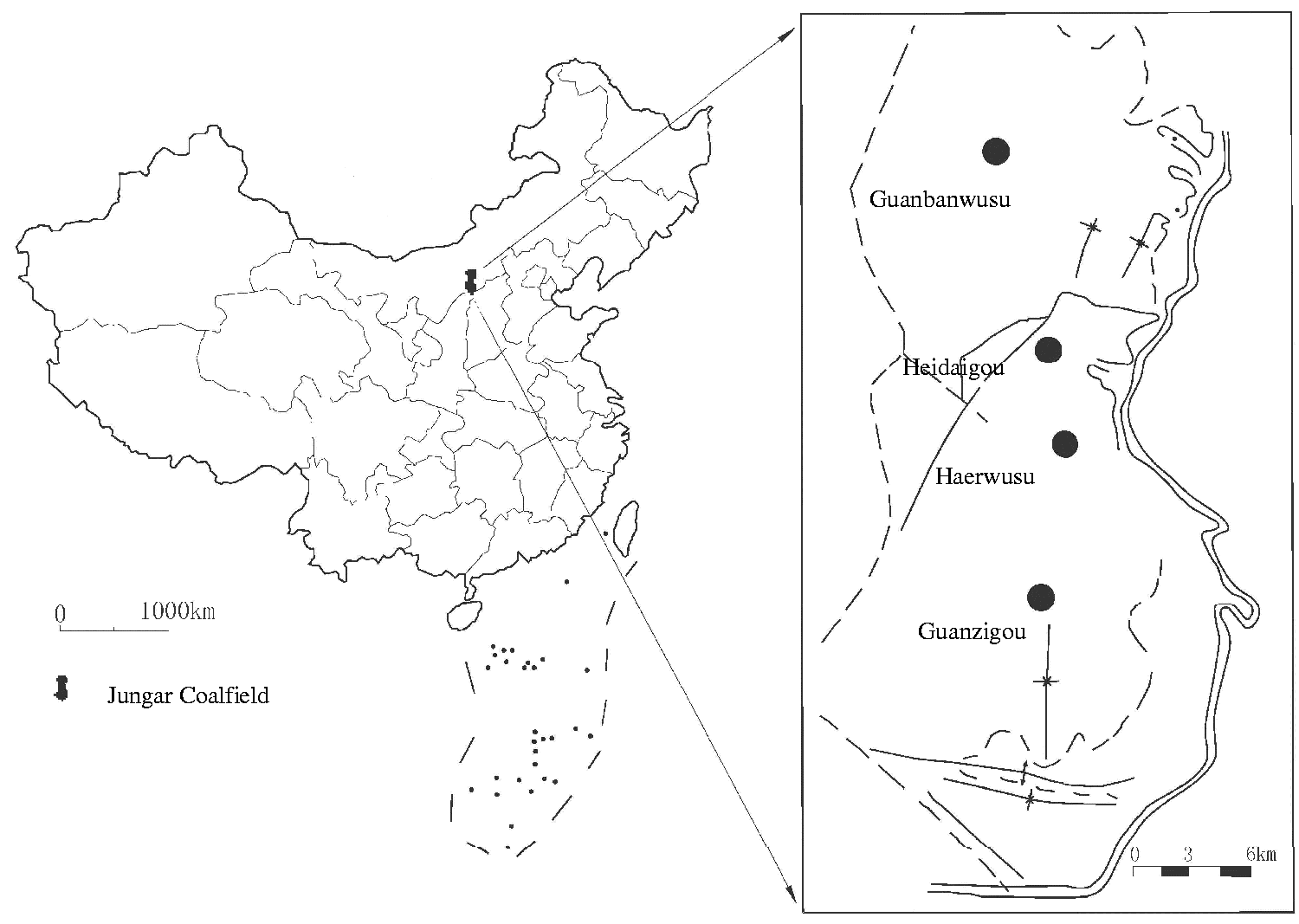
Location map of the Guanzigou Mine.
The coal-bearing strata in the Guanzigou Mine include the Benxi (Late Carboniferous), Taiyuan (Late Carboniferous and early Permian), and Shanxi (Middle Permian) Formations (Sun et al., 2017). The Benxi Formation is 15–35 m thick and overlies the Middle Ordovician limestone with an unconformable contact. The lower portion is mainly composed of mudstones and aluminous mudstones, and the lowest portion is a layer of grayish bauxite. The upper portion of the Benxi Formation is mainly composed of limestone thicker than 6 m, which is enriched in animal fossils and features partial intercalation of mudstone and coal seams (Sun, 2016).
The thickness of the Taiyuan Formation is 35–70 m. The lowest portion is sandstone, which marks the division between the Benxi and Taiyuan Formations. The lower portion is mainly sandy mudstone, with thin layers of mudstone, siltstone, and coal seam. The middle portion is dominated by mudstone and siltstone and also contains thin layers of limestone and No. 7, 8, and 9 Coals. Among these coals, the No. 9 Coal is mineable. The upper portion is dominated by the No. 6 Coal, which is located at the top of the Taiyuan Formation and is the main mineable coal seam in the Guanzigou Mine. The floor of the No. 6 Coal is mudstone (Sun, 2016; Sun et al., 2016).
The Shanxi Formation comprises quartzose sandstone, arkose, thin layers of sandy mudstone, and mudstone (Li et al., 2017). The No. 1 to No. 5 Coals are of 45–90 m in thickness. The lowest portion of the Shanxi Formation is a thick, stable layer of sandstone, which marks the division between the Taiyuan and Shanxi Formations. The strata overlying the Shanxi formation are noncoal bearing (Sun, 2016).
Sampling and analytical methods
Sampling
Twelve bench samples were collected from the workface in the Guanzigou Mine following Chinese Standard Method GB482-2008. These samples included 10 coal bench samples and two partings. No samples were collected from the roof or the middle and upper coal benches due to the conditions of the mine. Each coal bench sample was 30 cm ×10 cm × 10 cm in volume. All samples were immediately stored in plastic bags after collection to minimize contamination and oxidation.
Proximate analysis
Proximate analysis was conducted using Chinese Standard Method GB/T212-2001. Total sulfur was determined following Chinese Standard Method GB/T214-2007. The total sulfur content was very low and therefore was not further tested.
Vitrinite reflectance and minerals
A Leica DM2500P reflected light microscope (oil 32/0.65, 548 nm, 3 µm × 3 µm, EMI9592S-11) was used to determine the random vitrinite reflectance and mineral composition in the Key Laboratory of Resource Exploration Research of Hebei Province.
Analysis of major elements and trace elements
The samples were ground to 200 meshes and heated in an 815°C muffle furnace for nearly 4 h. X-ray fluorescence spectrometry (ARL ADVANT’XP+) was then performed to determine the oxides of major elements in the coal and parting ash (Zhao et al., 2012).
To determine the content of trace elements, 200 mg coal sample (40 µm), 2 ml of HF (50%), 2 ml of H2O2 (30%), and 5 ml of HNO3 (65%) were added to a polytetrafluoroethylene vessel. After microwave digestion for 1 h at 210°C, the liquid was transferred to a 125 ml FEP bottle, which was filled with deionized water to obtain a total mass of 100 g. The samples were then analyzed by inductively coupled plasma mass spectrometry in the Key Laboratory of Resource Exploration Research of Hebei Province (Sun et al., 2009; Zhao and Sun, 2008).
Results and discussion
Coal chemistry and vitrinite reflectance
The proximate analysis, total sulfur, and random vitrinite reflectance data of the parting, floor, and 10 coal bench samples from the Guanzigou Mine are summarized in Table 1. The weighted average contents of moisture, volatile matter, and total sulfur are 3.85, 26.8, and 0.33%, respectively. Compared with average content of high-aluminum coal (Sun, 2016), these values are lower. However, the weighted average ash yield and vitrinite reflectance are higher than those of high-aluminum coal in the Jungar Coalfield by 28.17 and 0.62%. The higher vitrinite reflectance and lower volatilize matter are consistent with the results of Sun et al. (2010).
Concentrations of major element oxides as determined by proximate analysis, total sulfur, and vitrinite reflectance of coal benches, parting, and floor from the lower part of the Guanzigou Mine (%).
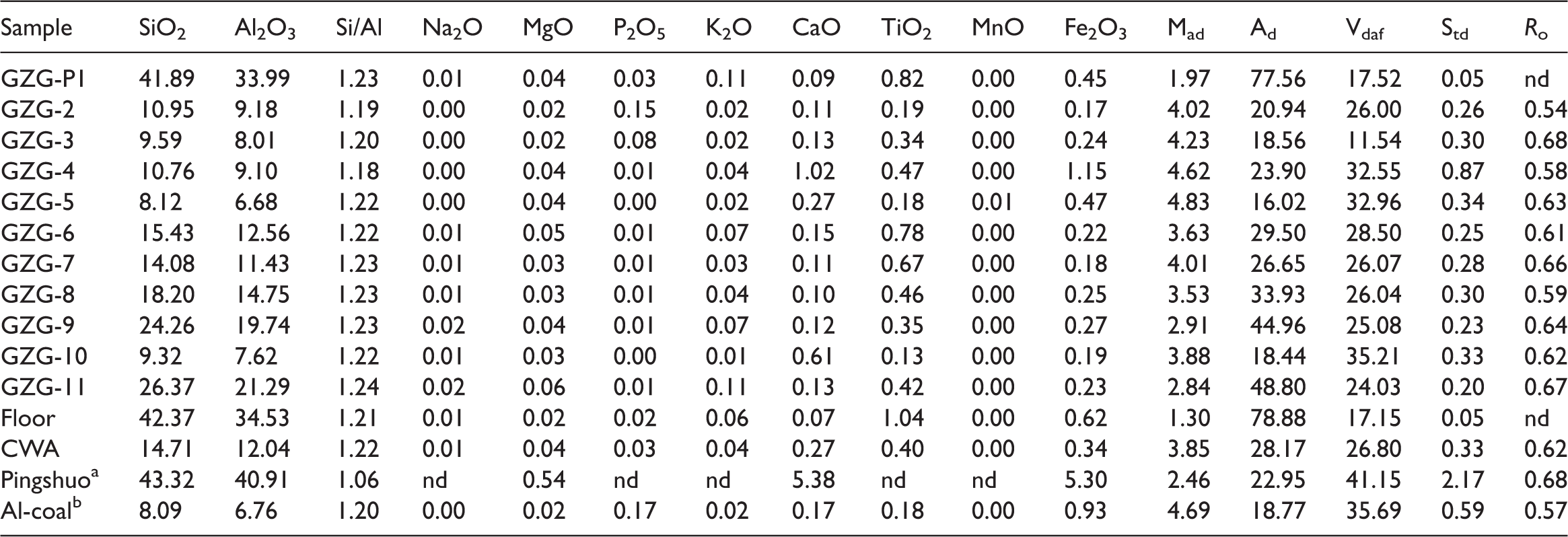
a: average content in the No. 9 Coal of the Pingshuo Mining District (Sun et al., 2016); Ad: ash; b: average content in high-Al coal (Sun et al., 2016); CWA: coal weighted average; Mad: moisture; nd: no data; Ro: vitrinite reflectance; Std: total sulfur; Vdaf: volatile matter.
Mineral contents
The category of mineral is unitary in the bottom of the No. 6 Coal and is dominated by clay (mainly kaolinite), followed by calcite, pyrite, and accessory minerals, as determined by optical microscopy. The clay minerals often occur as fine particles (Figure 2(a)) indicating a terrigenous origin, and a small proportion occur as cell fillings (Figure 2(b)), indicating formation by authigenic processes (Ward, 1989). The predominant mode of calcite occurrence is as cell fillings (Figure 2(c)) or fracture fillings, indicating an epigenetic origin (Kortenski, 1992; Sun et al., 2012a). Pyrite occurrence is dominated by fracture fillings (Figure 2(d)), indicating an epigenetic origin (Chou, 1997; Liu et al., 1999). Notably, no boehmite is observed, in contrast to other mines in the Jungar Coalfield. The lack of boehmite may reflect the collection of samples in the lower part of the coal, which accepted substances from the Yinshan Oldland (Liu et al., 1991), whereas boehmite is derived from the weathering crust of the Benxi Formation (Dai et al., 2006).
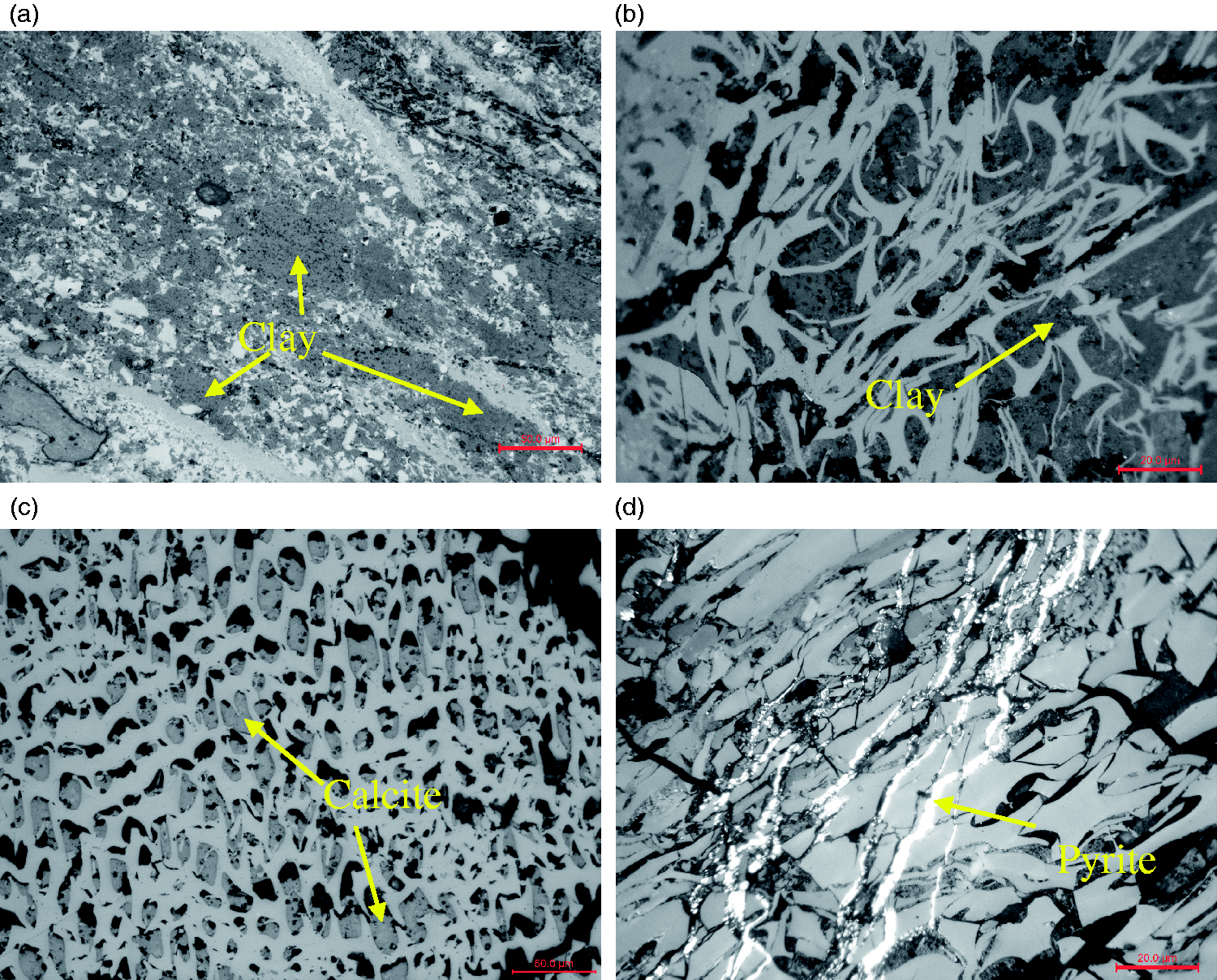
Minerals as viewed under a microscope with reflected white light. (a) and (c) reflected light, 200×; (b) and (d) reflected light from oil, 500×.
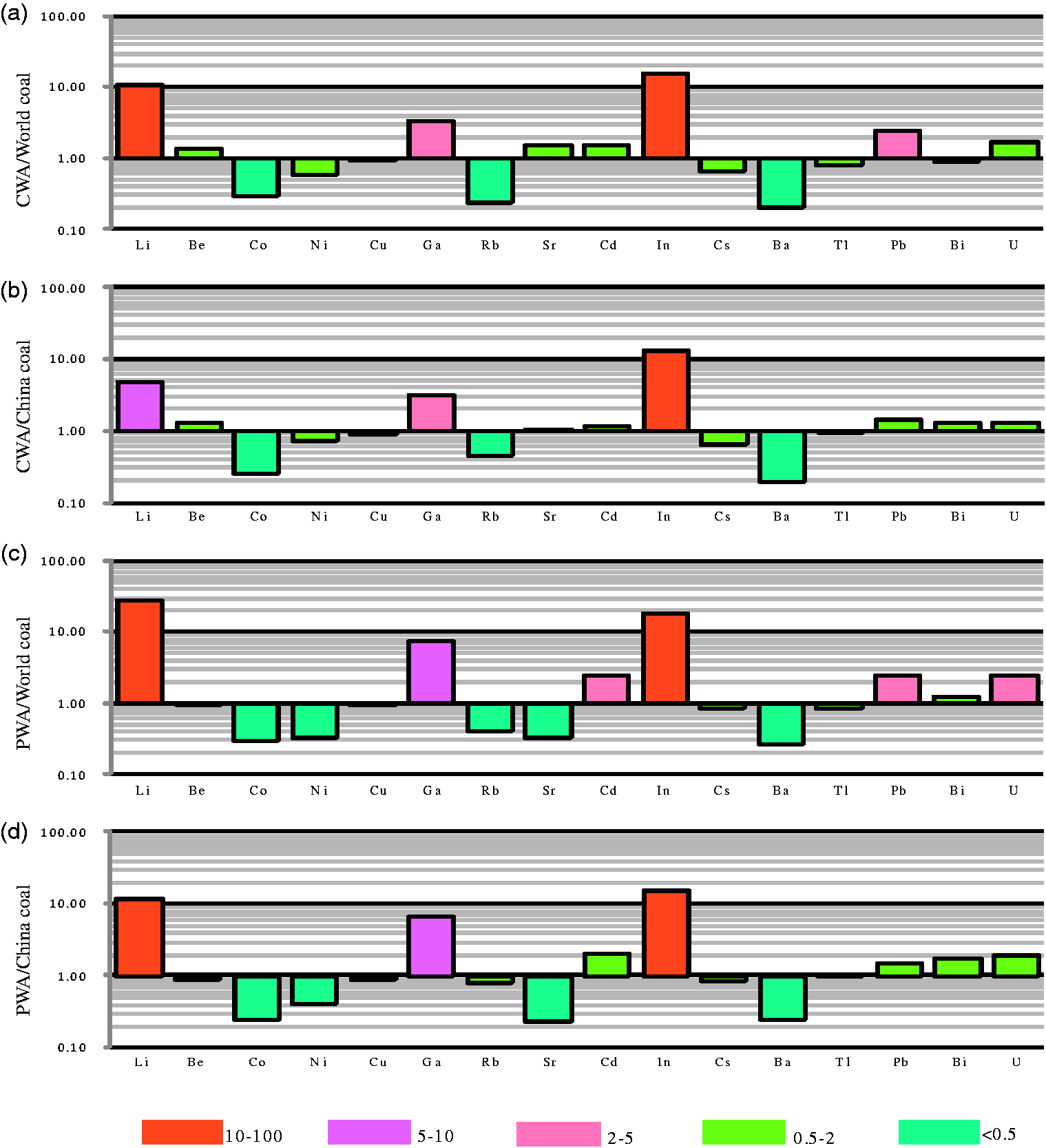
Concentration coefficients (CC) of trace elements in the Guanzigou Mine. (a) CC of trace elements in the Guanzigou coal compared with the world hard coal, (b) CC of trace elements in the Guanzigou coal compared with Chinese coal, (c) CC of trace elements in the Guanzigou parting and floor compared with the world average for hard coal, and (d) CC of trace elements in the Guanzigou parting and floor compared with Chinese coal. CWA: coal weighted average; PWA: parting and floor weighted average.
Abundance characteristics of elements
Abundance characteristics of major element oxides
The abundances of major oxides in the samples and comparisons with the “high-Al coal” are listed in Table 1. The major element oxides Al2O3 (12.04%) and SiO2 (14.71%) are dominant in coals of the Guanzigou Mine, and the abundances of these oxides are much higher than those in “high-Al coal” (Table 1). The contents of SiO2, Al2O3, MgO, CaO, and Fe2O3 are much lower, and Mad and Ad are higher (Table 1) compared with the coal in the Pingshuo Mining District of the Ningwu Coalfield. By contrast, Na2O (0.01%), MgO (0.04%), K2O (0.04%), CaO (0.27%), Fe2O3 (0.34%), and P2O5 (0.03%) are significantly depleted, and TiO2 (0.4%) is similar to that of the average values in Chinese coal. The SiO2/Al2O3 ratio in coal of the Guanzigou Mine (1.22, Table 1) is higher than that in “high-Al coal” (1.20). In addition, the SiO2/Al2O3 ratio is similar to that of pure kaolinite (1.18) because the minerals are dominated by clay (mainly kaolinite) in the Guanzigou Mine.
Abundance characteristics of trace elements
The concentrations of trace elements in the 10 bench samples, parting, and floor of the Guanzigou Mine and comparisons with other mines and the average values of the “high-Al coal” are presented in Table 2. The characteristics of the 16 trace elements in the No. 6 Coal can be described as follows.
Trace elements in coal benches, parting, and floor from the bottom of the Guanzigou Mine (elements in µg/g).
a: average content in the No. 9 Coal of the Pingshuo Mining District, from Wang et al. (2015); b: from Ketris and Yudovich (2009); CWA: coal weighted average; PWA: parting and floor weighted average.
(I) Compared with the average contents of trace elements in the world hard coals (Ketris and Yudovich, 2009), Li (CC = 10.68), Be, Ga (CC = 3.42), Sr, Cd, In (CC = 15.25), Pb (CC = 2.42), and U are enriched in the bench samples, whereas the abundances of Co (CC = 0.30), Ni (CC = 0.60), Rb (CC = 0.24), and Ba (CC = 0.21) are lower. The contents of Cu, Cs, Tl, and Bi are similar to the world averages (Figure 3(a)).
(II) Compared with the trace elements in Chinese coal, Be, Sr, Cs, Pb, Bi, and U are slightly more abundant, and Li (CC = 4.70), Ga (CC = 3.13), and In (CC = 12.99) are much more abundant. However, Ni, Cu, Cd, and Tl are slightly lower, and Co (CC = 0.25), Rb (CC = 0.46), and Ba (CC = 0.20) are much lower (Figure 3(b)).
(III) Notably, In and Tl are more abundant than those in the Jungar Coalfield coals (Dai et al., 2006), whereas Sr is lower.
(IV) Li, Ga, Cd, In, Pb, Bi, and U are enriched in the partings from the Guanzigou Mine. Compared with the average contents of trace elements in coal from the Pingshuo Mining District of the Ningwu Coalfield, Li, Be, Ni, Cd, Cs, Tl, Bi, and U are higher, whereas Co, Cu, Ga, and Ba are lower (Table 2). Compared with trace elements in coal from China (Figure 3(c)) and the world averages (Ketris and Yudovich, 2009; Figure 3(d)), Be, Cu, Cs, and Tl are lower, whereas the contents of the remaining trace elements are similar to the values for Chinese coal and the world averages.
(V) According to Sun et al. (2014), minimum mining grade of Li content in coal is 120 µg/g. The Li content (149.55 µg/g) in coal from the Guanzigou Mine has already reached the minimum mining grade. The enrichment of Li in the partings (376.92 µg/g) is even greater than that in the coal.
Vertical variations
The distribution of elemental content in the vertical section is influenced by many factors. The vertical variations of elements in coal provide important information about the coal-forming environment (Finkelman, 1993) and also indicate its various origins and relationships with peat-swamp environments or postdepositional sedimentary environments (Eskenazy and Stefanova, 2007). In this study, five groups of major element oxides and trace elements (named Group 1–5) can be recognized based on the variations in concentration through the seam section.
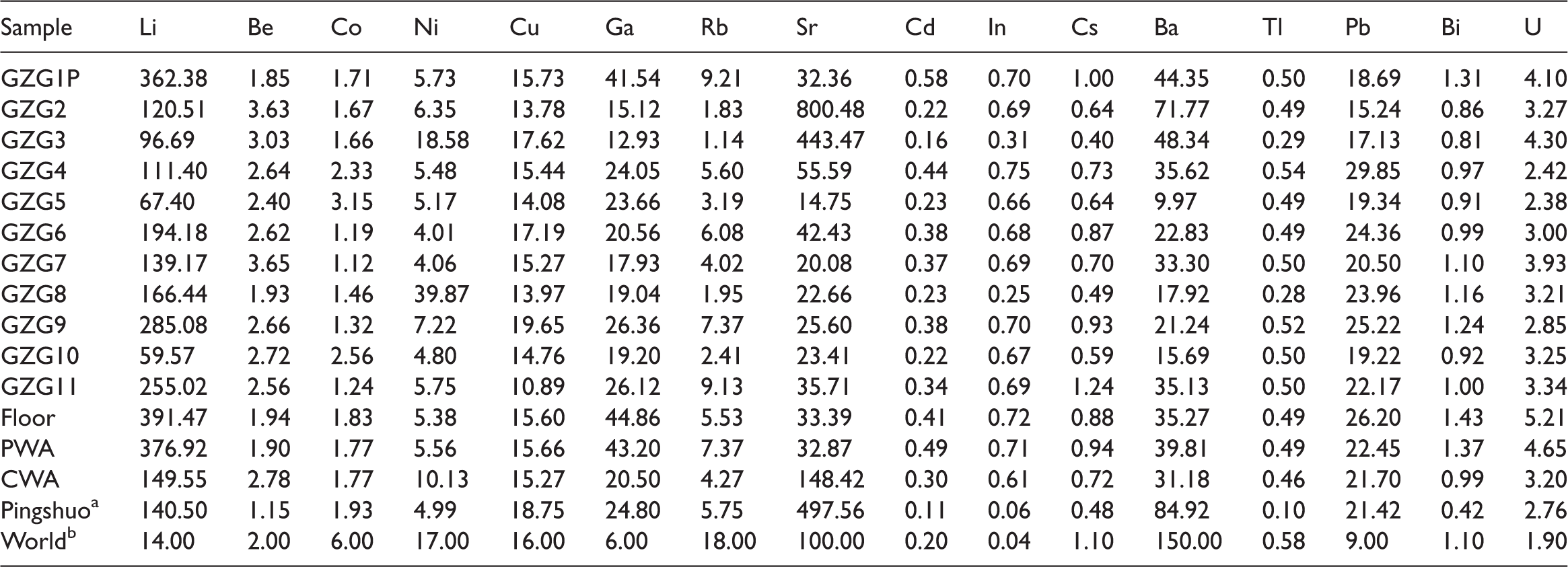
Group 1 includes SiO2, Al2O3, TiO2, Li, Ga, and Bi. The variations of these element oxides and trace elements, particularly SiO2, Al2O3, and Li, are similar to the ash yield variation through the section (Figure 4). This group is enriched in the parting and floor but is lower in the coal benches and gradually decreases from the bottom upward. The similarity of the vertical variations of these elements may indicate that they are from similar minerals, such as clay.
Variations of ash yield, SiO2, Al2O3, TiO2, Li, Ga, and Bi through the seam section.
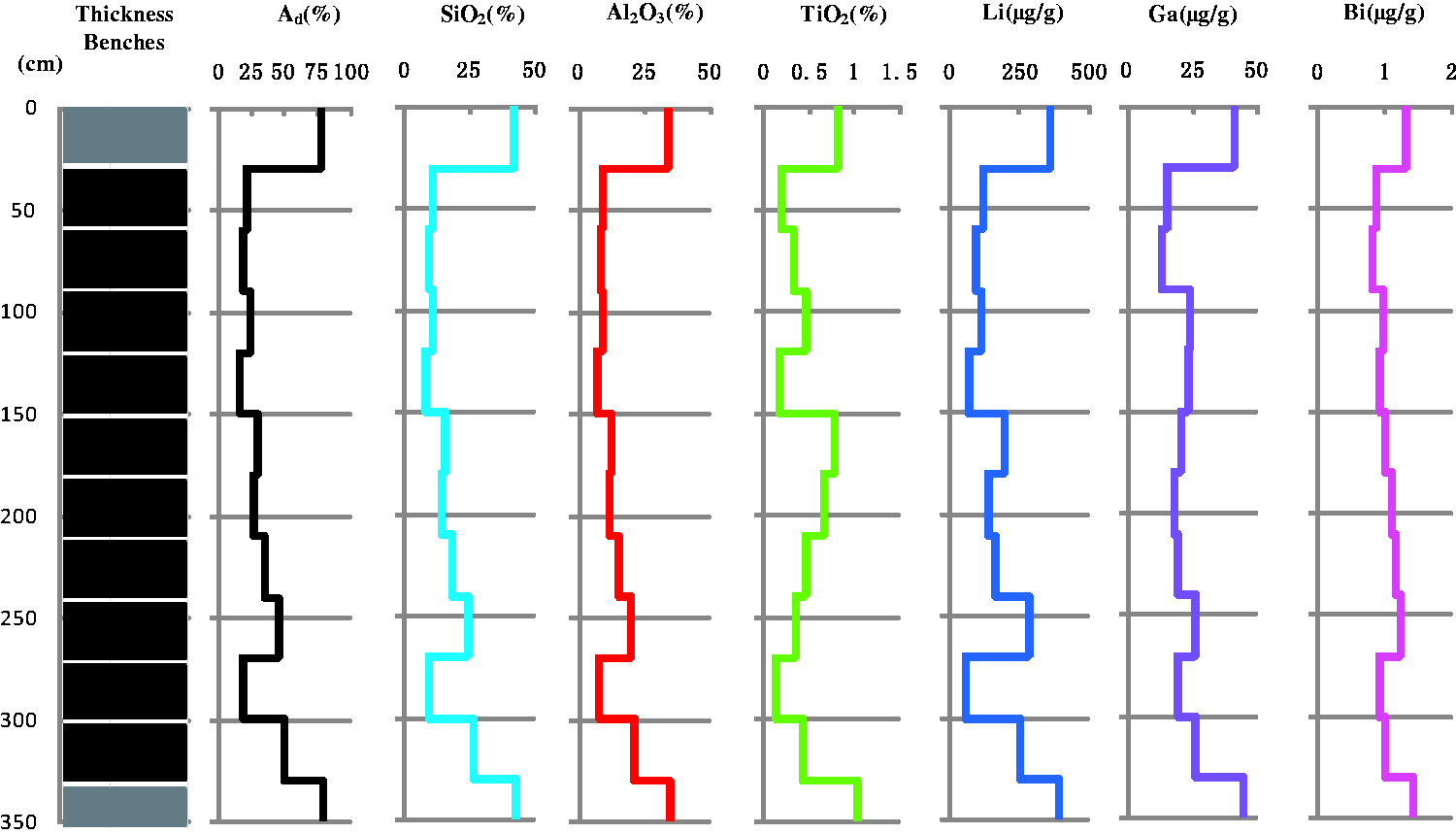
Group 2 includes Na2O, MgO, K2O, and Rb, which exhibit a saw-like distribution (Figure 5). These element oxides and trace elements are enriched in the parting but are not high in the floor. The contents decrease from the bottom upward more gradually than in Group 1.
Variations of Na2O, MgO, K2O, and Rb through the seam section.
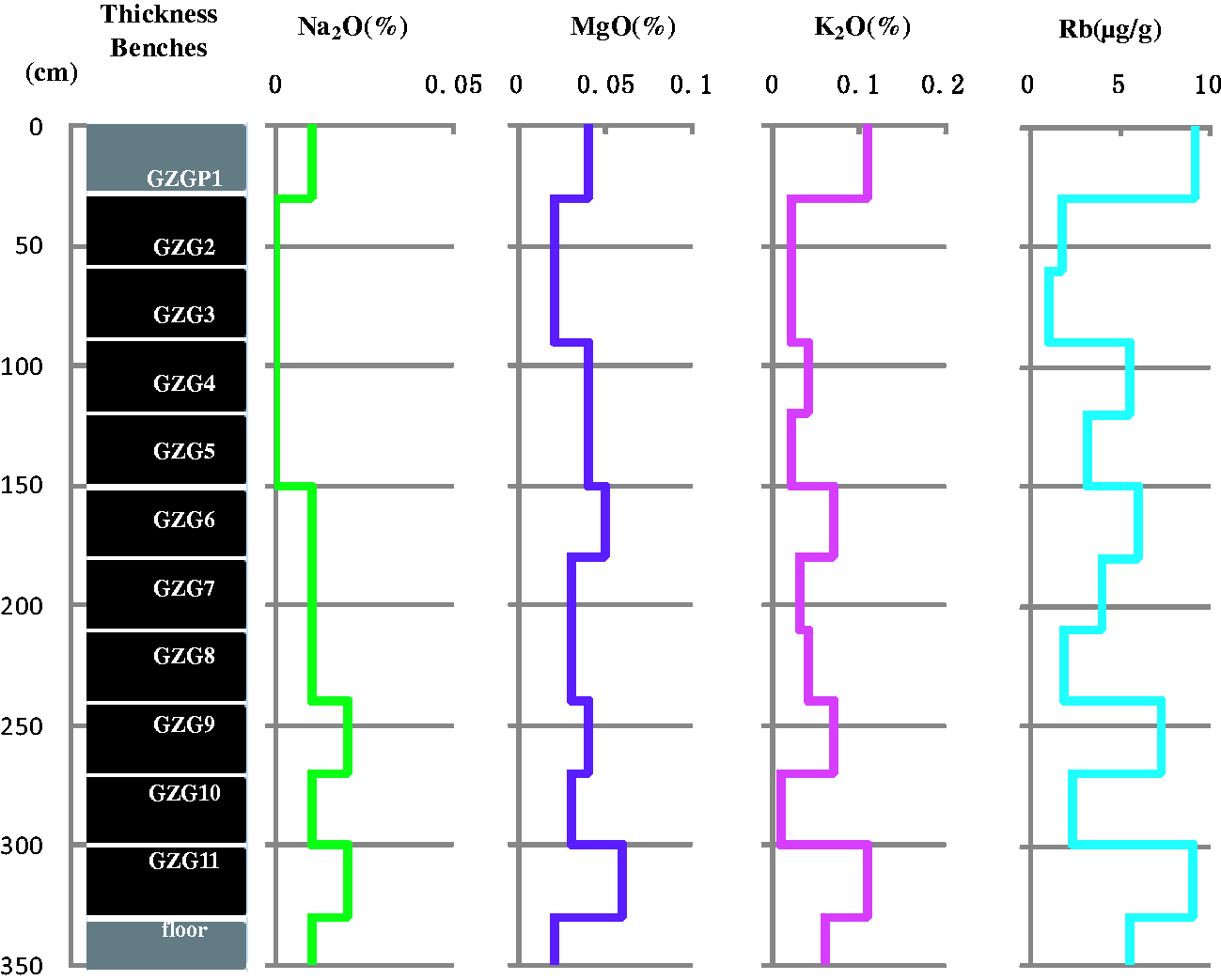
Group 3 includes CaO, Fe2O3, Std, and Co (Figure 6). These element oxides and trace elements are enriched in GZG4 and GZG10 and are lower in the other coal benches, parting, and floor. In addition, their content varies very little among the different parts, except in coal benches GZG4 and GZG10. The similarity of their vertical variations may indicate that these elements have a common terrestrial origin, such as pyrite.
Variations of CaO, Fe2O3, Std, and Co through the seam section.
Group 4 includes P2O5, Be, Sr, and Ba, which exhibit a pattern of variation similar to that of the Sr/Ba ratio through the section (Figure 7). These element oxides and trace elements are enriched in the upper part of the coal benches, GZG2 and GZG3, but are lower in the other coal benches, parting, and floor, with the exception of Be. The Sr/Ba ratio is higher than 1 in GZG2 and GZG3 but less than 1 in the other coal benches, suggesting that these elements were deposited in a marine environment.
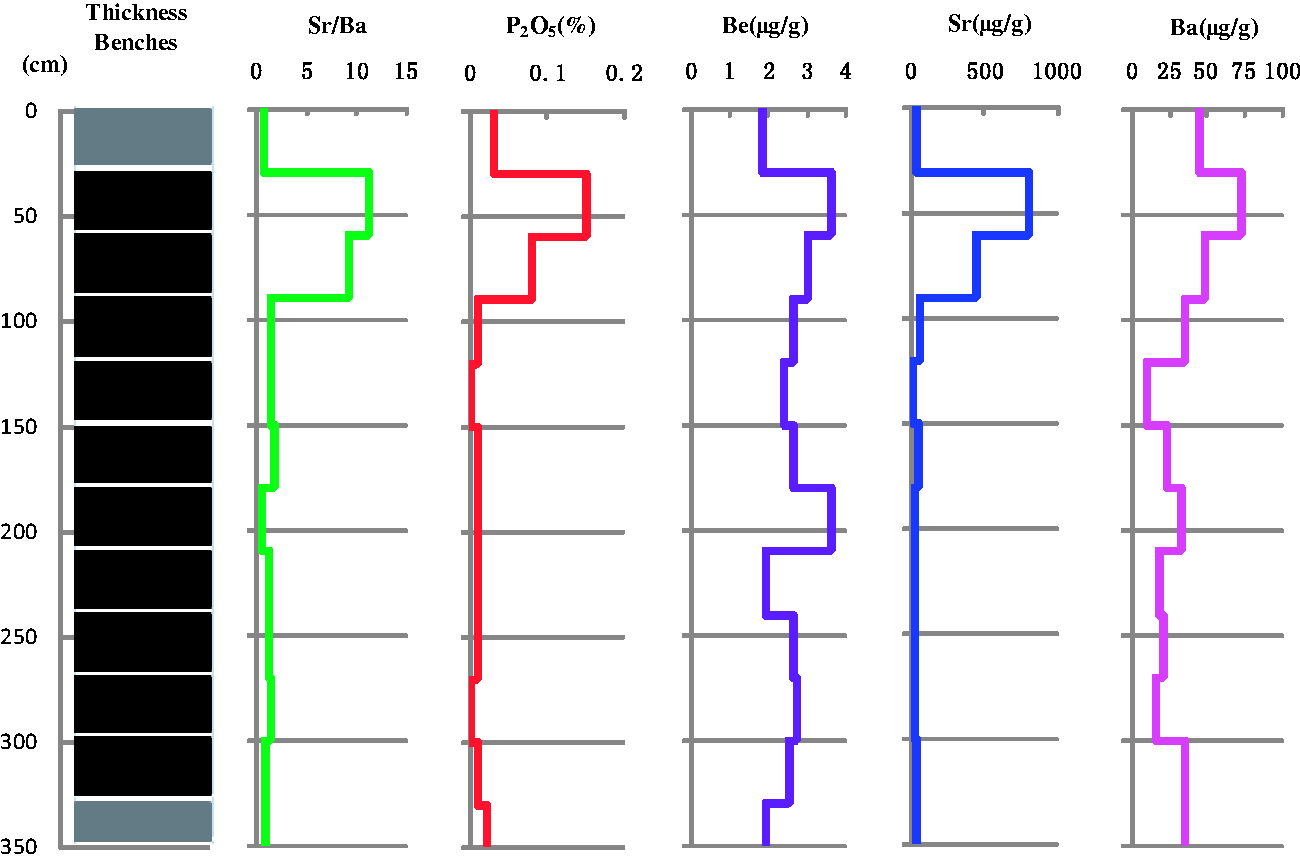
Variations of Sr/Ba, P2O5, Be, Sr, and Ba through the seam section.
Group 5 includes Cu, Cd, In, Cs, Tl, Pb, and U (Figure 8) and features a homogenous vertical distribution; only a few coal benches differ from the others.
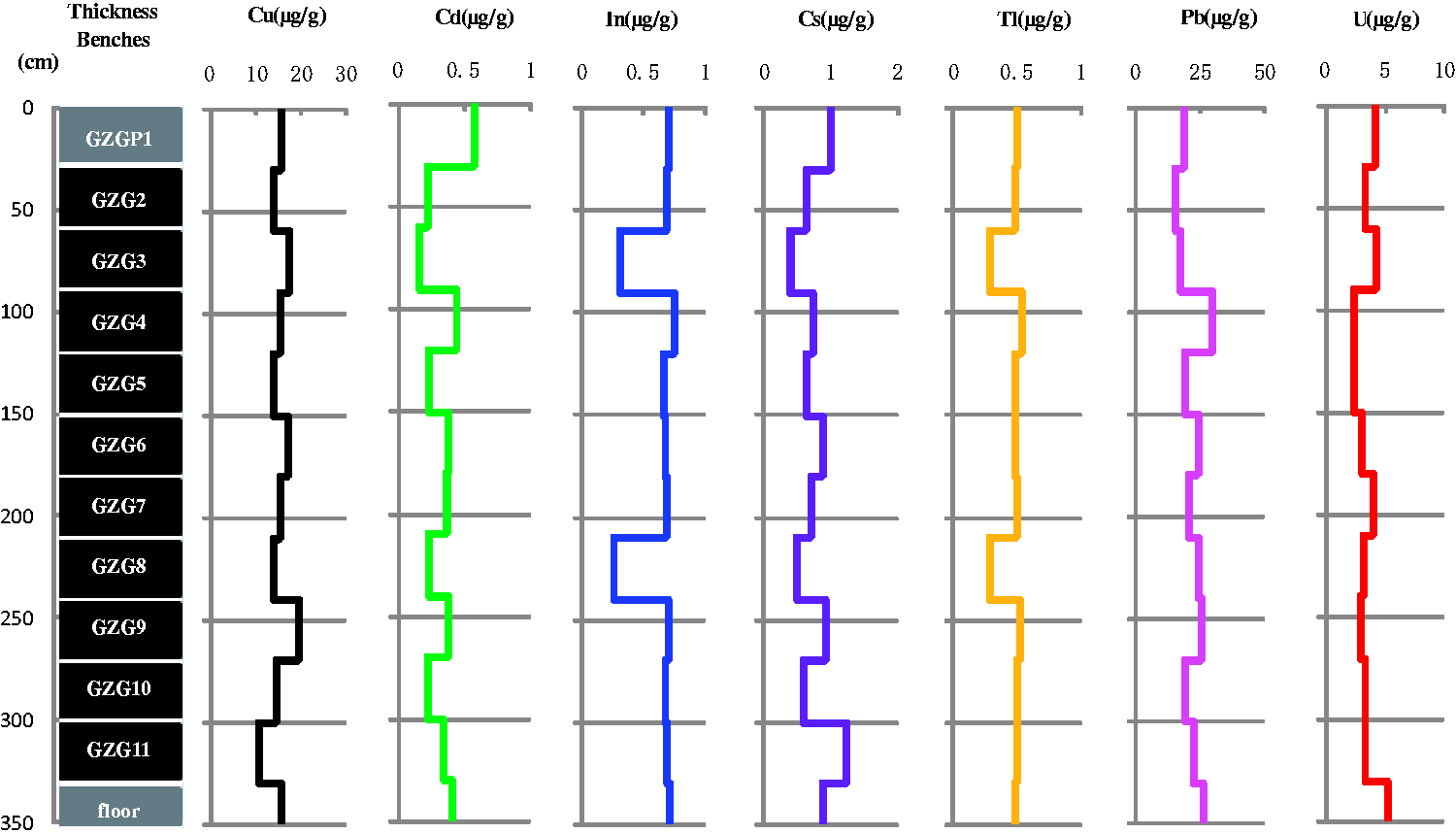
Variations of Cu, Cd, In, Cs, Tl, Pb, and U through the seam section.
Affinities of the elements
Correlation analysis can provide preliminary information about the affinities of elements (Kortenski and Sotirov, 2002) and is widely used to discuss the occurrence and genesis of trace elements in coal (Hu et al., 2006; Kortenski and Sotirov, 2002; Spears and Zheng, 1999; Xu et al., 2011). The correlations between the ash yield and elements or combinations of selected elements are presented in Table 3. Five groups of elements (named Group I–V below) can be differentiated according to their correlation coefficients with the ash yield (Table 3).
The correlation coefficients between ash yield and the content of each major element or selected combination of elements.

The elements of Group I include Bi, Cs, Rb, Na2O, K2O, Li, Al2O3, and SiO2 and they are strongly correlated with the ash yield (rash = 0.7–1.0; Table 3). Group A exhibits a dominant inorganic affinity. The major mineral is dominated by clay in coals from the Guanzigou Mine, and Bi, Cs, Rb, Na2O, K2O, and Li all have high correlation coefficients with aluminosilicate (rAl–Si > 0.7). Therefore, these elements may be associated with clay minerals.
The elements of Group II have high correlations with the ash yield, but the correlations are weaker than those of Group A. This group includes Ga and MgO. The correlations of these elements with the ash yield reflect an inorganic affinity. The correlation coefficients with aluminosilicate range from 0.5 to 0.69, indicating an association with clay minerals.
The elements of Group III have correlation coefficients with the ash yield ranging from 0.35 to 0.49. This group includes Pb and Cd. Pb has a strong affinity for sulfur (Table 3), while Cd has a lower but still relatively high sulfur affinity (Table 3). These elements may be associated with sulfide or sulfur.
Group IV includes MnO, Sr, CaO, Std, P2O5, Be, Fe2O3, Ba, Cu, U, In, Ni, and Tl. The correlation coefficients with the ash yield range from −0.34 to 0.34, indicating either organic or inorganic affinities. However, Fe2O3 has strong affinities for sulfur and carbonate (Table 3), suggesting an association of Fe with pyrite and siderite.
Group V contains only one element, Co, and the correlation coefficient with ash yield is −0.65, indicating a dominant organic affinity.
Geochemical associations
Cluster analysis can divide elements into different categories based on similarity (Chun, 2007; Wang et al., 2011). Elements are clustered into four groups (Group A–D) based on cluster analysis using centroid clustering. The spacing was determined by the Pearson correlation, and the standard conversion value had a maximum magnitude of 1.
Group A includes Al2O3, SiO2, Li, Na2O, Bi, MgO, K2O, Rb, Cs, Ga, and the ash yield (Figure 9). The elements in this group strongly correlate with ash yield. The correlation coefficients range from 0.61 to 0.99 (Table 3). The correlation coefficients of Al2O3–Ad (0.99), SiO2–Ad (0.99), Li–Ad (0.96), Na2O–Ad (0.89), and Al2O3–SiO2 (0.99) (Table 3) indicate that they may correlate with kaolinite (Sun et al., 2012a). Li, Rb, and Cs are all alkali metals and are similar in properties. Therefore, the elements in this group may be associated with clay minerals (Bai, 2003).
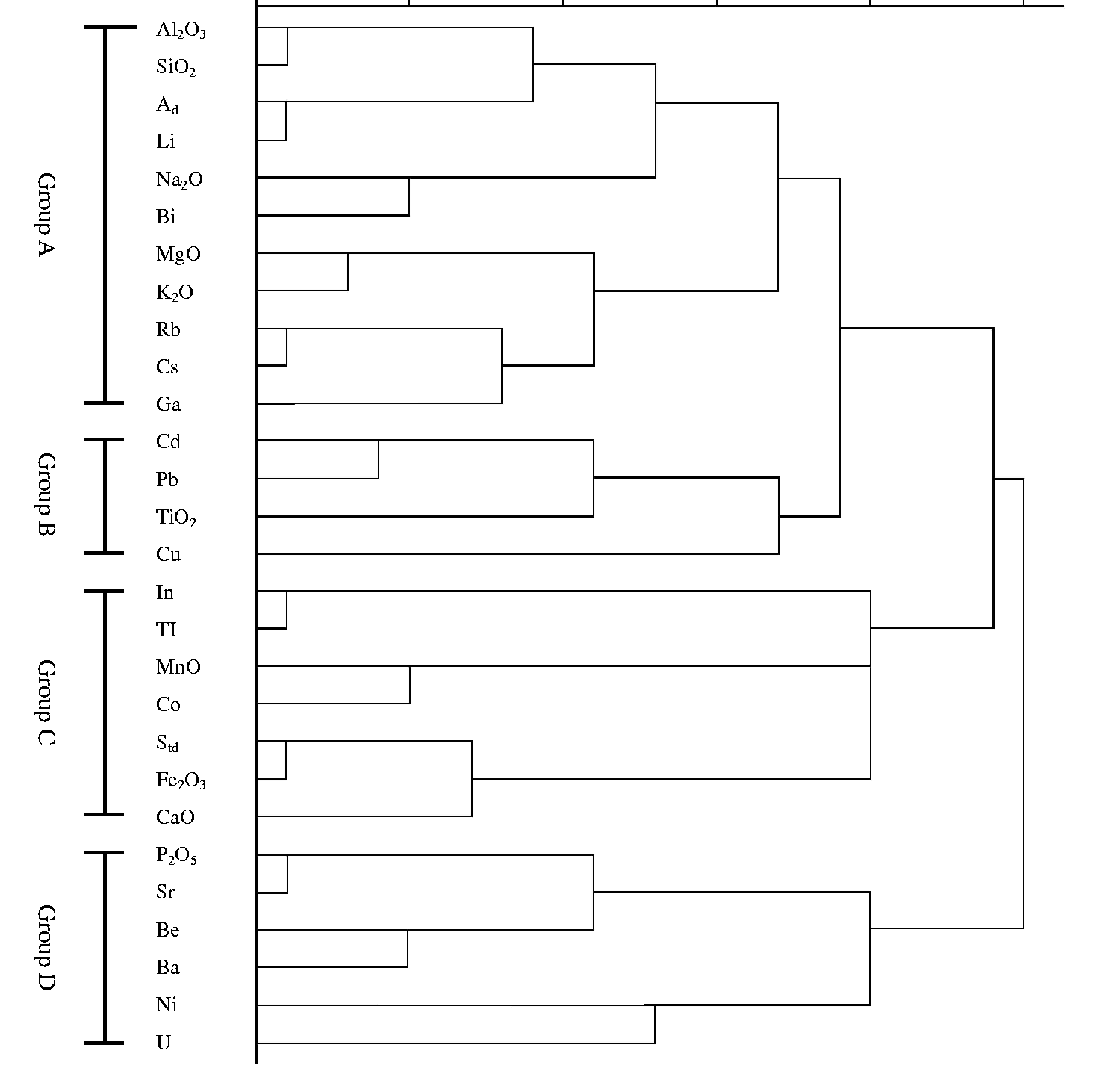
Dendrogram produced by cluster analysis of the analytical results for the 10 samples.
Group B includes Cd, Pb, TiO2, and Cu (Figure 9). This group exhibits positive correlations with ash yield, except for Cu (Table 3). The correlation coefficient of Pb–Std (0.58) indicates that Pb may be correlated with S, consistent with Querol’s et al. (1997) research. In addition, Cd (Swaine, 1990) and Cu (Pickhardt, 1989; Zhang et al., 1999) often occur in sulfides. However, copper can also associate with organic matter (Miller and Given, 1987a, 1987b). The elements in this group may be associated with unidentified traces of sulfide minerals or have an organic association.
Group C includes In, Tl, MnO, Co, Std, Fe2O3, and CaO (Figure 9). The correlation coefficients of Group C with ash yield range from −0.65 to 0.12, reflecting a certain organic affinity. Tl (Finkelman and Aruscavage, 1981; White et al., 1989), Co (Finkelman et al., 1990), S, and Fe often occur in pyrite and other sulfide minerals, and Mn and Ca often exist in carbonates. Mn, Co, S, Fe, and Ca are also distributed in organic matter and have negative correlations with the ash yield. Similar points of view are obtained in this study. All elements in this group may be associated with sulfides, carbonates, or organics.
Group D includes P2O5, Sr, Be, Ba, Ni, and U (Figure 9). All elements except Ni have negative correlations with ash yield. The correlation coefficients of P–Sr (0.99), P–Ba (0.91), Sr–Ba (0.90), and Ba–Be (0.71) indicate that they may be associated with gorceixite or goyazite.
Associated resources and harmful trace elements in the coal
Aluminum oxide
The content of Al2O3 in the ash of the Guanzigou Mine varies from 40.0 to 44.0%, and the average is 43.2%. The Geology and Ore Deposit Standard Specifications for Bauxite Exploration of the People’s Republic of China (DZ/T 0202-2002) stipulates that the content of an economic-grade Al2O3 deposit is >40%. Thus, the average Al2O3 content (43.18%) in the Guanzigou Mine has reached an associated deposit.
Lithium
Lithium is very useful not only as an important industrial raw material but also as an energy metal (Qin et al., 2015b; Sun et al., 2013b). A Li reactor requires only 10 t of Li to generate 10 billion kW h of energy (Metal, 2006).
The content of Li in the Guanzigou Mine (149.55 µg/g, Table 3) is much higher than that in the Heidaigou Mine and the average values in Chinese coal and world hard coals, and slightly higher than that in the Haerwusu Mine. However, the Li content in the Guanzigou Mine is slightly lower than that in the Guanbanwusu Mine. As previously mentioned, the coal in the Guanzigou Mine has already reached the cutoff grade for Li in coal and has great economic value (Sun et al., 2012b, 2014). Notably, the Li content in the partings of the Jungar coalfield is much higher than that in the coals of the Guanzigou Mine.
The modes of occurrence of lithium in coal have been studied (Finkelman, 1981; Sun et al., 2010, 2012a, 2013b; Tang and Huang, 2004). Studies of the occurrence of Li in the Haerwusu Mine (Dai et al., 2006) and the Guanbanwusu Mine (Sun et al., 2012a, 2013a) have shown that Li mainly occurs in aluminosilicates. In this study, Li correlates weakly with CaO, P2O5, Std, vitrinites, liptinites, and inertinites (Table 4), indicating that none of them are vectors of Li. However, Li correlates strongly with the ash yield (0.96), Al2O3 (0.96), and SiO2 (0.96) (Table 4), suggesting that Li may be associated with aluminosilicates, consistent with the results obtained by Dai et al. (2006) and Sun et al. (2013a) for the Jungar coalfield. In addition, the correlation coefficient of Li with MgO is 0.58, indicating that some Li may occur in the carbonate.
The correlation coefficients of Li and Ga in No. 6 Coal.

Ad: ash; Std: total sulfur.
Gallium
Gallium is also a very valuable and rare metal in coal (Qin et al., 2015a). The content of Ga is 20.50 µg/g (Table 2) in the Guanzigou Mine, and the CC value reaches 3.42. The Ga content in the Guanzigou Mine is much higher than that in the Guanbanwusu Mine, slightly higher than that in the Haerwusu Mine, but observably lower than that in the Heidaigou Mine. Like most mines, the Ga content in the Guanzigou Mine has not reached minimum mining grade. However, this does not mean that the Ga in the Guanzigou Mine has no commercial value. A study of the fly ash of the Jungar power plant by Dai et al. (2006) showed that the content of Ga in fly ash is much higher than that in coal and greatly exceeds the minimum mining grade of Ga. Therefore, Ga can be collected and extracted from fly ash. Like Li, the content of Ga is much higher in the partings and floor of the Guanzigou Mine than in the coals.
Gallium is generally associated with clay minerals in coal (Chou, 1997; Finkelman, 1981; Zhao et al., 2009) but can also occur in other minerals and organic compounds of coal (Seredin et al., 2013). For instance, in the Heidaigou (Dai et al., 2006), Ga occurs in boehmite and organic matter, boehmite, and goyazite, respectively. In this study (Table 4), Ga in the Guanzigou Mine has a strong negative correlation with goyazite based on the correlations between Ga and P2O5 (r = −0.66), Sr (r = −0.67), and Ba (r = −0.58), in marked contrast to the Guanbanwusu Mine. However, organic matter may not be a carrier of Ga because Ga also has strong negative correlations with vitrinites (r = −0.62), liptinites (r = −0.59), and inertinites (r = −0.02). However, Ga may occur in clay minerals or carbonate minerals based on the correlations of Ga–Ad (r = 0.61), Ga–SiO2 (r = 0.57), Ga–Al2O3 (r = 0.57), and Ga–MgO (r = 0.82).
Beryllium, uranium, cadmium, and plumbum
Be, U, Cd, and Pb are all harmful to the environment or human health (Ding et al., 2001; Swaine, 2000; Swaine and Goodarzi, 1995; Zhao and Sun, 2008). The contents of Be, U, Cd, and Pb in the Guanzigou Mine are 2.78, 3.2, 0.3, and 21.7 µg/g, respectively (Table 2). The contents of Be and Cd are similar to those in Chinese coals and do not pose major hazards to the environment or human health. However, the contents of Be, U, Cd, and Pb in the Guanzigou Mine are higher than world hard coals. Beryllium is combined with organic matter in coal, but it cannot be excluded from the combination of clay minerals (Finkleman, 1994). Uranium in coal is mainly in the form of complexes with organic matter combination, which is generally associated with clay minerals and pyrite in coal (Hu et al., 2016). Lead and cadmium are mainly in coal in inorganic form and Pb is related to sulfide minerals, Cd is related to oxide minerals (Gayer et al., 1999). Be and U have negative correlations with ash yield, but Pb and Cd have positive correlations with ash yield. Pb has a strong affinity for sulfur (Table 3), while Cd has a lower but still relatively high sulfur affinity. In addition, Cd (Swaine, 1990) often occurs in sulfides.
Consequently, the usage and treatment of this coal will have certain impacts on the environment or human health, which should be paid attention to.
Conclusions
Compared to Chinese coals and the world averages for hard coals, the contents of SiO2, Al2O3, TiO2, Li, Ga, In, and Pb are higher in the Guanzigou Mine, whereas other major elements, i.e. Co, Ni, Rb, Cs, and Ba, are lower in the No. 6 Coal (the bottom) in the Guanzigou Mine. The SiO2/Al2O3 ratio is lower than the average values in Chinese coal and is higher than the values in other mines in the Jungar Coalfield. The SiO2/Al2O3 ratio is closer to the ratio in pure kaolinite. This profile may reflect the lack of quartz and boehmite in the coals at the bottom of the Guanzigou Mine.
In the vertical section, SiO2, Al2O3, TiO2, Li, Ga, and Bi are enriched in the parting and floor; Na2O, MgO, K2O, and Rb are enriched in the parting; CaO, Fe2O3, P2O5, Std, Co, Be, Sr, and Ba are enriched in the coal. The remaining elements exhibit homogeneous vertical distributions.
The correlations with the ash yield and cluster analysis indicate that Al2O3, SiO2, Li, Na2O, Bi, MgO, K2O, Rb, Cs, and Ga are associated with clay minerals; Cd, Pb, TiO2, and Cu are associated with unidentified traces of sulfide minerals or organic matters; In, Tl, MnO, Co, Std, Fe2O3, and CaO are associated with sulfide, carbonate, or organic matters; and P, Sr, Be, Ba, Ni, and U are associated with gorceixite or goyazite.
Among the potential associated minerals, Al2O3, Li, and Ga have economic value, and comprehensive development and utilization are possible. The total content of harmful elements in the Guanzigou Mine is low, but the contents of Pb and U are slightly high and warrant attention.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National science and technology cooperation project (2014DFR20690) and the National Science Fundamental of China Project (41440019).
