Abstract
The occurrence of Rb and Cs in coal samples from Iqe coalfield was analyzed by a sequential chemical extraction experiment. Five types of Rb and Cs were determined in the coal: water soluble, ion exchangeable, organic bonded, carbonate, and silicate. The results indicated that the occurrence of Cs is similar with Rb in coal, and nearly all Rb and Cs are distributed in silicate. The occurrence of Rb and Cs is most likely associated with silicate minerals in coal. With the high correlation between Rb, Cs, and K, and the high content of kaolinite and illite in these coal samples, it can be inferred that illite probably is the main carrier of Rb and Cs. Minor amounts of Rb and Cs may also be adsorbed by kaolinite in Iqe coal, and Cs may exist in other states, such as an exchangeable ion.
Introduction
More and more valuable trace elements, such as Li, Ga, rare earth elements, Ge, Nb, Ta, Sc and so on, have been found in coal (Qin et al., 2015; Qin and Li, 2015; Sun 2015; Sun et al., 2010, 2012a, 2012b, 2013a, 2013b, 2015, 2016; Xiao et al., 2016; Xu et al., 2011; Yi et al., 2015; Zhao et al., 2009, 2014). Anomalous enrichments of Rb and Ce have been found in coals from Far East Russia, Qinghai-Tibet Plateau (Dai et al., 2014; Eskenazy, 2009; Seredin, 2003; Sun et al., 2015). The content and occurrence of Rb and Cs have been studied by some geologists (Dai et al., 2014; Eskenazy, 2009; Eskenazy and Ivchinova, 1987; Ketris and Yudovich 2009; Seredin, 2003; Swaine, 1990). Most of them believe that Rb displays an inorganic mode of occurrence in coal (Eskenazy, 2009; Yossifova et al., 2011). Seredin (2003) pointed out that Rb was mainly absorbed by clay material rather than organic matter, whereas Cs was presumably absorbed by both components in the Spetzugli brown coal, and Yossifova et al. (2011) reported that Rb in the Pernik coal has a distinctly inorganic mode of occurrence. Cesium can isomorphously substitute K, so it generally occurs in K-bearing minerals (Swaine, 1990; Tang and Huang, 2004). Illite is the major carrier of Cs in Wulantuga coal (Dai et al., 2014). Sequential chemical extraction experiment (SCEE) is an effective method to study the mode of occurrence of elements (Bacon and Davidson, 2008; Dai et al., 2002; Dreher and Finkelman, 1992; Quevauviller et al., 1997; Sun et al., 2013, 2016; Tessier et al., 1979). The concentrations of Rb (average 128.56 ppm) and Cs (average 16.60 ppm) in the No. 7 Coal from Iqe Coalfield are 10.9 and 15 times higher than those of world coals, respectively (Sun et al., 2015). The purpose of this paper is to study the mode of occurrence of Rb and Cs in Iqe coal.
Geological setting
Iqe Coalfield is a long and narrow continental basin, which is located in the northeast part of Qaidam Basin, Qinghai-Tibet Plateau (Figure 1(a)). The coal-bearing sequences include the Dameigou Formation and Shimengou Formation, which belong to the Middle Jurassic, have an average thickness of 110 m of the two formations. There are seven seams in the coal measures. No. 5 and No. 7 seams are the main minable seams, with an average thickness of 12.4 m, while No. 3, No. 4, and No. 6 are partly minable seams (Figure 1(b)). Anomalous rare elements were found by Sun et al. (2015) in the No. 7 coal from Iqe coalfield.
Location and sedimentary sequences of Iqe Coalfield. (a) Location of Iqe Coalfield; (b) generalized sedimentary sequences of Iqe Caolfield.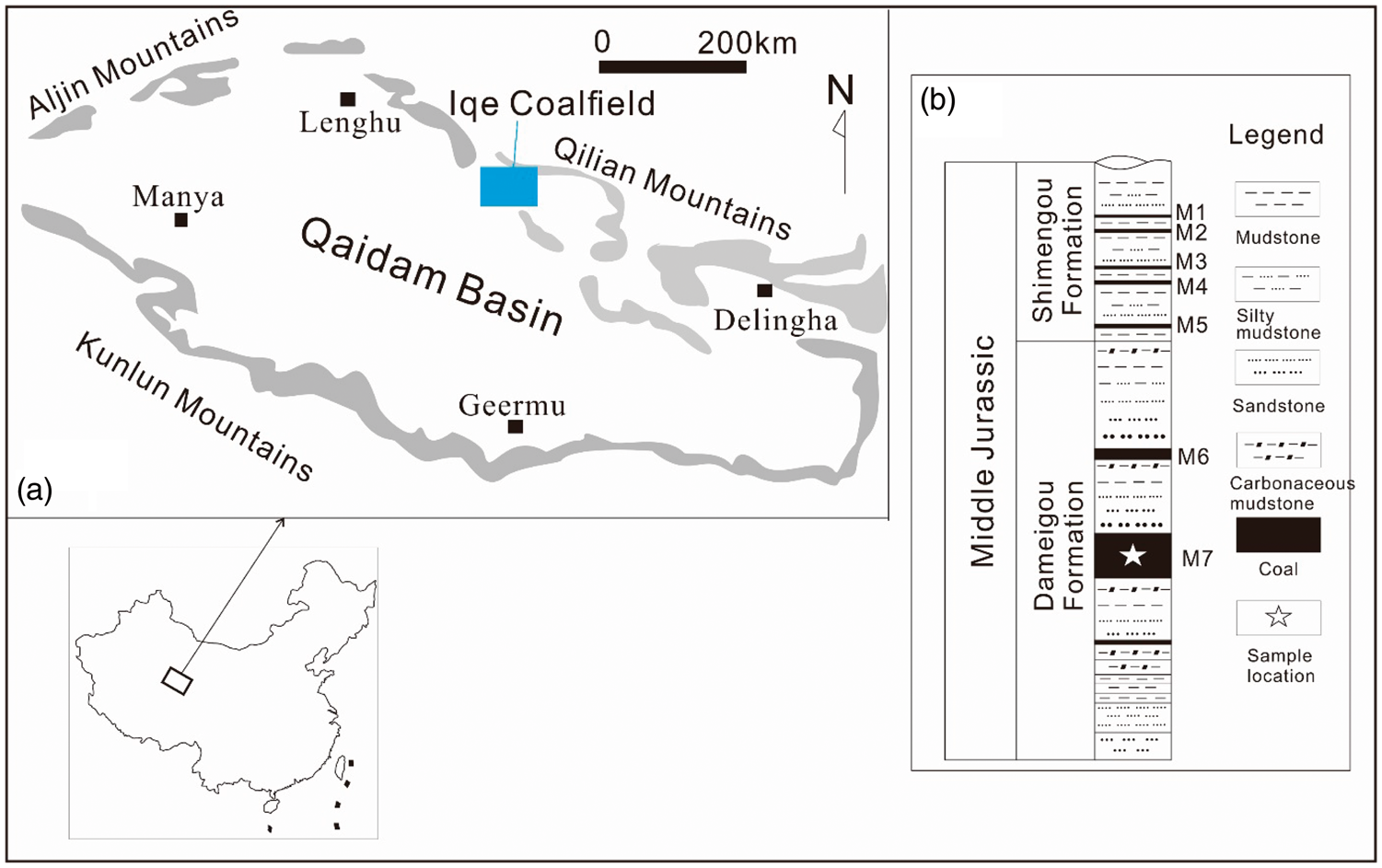
Sampling and analytical procedure
Sampling
A total of 23 bench samples were taken from the drillcore of the No. 7 coal in the Iqe Coalfield, following Chinese Standard Method GB/T 482-2008. All collected samples were immediately stored in plastic bags to minimize contamination and oxidation. The samples were divided into two groups: coal (Ad < 50%) and parting (Ad > 50%). The random vitrinite reflectance, ash yields, volatile matter (daf: dry and with ash basis), and moisture (ad: air-dry basis) are 0.659%, 40.5%, 23.05%, and 3.2% of the coal samples, respectively. It belongs to higher ash (40.5%) and lower moisture (3.2%) coals. The average total sulfur content of the coal samples is 0.43% (Sun et al., 2015). All analyses were performed in the Key Laboratory of Resource Exploration Research of Hebei Province.
ICP-MS analysis
The element content was determined by inductively coupled plasma mass spectrometry (ICP-MS) in the Key Laboratory of Resource Exploration Research of Hebei Province. For the ICP-MS analysis, 40 mg sample (<200 mesh) were put into a poly tetra fluoro ethylene (PTFE) vessel, and 2 ml HF (50%) + 5 ml HNO3 (65%) + 2 ml H2O2 (30%) were added into the vessel, then microwave-digested for 36 h at a temperature of 200℃. The vessels were left overnight to cool down. Then the digested liquid was evaporated at 90℃ under vacuum, which took about 2 h. The residue was then dissolved in 1 ml HCl (32%) + 8 ml deionized water + 1 ml HNO3 (65%). This solution was then transferred into 125 ml fluoro-ethylene polymer (FEP) bottles which were filled up to 100 ml with deionized water. The signal collection of ICP-MS lasted for 60 s, and the scanned mass ranges were set at 6–235 amu to cover the target elements.
X-ray powder diffraction analysis
The mineralogical composition was determined by powder X-ray diffraction (XRD). The samples were crushed and ground to 200 mesh and oven-dried overnight before ashing in a low-temperature asher. Low-temperature ashing (LTA) of coal was performed on an EMITECH K1050X plasma Asher. The temperature for LTA was lower than 200℃ (75 W power). XRD analysis of the low-temperature ashes and the parting samples was performed on a D/max-2500/PC powder diffractometer with Ni-filtered Cu-Kα radiation and a scintillation detector. The XRD pattern was recorded over a 2θ interval of 2.6–70°, with a step size of 0.01°.
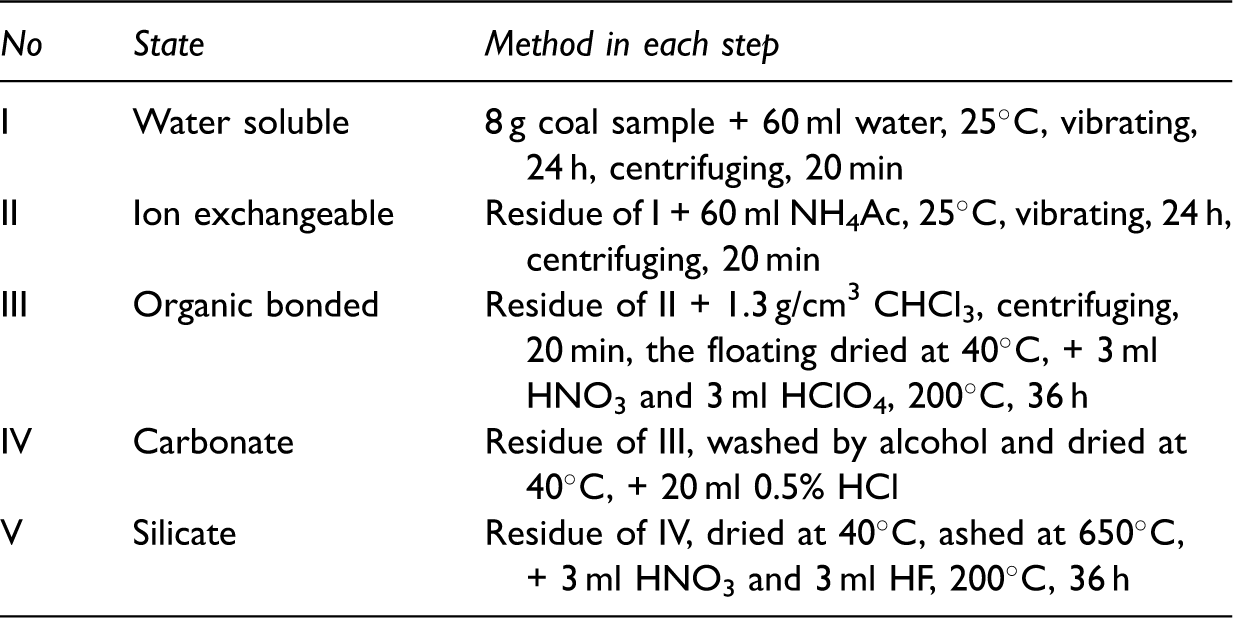
SCEE analysis
The five-step sequential chemical extraction experiment (SCEE).
SEM-EDS analysis
A scanning electron microscope (HITACHI UHR FE-SEM, SU8220) equipped with an energy-dispersive X-ray spectrometer (SEM-EDS) was used to study the distribution characteristics of the minerals, and the distribution patterns of some elements of interests in the coal.
Results and discussion
Concentrations of Rb and Cs
Concentrations of Rb, Cs, and major element oxides in samples from the core of ZK32-5N (%; on whole-coal basis). a
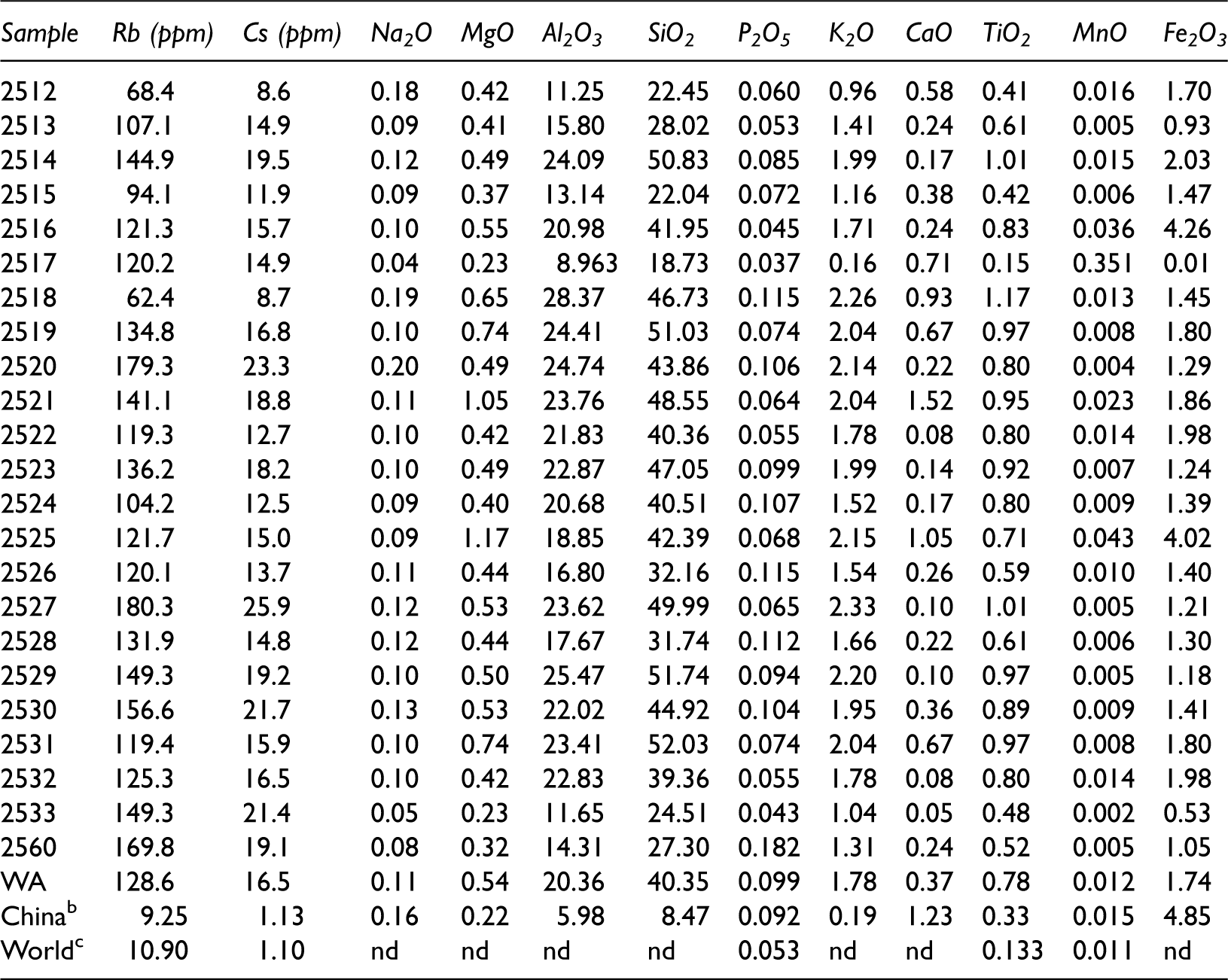
WA: weighted average; nd: no data.
Data from Sun et al. (2015).
Data from Dai et al. (2012)
Data from Ketris and Yudovich (2009).
Mineral composition
The data of XRD show that the minerals in the coals are mainly kaolinite, quartz, and illite. Some traces of pyrite, siderite, calcite, and rutile were also detected (Figure 2).
Optical photomicrographs of minerals in the Iqe coals (reflected light). (a) Cell-filling clay; (b) subangular detrital quartz; (c) framboidal pyrite and pyrite vein; (d) calcite vein.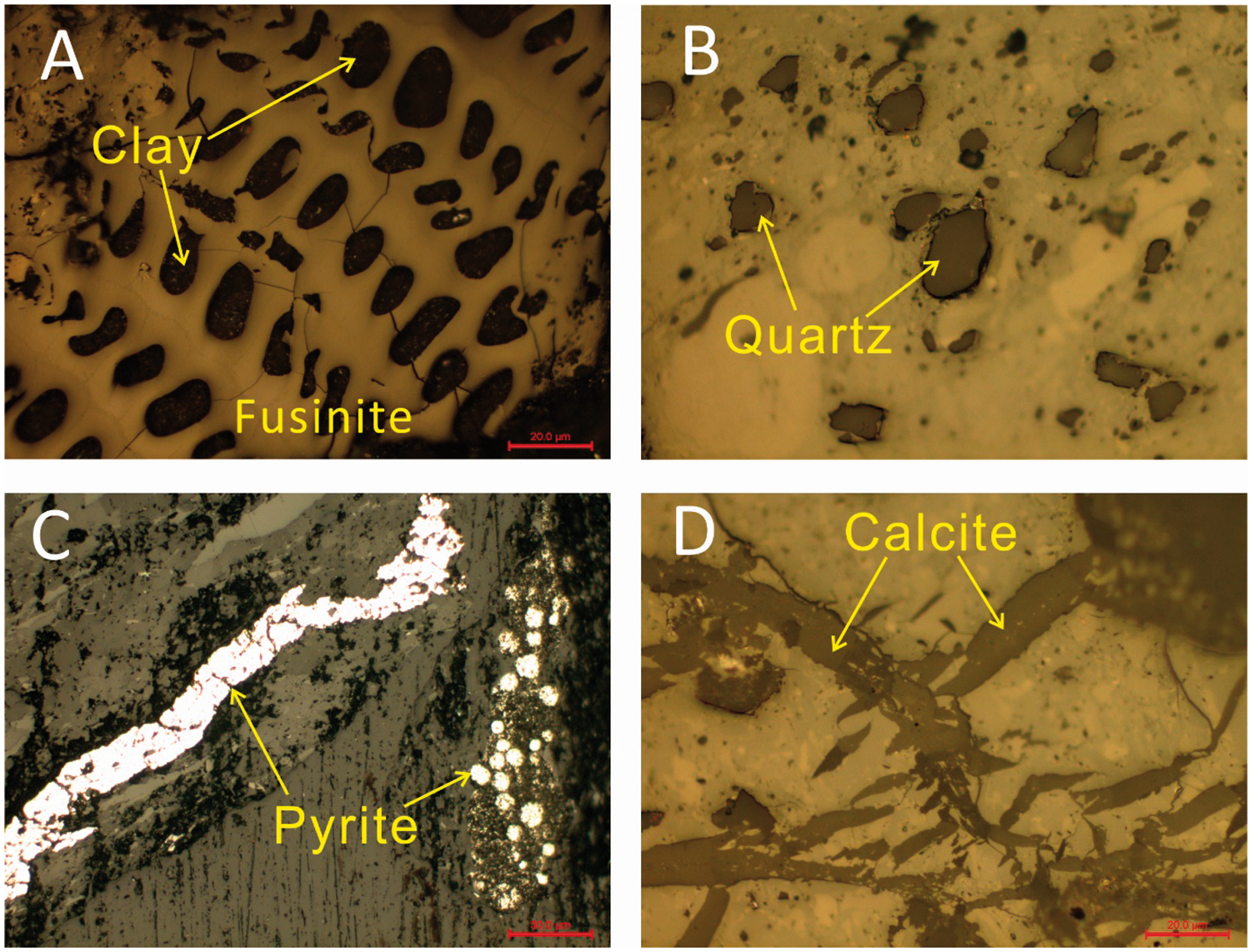
Kaolinite, the main clay mineral in Iqe coal, was determined by XRD. The XRD pattern shows that the kaolinite in the coal LTAs has a well-ordered structure (Figure 3). Kaolinite often occurs in cell cavities and fusinite, or belt shape (Figure 2). Kaolinite may be formed by weathering from illite.
X-ray powder diffraction patterns of the coal low-temperature ashing (LTA).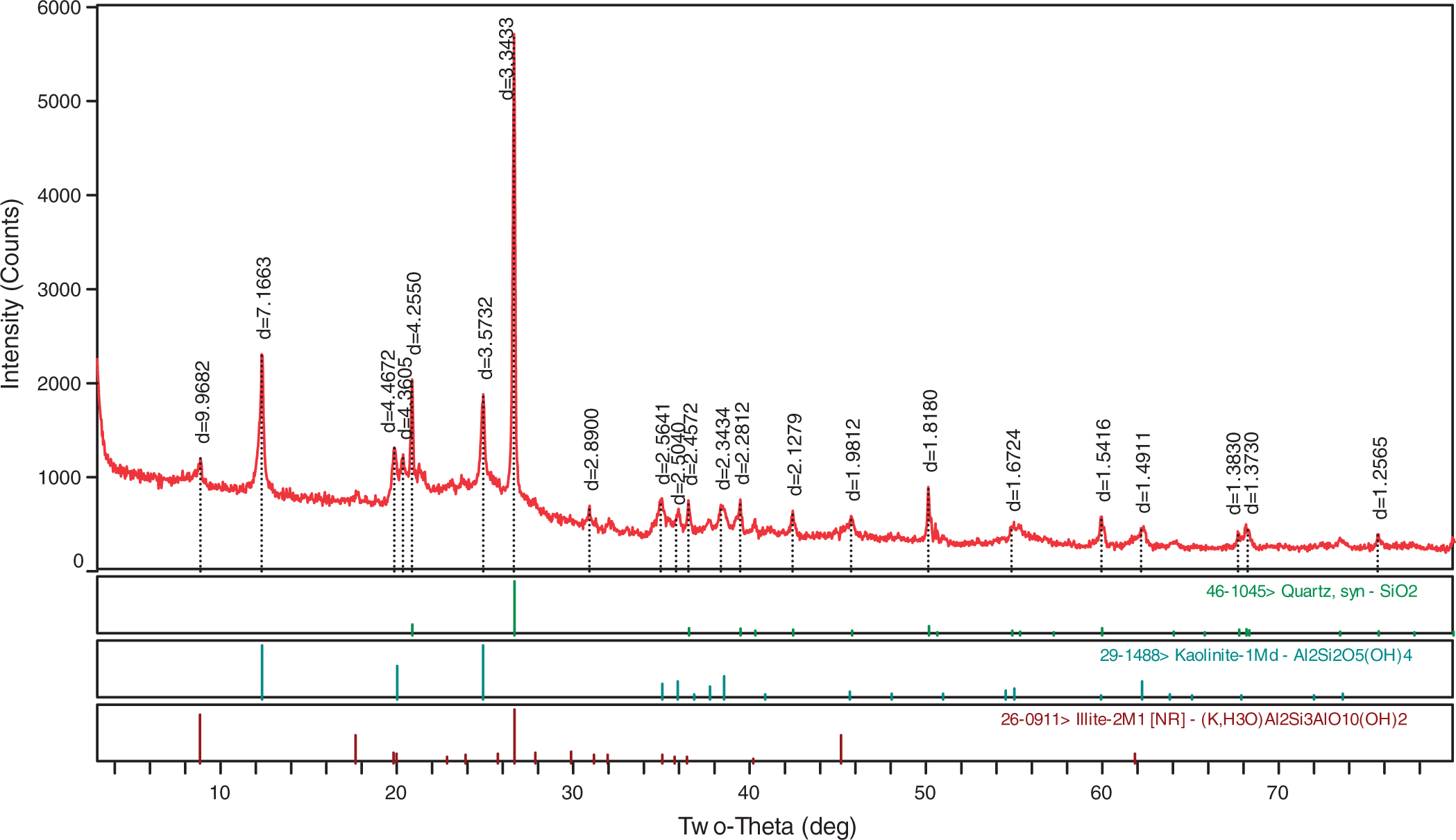
Illite is often associated with quartz and kaolinite (Figure 4), and distributed in the collodetrinite or fusinite. Muscovite is mostly derived from the source area—Qilian Mountain, which mainly consists of granite and gneiss. These rocks were made up of feldspar, quartz, and mica and because Qilian Mountain is very close to the northern edge of the Iqe coalfield, it is probable that illite in the Iqe coalfield is of terrigenous origin. As the only K-bearing mineral in Iqe coal, illite may be the most likely carrier mineral of Rb and Cs in Iqe coals.
Modes of occurrence of minerals in the coal. Spot 1: energy-dispersive X-ray spectrometer (EDS) of illite; spot 2: EDS of quartz; spot 3: EDS of rutile.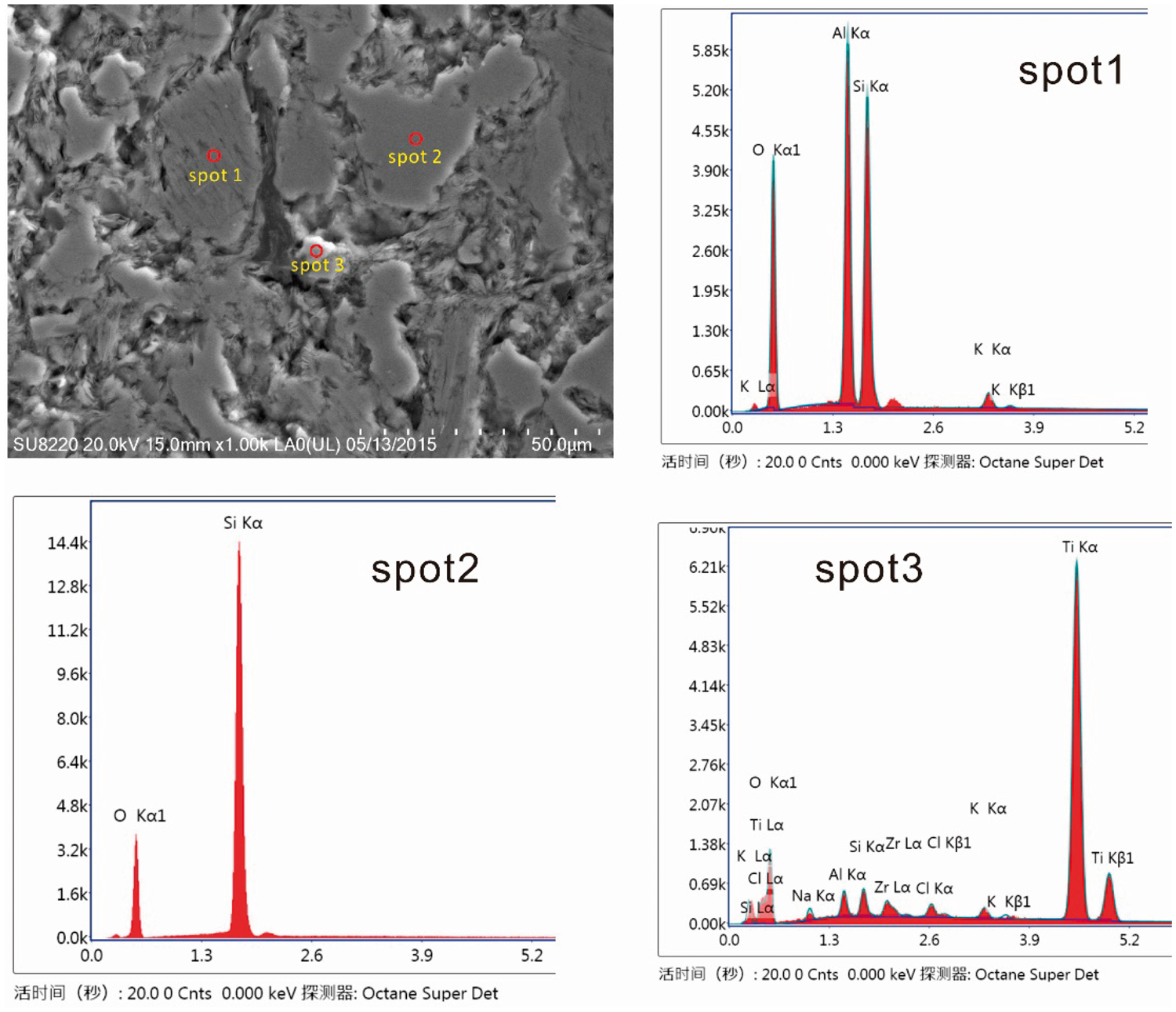
Quartz is the most important oxide mineral in coal. The content of quartz is a marginally higher in the Iqe coal than that of the common Chinese coal, and most of the quartz particles are irregular with low sphericity (Figure 2), possibly due to the closeness of the source region.
Apatite and rutile are rare in the coal and below the detection limit of the XRD, they can only be observed under SEM (Figure 4). Apatite generally occurs in the cells of fusinite and in collodetrinite with kaolinite or muscovite. The apatite is chlorapatite, which is determined by energy dispersive X-ray analysis. Rutile is always associated with kaolinite or muscovite as irregular particles, distributing in the mixed-layer clay or collodetrinite.
SCEE results
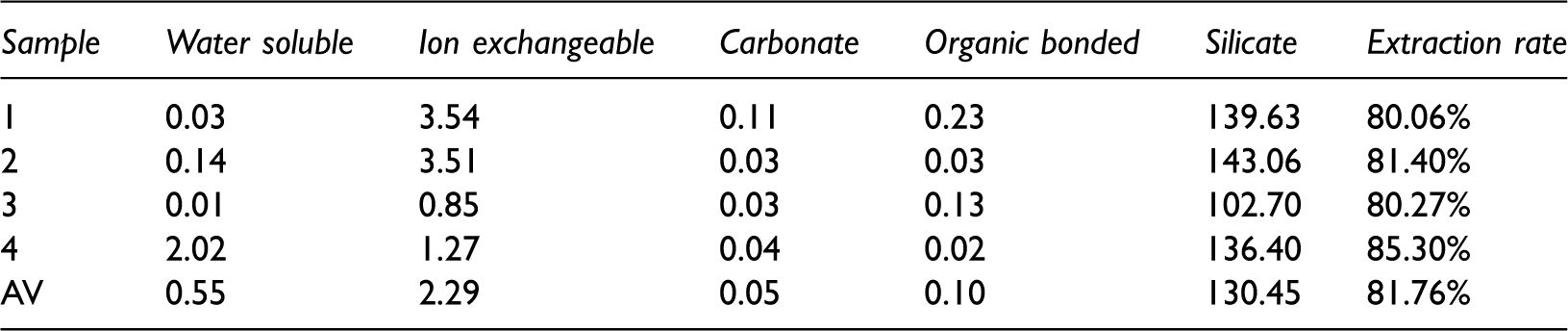
Rubidium contents in different fractions by SCEE (ppm).
SCEE: sequential chemical extraction experiment.
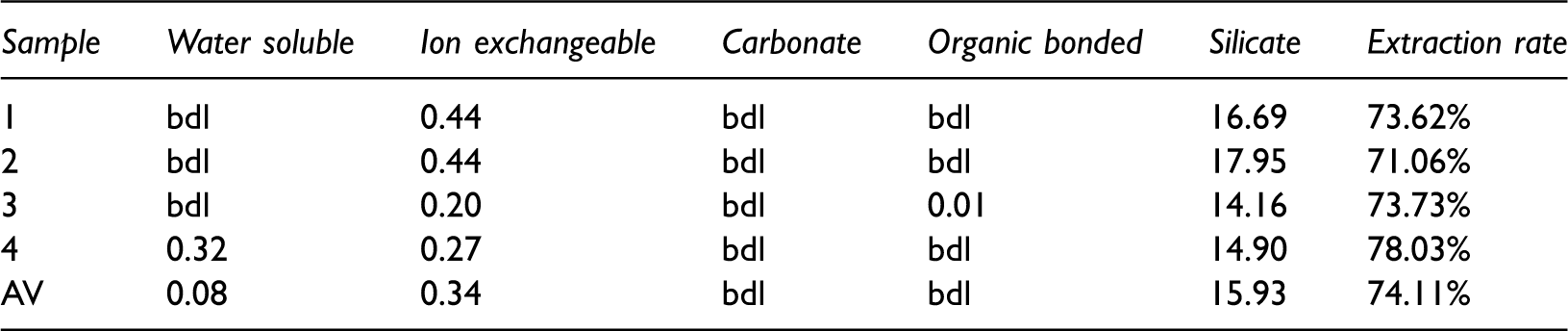
Cesium contents in different fractions by SCEE (ppm).
bdl: below the detection limit; SCEE: sequential chemical extraction experiment.
The data show that rubidium contents are much higher in the inorganic fraction than those in the organic fraction. The dominant Rb content is in the silicate of every sample, and more than 130 ppm (97%) Rb is in a silicate state (Table 3). Only 2.29 ppm (1.6%) Rb is in an ion exchangeable state. Water soluble, carbonate, and organic bonded fractions are lower than 1 ppm. This phenomenon indicates that nearly all rubidium (99%) in the Iqe coal has an affinity with inorganic matter, especially silicates, and has a much lower affinity with organic matter.
Cesium has the same distribution with rubidium in each fraction. There are more than 97% (15.9 ppm) Cs in silicate state, showing the high affinity between Cs and inorganic matter. The content of Cs in the rest of fractions is lower than 1%. The results suggest that cesium may have the same occurrence with rubidium, and both of them mainly occur in a silicate state.
In addition, about 20% of Rb and 25–30% of Cs were not extracted from the coal samples by SCEE (Tables 3 and 4), maybe these ‘missing’ elements are due to insoluble phases, minerals shielded from the solvents and organic complexes.
The occurrence of Rb and Cs in Iqe coal
The SCEE results show that most of the Rb and Cs is associated with silicates with only minor amounts of both elements occurring in other forms. Kaolinite and illite are the common silicate minerals in Iqe coal, and the mode of occurrence of Rb and Cs is most likely related to these two minerals.
In addition to the high proportion in the silicate fraction in SCEE, Rb and Cs have a high correlation coefficient with SiO2, Al2O3, the ash yield, and K2O (Figure 5) based on analysis of the samples. As shown in Figure 5, the correlation coefficient between Rb and SiO2, Al2O3, ash yield and K2O is very high, and this is consistent with the SCEE, indicating that the occurrence of Rb must be associated with silicate minerals in Iqe coals. The correlation coefficient between Rb and K2O is higher than that with SiO2, Al2O3, and ash yield, which shows that Rb may have a better affinity with K than SiO2 and Al2O3. Cesium regulates similar magnitude of the correlation coefficient; thus suggesting that their carriers are K-bearing clay minerals (Eskenazy and Ivchinova, 1987; Filby et al., 1997; Yudovich et al., 1985), which is illite in Iqe coal.
The correlation coefficient between Rb, Cs and SiO2, Al2O3, ash yield, and K2O.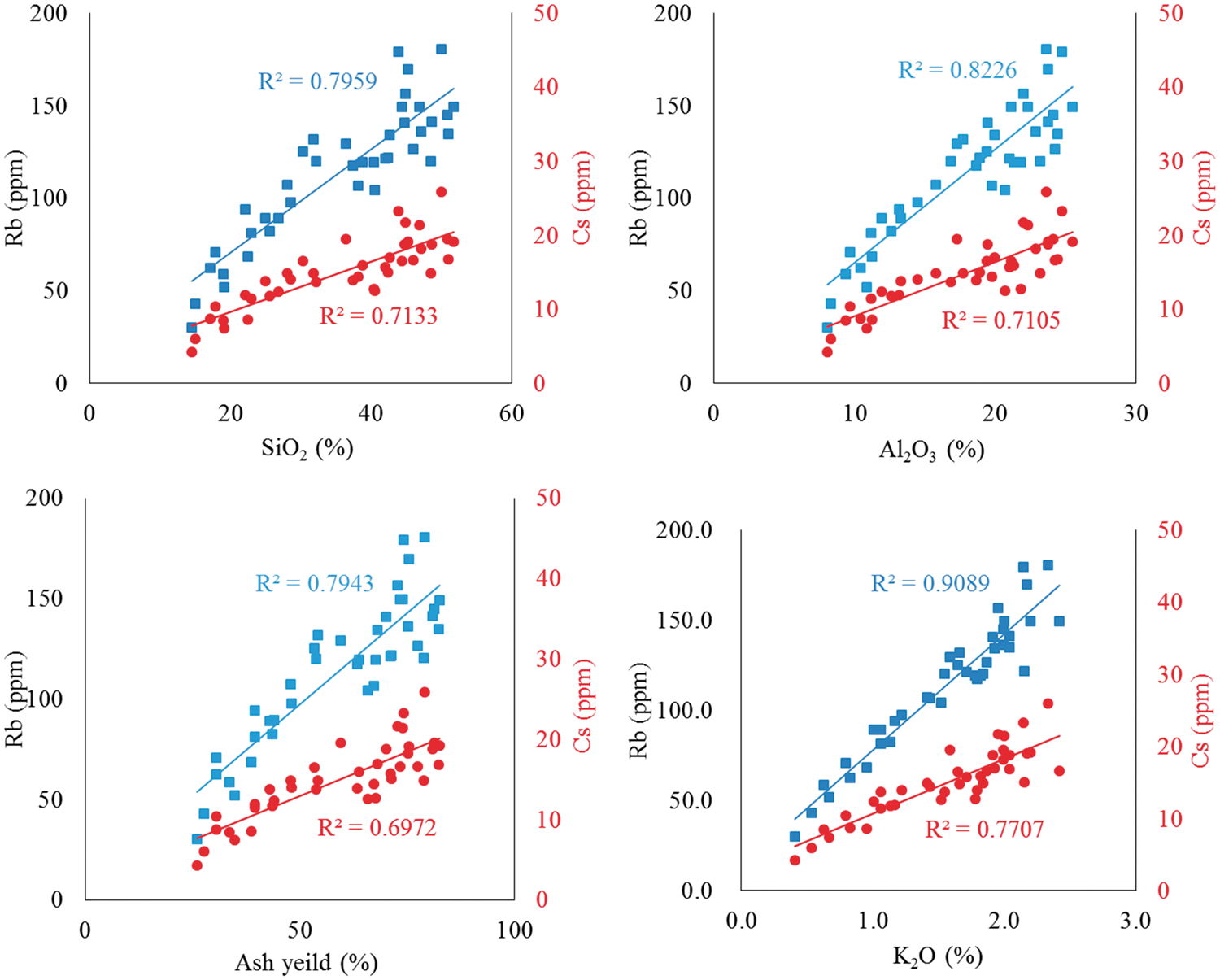
Rubidium has a higher correlation coefficient with SiO2, Al2O3, ash yield, and K2O than cesium (Figure 5). The correlation coefficient mostly depends on the geochemical characteristics of Rb and Cs themselves. Both Rb and Cs can isomorphically replace potassium, and rubidium has a similar ionic radius to potassium (Bastide et al., 1995; Doyle et al., 1998; Ganzeyev et al., 1969); therefore, most of the Rb can be substituted with K isomorphously in K-bearing minerals. However, the ionic radius of cesium is larger than that of potassium, limiting the amount of cesium that may be incorporated, in K-bearing minerals, possibly explaining the lower correlation coefficient of Cs with K2O.
Conclusion
SCEE results show that rubidium and cesium occur mainly in the silicate fraction (more than 97%) in Iqe coal, and only minor amounts occur in the ion exchangeable fraction. Almost no Rb and Cs are in a water soluble state, an organic bonded state or a carbonate state under the experimental conditions. Both Rb and Ce have a high correlation coefficient with SiO2, Al2O3, and K2O, the highest for both elements being with K2O and Al2O3, indicating that the occurrence of Rb and Cs must be closely associated with K-bearing silicate minerals, which is isillite in Iqe coal. Both Rb and Ce can isomorphically replace K. Due to the difference of ionic radius with K, the amount of Rb which can substitute isomorphically for K in K-bearing mineral is higher than that of Cs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National Science Fundamental of China (Nos. 41330317, 41402318, 41641019 and 41611130046) and Science Fundamental of Hebei (Nos. D2016402104 and D2015402029).
