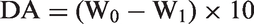Abstract
Studying the interactions between CO2-rich fluid and reservoir rock under reservoir temperature and pressure is important for investigating CO2 sequestration and the CO2-enhanced oil recovery processes. Using high-concentration CaCl2-type formation water as an example, this study performed a CO2-rich fluid–rock interaction experiment at 85℃ and compared the dissolution of calcite and sandstone samples, as well as sandstone powder and thin-slice samples. This study also investigated the effects of the sample surface area, the CO2 partial pressure (P
Introduction
Geological sequestration of carbon dioxide (CO2) can reduce the CO2 content of the atmosphere and is a feasible means to mitigate the greenhouse effect (Balat and Öz, 2007; Bertier et al., 2006; Cantucci et al., 2009; Chen et al., 2014; Holloway, 1997; Jun et al., 2012; Krevor et al., 2012; Lu et al., 2012; Parthasarathy et al., 2016). Depleted or nearly depleted petroleum reservoirs are important media for CO2 geological sequestration (Leung et al., 2014; Worden and Smith, 2004). All petroleum reservoirs are proven to have the capacity to store fluids (Lu et al., 2012; Shukla et al., 2010). In addition, the injection of CO2 into a petroleum reservoir will increase the internal pressure of the reservoir (Leung et al., 2014), reduce the viscosity of the crude oil (Holm and Josendal, 1974), and facilitate the flow of oil and gas in the rock pores, which could achieve CO2-enhanced oil recovery (EOR) and simultaneously offset some of the economic costs of CO2 geological sequestration (Bachu and Bennion, 2008; Chiaramonte et al., 2011). Thus, CO2 sequestration in petroleum reservoirs has been widely used and successfully implemented in several oil and gas fields (Holtz et al., 2001). However, after being injected into a geological formation, large amounts of CO2 can be easily dissolved in the formation water and form an acidic fluid, which will alter the chemical equilibrium between the formation water and the rocks and induce chemical reactions (dissolution or precipitation) that can change the geochemical characteristics of the formation water and the mineral composition and physical properties of the reservoir rocks (André et al., 2007; Gao et al., 2007; Kampman et al., 2014; Shao et al., 2010). These changes might have significant impacts on the effective implementation of CO2 flooding and the long-term safety of CO2 geological sequestration (Bertier et al., 2006). Therefore, before implementing CO2-EOR or CO2 geological sequestration, it is necessary to assess the effects of CO2 injection on the properties of the formation water and the physical properties of the reservoir.
Previous researchers have performed extensive experimental studies on the mechanisms and characteristics of the water–rock reactions that occur during CO2-EOR and CO2 geological sequestration. Pressures of 1–30 MPa and temperatures from 21 to 120℃ have been used in experiments regarding the interactions between rock, CO2, and saline water (Bertier et al., 2006; Fischer et al, 2010; Lamy-Chappuis et al., 2014; Lu et al., 2012; Shao et al., 2010; Zheng et al., 2015). In several studies, the experimental temperature (200℃) was set higher than the reservoir temperature to increase the reaction rate (Kaszuba et al., 2003, 2005; Lu et al., 2011). The main component of the reaction solution was sodium chloride (NaCl), and relatively low concentrations of NaCl have been used. Because of the differences in the reaction conditions, there have been several discrepancies between previous experimental results that affect the understanding of the actual water–rock reaction process. For example, several studies have demonstrated that the acidic fluid that forms after the injection of CO2 into a geological formation will dissolve carbonate minerals such as calcite, dolomite, and siderite (e.g. Emberley et al., 2004; Kharaka et al., 2006; Sayegh et al., 1990; Shiraki and Dunn, 2000; Wigand et al., 2008). The dissolution of carbonate minerals is an important mechanism for the formation of secondary pores and can even lead to the formation of secondary dissolution pathways, which can increase the permeability of the rocks (Cao et al., 2016; Farquhar et al., 2015). This geological process can increase the CO2 storage space and facilitate the flow of CO2 in the reservoir (Lamy-Chappuis et al., 2014), which improves the efficiency of CO2-EOR, but increases the risk of CO2 leakage (Kharaka et al., 2006; Rathnaweera et al., 2016). In contrast to the dissolution of carbonate minerals, several researchers believe that the carbonate ions (
Previous studies have mainly used low-concentration NaCl reaction solutions. However, the formation water of an actual oil reservoir is generally characterized by high salinity that can reach more than 300 g/l (Hanor, 1994). In addition, most formation water is calcium chloride (CaCl2) type, and the concentration of CaCl2 can reach 166.64 g/l (Guo, 2012). Thus, this study prepared two high-concentration reaction solutions: 3 mol/kg NaCl and 1 mol/kg CaCl2–2 mol/kg NaCl solutions. Sandstones are common petroleum reservoir rocks, and sandstone aquifers are also the preferred target for CO2 sequestration (Rathnaweera et al., 2016). The dissolution and precipitation of carbonate cements have a significant impact on the physical properties of sandstone reservoirs (Taylor, 1990). Calcite is the most common carbonate cement (Kaszuba et al., 2003). This study performed water–rock (CO2-rich fluid–calcite and CO2-rich fluid–sandstone with carbonate cement) reaction experiments at 85℃ and CO2 partial pressures (P
Experimental section
Experimental samples
The experimental samples included calcite and sandstone powder and thin slices. Sandstones were collected from drill cores in the lower part of the first member of the Paleogene Shahejie Formation in the Pucheng oilfield. The Pucheng oilfield is in Fanxian County in Henan Province, which is in the northeastern part of the central uplift belt in the Dongpu Depression (Figure 1). The Dongpu Depression is a Cenozoic rift basin that developed on the Mesozoic–Paleozoic basement. It trends NNE over 5300 km2. Burial history analysis showed that the Dongpu Depression uplifted in Mesozoic (depth of < 4000 m) and subsided in Cenozoic (depth of > 8000 m). The oilfield in the lower part of the first member of the Paleogene Shahejie Formation is in the northeast region of the Pucheng anticline; the trap type is a tectonic-lithologic trap (Peng et al., 2010; Xue and Gao, 2011; Zhou and Xu, 2007). The Pucheng oil field has already been in a high water-cut stage (Li and Cheng, 2011); therefore, CO2 flooding is an effective means to enhance oil and gas recovery. The age at the bottom of the lower part of the first member of the Paleogene Shahejie Formation is ∼35 Ma. A tectonic uplift occurred in ∼22 Ma and then subsided in 17 Ma. The present buried depth is 2100–2437 m (Peng et al., 2010). The grain sizes of sandstones were in the range of 0.01–0.1 mm, and the roundness varied from subangular to subrounded. Sandstones are typically arkoses, exhibiting a grain-supported framework and a high degree of compositional and textural maturity (Figure 2). Microscopic identification showed that the sandstone was composed of ∼50% quartz, 25–30% feldspar, 10–15% carbonate cement, and small amounts of siderite and pyrite. The most important diagenesis that the reservoir experienced includes compaction and carbonate cementation. Mechanical compaction led to reorientation of detrital grains, resulting in planar to concavo-convex grain contacts. Some rigid grains like quartz are fractured under overburden pressure. The carbonate cementation in the sandstone reservoir was strong, and calcite was the main cement that filled the sandstone pores. Both compaction and cementation were the main reasons that primary pores reduced greatly.
Schematic map of local structure in Dongpu Depression and the position of the Pucheng oilfield. Microscopic (a, perpendicular polarized light) and back scatter electron (b) images of the sandstone samples. Kfs represents potassium feldspar (K-feldspar). Cal, Qtz, and Ab denote calcite, quartz, and albite, respectively.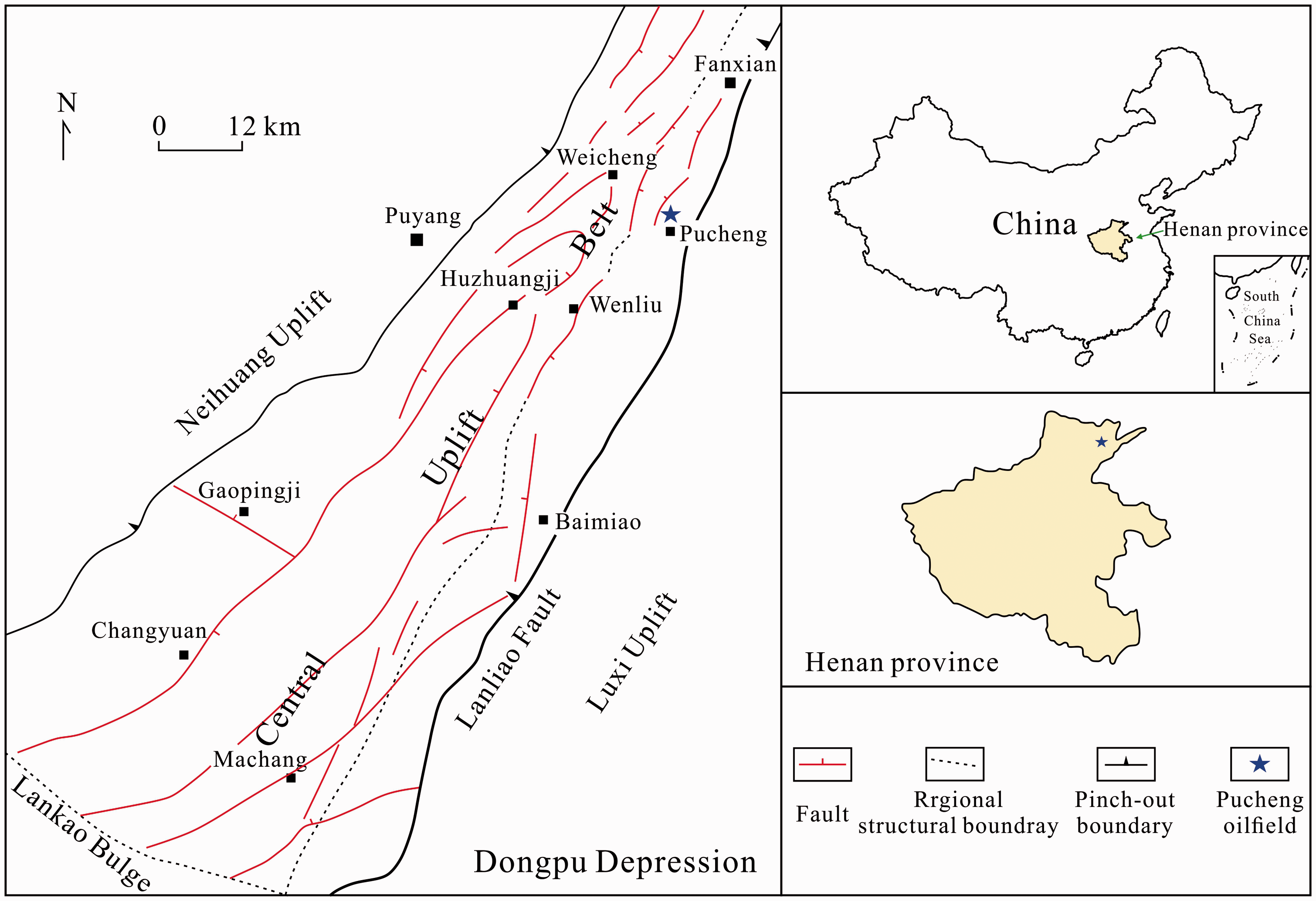

Calcite and sandstone samples were ground into a powder with particle sizes of 0.8–2 and <4 mm, respectively. Thin slices with thicknesses of 5–8 mm and a diameter of 25 mm were prepared from the sandstone core plug to observe the changes in the morphology of the minerals on the sandstone surface after dissolution. The dissolution of the sandstone powder and thin slices was also compared. Prior to the start of the experiment, all samples were placed in an oven (Hengchang Instrument Factory of Shanghai Hualian Environmental Test Equipment Company) and dried for 72 h at 55 ± 1℃.
The reaction solutions were prepared using chemical reagents (NaCl: purity ≥ 99.5%, Sinopharm Chemical Reagent Co., Ltd; CaCl2: purity ≥ 96.0%, Nanjing Chemical Reagent Co., Ltd) and deionized water at different molalities, mol/kg (m). The compositions of the reaction solutions, which were prepared to simulate the formation water, were 3 m NaCl (175.32 g/l) and 1 m CaCl2–2 m NaCl (111 g/l CaCl2 and 227.88 g/l total mineralization). The CO2 used in this study was 99.999% pure (Yangzhou Nanyang Gas Co., Ltd).
Experimental methods
Figure 3 is a schematic diagram of the experimental apparatus. During the experiment, the dried calcite particles, sandstone powder, and sandstone thin slices were measured using a Mettler-Toledo AL204 electronic balance with an analytical precision of 10−4 g. Then, the measured sample was placed in a reaction flask, which was filled with 100 ml reaction solution (3 m NaCl or 1 m CaCl2–2 m NaCl). The flask was then placed in a high-pressure autoclave and sealed. The inner wall and lid of the autoclave were made of 1Cr18Ni19Ti alloy so that it could withstand the corrosion from weak acids and bases. The total volume of the autoclave is ∼1000 ml. The autoclave can hold pressures up to 50 MPa. A pressurization system was used to control the inner pressure of the autoclave. The detailed procedures of the pressurization were summarized as follows: (1) CO2 was discharged from a CO2 cylinder into the autoclave, which was equipped with a piston; (2) water was injected into the bottom of the autoclave using a pressure pump to reduce the volume of CO2 above the piston, which can increase the CO2 pressure; (3) high-pressure CO2 was discharged into the autoclave to allow the pressure to be slightly lower than the target pressure (i.e. < 10 or 20 MPa). The pressure pump can generate pressures ranging from 0.1 to 70 MPa. All pressure control devices were purchased from Yangzhou Huabao Petroleum Instrument Co., Ltd. The pressurized autoclave was placed in a heating oven (Guangpin Test Equipment (Shanghai) Co., Ltd) and heated to 85℃. In addition, the pressure inside the autoclave was adjusted to the target value (10 or 20 MPa). The reactions ran for 20–200 h.
Apparatus used for the rock–CO2–saline water interaction experiments.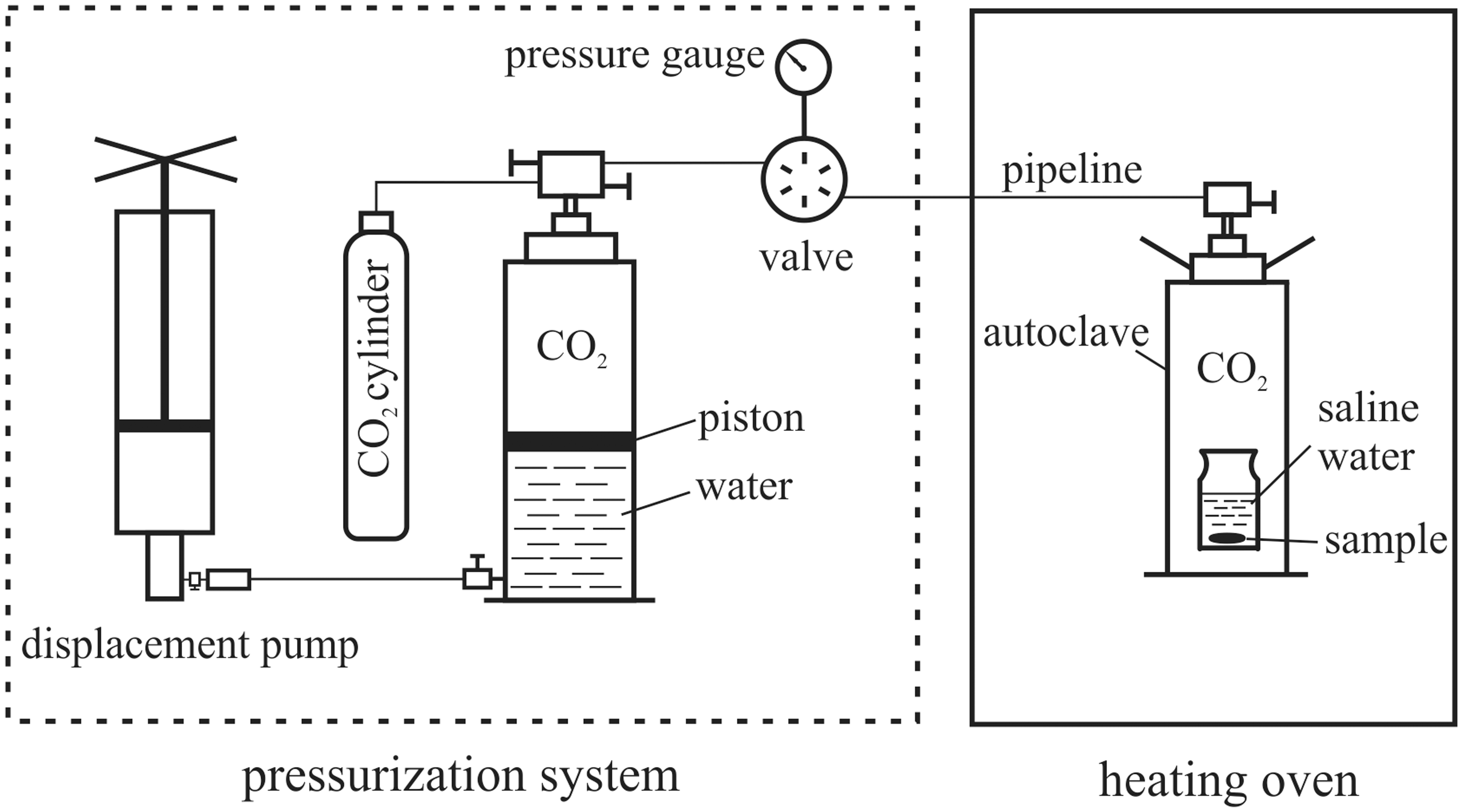
When the reaction time reached the preset value, rapid quenching and depressurization were performed. The autoclave was opened, and the reaction flask was removed from the reactor. The solution inside the flask was rapidly filtered using a Buchner funnel. The filtrate was collected, and 10 ml of diluted hydrochloric acid (HCl) was added to prevent the formation of carbonate precipitates (Kaszuba et al., 2005). The concentrations of sodium ions (Na+), potassium ions (K+), calcium ions (Ca2+), and magnesium ions (Mg2+) in the solution before and after the reaction were determined using a Thermo ICS-1100 ion chromatograph (analytical precision: 10−6 g). The calcite and sandstone powders and sandstone thin slices were washed 3–5 times using deionized water and then dried in a heating oven (55 ± 1℃, 72 h). The dried calcite and sandstone powders and sandstone thin slices were then weighed to determine the mass loss of each sample after the reaction. Subsequently, based on the volume of the reaction solution, the mass loss of each sample was converted to the dissolved amount (g/l), i.e. the mass of the minerals dissolved in each liter of the solution was used to reflect the intensity of dissolution
Results
Calcite–CO2–saline water system
Experimental results of the dissolution of calcite in the 3 m NaCl and 1 m CaCl2–2 m NaCl solutions.
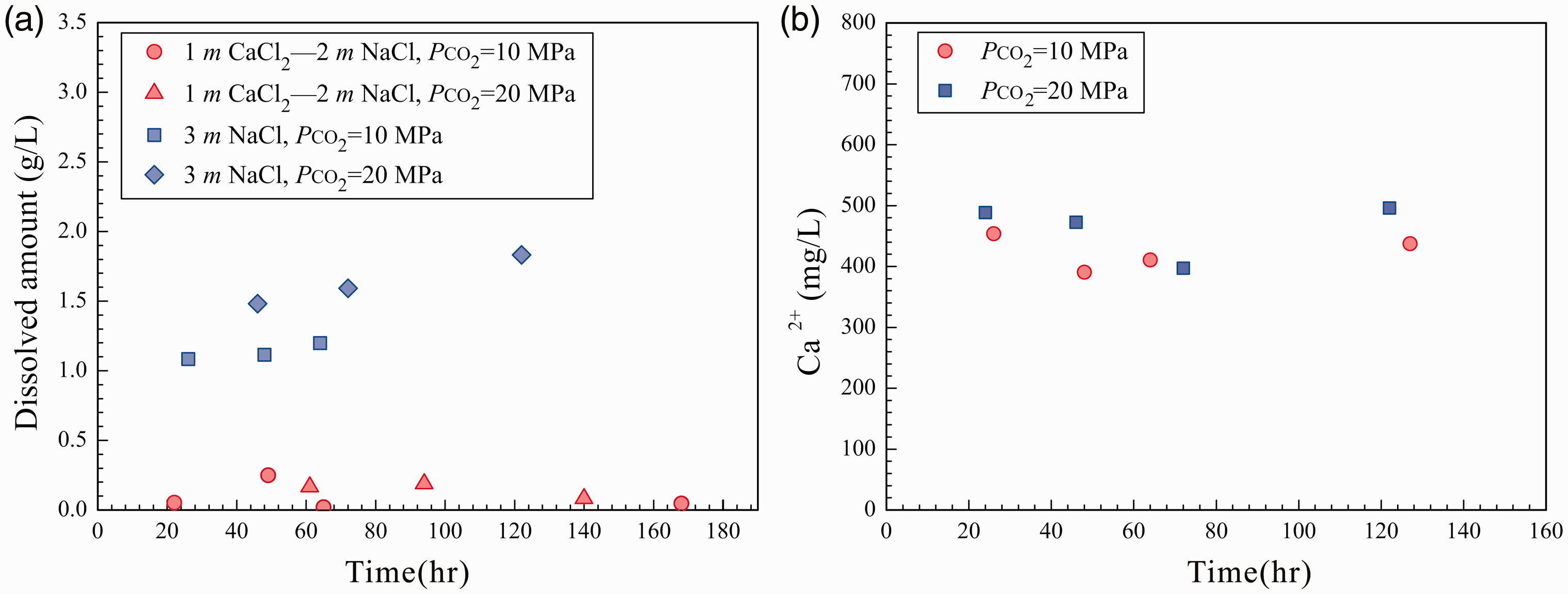
Dissolution of calcite in 3 m NaCl and 1 m CaCl2–2 m NaCl solutions at 85 ℃ and 10, 20 MPa (a) weighing results expressed by dissolved amount; (b) IC analysis results for 3 m NaCl solution. IC: ion chromatography.
At the same P
Sandstone–CO2–saline water system
Weighing and IC analyses
Experimental results of the dissolution of sandstone samples in the 3 m NaCl solution.
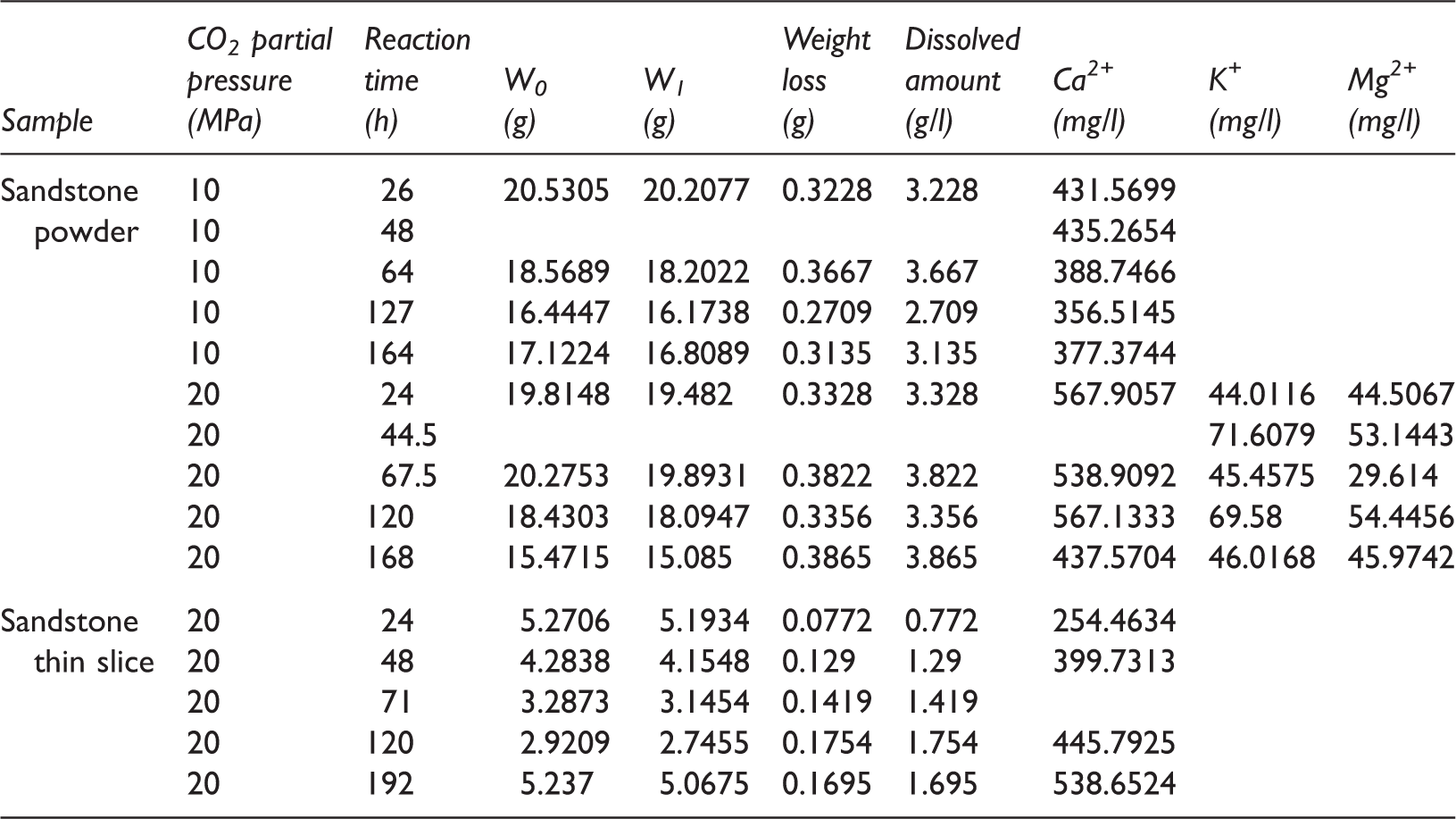
Dissolution of sandstone powders and sandstone thin slices in 3 m NaCl solution (a) weighing results and (b) IC analysis results. IC: ion chromatography.
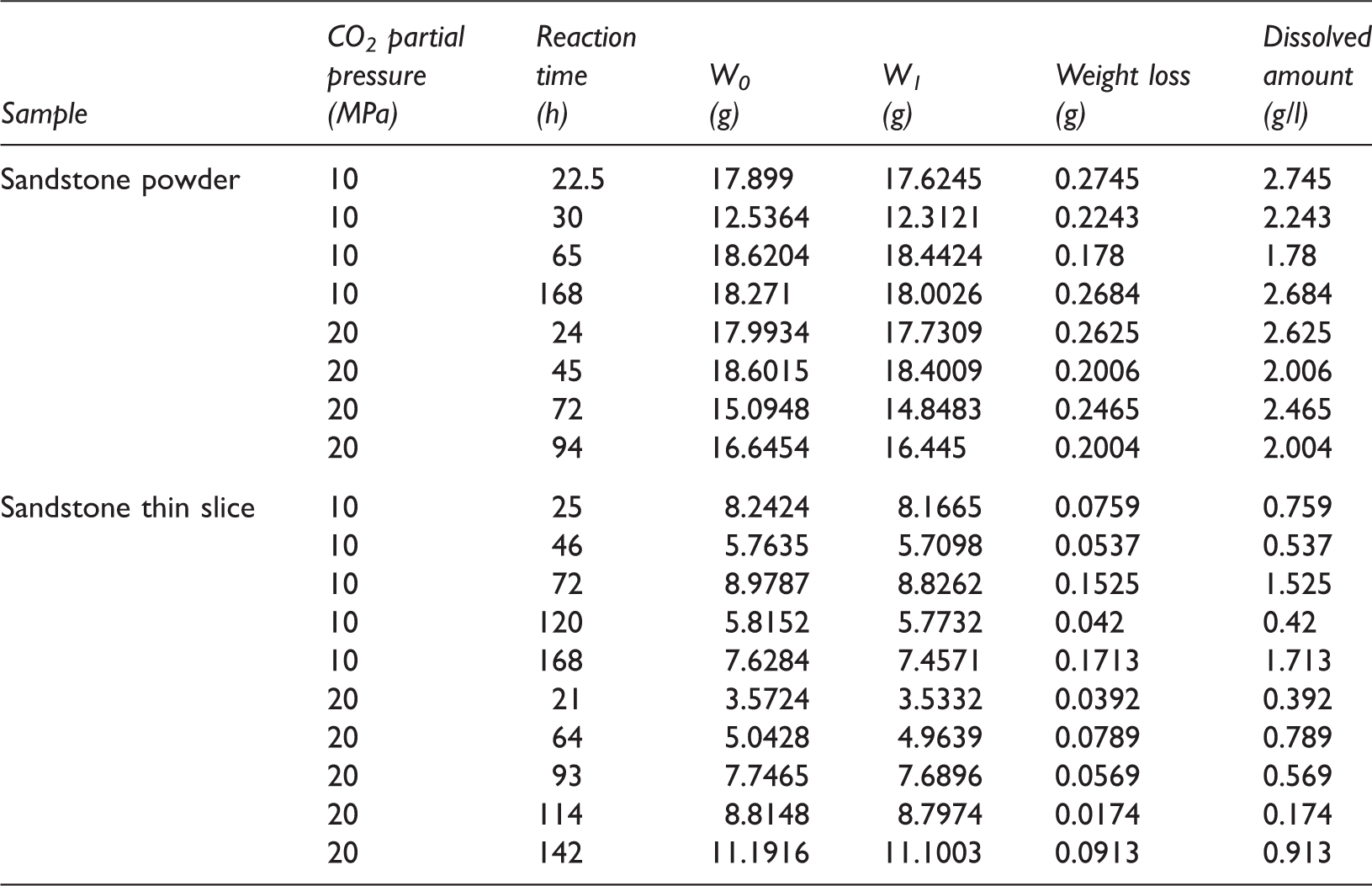
Compared to the sandstone powder, the contact area between the sandstone thin slices and the solution was relatively small. Therefore, although the dissolved amounts of the sandstone thin slices continuously increased with increasing reaction time, the dissolution rate gradually decreased. In addition, the dissolution equilibrium could not be reached within a short period of reaction time (Table 2 and Figure 5(a)). At P
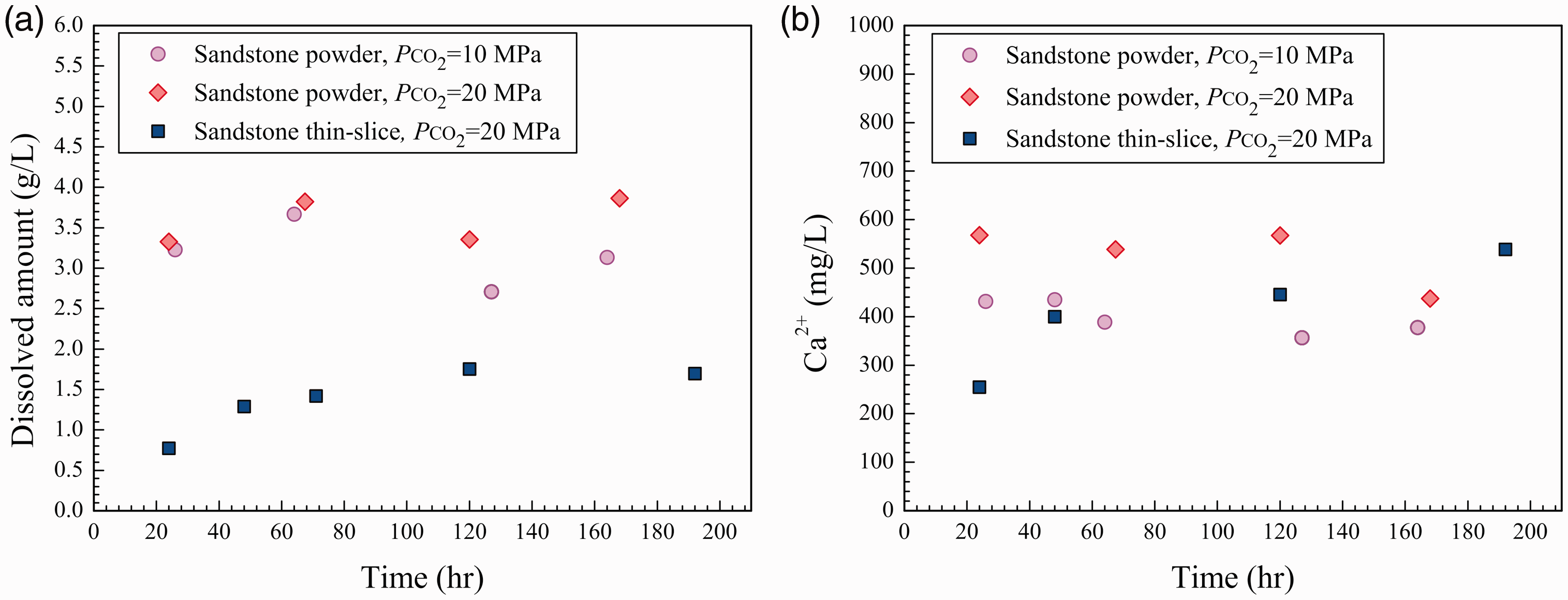
Experimental results of the dissolution of sandstone samples in the 1 m CaCl2–2 m NaCl solution.
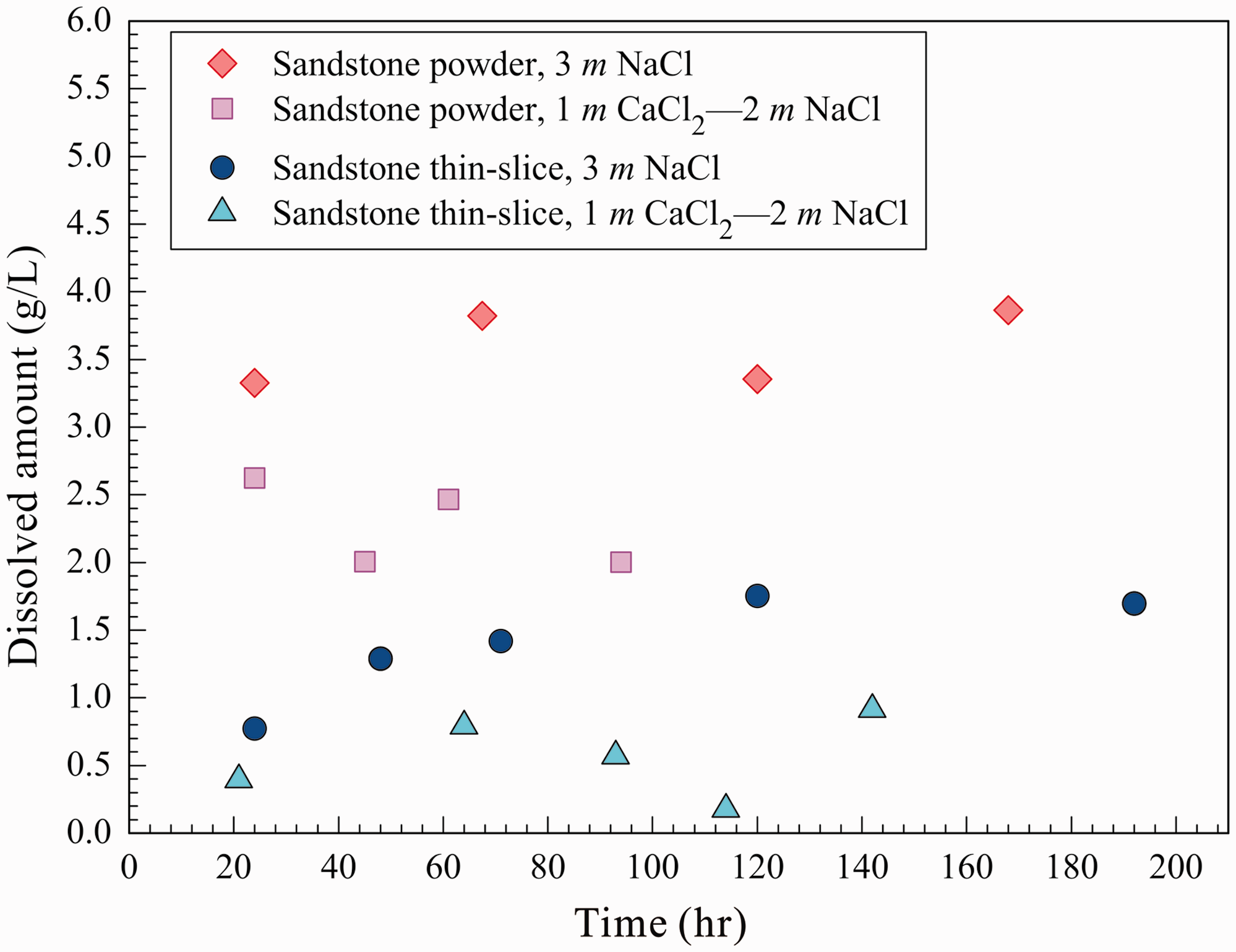
Dissolution of sandstone powder and thin slice in the 3 m NaCl and 1 m CaCl2–2 m NaCl solutions at PCO2 = 20 MPa.
IC analyses showed that, in addition to Ca2+, K+ and Mg2+ also dissolved from sandstone. For example, at P Concentration of ions that dissolved from sandstone powder in the 3 m NaCl solution ((a) concentration of K+ and (b) concentration of Mg2+).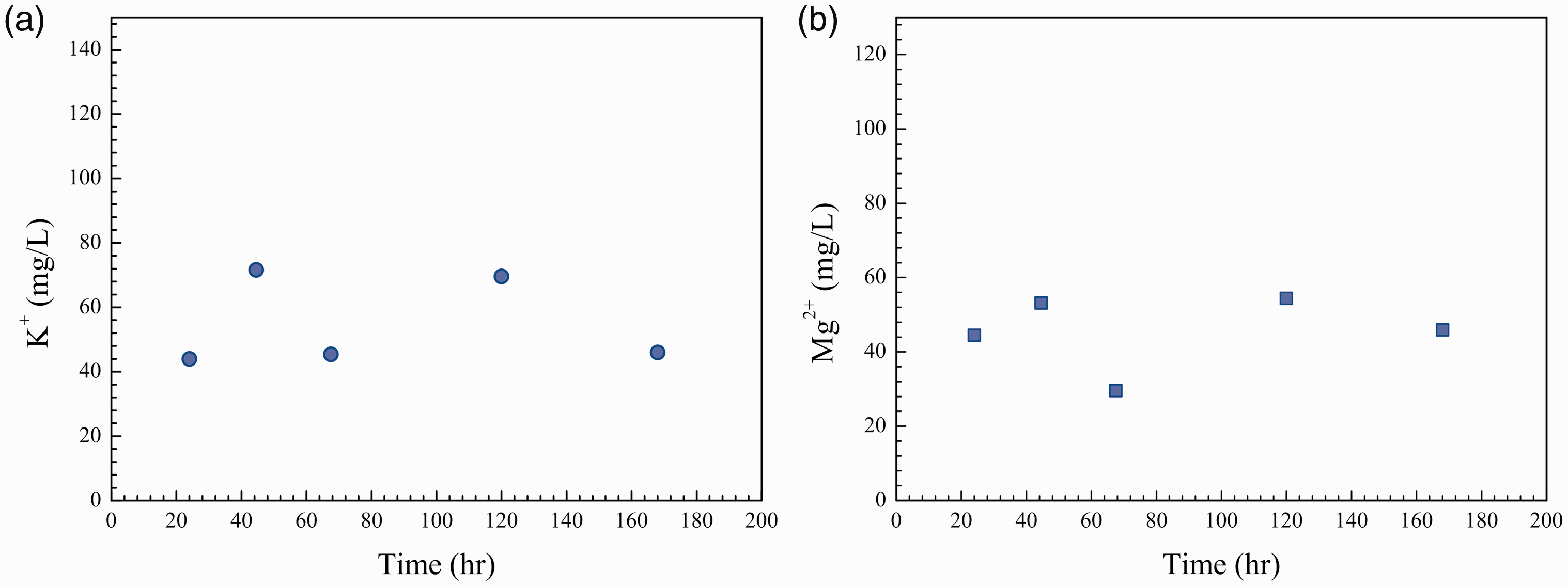
Scanning electron microscopy (SEM) observations
To further investigate the changes in the mineral morphology and porosity during the water–rock interaction, the surfaces of sandstone thin slices were observed using SEM before and after the reaction. The results showed that the calcite cements exhibited nice crystalline form before the water–rock interaction (Figure 8(a) and (b)); the crystal surface was smooth and clean. However, the calcite was intensely dissolved in the 3 m NaCl solution. For example, at P SEM images of calcite before and after dissolution in the 3 m NaCl and 1 m CaCl2–2 m NaCl solutions when PCO2 = 20 MPa. (a, b) Calcite cements before the reaction, (c) and (d) are calcites reacted in the 3 m NaCl solution for 25 and 72 h, respectively, and (e) and (f) are the calcites reacted in the 1 m CaCl2–2 m NaCl solution for 21 and 114 h, respectively. Inserted are the EDS spectra for calcite. EDS: energy dispersive spectrometer; SEM: scanning electron microscopy.
The dissolution of K-feldspar was also observed. The K-feldspar had smooth and clean surfaces before the reaction (Figure 9(a)). However, after the reaction in the 3 m NaCl solution, the K-feldspar was dissolved by the acidic CO2-rich fluid, and many pits formed on its surface (P SEM images of sandstone thin slices in the 3 m NaCl solution when PCO2 = 20 MPa (a) K-feldspar before dissolution, (b) K-feldspar after dissolution, (c) albite before dissolution, and (d) albite after dissolution. Inserted are EDS spectra for K-feldspar and albite. EDS: energy dispersive spectrometer; SEM: scanning electron microscopy.
Discussion
Processes and mechanism of CO2-rich saline water–sandstone interaction
Previous studies reported that carbonate minerals dissolve relatively quickly in an acidic CO2-rich fluid and that reaction equilibrium can be reached within several months or even several weeks (Hangx et al., 2013; Marbler et al., 2013). Miller (1952) found that the dissolution rate of calcite increased rapidly when CO2 was injected into the CaCO3–water system and then gradually decreased after 1 h, and the CaCO3–CO2–water system could reach complete equilibrium within several hours. The experimental results of this study show that the dissolution of calcite in the CO2-rich formation water at the temperature and pressure conditions of a petroleum reservoir could reach equilibrium within 1–2 days. The solubility of CaCO3 is generally believed to increase with increasing P
The chemical composition of the reaction solution also had an obvious impact on the dissolution of sandstone. The inhibiting effect of Ca2+ on the dissolution of calcite could be used to explain why the dissolved amount of sandstone in the 1 m CaCl2–2 m NaCl solution was lower than that in the 3 m NaCl solution. However, the dissolution of silicate minerals such as feldspar was nearly unaffected by the inhibiting effect of Ca2+. The reaction rate was largely controlled by the external surface area of the sample (Fan et al., 2007). Under the same conditions, the amount of dissolved sandstone powder was significantly higher than that of sandstone thin slices, but there was no significant difference between the concentrations of Ca2+ that dissolved from the sandstone powder and thin slices. The time that was required for the reaction to reach equilibrium varies among different minerals. Silicate minerals, such as feldspar, require more time to reach reaction equilibrium than carbonate minerals (Rathnaweera et al., 2016). Due to the relatively smaller surface areas of the sandstone thin slices, the dissolution of carbonate minerals could reach equilibrium quickly, but the silicate minerals were only dissolved slightly; the dissolution of silicate minerals did not reach equilibrium. As for the sandstone powder, its dissolution rate was significantly increased compared with that of the sandstone thin slice because the sandstone powder was in full contact with the reaction solution. Then, the reaction reached equilibrium relatively quickly. The dissolution process of the sandstone thin slices is very similar to the actual geological process; under geological conditions, the sandstone–CO2–saline water interaction is slow. This result also indicates that the water–rock interaction is favored in porous stratum.
CO2 was reported to exist in two forms after it is injected into the formation water: (1) interacting with the reservoir rocks and forming secondary minerals; and (2) dissolving in the formation water (Gunter et al., 2000). Whether carbonate precipitates can form after CO2 is injected into formation water is mainly determined by the reservoir lithology (Xu et al., 2004). For example, Wigand et al. (2008) investigated water–rock interactions in the sandstone–CO2–NaCl–H2O system and found that the dolomite cement dissolved, but no secondary carbonate minerals formed. They believed that the formation of carbonate precipitates reported in previous simulation experiments was in close association with the dissolution of Ca-rich minerals such as anorthite and gypsum. The dissolution of these Ca-rich minerals provided a large amount of Ca2+ for the precipitation of carbonate minerals. The sandstone samples that were used by Wigand et al. (2008) did not contain anorthite and gypsum, and no other minerals could provide an excessive amount of Ca2+. Therefore, if the Na- and/or K-silicate minerals are the main silicates in a sandstone reservoir, the injection of CO2 into the reservoir will not result in the generation of carbonate precipitates, and the injected CO2 will exist mainly in the form of dissolved CO2 (Emberley et al., 2004). However, our experimental results suggest that even if there is a relatively high concentration of Ca2+ in the formation water (i.e. 1 m CaCl2), the injection of CO2 will still result in the dissolution of carbonate minerals. Of course, the relatively high concentration of Ca2+ in the formation water will prevent the dissolution of calcite, indicating that the acidic fluid that forms after CO2 is injected into CaCl2-type formation water has a limited dissolution effect on carbonate minerals (e.g. calcite) and thus cannot significantly increase the storage space in the reservoir. CO2 injection can induce the dissolution of carbonate minerals because H+ ions are generated from the ionization of the H2CO3 that forms from CO2 and water. This process can decrease the pH of the formation water. PHREEQC simulation results (Tables S1 and S2) showed that the pH of both reaction solutions decreased with increasing P
The dissolution of K-feldspar was observed, whereas albite appeared to be resistant to the acidic solutions during the experiment. To investigate the factors controlling the dissolution behaviors of these two silicate minerals, the dissolution of the major minerals of the reservoir rocks in 3 m NaCl solution was simulated using PHREEQC software (Table S3). The simulation conditions were as follows: T = 85℃; P
Implications for CO2-EOR and CO2 sequestration
Previous studies and the PHREEQC simulation results (Tables S1 and S2) indicated that the injection of CO2 into a reservoir will cause a significant decrease in the pH of the formation water (Carroll et al., 2014; Kaszuba and Janecky, 2009; Rosenbauer et al., 2005; Yu et al., 2015). The acidic CO2-rich fluid that forms from the CO2 injection will dissolve minerals such as carbonates and silicates. This process results in the formation of secondary pores and changes in the pore structure, which can improve the physical properties of the reservoir. Our experimental results are consistent with those documented in Farquhar et al. (2015), where the dissolution of carbonates and K-feldspar was suggested to be the major result of the interaction between CO2-rich acidic fluid and sandstone. The carbonate cements in the sandstone reservoir rocks dissolved in the acidic CO2-rich fluid, which increased the permeability of the reservoir (Ross et al., 1982; Sayegh et al., 1990). Geological observations also indicate that the occurrence or dissolution of carbonate cement is vital to the physical properties of sandstone reservoirs. The Huangqiao region in the North Jiangsu Basin is a well-known CO2 gas field. The interaction between the formation water rich in mantle-derived CO2 and the feldspathic quartz sandstone in the Permian Longtan Formation resulted in the dissolution of minerals such as calcite and feldspar, which increased the porosity and permeability of the reservoir. For example, the Longtan Formation in the Xi-2 well is relatively insignificantly affected by the CO2-rich fluid. Its sandstone section has an average porosity of 6.91% and an average permeability of 0.958 md, making this section an ultralow to low porosity and ultralow permeability reservoir. In contrast, the Longtan Formation in the Xi-3 well is significantly affected by the CO2-rich fluid and has an average porosity of 9.92% and an average permeability of 128.017 md. Thus, the physical properties of the Longtan Formation reservoir in the Xi-3 well are much better than those in the Xi-2 well. The porosity and permeability of the reservoir rocks of the Kela-2 Gas Field in the Kuche Depression in the Tarim Basin (NW China) are negatively correlated with the amount of carbonate cements; the greater the amount of carbonates, the poorer the physical properties of the reservoir (Yu and Lai, 2006). During investigation of the distribution of the carbonate cements in the Paleogene Zhuhai Formation reservoirs in the Wenchang A Sag in the Pearl River Mouth Basin, You et al. (2012) found that when the content of the carbonate cement was greater than 5%, the porosity and permeability decreased with the increasing content of carbonate cement. Carbonate cementation was suggested to be a major cause of the deterioration in the physical properties of the deep Zhuhai Formation reservoir. You et al. (2012) also thought that the physical properties of the reservoir were significantly improved through the dissolution of carbonate cements by diagenetic acidic fluid. In summary, the dissolution of alkaline minerals, such as carbonate and feldspar, in a sandstone reservoir by a CO2-rich fluid is favorable to the development of secondary pores. Dissolution of these alkaline minerals, especially the carbonate cements, can improve the physical properties of the reservoir and is favorable for the transport and extraction of oil and gas during the CO2-EOR process.
Storing dissolved CO2 in a trap is an important means of CO2 sequestration. In a reservoir, the dissolution of minerals such as calcite in a CO2-rich fluid can improve the porosity and permeability of the reservoir, which facilitates the transport and storage of CO2 in the stratum (Lamy-Chappuis et al., 2014). When evaluating a CO2 sequestration site, the lithology and the chemical compositions of the formation water should be considered. For example, under the same conditions, more sandstone dissolved in the NaCl-dominated solution than in the CaCl2-dominated solution. If the formation water is rich in CaCl2 (e.g. CaCl2 type), the effect of mineral dissolution on the reservoir properties is limited because the high concentration of Ca2+ prevents the dissolution of calcite. Therefore, sandstone formations in which NaCl is the major component of the formation water are preferred for CO2 geological sequestration. In addition, after a CO2-rich fluid dissolves the alkaline minerals (e.g. carbonates) in a reservoir, dissolution pathways may form and lead to the transportation of the CO2-rich solution. When CO2-rich solution is transported to a fault or a fault zone, CO2 will escape from the solution due to a sudden decrease in pressure. Then, the pH of the solution will increase, resulting in the precipitation of alkaline minerals such as carbonates in the fissures. This process can seal the fissures (Zhu et al., 2012), block the dominant migration pathways, and prevent further CO2 leakage (Shukla et al., 2010). CO2 will also be transported to the region below the caprocks of the reservoir and then start to accumulate in this region due to buoyancy. If the caprocks are somewhat permeable, the abnormally high local pressure that results from the continuous accumulation of CO2 can be mitigated (Smith et al., 2012). Keating et al. (2014) thought that a certain level of CO2 leakage did not affect the effectiveness of CO2 sequestration and the water quality of the overlying aquifer. However, if the caprocks contain minerals that can easily be dissolved by a CO2-rich fluid, such as calcite and K-feldspar, long-term water–rock reactions will result in a gradual increase of the permeability of the caprocks and a decrease in the compressive strength of the rocks (Zheng et al., 2015), which could reduce the sealing capacity of the caprocks. Consequently, CO2 will leak and flow into the overlying shallow freshwater aquifer and even return to the ground (Xu et al., 2011). In addition to CO2 leakage, deep saline water will also flow into the shallow rock layers. Then, the salts, organic matter, and trace metals that are carried by the deep saline water will severely pollute the freshwater aquifer (Jung et al., 2013; Keating et al., 2012). In summary, dissolution of the alkaline minerals in the reservoir rocks by CO2-rich acidic formation water is favorable to CO2-EOR and CO2 geological sequestration, but it is also necessary to investigate the changes in the sealing capacity of the caprocks after the injection of CO2 and assess the risk of CO2 leakage.
Conclusions
Using calcite and carbonate-cement-bearing sandstone as the experimental materials, we performed CO2-rich fluid–rock interaction experiments at the reservoir temperature (85℃). Combined with PHREEQC simulations, the effects of the sample surface area, the P The CO2 injection resulted in a significant decrease in the pH of the formation water, which induced the dissolution of alkaline minerals such as carbonate cements and K-feldspar. The type of formation water showed a significant impact on the dissolution of carbonate minerals such as calcite. The dissolution of calcite was significantly inhibited in the CaCl2-type formation water. During the experiment, the amount of sandstone that dissolved was directly proportional to the surface area of the sample and increased with increasing P Under the experimental conditions, dissolution of the alkaline minerals in the reservoir rocks was the major result of the interaction between the CO2-rich fluid and the reservoir rock, indicating that the injection of CO2 into a stratum will improve the porosity and permeability of the reservoir and facilitate the progression of the CO2-EOR process. In addition, increases in the porosity and permeability of a reservoir lead to an increase in the capacity of the reservoir to store CO2. However, CO2 leakage will result in an increase in the pH of the formation water and the precipitation of minerals such as carbonates, which may block the CO2-EOR pathways. If the caprocks contain alkaline minerals such as calcite, CO2-rich formation water will dissolve these minerals. This process may result in an increase in the porosity and permeability of the caprocks and affect their sealing performance, thereby inducing the leakage of CO2 and formation water. Consequently, the effectiveness of CO2 sequestration will be weakened, and shallow freshwater aquifers may be polluted. Future research should further assess the effect of CO2-rich fluids on the sealing capacity of the caprocks of reservoirs.
Footnotes
Acknowledgements
We thank Mr Haihong Cao from SINOPEC for his assistance during the high P–T experiments of CO2-rich water–rock interaction. We also thank Prof. Yuzhuang Sun for his handling this manuscript, and Dr Pedram Masoudi and an anonymous referee for their careful review and constructive comments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Grant nos. 41230312 and 41573054) and the National Science and Technology Major Project of China (Grant no. 2016ZX05002-006-005).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.