Abstract
When enhancing coalbed methane recovery using CO2 or N2 injection, injected gas flows into coal matrix by diffusion. Gas diffusion velocity varies, depending on gas molecular size and pore geometry which causes different sorption rates of the gas in coal seam. In this aspect, this study provides the fundamental reason for the reduction in gas permeability through cleats and methane recovery during enhanced coalbed methane (ECBM) processes. This reduction occurs not only because of the sorption affinity as reported in previous works, but also because of the characteristics of gas diffusional flow which this study attempted to examine experimentally. From the results obtained by diffusional flow experiment, diffusion coefficient is no longer increased at high pressure. Although CO2 injection rate is very high, a large amount of CO2 moves through cleat instead of adsorbed in matrix, which causes early CO2 breakthrough. In ECBM, N2 mostly acts as a displacing agent of methane, because co-diffusion of N2 with methane is more dominant than counter-diffusion owing to its extremely low adsorption affinity. On the other hand, CO2 is rapidly adsorbed due to its fast increasing rate of diffusion coefficient with pressure increase. Consequently, CO2 permeability is greatly reduced at the beginning of injection and ultimately becomes the lowest value at the maximum adsorption pressure. Also, delayed methane recovery by fast diffusion and high adsorption affinity of CO2 occurs accordingly. This study confirms that the CO2–N2 mixed gas injection is advisable comparing to only injecting CO2 to pursue the prevention of CO2 injectivity reduction and enhanced methane recovery, simultaneously.
Introduction
Previous researches on enhanced coalbed methane recovery are mainly composed of analysis on adsorption characteristics of sorbed gas to coal particles and gas diffusion in coal seam (Shi and Durucan, 2008; Sung et al., 2000; Wei et al., 2007). When injected gas flows into micropore in matrix through cleat by diffusional flow, the matrix swells depending on the magnitude of adsorption affinity onto coal particles, and this causes reduction of cleat permeability (Kim et al., 2012). Shi and Durucan (2005) proposed an improved model on Gray’s model first quantifying the permeability change caused by matrix swelling in accordance with gas adsorption, and Palmer and Mansoori model most commonly used to describe the permeability of coal seam in coalbed methane. They intended to describe the permeability change more accurately by applying the new dynamic permeability model consisted through a direct link between the amount of desorbed gas and volumetric matrix strain. CO2 has the highest adsorption affinity among CO2, CH4, and N2, which are adsorbed gas to coal, but CO2 injectivity is decreased by the highest reduction in cleat permeability resulting from matrix swelling (Busch and Gensterblum, 2011; Chen et al., 2009; Guo et al., 2008). Thus, mixed gas injection that CO2 to be injected is mixed with N2 having the lowest adsorption affinity is proposed as a way to supplement the problem of efficiency reduction in CO2 injection (Seto et al., 2006; Zhou et al., 2011; Zhu et al., 2002). Lin et al. (2008) conducted experiments of permeability alteration by injecting pure CO2, CH4, and N2 gas, and various mixing ratios of CO2–N2 mixed gas using crushed coal sample. They ascertained that the higher content of N2 in the gas mixture, the less permeability reduction occurred through calculating the permeability reduction by adsorption with pressure increase. In the research conducted by Kumar et al. (2012, 2015) and Wang et al. (2012), they intended to investigate permeability evolution more accurately in sorbing materials of coal and shale through observing permeability change by gas adsorption and external stress. Shale is also relevant to sorbing media with methane as adsorbed state like coal. Thus, according to the research by Kumar et al. (2010, 2016), shale is a promising area because of geological sequestration of carbon dioxide, but permeability reduction is very small than that of coal due to adsorption characteristic of matrix.
The analysis of diffusional flow as well as characteristic of CO2 adsorption to coal is extremely important to enhance methane recovery (Kwon et al., 2001). The diffusional flow in matrix of coal seam is largely influenced by gas molecular size, pore geometry, and pore diameter. In particular, the process of methane production can be influenced by the pore structure (Sun et al., 2015). Shi and Durucan (2003) tried to interpret the flow mechanism of methane by CO2 injection through CO2–CH4 displacement flow modeling applying bidisperse model composed of micropore and macropore. They demonstrated that uniform pore structure of unipore model did not accurately describe the behavior of gas diffusion in matrix of coal seam. Fathi and Akkutlu (2008) observed that the surface diffusion in the micropore is important mechanism in ECBM by injecting CO2 through numerical modeling on counter-diffusion and competitive adsorption with methane during CO2 injection by separating the pore within coal seam into micropore, macropore, and fracture. In addition, Cui et al. (2004) proposed that for CO2, it is possible to diffuse more fine pore in a complex system of micropores in coal seam because kinetic diameter affecting the diffusional flow of gas molecules is 0.33 nm, it is relatively small compared to 0.36 nm for N2 and 0.38 nm for methane.
Jessen et al. (2008) analyzed the effect of enhanced methane recovery by N2 injection using crushed coal. They compared results in experiment of displacement flow with those of simulation using pure CO2 and CO2–N2 mixture. Mazumder et al. (2008) conducted displacement experiment by using coal core sample in order to preserve the characteristics of heterogeneity and dual porosity in coal. They tried to simulate the process of methane displacement more actually, when pure CO2 or CO2–N2 mixture was injected through the analysis reflecting effects of competitive adsorption, multiphase flow, and geomechanics. Schepers et al. (2010) analyzed the aspect of displacement flow caused by difference in adsorption affinity for each gas composition during CO2–N2 mixed gas injection in coal seam and aimed to calculate the optimum mixed ratio of injection gas for different coal rank.
As described above, CO2 can be displaced with methane effectively due to its high adsorption affinity has been proved through several experiments and simulation studies as previous works. The injection of CO2–N2 mixed gas is presented to supplement permeability reduction of cleat due to high adsorption affinity of CO2 as well. However, very few studies have been conducted to apply the effect of diffusion characteristics of each sorbed gas on displacement flow behavior experimentally.
Accordingly, in this study, the experimental apparatus was set up to analyze enhanced methane recovery as shown in Figure 1, and we performed experiments of diffusional flow using CO2, CH4, and N2. The analysis of methane recovery was executed through experiments on permeability change and methane displacement for various injection condition of pure CO2 and N2, and CO2–N2 mixture. Ultimately, we investigated the effect of CO2–N2 injection considering gas diffusion phenomenon on enhancement of methane recovery.
Experimental apparatus for analysis of enhanced methane recovery.
Coal sample preparation
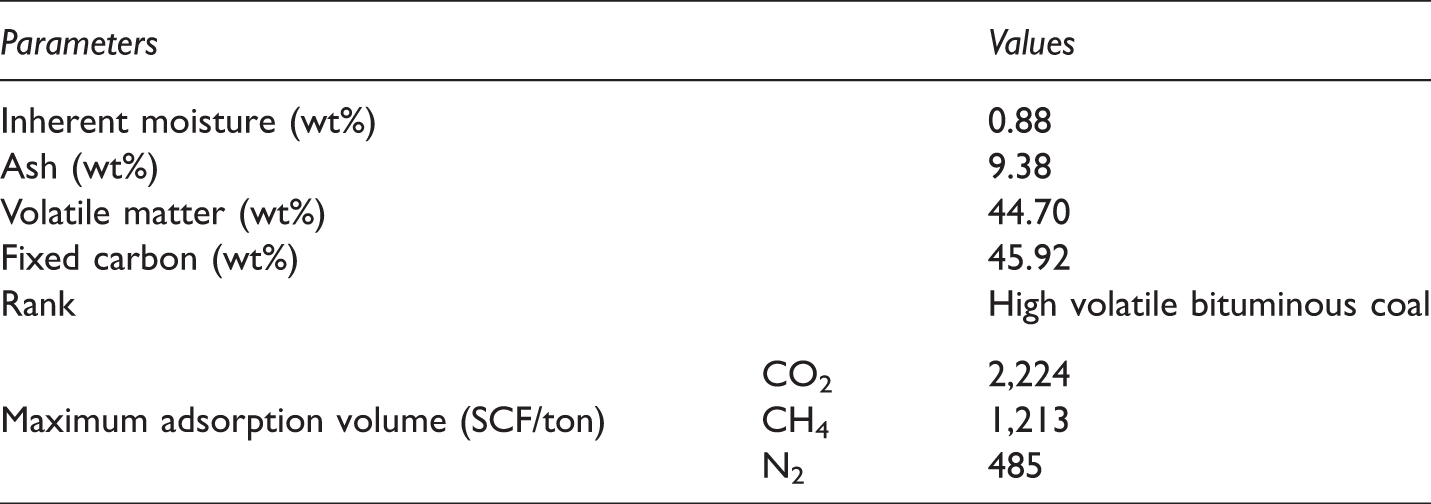
The results of proximate analysis and measured Langmuir constant (VL).
Experimental apparatus and procedures
Diffusional flow

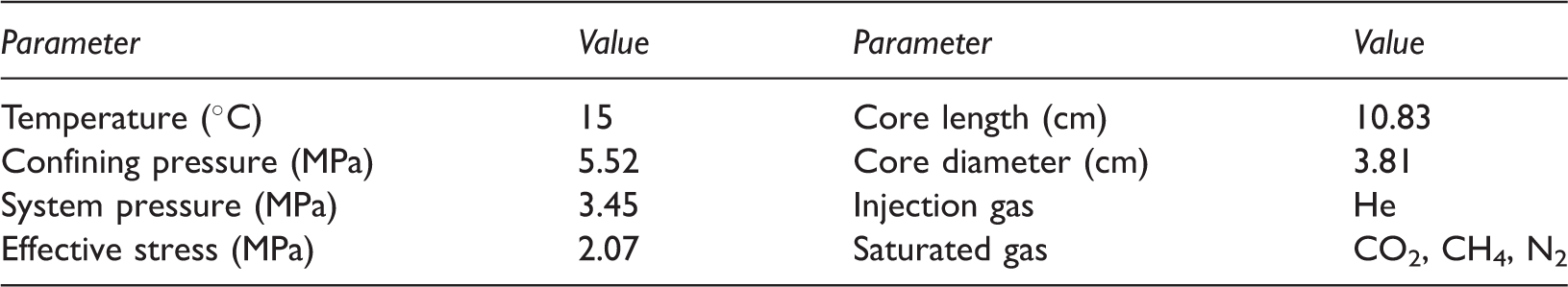
Experimental conditions for diffusion coefficient using coal core.
Additionally, initial and boundary conditions applied to the modeling are as follows:
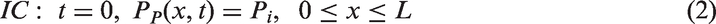
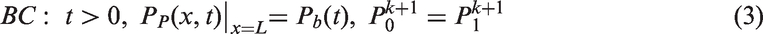
Gas displacement
Experimental conditions for methane displacement by gas injection.
Coal permeability
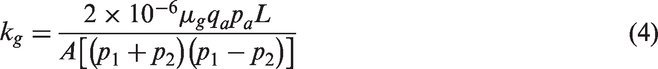
Experimental conditions of permeability change for coal core.
Results and discussion
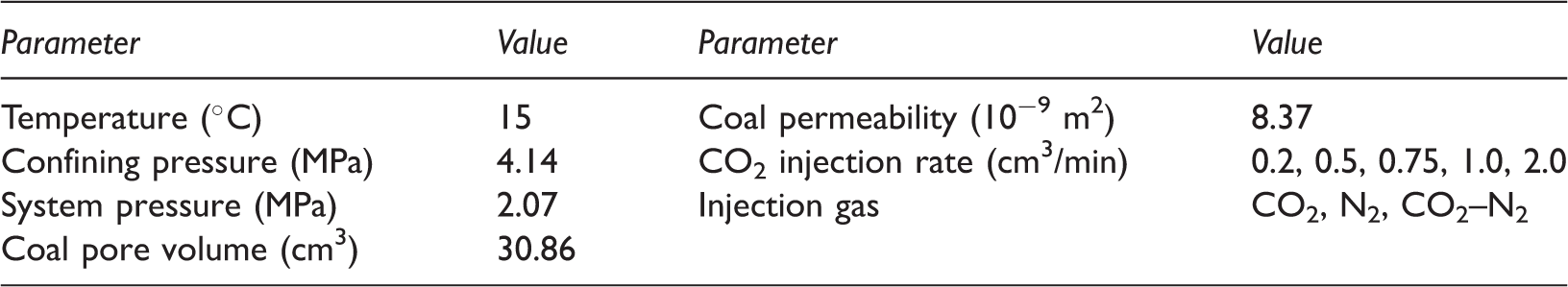
Effect of CO2 injection rate on diffusional flow
Calculation of diffusion coefficient
Helium, a non-adsorbing gas, was injected to the coal sample saturated with sorbed gas including CO2, CH4, or N2 at 3.45 MPa of pressure. Helium was injected at the constant pressure of 3.45 MPa through the inlet. At the outlet that means the boundary condition in the numerical model, the measured gas partial pressure was set to the last cell. The calculated optimum diffusion coefficient was determined by numerical matching of the estimated gas partial pressure in the last cell, which was calculated with a random diffusion coefficient, and the measured gas partial pressure (Seomoon et al., 2015).
Figure 2 shows the results of diffusion coefficient with increase of the mean partial pressure for each sorbed gas. Li et al. (2013) and Zhao (1991) showed that diffusion coefficients were increased non-linearly with increase in gas partial pressure due to change in pore size caused by gas adsorption and desorption using CH4 and CO2, respectively. The results of diffusional flow experiment using CO2, CH4, and N2 in this study were of similar pattern to those of the references above mentioned as to change in diffusion coefficient. Because gas diffusion in coal seam has specificity which is caused by sorption-induced concentration gradient into coal particles of matrix, the increasing rate of diffusion coefficient for CO2 was calculated as 15.53% which was about 1.7 times higher than those of CH4 and N2. The diffusion coefficient, however, has a certain value determined by gas molecular size and pore structure. Although CO2 injection rate is increased, diffusion velocity is limited, thus remaining CO2 is moved into cleat and it causes early CO2 breakthrough (BT). Accordingly, experiments were performed judging from that change of diffusion coefficient affects CH4 recovery which is calculated differently depending on CO2 injection rate.
The results of diffusion coefficient with mean partial pressure of CO2, CH4, and N2.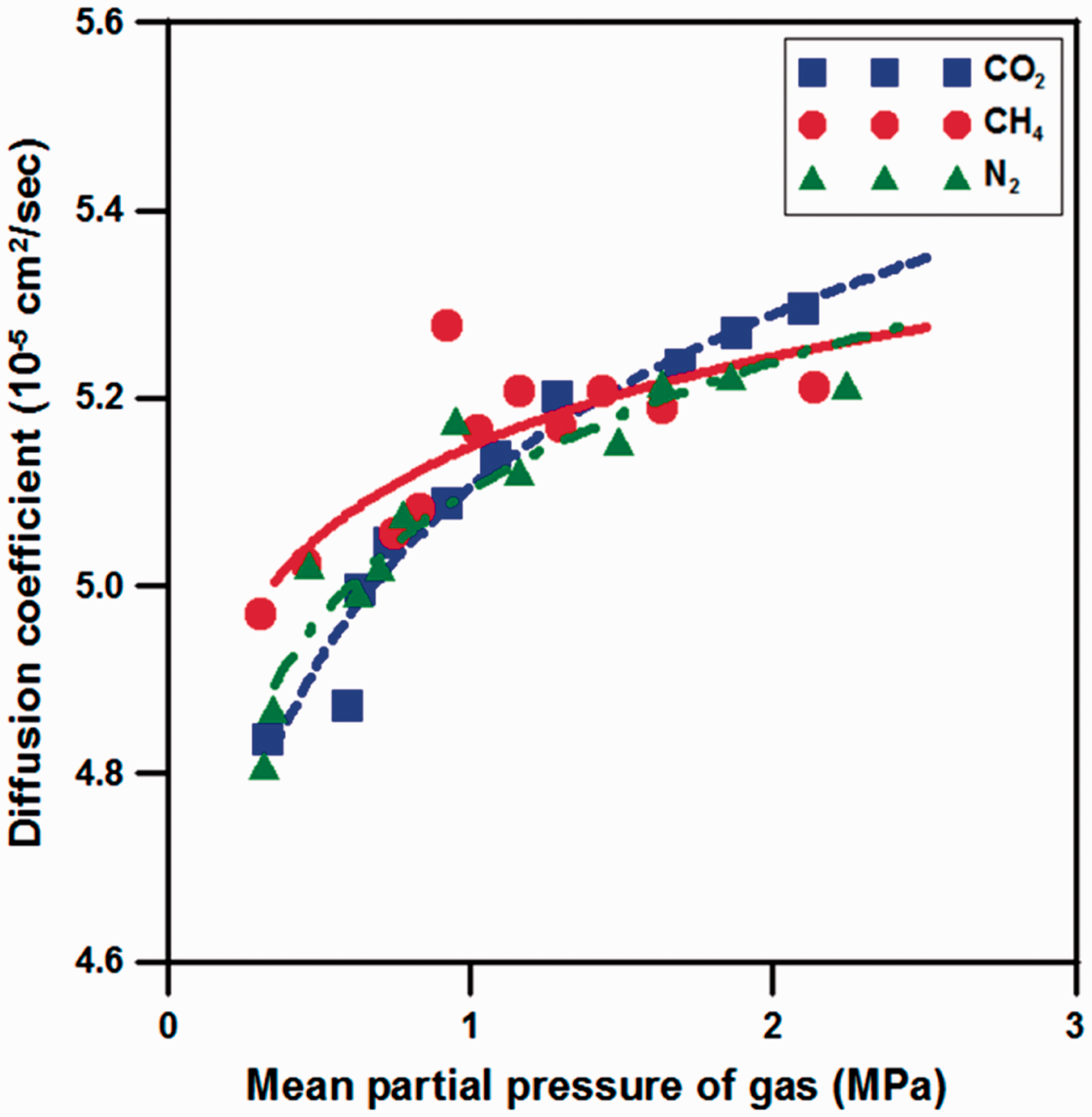
Diffusional flow in CO2-ECBM
As shown in Figure 3, for CO2 injection at a rate 0.2 and 0.5 cm3/min, CO2 BT occurred about 4.6 and 2.1 PVI, respectively. This is because adsorption proceeded to micropore due to CO2 diffusion into the inside of matrix by low CO2 injection rate. As a result of increased CO2 injection rate at a 0.75 cm3/min, CO2 BT was shown at approximately 1.77 PVI, and it can be identified that CO2 BT was accelerated about 0.33 PVI than that of 0.5 cm3/min. It is judged that because CO2 injection rate was relatively higher than that of diffusion into the matrix, early CO2 BT occurred. Next, a displacement result from CO2 injection rate at 1.0 cm3/min was compared to those at previous injection rates. In this case, CO2 BT was generated about 1.18 PVI, and it represents that CO2 BT was faster about 0.59 PVI than that of 0.75 cm3/min. In addition, for significantly increased CO2 injection rate of 2.0 cm3/min, CO2 BT occurred at about 0.6 PVI, and it tends to be even faster ahead with increasing CO2 injection rate. Therefore, it is necessary to consider diffusional flow effect in estimating the optimum injection rate.
The results of methane displacement behavior for various CO2 injection rates.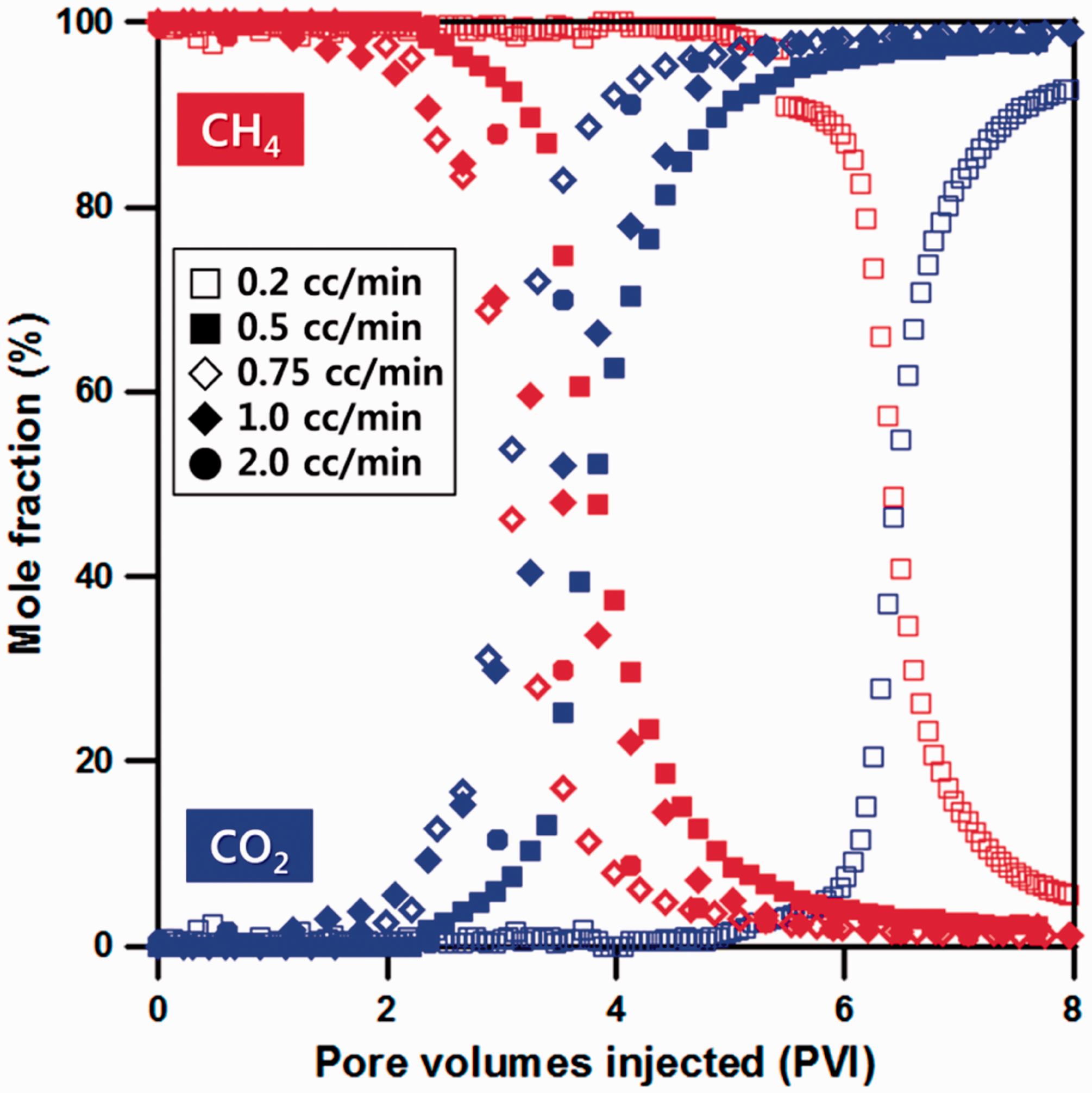
Figure 4 shows the methane recovery with increase of PVI depending on CO2 injection rate. When CO2 was injected at 0.2 cm3/min rate, most of CO2 were diffused to internal micropore, and displacement with CH4 was delayed until approximately 4.5 PVI. Thus, it took a long time to recover methane initially and exhibited extremely low displacement efficiency. In addition, for 0.5 cm3/min, it shows a tendency that quite a little amount of methane was recovered until about 0.59 PVI due to the delayed displacement phenomenon as well. Comparing the results at 0.75 cm3/min and 1.0 cm3/min, it can be confirmed that displacement efficiency was reduced at 1.0 cm3/min compared to 0.75 cm3/min due to the intensified CO2 BT. In particular, for 2.0 cm3/min, increasing rate of CH4 recovery appears to be conspicuously reduced in the early injection period than that of 0.75 or 1.0 cm3/min. Thus, an appropriate injection rate is determined as 0.75 cm3/min considering methane recovery by injection of pure CO2 gas.
The results of methane recovery for various CO2 injection rates.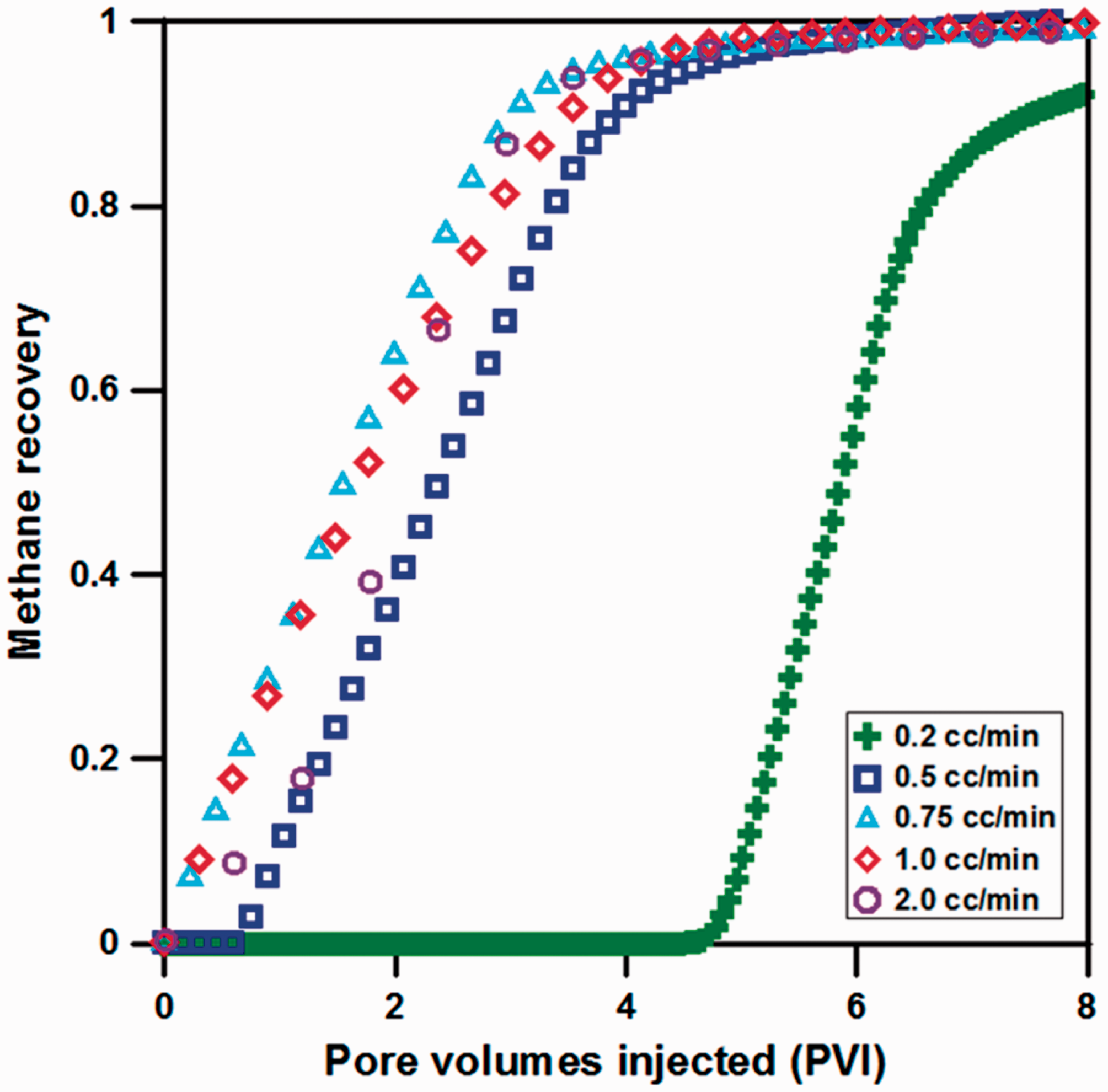
Methane displacement behavior considering diffusion characteristics
Delayed displacement by pure CO2 injection
Jessen et al. (2008) performed the displacement experiments using crushed coal sample, and the results were compared to those of the simulation. In this result, a very fast diffusion occurred due to the destroyed pore structure by using crushed coal. Also, Mazumder et al. (2008) conducted experiments for displacement analysis by the differences of adsorption affinity for each gas and moisture content using coal core, but they did not consider diffusional flow in coal core. In this study, for CO2, delayed displacement with CH4 was confirmed due to fast diffusion into the micropore and high adsorption affinity considering the diffusion phenomenon caused by concentration gradient in the preserved pore structure of the core sample. As a way to compensate the defect, through experiment of mixed gas injection including N2 which is slow diffusion than CO2 and has also low adsorption affinity, it was intended to compare with the result of pure CO2 gas injection. Therefore, CO2 BT time and methane recovery were compared on the basis of the injection rate of 0.5 cm3/min that generated delayed methane recovery by injection of pure CO2 gas.
Diffusional flow in CO2–N2 ECBM
Figure 5 shows displacement behavior of pure CO2 and N2, and various mixing ratios of CO2–N2 mixture at a rate of 0.5 cm3/min. For pure N2 gas injection, N2 BT occurred at about 1.48 PVI, and it can be ascertained that N2 increased sharply after that point. This is because co-diffusion with methane of N2 was dominant rather than displacement with methane, and it acted as a displacing agent of methane. In the analysis result of methane displacement behavior using pure CO2 with the same injection rate, CO2 BT occurred at 2.07 PVI, and it is verified that effective displacement of methane by counter-diffusion between CO2 and methane was possible. This means that CH4 recovery by pure N2 injection is a slow process, and thus, it indicates that a large amount of N2 is to be injected as compared to CO2. In addition, in the case of pure N2 injection, additional cost is needed to separate CH4 and N2 due to extremely fast BT of N2. Next, it is examined for the result of displacement experiment by using 75:25 CO2–N2 of high purity mixture. In this case, CO2 BT occurred at 2.2 PVI of mixed gas, and it is confirmed that CO2 BT time was delayed about 0.1 PVI than that of pure CO2 injection. Therefore, the CO2 BT time was somewhat delayed by enhanced methane production through N2 component included in mixture. For 50:50 CO2–N2, CO2 BT time occurred at about 2.95 PVI, and it is found that delayed effect of CO2 BT happened by reduction of CO2 ratio in mixed gas. Meanwhile, for 25:75 CO2–N2 as a rate of 0.5 cm3/min, it is identified that the increasing amount of N2 generated N2 BT rapidly, and it caused further ahead the time as displacing agent one of the important roles of N2. Furthermore, CO2 BT occurred at about 2.80 PVI of mixed gas, and it was delayed about 0.7 PVI than pure CO2 injection under the same condition. From this result, it is elucidated that co-diffusion with methane caused by low adsorption affinity of N2 in ECBM acts as a factor of enhanced displacement efficiency.
The results of CH4 displacement behavior by injecting mixed gas. (a) N2 100%, (b) CO2 100%, (c) CO2:N2 = 75:25, (d) CO2:N2 = 50:50, (e) CO2:N2 = 25:75.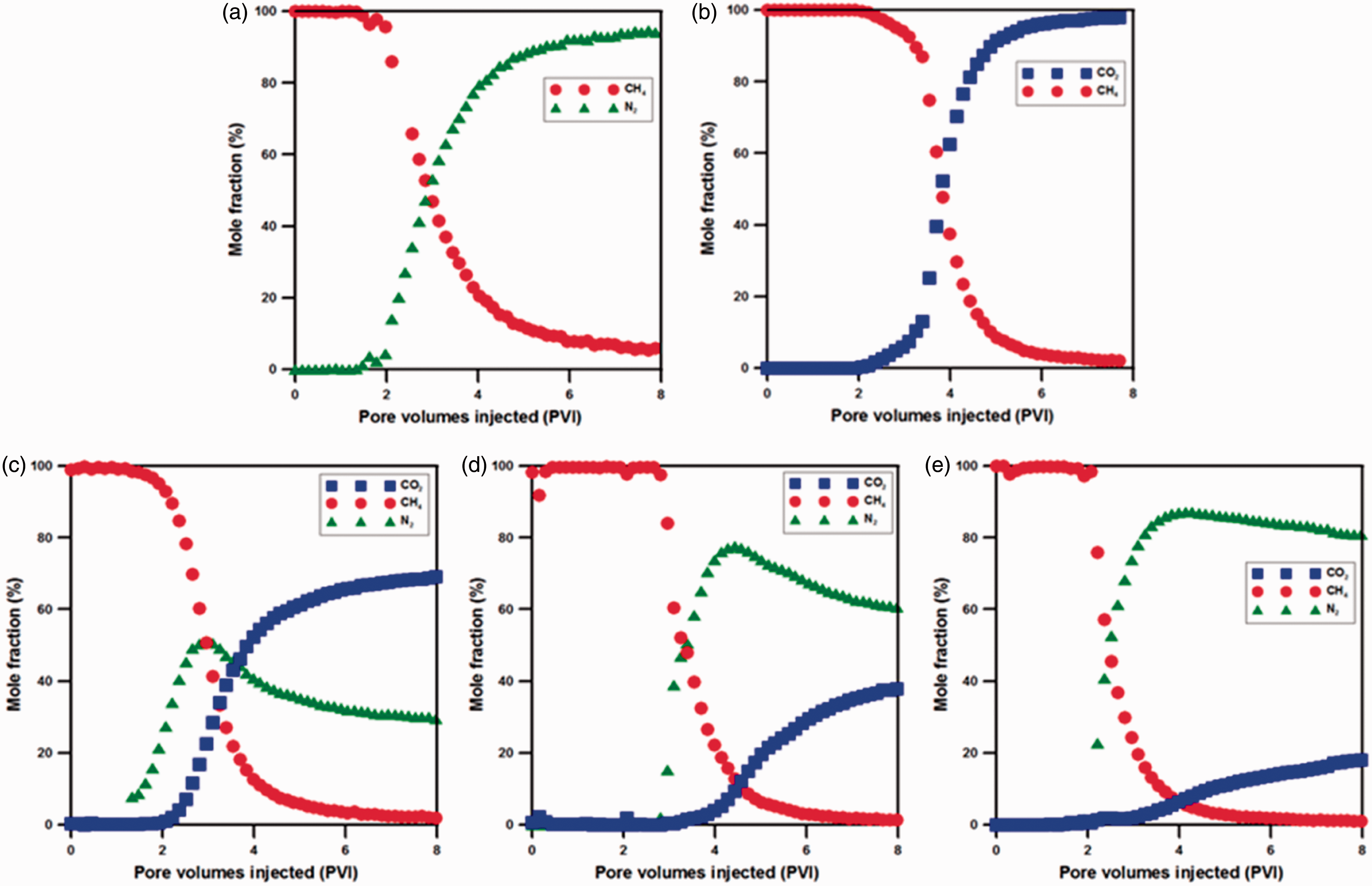
Improvement of CO2 injectivity and methane recovery
Permeability reduction considering gas diffusion
This study was based on a permeability change in the Palmer and Mansoori model represented by the sum of pore and sorption strain components (Palmer and Mansoori, 1998).
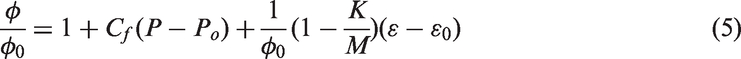
According to the previous research, it is known that the reduction in permeability halts at a critical pressure corresponding to the point at which maximum adsorption is achieved and then increase as a consequence of diminishing effective stress (Kumar et al., 2010, 2016; Wang et al., 2012). In this study, analysis of permeability change was based on the Palmer and Mansoori model which consists of the sum of pore strain and sorption strain, but experiments were conducted under same effective stress to analyze the permeability change by only sorption strain considering diffusion phenomenon in coal. Therefore, because the effect of external stress is excluded, permeability is reduced continuously with increasing pore pressure until it reaches the maximum amount of adsorption, and after that, the permeability change did not occur due to constant effective stress. From the experimental results shown in Figure 6, the drastic permeability reduction at the early time of injection is induced by diffusion characteristic shown in Figure 2. In other words, because fast increase in diffusion coefficient increases adsorption rate, it results in a drastic permeability reduction. In addition, the permeability at the maximum adsorption pressure is ultimately determined by the difference in adsorption affinity of each gas. For the case of injecting N2, movement of gas molecules caused by diffusion is slower than that of CO2, and early BT occurs due to low adsorption affinity. It leads to the result that permeability decreases gradually. For CO2–N2 mixed gas injection, adsorbed N2 is desorbed easily due to the high sorption capacity of CO2. Accordingly, permeability change appears somewhat irregularly, especially for 25% CO2. For injecting mixed gas including N2, permeability reduction in the experimental pressure range were shown as 73.2% for 75% CO2, 70.7% for 25% CO2. It was improved as compared to permeability reduction as 76.5% for injecting pure CO2 case. It is confirmed that N2 delays CO2 BT and acts as a factor for inhibition in permeability reduction simultaneously. As a result, since the increase in diffusion coefficient with gas partial pressure is different depending on diffusion properties, gas flow also occurs differently, and then it affects the aspect of permeability change and displacement flow. It can be seen that diffusional flow must be considered in permeability change and displacement flow in coal seam.
The results of permeability change by sorbed gas injection.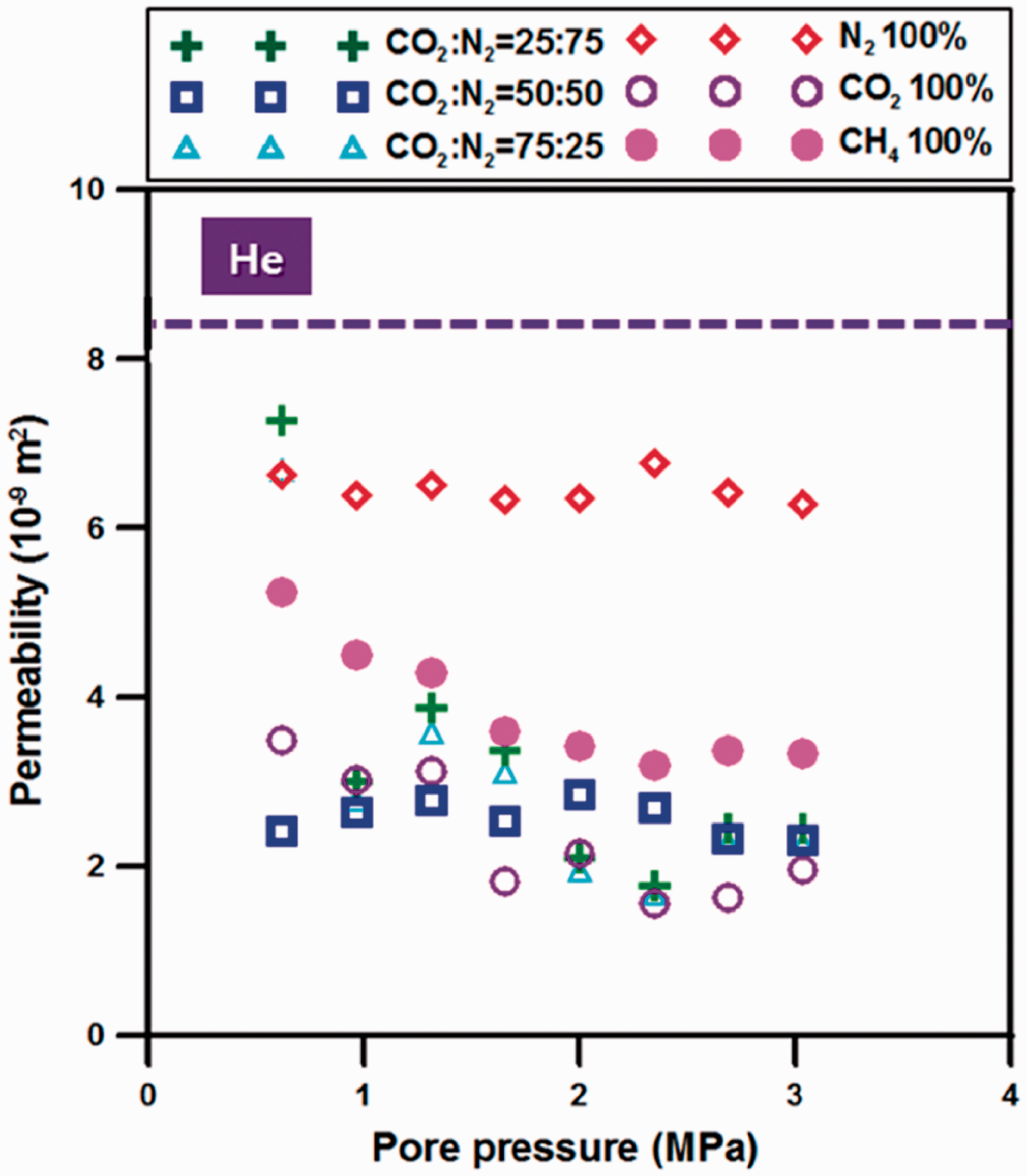
Enhanced methane recovery by CO2–N2 injection
Figure 7 shows the result of methane recovery for pure CO2 and N2, and CO2–N2 mixture with various mixing ratios. For pure CO2 injection, although CH4 recovery increases slowly, displacement efficiency is enhanced by only CH4 production due to complete displacement between CO2 and CH4. However, injectivity problem occurs near injection well owing to the high adsorption affinity of CO2. As known from previous studies, coal is more vulnerable to the injectivity problem and reduction of displacement efficiency with CH4 due to high matrix swelling of coal caused by sorption-induced strain in CO2-ECBM. Also, diffusion phenomenon depending on pore structure in coal seam affects aspects of permeability change and displacement flow conclusively, diffusion properties of each sorbed gas are very important in ECBM accordingly. Therefore, this study was conducted with a focus on CO2–N2 mixed gas injection using the low adsorption property of N2 considering diffusion phenomenon. As shown in Figure 7, for mixed gas injection consisted of 25%, 50%, and 75% in N2, it tends to increase methane recovery than that of pure CO2 injection, and N2 serves as a displacing agent heavily for 75% N2 than 25% N2. Because CO2 content is low among the compositions of injected gas, however, it reveals rather low increasing behavior of methane recovery compared to 25% N2. Furthermore, for 50% N2, it shows a similar increasing behavior of methane recovery for 75% N2 during initial injection period, but difference in methane recovery occurred gradually on account of delayed displacement depending on counter-diffusion with methane which was generated by CO2 content as increase of injection volume. Nevertheless, the recovery difference appears to be decreased progressively toward the latter. Thus, it is confirmed that the co-diffusion with methane by N2 at the initial period of injection and the counter-diffusion by CO2 in the latter affected methane production significantly. For pure N2 gas injection, methane was produced through role of N2 as a displacing agent for desorbed methane with reduction of partial pressure so that methane recovery tends to increase rapidly. Nitrogen, however, has a disadvantage that a large amount of N2 must be injected compared to the other gases due to the increased role of co-diffusion with methane as well as the most rapid BT time of N2. Accordingly, it is ascertained that N2 is effective for enhanced methane recovery when it is injected as CO2–N2 mixture than pure gas resulting in generating a greater concentration gradient between CO2 and methane, and facilitating counter-diffusion further.
The results of methane recovery by sorbed gas injection.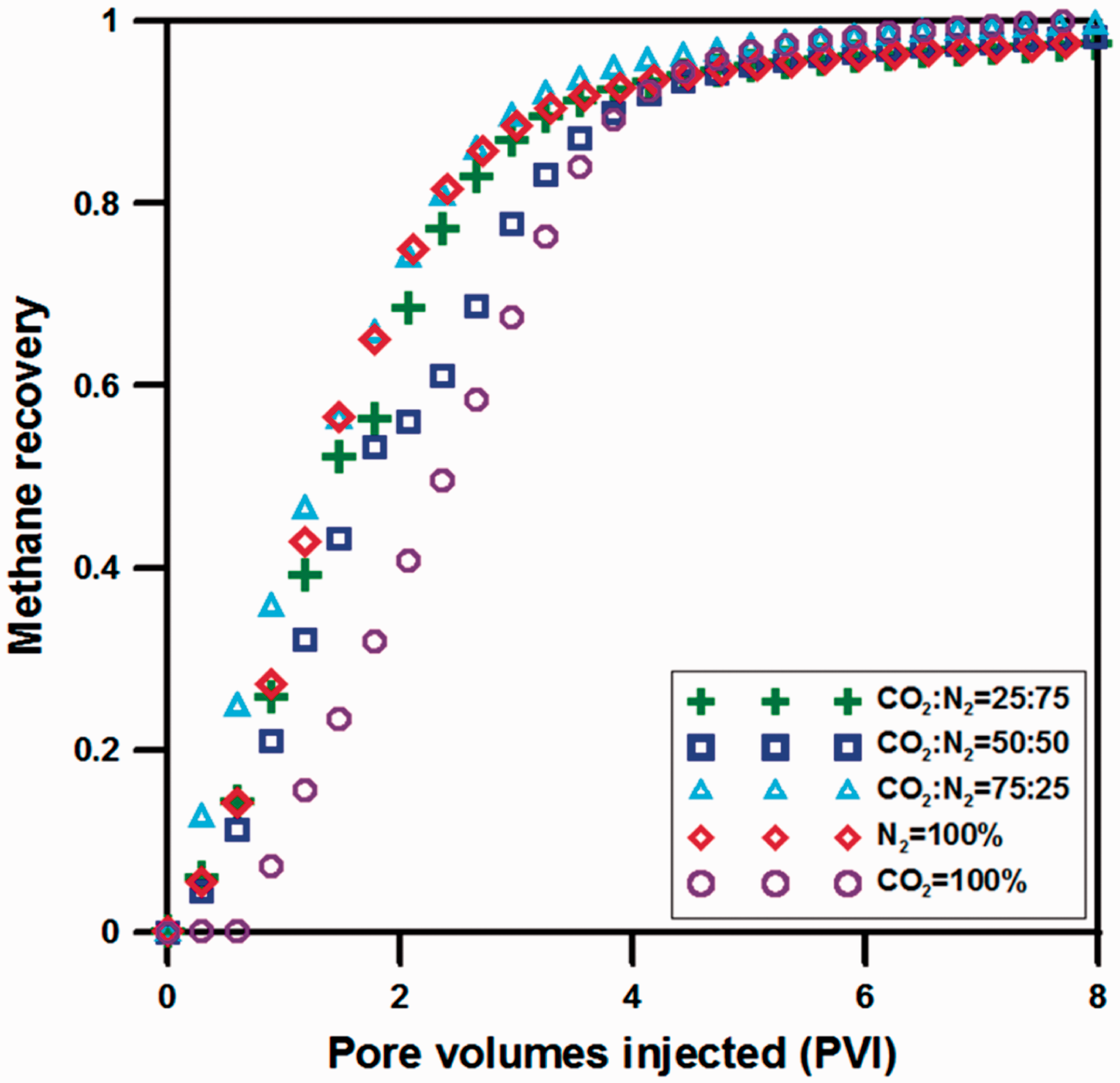
Conclusions
This study provides the fundamental reason for the reduction of gas permeability and methane recovery during ECBM processes. From the results obtained from the experiments of diffusional flow, displacement flow, and gas permeability, we conclude the followings:
Because the diffusion coefficient is in general a function of gas molecular size and pore geometry of coal matrix, even if CO2 injection rate is very high, it is no longer increased at high pressure. Therefore, a large amount of CO2 moves through cleat causing an early CO2 breakthrough, and correspondingly, it is necessary to consider diffusional flow effect in estimating the optimum injection rate. When injecting a CO2–N2 mixed gas into coal seam, N2 acts as a displacing agent of methane, because co-diffusion of N2 with methane is more dominant than counter-diffusion owing to its extremely low adsorption affinity. It can be seen that such diffusional flow behavior of N2 is important in enhanced methane recovery. CO2 is rapidly adsorbed due to its fast increasing rate of diffusion coefficient with pressure increase. Thus, CO2 permeability is reduced rapidly and ultimately becomes the lowest value at the maximum adsorption pressure. Also, delayed methane recovery by fast diffusion and high adsorption affinity of CO2 occurs accordingly. This study confirms that the CO2–N2 mixed gas injection is advisable compared to CO2 injection to pursue the prevention of CO2 injectivity reduction and enhanced methane recovery, simultaneously.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Korea Evaluation Institute of Industrial Technology grant from the Korean Government’s Ministry of Knowledge Economy (No. 10039231).
