Abstract
Antimicrobial resistance is now recognised as a threat to health worldwide. Antimicrobial stewardship aims to promote the responsible use of antibiotics and is high on international and national policy agendas. Health information technology has the potential to support antimicrobial stewardship in a number of ways, but this field is still poorly characterised and understood. Building on a recent systematic review and expert roundtable discussions, we take a lifecycle perspective of antibiotic use in hospitals and identify potential targets for health information technology-based interventions to support antimicrobial stewardship. We aim for this work to help chart a future research agenda in this critically important area.
Introduction
Antimicrobial resistance is now recognised as an urgent global threat to health. 1 The prevalence of single- and multi-drug-resistant organisms is increasing 2 as is mortality due to infections caused by these organisms. 3 Across the European Union and the Unites States, antimicrobial-resistant infections are responsible for at least 50,000 deaths annually.4,5 Globally, these infections claim an estimated 700,000 lives each year. 6
Antimicrobial stewardship is the international response to the threat of resistance. As the inappropriate use of antibiotics is a key contributor to resistance, 7 there is increasing policy and clinical interest in promoting appropriate use of antibiotics 8 through a set of concerted interventions, including: health information technology; 9 guidelines and protocols; 10 education and training; and novel staffing structures in the form of antimicrobial stewardship committees and champions. 11 Often used in combination, these interventions aim to optimise the use of antibiotic therapy, 12 reduce resistance, minimise hospital acquired infections and improve clinical outcomes. 13
Health information technology has much to offer the antimicrobial stewardship agenda agenda as can be seen by the increasing number of studies that have looked into the feasibility, effectiveness and accessibility of various health information technology-mediated antimicrobial stewardship interventions.9,14,15 There is, however, a lack of any comprehensive overview of the potential targets for health information technology-based interventions. This may hamper progress in this still nascent, but critically important field.
The aim of this article is to highlight areas of the antibiotic lifecycle that are potentially most amenable to health information technology interventions in hospital settings. This review also outlines health information technology that has the potential to be deployed in relation to antimicrobial stewardship, and is currently, or soon to be, available. We do not, however, make claims that this health information technology is unequivocally helpful. Furthermore, this review is not exhaustive but intends to invite discussion around which of these areas may be implementable and subsequently scalable in hospital antimicrobial stewardship programmes in the UK and to identify areas for further research enquiry.
Methods
We began by developing a conceptual schema of all stages of an archetypal antibiotic lifecycle from the initial decision to prescribe an antibiotic for a patient with suspected infection, through to the review of treatment within a typical United Kingdom hospital setting (Figure 1). This antibiotic lifecycle was informed by a review of the published and grey literature, and multidisciplinary discussions involving patients, hospital clinicians and management staff undertaken as part of our recently completed programme grant on hospital electronic prescribing (ePrescribing) systems. More specifically, we drew on our recent systematic review
14
and expert multi-stakeholder roundtable discussion,
16
which highlighted the many ways in which health information technology already supports, and could potentially support, antimicrobial stewardship by applying best practice technologies and procedures during the antibiotic lifecycle in British hospital care. We conceptualised this work within the context of the UK policy framework for antimicrobial stewardship: Start smart – then focus.
17
Conceptual schema of an archetypal antibiotic lifecycle through prescription and review within a British hospital setting.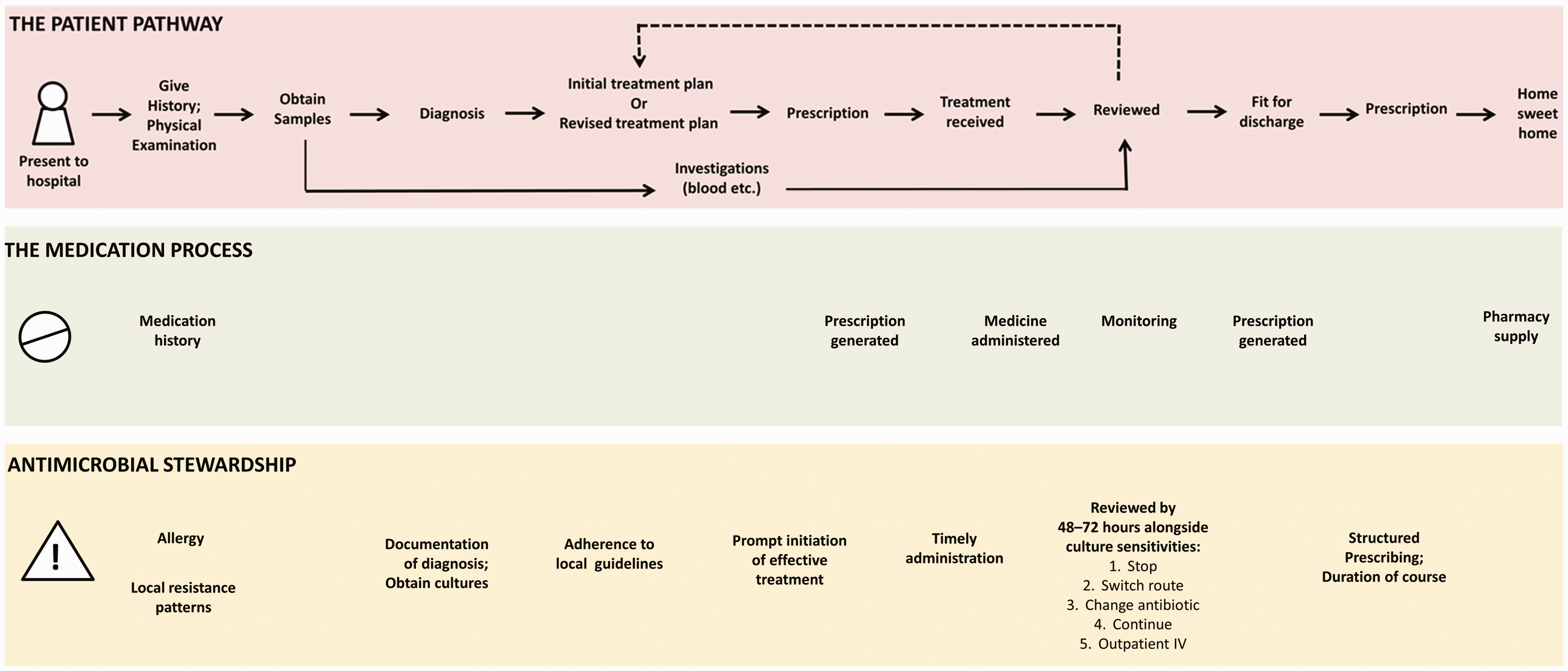
Lifecycle of hospital antibiotic use
The lifecycle of antibiotic management in hospital starts when a patient presents to hospital with or develops during their hospital stay a suspected infection. The lifecycle (Figure 1) proceeds through an initial differential (working) diagnosis from the initial presentation, obtaining specimens for microbiological cultures and sensitivity testing (where applicable), initial empirical treatment decision and generation of a prescription order, followed by timely review of treatment and subsequent amendment where applicable. This is far from a linear process; it typically involves many different ‘actors’ at different points in the care process, and each process is variable depending on patient demographics such as prior history and comorbidities. As such, to understand how the antimicrobial stewardship agenda can be supported with health information technology, it is important to consider the clinical situations that fall outside the lifecycle and how these too may be supported through appropriate health information technology intervention. In the sections below, we discuss the lifestyle components that are already supported and those that can be supported by health information technology to better align care with the antimicrobial stewardship agenda. At present, some of these health information technology interventions are commonly seen across hospitals in the United Kingdom, European Union and United States, while others have been employed by only a few benchmark hospitals.
Potential for health information technology interventions to promote antimicrobial stewardship
Health systems are moving towards digitisation. 18 The ability to store, share, access and proactively use information at the point of care as well as through prospective and retrospective auditing and feedback is being used to drive forward efforts to enhance the safety, quality and efficiency of healthcare. 19 Health information technology that supports antimicrobial stewardship can come in many forms: electronic health records that record patient information such as diagnoses and previous medication sensitivities; ePrescribing systems, such as computerised physician order entry and/or clinical decision support systems 20 ; and monitoring systems that draw together information from disparate parts of the hospital, enabling, for example, the triaging of patients who require clinical intervention. These systems can make data accessible to support diagnosis, treatment decisions and review of patients who may for example need the route of delivery of antibiotics to be changed (e.g. intravenous to oral) or those in whom treatment can be discontinued. 13
In the subsequent sections, we describe particular points in the antibiotic lifecycle that may be supported by and optimised through health information technology intervention (also delineated in Figure 2). These sections reflect the Start smart – then focus
17
framework as it is the recommended approach by Public Health England for all antibiotic prescribing. The interventions described mainly target clinical staff: doctors, pharmacists and nurses at the point of care, as well as at the level of an antimicrobial stewardship committee or hospital management. They may in the future also target patients/carers.
Health information technology support of the antimicrobial stewardship agenda.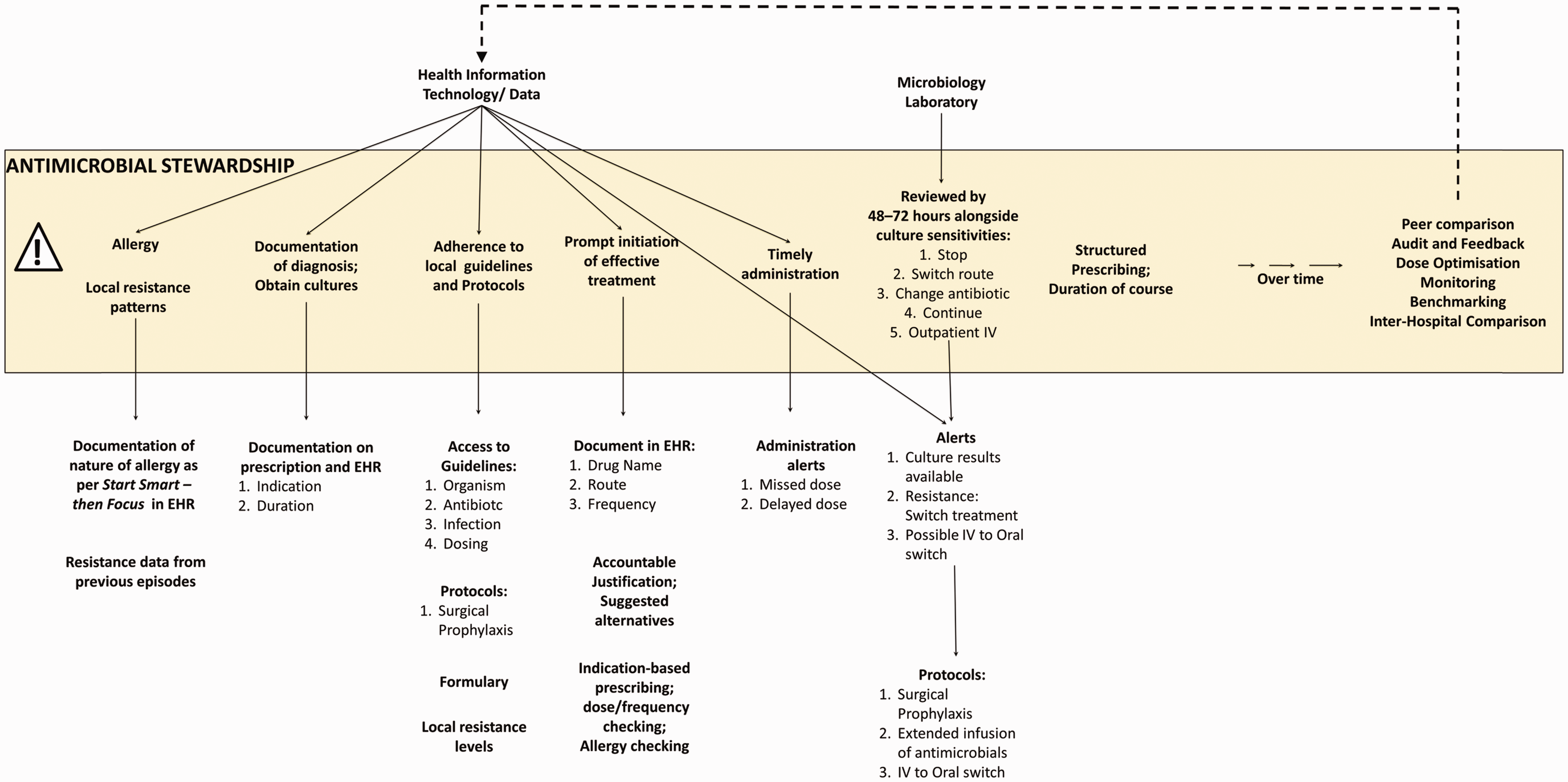
Start smart
Patient presentation
At its simplest, the lifecycle of a hospital administered antibiotic starts when a patient presents with symptoms suggestive of an infection or develops a new infection while already in hospital. A clinician will take (or review) the patient’s medical and drug history, undertake a physical examination and perform relevant investigations, including blood tests. This process and information can be documented in the electronic health record alongside the patient’s current regimen of medication and any known allergies to medications. Through the electronic health record, the clinician may also access the patient’s previous microbiological results with any antimicrobial resistance data that may help inform assessment of the likely effectiveness of the antibiotic(s).
Diagnosis
The clinician will make a differential diagnosis, and if an infectious process is suspected then a decision will be made whether to commence empirical treatment with a relevant antibiotic. Decisions can be documented in the electronic health record along with the suspected indication and proposed duration of any treatment. The diagnosis may also be documented in the ePrescribing system if this is separate to the electronic health record. As electronic health records and ePrescribing systems are often integrated, the patient information and indication for treatment 21 can be easily accessed as is the ability to track the progress of the patient and the clinical decision-making over time. However, not all information may be in a form that allows for this tracking of decision-making processes over time – this is because the initial diagnosis may not be recorded or indications may not be reported in a structured way that can be used to drive reports. Further research using natural language processing may prove helpful in such scenarios.
Obtain cultures
An important step in the antibiotic prescribing process is, where appropriate, obtaining cultures to determine the organism responsible for the infection. Importantly, cultures should be obtained before starting treatment, although this should not delay the initiation of antibacterials in cases of suspected sepsis. Culture results can confirm whether any empirical treatment commenced is appropriate to continue, or whether treatment needs to be changed for optimal management (or started if not done so previously). Through understanding local resistance patterns and susceptibility rates of particular organisms, clinicians can determine which antibiotics might work for the particular infection and also whether the chosen antibiotic(s) is likely to work in any individual patient.
Health information technology can play an important role here. By linking culture orders, medication orders and administration records, health information technology supports communication between the prescribers on the wards and the microbiologists in the laboratory. Furthermore, microbiology results can be incorporated into the electronic health record and made available at the point of care. Systems can alert prescribers to results that have recently been made available, in order to prompt action for review. However, as these results often rely on free text data, the multiplicity of report formats mean that microbiology results are often not yet in a form that can be used to drive more specific alerts, such as when the organism causing a patient’s infection is resistant to the antibiotic the patient has been prescribed. Coding for urgency in alerts prompted by available microbiology data is an important area of future health information technology development.
Initial treatment decision
While awaiting the results of cultures and antibiotic sensitivity tests, clinicians will typically institute an empirical treatment plan based on the differential diagnosis. Minimising exposure to broad-spectrum antibiotics and maximising exposure to optimal therapies is an important component of antimicrobial stewardship. 22 The initial treatment decision and prompt initiation for specific indications (e.g. ventilator-associated pneumonia) 23 and in specific hospital care contexts (e.g. critical care units) 24 is another strategic area for antimicrobial stewardship; however, this may be in tension with the judicious use of antibiotics until culture results are available. 13
Hypothetical antibiogram of Gram-negative and Gram-positive isolates from urine cultures.
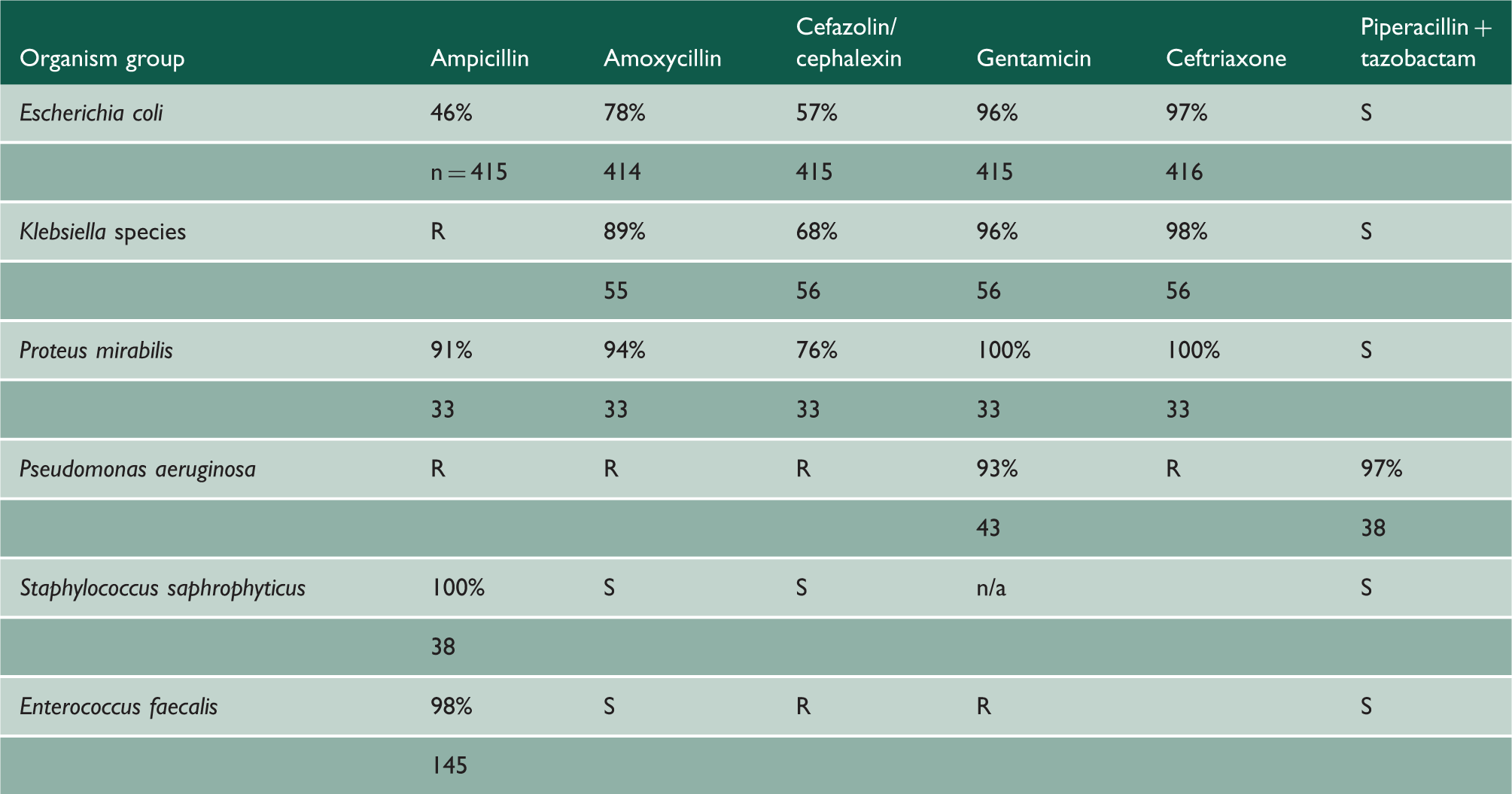
Antibiograms summarise antibiotic susceptibilities in order to facilitate the selection of appropriate antibiotic therapy. In the past, antibiograms were updated annually. Presently, through the help of health information technology, antibiograms are updated continually as new susceptibility data become available. Antibiograms can be filtered to show prescribers susceptibility patterns for the region, the institution and the ward.
n/a: not available (not routinely tested in local laboratory); S: susceptible by extrapolation; R: intrinsically resistant; %: % isolates susceptible; n: number of strains tested.
Clinical examples of inappropriate use of antibiotics targeted and remedied by health information technology intervention.

Health information technology can further support initial treatment decisions by enabling the clinician to document their decision including the drug name, route, dose and frequency in the electronic health record and/or ePrescribing systems. With this information, health information technology can aid with indication-based prescribing, dose/frequency checking, as well as allergy, drug–disease interaction and drug–drug interaction checking. ePrescribing systems can also encourage the use of appropriate, evidence-based antibiotic therapies through behavioural nudges. 25 At the point of care, nudges can come in the form of ‘accountable justification’ and ‘suggested alternatives’, as described by Meeker et al. 25 Accountable justification, through prompts within the ePrescribing system, requires clinicians to explain their selection of antibiotics in a free text box that is ultimately included in the patient’s electronic health record. Suggested alternatives, through pop-up alerts, are triggered when antibiotics are prescribed for conditions that may be treated through other therapeutic routes. These alerts can provide prescribers with a list of alternative therapies as well as easy access to electronic order sets for these therapies.
Prompt initiation of effective treatment supported by electronic health record
The interoperability and automation of health information technology systems aim to minimise the time from the diagnostic order to the patient receiving the first dose. 26 During prescription preparation, health information technology can support pharmacists through a process of controls: checking whether the prescription is appropriate for the indication and the patient as well as facilitating contraindication checks such as allergies, drug interactions and conditions such as renal impairment which may necessitate a modification of the antibiotic dose. Electronic health record and ePrescribing systems can also support nurses during the administration of medication through reminders for timely administration as well as alerts for missed and delayed doses.
Then focus
Reviewing the clinical diagnosis and assessing the need for continuing antibiotics
The National Institute for Health and Care Excellence recommends that prescriptions for antibiotics are reviewed 48 to 72 h after initiation.
27
The purpose of the review is to determine:
whether the patient has an infection that will respond (or is responding) to antibiotics; whether the patient is on the correct antibiotics (as well as if the therapy is at the right route and dose of administration); if a more targeted antibiotic can be used (especially if a narrower spectrum antibiotic will be sufficient); and how long the patient should receive the antibiotic treatment.
Importantly, this review is performed alongside the culture and sensitivity data provided by the microbiology laboratory. These data will also provide information on drug resistance as well as the sensitivity of antibiotics to the isolated organism. If changes are deemed necessary following the review, there are several potential actions available: continuing treatment; stopping therapy; switching the route; changing the antibiotic; or discharging the patient with a continued course of antibiotics.
The review and subsequent changes to antibiotic treatment should occur in a timely manner, as soon as culture results are available or if the patient’s condition is altering. Health information technology can support this process by alerting clinicians to view the results or by providing reminders that a review is required. Health information technology can further alert clinicians to consider potential actions: for example, switching to an alternative therapy in the presence of a drug–organism mismatch, or switching from intravenous to oral antibiotics when the patient is eating/drinking/taking other treatments orally and appropriate treatments are available to take by mouth. These alerts can help to reduce the time between the availability of culture results and the review of antibiotic treatment. This is particularly important for antimicrobial stewardship efforts as switching from initial therapy to a more optimal, appropriate therapy can minimise deleterious effects to patient health, 15 contribute to an earlier discharge, 9 minimise cost 13 and reduce the risk of resistance. 13 Health information technology can further support review and subsequent therapeutic changes by facilitating access to relevant protocols: e.g. extended infusion of antibiotics, parenteral to oral switch or Out-Patient Antibiotic Therapy. However, further research is needed to explore how health information technology can support the behavioural components involved in reviews as healthcare professionals can be reluctant to revisit and change a decision previously made by a doctor.
Review by antimicrobial stewardship champions
Alongside the work of individual practitioners in the medication process, antimicrobial stewardship programmes are more effective when driven by a focused group of healthcare professionals.
13
Often termed the antimicrobial stewardship Committee, or antimicrobial stewardship Champions, this group supports many components of the antimicrobial stewardship agenda (Figure 3) including the provision of face-to-face specialist review or support to clinical teams during complex patient cases. Important to the work of committees and champions is the ability to have oversight of all antibiotic prescriptions across the hospital. This oversight can be considerably enhanced through the use of monitoring systems (Box 1, Example 2). These systems can aggregate and organise data from many sources across the hospital into actionable alerts. Data can come in the form of resistance and susceptibility information, as well as information about patients, treatments and indications. Antimicrobial stewardship committees can gain an overview of all patients on antibiotic therapy through these systems. The most urgent cases can also be made visible, enabling the prioritisation of patient review.
Support of the antimicrobial stewardship agenda through antimicrobial stewardship committees and champions.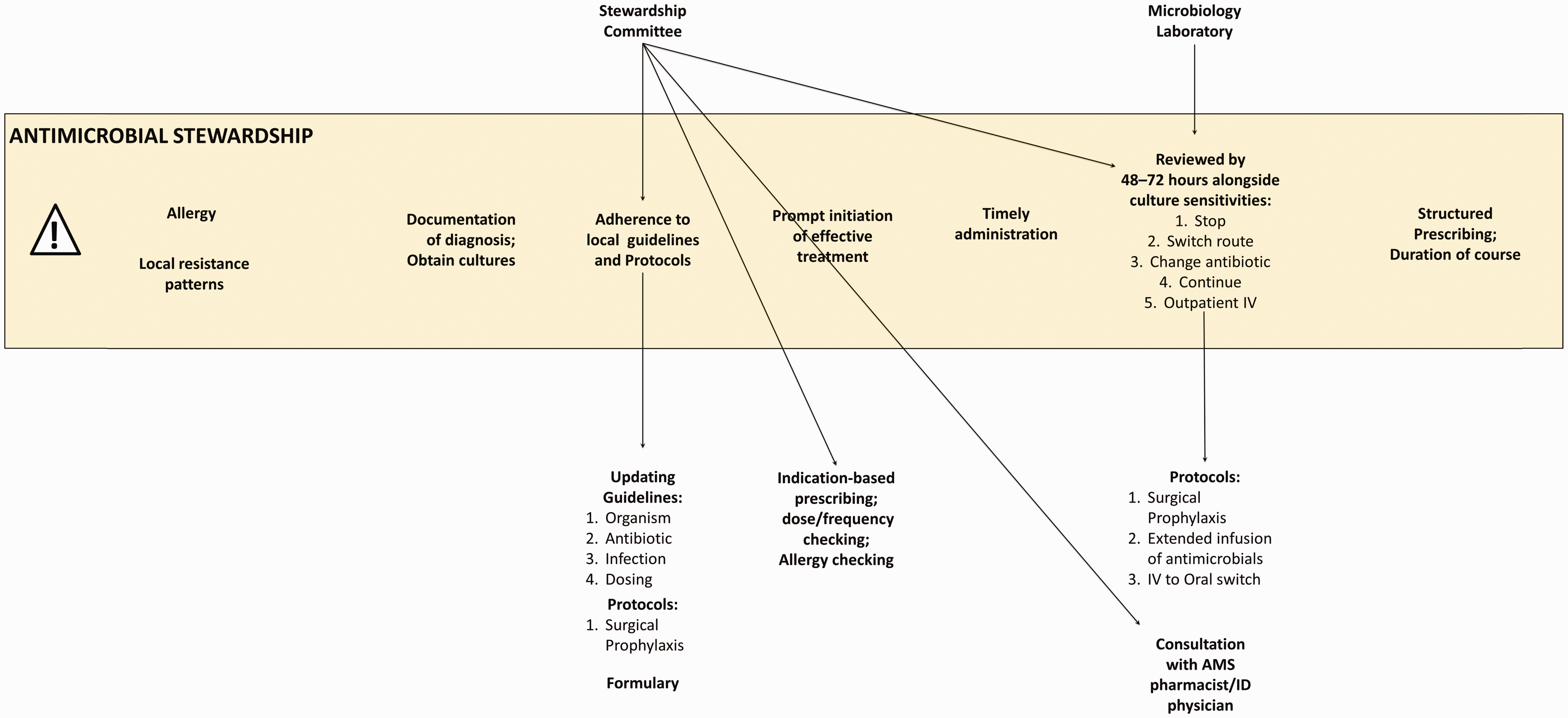
Antimicrobial stewardship committees also update antimicrobial guidelines and protocols, and inform the formularies that can be made available within the electronic health record. Furthermore, committees can monitor adherence to guidelines and protocols with the help of health information technology to identify wards, clinical teams or individual prescribers who may require further support as well as to inform the update of hospital policies.
Over longer timeframes (months to years), collecting and reusing data
As discussed above, health information technology has much to offer the antimicrobial stewardship process at the point of care. Many components of the antimicrobial stewardship agenda can also provide benefits over longer timeframes, especially those supported by health information technology. Electronic health records and ePrescribing systems can aggregate longitudinal prescribing data at multiple levels: individual clinicians; clinical teams; wards; and hospitals. This enables comparison of inappropriate prescribing rates (Box 1, Example 3). Meeker et al. 25 describe an intervention based on behavioural economics that contacted each prescriber via email reporting whether the prescriber was a ‘Top Performer’ or ‘Not a Top Performer’, as defined by their proportion of inappropriately prescribed antibiotics. Due to the positive reinforcement and social motivation behind this intervention, inappropriate antibiotic prescribing was significantly reduced.
Over longer timeframes, health information technology can also enable further audit and feedback, monitoring and benchmarking of antimicrobial prescribing practice. These functions can support clinicians, pharmacists and antimicrobial stewardship committees in further promoting the antimicrobial stewardship agenda as well as lead to inter-hospital comparison. For example, in the UK, there is a national benchmark indicator derived in many hospitals from health information technology data to compare the total consumption of carbapenem antibiotics (a broad spectrum class agent) per 1000 admissions as part of the NHS Commissioning for Quality and Innovation targets in 2016/2017. 28 Ultimately, data-driven surveillance can help to better define and direct effective antimicrobial stewardship strategies, as well as continuously assess hospitals systematically at the local, regional, national and international levels.
These developments over longer timeframes can all feed back into health information technology systems to further optimise future therapeutic decisions, improve outcomes and reduce resistance to antibiotics.
The future of health information technology-mediated antimicrobial stewardship
There exist many further areas of health information technology support that are either in the early stages of evaluation in pilot hospitals or are still in the development pipeline. Many of these involve the improvement of existing system design to more accurately transmit actionable information throughout the hospital (such as the potential for specific microbiology results to prompt urgent alerts, as discussed above).
Predictive modelling 29 is another emerging possibility for antimicrobial stewardship support that has the potential to optimise the initial prescription of antibiotics. By bringing together information regarding an individual patient’s risk factors for resistance and susceptibility to certain antibiotics, predictive modelling tools may aid in the selection of optimal antibiotic therapy at the level of the prescriber. Point-of-care diagnostic tests also offer the possibility of selecting optimal routes of care on the ward through the rapid identification of infectious agents.
Health information technology can also enable further data collection for audit and feedback. These data are important for benchmarking hospitals against others to evaluate antimicrobial stewardship interventions and to better understand the barriers and facilitators to implementation.
Cloud-based electronic health records are on the horizon which will raise the possibility of expanding health information technology support from the antibiotic prescription and review lifecycle on the ward to an extended antibiotic lifecycle: one that follows patients through their hospital admission, discharge and back into the community. Further support in the community is expected to come in the form of patient access to information and education about appropriate treatment and avoiding overuse of antibiotics. These approaches can also support those in the community who have different needs around antibiotics, such as expert patients with long-standing conditions (e.g. for a patient with cystic fibrosis, the timely administration of antibiotics is often more important than avoiding overuse of antibiotics). With the aim of empowering patients and involving them in the decision-making process around antibiotics, patients will soon be able to access information about their resistance patterns. 30
Conclusions
Antimicrobial stewardship is a national and global priority being implemented to reduce the risk of antimicrobial resistance. Health information technology is both maturing and converging, and as a result offers considerable opportunities by supporting: access to relevant data (e.g. personal and local antibiotic resistance patterns) and antimicrobial guidelines, documentation of diagnoses, initial selection of empirical treatment, review, audit, and feedback and monitoring. The most effective health information technology-enabled antimicrobial stewardship interventions are likely to be multi-faceted, consisting of many of these different components in tandem with behavioural changes. Importantly, any implementation requires careful planning and integration with existing systems, as well as staff engagement and participation. Health information technology processes alongside education, training and person–person interaction between staff as well as with patients will be most effective in comprehensively supporting antimicrobial stewardship.
This paper provides a starting point for how these components may be conceptualised alongside the existing processes surrounding antibiotic prescription and review. Research in this area needs to be prioritised in order to ensure that the considerable potential offered by developments in health information technology in relation to antimicrobial stewardship are realised in a timely fashion.
