Abstract
The use of pheromones is a promising alternative in insect management and control. Recently, the incorporation and release of active ingredients from nanofibers has been of interest for agricultural purposes due to their higher surface area to volume ratios. The objective of this study was to produce nanofibers incorporating synthetic sex pheromones from the oriental fruit moth, Grapholita molesta (OFM), using different polymers. The nanofibers with pheromones were produced by electrospinning and were evaluated for: i) homogeneous distribution of OFM pheromone in nanofibers by electroantennography (EAG) tests; ii) dose-responses of OFM males to acetate cellulose nanofibers; iii) EAG responses of OFM males to different polymer nanofibers exposed under controled conditions for up to 5 weeks: iv) attractiveness of G. molesta to nanofibers containing synthetic OFM in field conditions; v) quantification of incorporated pheromone in PCL and PVAc nanofibers using gas chromatography and vi) morphology using scanning electron microscopy. The best field results were achieved in smoother fibers with higher impregnation. Pheromone incorporating nanofiber vehicles were tested that achieved the controlled release of the pheromone for up to three weeks in nanoscale and microscale. The polymer, solvent, and dosage incorporated in the nanofibers were important for controlled delivery. Understanding these factors is important for the development of better pheromone dispensers.
Introduction
The use of pheromones is a promising alternative in the management and control of agricultural pests. These substances have the advantage of high specificity and its use does not harm the environment, compared to traditional methods based on the use of insecticides (Vilela and Mafra-Neto 2001). These attractive compounds can be used in agricultural systems in order to monitor or control insects. The control can be obtained by collecting mass of males, as well as confounding by sexual mating disruption or when the male confusion is caused due to the excess chemical stimulus in the field, preventing mating (Degen et al. 2005; Kovanci et al. 2005; Charmillot et al. 1997).
In Brazil, the semiochemicals market has increased considerably, bringing the use of pheromones in many cultures as an alternative to control pests important species, such as pink worm Pectinophora gossypiella (Saunders, 1843) (Lepidoptera, Gelechiidae), tomato borer Tuta absoluta (Meyrick, 1917) (Lepidoptera, Gelechiidae), cotton boll weevils Antonomus grandis Boheman, 1843 (Coleoptera, Curculionidae), sand fly Lutzomyia longipalpis Lutz and Neiva, 1912 (Diptera, Psychodidae), sugar cane borer Diatraea saccharalis (Fabricius, 1794) (Lepidoptera, Pyralidae) among others (ISCA 2013).
Numerous forms of dispersal have been developed for capturing Grapholita molesta (Busk, 1916) (Lepidoptera, Tortricidae), especially pastes, polymer sachets and microencapsulated formulations (ISCA 2013). Recently, the incorporation and release of active ingredients from nanofibers has been of great interest for pharmaceutical and agricultural purposes. Due to their higher surface area to volume ratio, nanofibers have been studied for applications such as drug delivery carriers (Ramakrishna 2005). There is increasing interest in better dispersal of agricultural inputs in crops aiming to increase productivity and lower costs. Electrospinning (ES) is one of the most interesting new methods for drug delivery (Liao et al. 2008; Xu et al. 2008; Chakraborty et al. 2009; Tamaro et al. 2009; Cui et al. 2006) and the nanofibers produced can also bring benefits to the agriculture area.
The novel application of nanotechnology, specifically polymeric nanofiber as vehicles for semiochemicals dispensing in agriculture was based on a pioneering study conducted in Germany by Hellmann et al. (2009). In this study, the authors used the electrospinning technique for producing cellulose acetate nanofibers containing the grape berry moth Lobesia botrana (Denis & Schiffermüller, 1775) (Lepidoptera, Tortricidae) synthetic sex pheromone. The authors examined the possibility of incorporating the pheromone of this species within the nanofibers for their use in mating disruption in grapevine orchards.
Subsequently, Lindner et al. (2011) realized laboratory tests to characterize the nanofibers containing the L. botrana pheromone. Hummel et al. (2011) performed bioassays in the wind tunnel, also quantifying the mass loss of nanofibers with the aid of analytical balances and by conducting thermogravimetric analysis (TGA). According to Hummel et al. (2011), in the future, these nanofibers vehicles containing pheromone could be deposited directly on the plants, with the aid of portable electrospinning machine, which would allow the coverage of orchard large areas, while increasing the efficiency of the sexual confusion technique. Thus, in theory, it would be possible to produce nanofibers containing an emulsion encapsulating pheromone molecules. These small scale capsules dispersed in continuous polymer phase produced by electrospininng method would have a large area relative to the total, lowering cost and increasing efficiency of the mating disruption strategy.
To date, information about the use of nanofibers as vehicles for dispersing insect pheromones are scarce and largely based on the results obtained by Hellmann et al. (2009), Hummel et al. (2011) and Lindner et al. (2011), which served as parameters in the selection and polymer concentrations pheromones to be investigated. The OFM synthetic pheromone was chosen as a model compound for the study of a nanofibers vehicle because of its known efficiency in the pest management when used in other formulations. Thus, the objectives of this study were: i) evaluate the distribution of OFM pheromone in nanofibers through electroantennographic bioassays; ii) estimate the dose-responses of the OFM males to acetate cellulose nanofibers; iii) measure the EAG responses of OFM males to nanofibers exposed at controled conditions for until 5 weeks; iv) evaluate the attractiveness of adult G. molesta to nanofibers containing synthetic sex pheromone in field conditions; v) quantify the pheromone in PVAc and PCL nanofibers by gas chromatography and; vi) determine the morphology of the nanofibers produced by scanning electron microscopy (SEM).
Materials and methods
Insects rearing.
The G. molesta individuals used in the bioassays were obtained from rearing in BIOECOLAB-UFRGS, Department of Crop Protection, Faculty of Agronomy, Federal University of Rio Grande do Sul, kept under controlled conditions (25 ± 1 °C, 60 ± 10% relative humidity, and a 16-8 L-D hour photoperiod). The larvae were artificial diet fed, following the methodology of Arioli et al. (2007) and the adults were fed with water honey solution (15% honey and 5% methylparaben). Only male insects were used in the bioassays.
Electroantennography (EAG).
Insects used in the bioassays had the antennas sectioned at the pedicel and were connected to a bifurcated silver electrode. The antenna's apical and basal ends were attached between the registration and the neutral electrode, respectively. Both were immersed in Spectra 360% salt-free gels to enhance the electrical conductivity of the sample. The analogical signal response, measured in millivolts, was captured, amplified and processed with a data acquisition controller (IDAC -4 Syntech*) and subsequently recorded by software (EAG2000, Syntech).
All treatments were individually inserted into Pasteur pipettes. The front end of the pipette was placed into a hole in a metal tube (1cm diameter x 18 cm in length) oriented toward the antenna, having an approximated 1 cm distance. The antennas were subjected to air pulses generated by a flow controller (CS-02, Syntech*) in a volume of 2.5 mL/0.5 s with different treatments. It has been stipulated time of one minute between successive stimuli so that the antenna regained their capacity to perceive odorant. The electrophysiological responses of each treatment were recorded in millivolts and the EAG average responses were compared by ANOVA (Tukey test; α = 0.05) using the BioEstat 5.0 statistics program (Ayres et al. 2007). The antennas were used only once in each repetition. For each EAG response test, 15 virgin males aged between three to seven days were used.
Production of electrospinning nanofibers containing the OFM synthetic sex pheromone.
The polymer nanofibers were produced at Tecnano Research and Services Ltd., using a custom made electrospinning machine with a 60 kV HV supply. The syringe pump consisted of 3 ml syringe and a rotary aluminum collector was adjusted to about 60 rpm. In this process, polymer dispersions were prepared dissolved in specific solvents, with and without the synthetic sex pheromone of G. molesta (P9000 - 90 Bedoukian OFM Technical Pheromone). The polymers used were cellulose acetate (CA), polyvinyl acetate (PVAc), polycaprolactone (PCL), polyvinyl pyrrolidone (PVP) and styrene-butadiene-styrene (SBS) copolymer, and its blends. The solvents used were dichloromethane (DCM), dimethylformamide (DMF), ethanol (EtOH), tetrahydrofuran (THF) and water, either alone or in solvent blends. The polymer, or polymer blend, was dissolved using the solvent mix in order to obtain a final polymer concentration ranging from 0.02 to 10% (w/v) of OFM synthetic pheromone that was fed into the ES machine with a syringe pump at a rate of 0.05 ml/min using a 3 ml syringe with a blunted needle. A voltage of 1.81 kV/cm was applied between the needle and the earthed collector. The nanofiber films were deposited onto the glass slides and directly onto the metal stub for microscope analysis. The produced nanofibers were isolated and placed at -10 °C until the time of the bioassays.
Bioassays were conducted in laboratory (EAG) and in field conditions. Initially, based on Hellmann's work (2009), cellulose acetate nanofibers were produced and tested to verify the possibility of incorporating the G. molesta synthetic sex pheromone in these nanoscale matrixes.
Bioassays
i) Distribution of OFM pheromone in cellulose acetate nanofibers through electroantennographic tests.
Cellulose acetate nanofibers having dimensions of 100 cmβ were produced containing about 10% OFM pheromone based on polymer concentration. To check the homogeneity of the distribution of the pheromone within the matrixes, each nanofiber was divided into 25 quadrants measuring 4 cmβ and identified for use in electroantennographic bioassays. The G. molesta male antennal stimuli from each of the quadrants removed from nanofibers was evaluated by the bioactivity test and subjected to ANOVA determinations by Tukey test (α = 0.05).
ii) Dose-responses of the OFM males to cellulose acetate nanofibers.
EAG responses of G. molesta males were evaluated after stimuli of CA nanofibers containing 0.02, 0.2, 1, 2, 5 and 10% (w/v) synthetic sex pheromone. The EAG dose-response effect was observed after stimuli of the antennae by 2 cmβ cut pieces nanofibers having different concentrations and offered separately from the lowest to the highest dose. The bioassays data were subjected to ANOVA determinations by Tukey test (α = 0.05).
iii) EAG responses of OFM males to different polymer nanofibers exposed at controled conditions for until 5 weeks.
The different polymers nanofibers containing 0.2% OFM pheromone and without pheromone were exposed in a controlled environment (25°C, 14 h photoperiod, 70% RH) for up to five weeks nanofibers: 1) Cellulose acetate; 2) PVAc/PVP; 3) PCL+THF; 4) PCL+DCM. The electrophysiological responses of G. molesta males were compared in the same polymer nanofibers containing pheromone between different exposure times and control (not exposed). The bioassays data were subjected to ANOVA determinations by Tukey test (α = 0.05).
iv) Attractiveness of adult G. molesta to PVAc and PCL nanofibers containing synthetic OFM sex pheromone in field conditions.
The nanofibers containing the OFM sex pheromone attractiveness was evaluated in peach and plum orchards located in the district of Charqueadas and Eldorado do Sul, (RS- Brazil). Delta traps were baited with PVAc/PVP blend and PCL nanofibers, produced with 2 different solvents, measuring 4 cmβ, both made in the electrospinning with containing 10% OFM synthetic sex pheromone additivated with antioxidant and UV protector. Delta traps were also for means of comparison, baited with 2 different controls: nanofibers without pheromone, as negative controls; and commercial bait (Isca Lure Grafolita®), as positive control in order to evaluate the presence of the oriental fruit moth inner the orchards. The nanofibers were placed inside screened sachets and hanged on the inside top of the traps. These were installed in the plants branches at an approximate height of 1.70 m above the ground and spaced 30 meters apart. The five treatments were arranged in randomized blocks within areas of approximately 0.5 hectares, and had eight repetitions. There were two periods of installation (11/01/11 and 01/03/12), from which the attractive field remained for 30 days, and weekly number of registered individuals of G. molesta captured. The bioassays data were subjected to Kruskal-Wallis test (Student-Newman; α = 0.05) using the BioEstat 5.0 statistics program (Ayres et al. 2007).
v) Quantify the pheromone in PVAc and PCL nanofibers by gas chromatography.
Oriental fruit moth (OFM) pheromone (Bedoukian Research, Danbury, CT) was used for most of the lab and field tests in this research. This pheromone was chosen because synthetic OFM pheromone is readily available. Synthetic OFM pheromone is a mixture of (Z)-8-dodecen-1-yl acetate, (E)-8-dodecen-1-yl acetate, and (Z)-8-dodecen-1-ol, in a 93:6:1 ratio. The amount of pheromone in the samples was calculated by gas chromatography (GC) Dani model Master A, equipped with a flame photo-ionization detector (PID) and a megabore capillary column (DB-1. 30 m x 0.25 mm, 0.53 mm diameter, Agilent Technologies). The initial temperature of the injector was 250 °C and the detector was heated to 200 °C. After the injection of the sample, the temperature of the oven was maintained at 135 °C for 2 minutes and then heated to 200 °C with a margin of 10° C.min-1. The samples were analised in triplicate and the quantity of volume injected was 1 µL.
vi) Morphology of the nanofibers produced by scanning electron microscopy (SEM).
Images from SEM of the electrospun samples of PCL and PVAc nanofibers were performed on a JEOL JSM 6060 (JEOL Ltd.) electron microscope. The piece of aluminum foil containing the fibers was mounted on an aluminum stub and sputter-coated with 15 nm of gold for analysis. Three SEM photographs from every sample were used for analysis. The analysis was performed by measuring the diameter in every photograph 30 times. The software used for the measurements and statistical analysis was ImageJ 1.47x.
The all data were submitted to normality and homoscedasticity tests.
Results
i) Distribution of OFM pheromone in cellulose acetate nanofibers through electroantennographic bioassays.
There was no significant difference (P > 0.05) between the electrophysiological responses of G. molesta antennae male within the different portions coming from the different quadrants of the nanofibers. This result indicates that the synthetic sex pheromone of G. molesta was evenly distributed in all parts of the cellulose acetate nanofiber. That is an important find since the electrospinning is a random mat of nanofibers and validate the fact that we are using cut pieces of an entire mat.
ii) Dose-responses of the OFM males to cellulose acetate nanofibers.
The electrophysiological responses of G. molesta males subjected to stimuli with nanofibers containing different percentages of pheromone were statistically significant (F = 24.27, df = 6, P < 0.0001) (Fig. 1). The sizes of electrophysiological responses were similar among treatments nanofibers containing 0.02, 0.2 and 1% and between treatments containing 2, 5 and 10% pheromone. This result demonstrates the presence of pheromone in the nanofiber, whereas the antennae of G. molesta males demonstrated bioactivity to stimuli, increasing the size of EAG response as a function of the concentration of pheromone in the nanofiber.
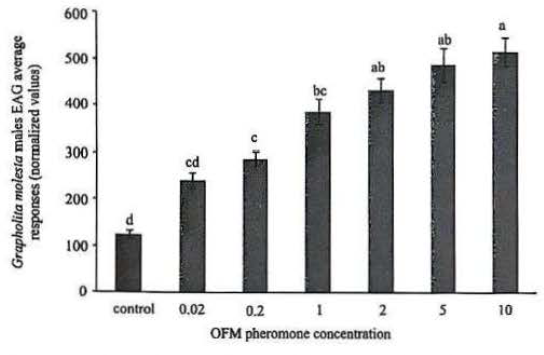
Grapholita molesta males electrophysiological (EAG) average (± SE) responses of the cellulose acetate nanofibers containing different dosages of sex pheromone attractant made from cellulose acetate. Means followed by different letters differ by Tukey test (P < 0.05).
iii) EAG responses of OFM males to different polymer nanofibers exposed at controled conditions for until 5 weeks.
The EAG responses of G. molesta males showed a significant difference only between 10% CA polymer in contrast with (8:2% PVAc/PVP polymers) and 10% PCL from DCM polymer, while the other had no difference (F = 3.05, df = 4, P > 0.05) (Fig. 2). When compared to their respective controls, the treatments containing pheromone produced significantly greater EAG responses, except for treatment 5 (6:4% SBS/ PVAC).
The formulation with the polymer 10% PCL with THF (Polymer 3) showed the greater retention of the pheromone producing electrophysiological responses equal to the new fiber until the third week of exposure, after being significantly equal to the control matrix. Thus, this formulation was one of those chosen for the preparation of nanofibers that were used in the experiments under field conditions. The formulation 10% PCL with DCM (Polymer 4) triggered electrophysiological responses similar to the new fiber only in the first week, decreased in relation to this, the other exposure times. With respect to 8:2% PVAc/ PVP (Polymer 2), although all treatments exhibited have lower response than the new fiber, these were stable and did not differ between itself from the first to the fifth week (Fig. 3). Furthermore, this was the only polymer which triggered electrophysiological responses were significantly higher after the fifth week of exposure compared to its control (no addition of pheromone in the fiber).
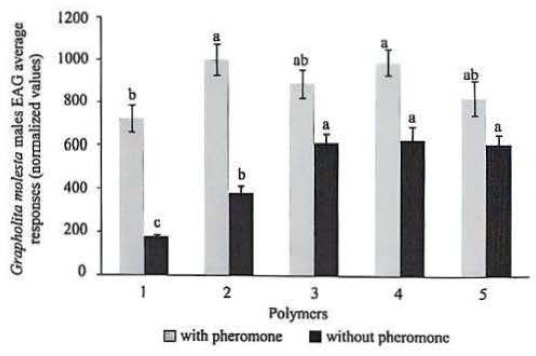
Electrophysiological (EAG) average (± SE) normalized responses of Grapholita molesta males antennas with different polymers nanofibers (gray bars with pheromone) and negative control (black bars – without pheromone). Different letters on the bars of the same color indicate significant differences by Tukey test (P < 0.05). Polymers (1) 10% cellulose acetate, (2) 8:2% PVAc/ PVP, (3) 10% PCL with THF; (4) 10% PCL with DCM, (5) 6:4% SBS/ PVAC.
For field conditions bioassays, only the nanofibers that showed the best characteristics of homogeneity, retention and gradual release of pheromone, under laboratory conditions, were selected (PVAc and PCL), while the others made from cellulose acetate and SBS/ PVAc were eliminated for these experiments.
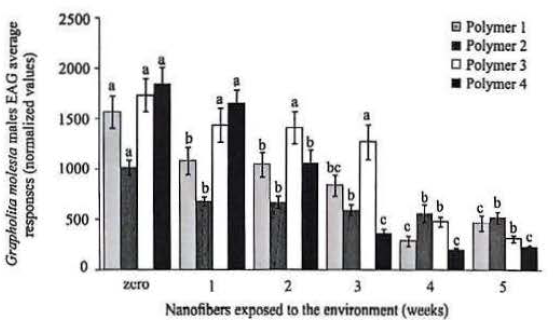
Electrophysiological (EAG) average (± SE) normalized responses of Grapholita molesta males antennae to pheromone-containing nanofibers made from different polymers and different exposure periods. Different letters on the same color bars indicate significant difference between the exposure time within the same polymer by Tukey test (P<0.05). Polymers (1) 10% cellulose acetate, (2) 8:2% PVAc/ PVP, (3) 10% PCL with THF; (4) 10% PCL with DCM.
iv) Attractiveness of adult G. molesta to PVAc and PCL nanofibers containing synthetic OFM sex pheromone in field conditions.
The Delta traps baited with nanofibers made of 8:2% PVAc/ PVP and 10% PCL with THF captured 13.75 ± 0.38 males and 9.78 ± 0.32 on average, respectively, did not differ from each other (P > 0.05). These averages were significantly higher than their respective controls (2.12 and 2.75 males on average), however, were lower than the average obtained with the commercially available bait (Isca Lure Grafolita®) (39.75 ± 0.51) (P<0.05, H = 26.75, dr = 4) (Fig. 4).
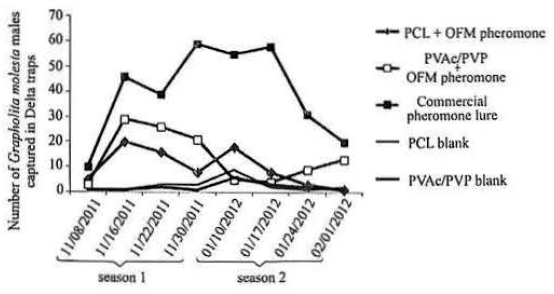
Population fluctuation of adult Grapholita molesta determined by the capture with a Delta traps baited with PCL and PVAc/ PVP, with and without pheromone (blank), and septum pheromone commercial orchards of peach and plum, in the period from November 2011 (season 1) and January to February 2012 (season 2) in Eldorado do Sul-RS and Charqueadas States of Brazil.
Mean number (± SD) of the diameter of three nanofibers formulation acquire by image SEM.
v) Quantify the pheromone in PVAc and PCL nanofibers.
Pheromone content in the tested nanofibers. The calibration curve showed a good repetition of the method (R2 = 0.99). After validating the GC measurement for measuring the pheromone content in nanofibers, the following nanofibers were evaluated: PVAc/ PVP, PCL with THF and PCL with DCM.
The three nanofibers samples with impregnated dodecenyl acetate were evaluated by GC analysis (Table 2). The PCL nanofibers produced using the solvent THF had the higher pheromone content. The different percentage of pheromone based on polymer mass found using the same polymer blend suggests that the solvent and co-solvent selection had importance to the encapsulation efficiency.
vi) Morphology of the nanofibers produced by scanning electron microscopy (SEM) and through this morphology.
Following are presented the images obtained for the samples analyzed in scanning electron microscopy. The figure 5 shows that the electronic microscope images of PCL nanofibers produced using the THF solvent had the format of beads-on-strings (A), while smoother fibers (B) were obtained with the pheromone addiction. The nanofibers containing the active (0.835 ± 0.4 mM), had higher mean diameter greater than the control (0.370 ± 0.2 mM) (Table 1). The increase in the average diameter of the nanofibers was statistically significant P < 0.05. Figures 5 and 6 show photomicrographs of fibers from polycaprolactone dissolved in organic solvent mixture. The same features were seen in figure 6 for the fibers made using the solvent DCM. In that second case, there were more beads than the first. The nanofibers containing the active (0.681 ± 0.3 mM), showed larger diameter compared to control (0.442 ± 0.1 mM). The increase of average diameter of nanofibers showed at table I was statistically significant (P<0.05).
On the other side, the nanofibers produced with PVAc/ PVP were smoother (Fig. 7). The fibers containing the active (1.217 ± 0.5 mM) had greater average diameter compared to control (1.630 ± 0.7 mM). This increase in diameter was also statistically significant (P<0.05) (Table 1).
Dodecenyl acetate content (%) of the three samples of each nanofibers gain by GC measurement.
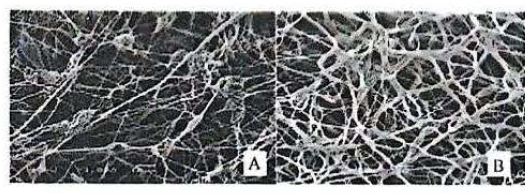
SEM images of nanofibers: 10% PCL with THF without the addition of active
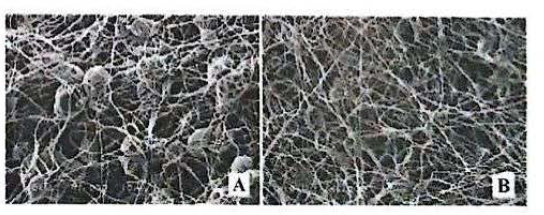
SEM images of nanofibers: 10% PCL with DMF without the addition of active
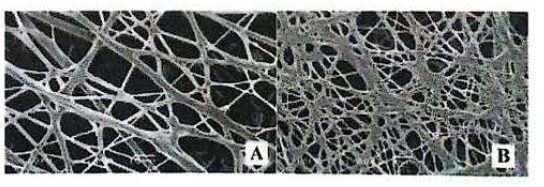
SEM images of nanofibers: 8:2% PVAC/PVP without the addition of active
Discussion
The present study was based upon the few papers using nanofiber as pheromone vehicles from the literature. The nanofibers polymer selection was firstly based on study carried out by Hellmann et al. (2009), where cellulose acetate film were tested in vitro and found to be an excellent vehicle for the transport of European grape berry moth pheromone Lobesia botrana due to features such as the ability to incorporate pheromone molecules and its rate of release for weeks straight. In the present study, the efficacy of incorporation and release of pheromone from nanofiber mats and its even distribution inside the matrixes was evaluated by the bioassays proposed. Pheromones impregnation dosages that caused major electrophysiological responses of G. molesta males concentrations were 2 to 10%.
Because of the short duration of the cellulose acetate nanofiber in the initial tests, new polymers and its blends were also tested. Because of the chemical similarity between PVAc and PCL, both esters, with the major G. molesta component pheromone, dodecenyl acetate, those polymers were chosen. Styrene-butadiene-styrene was selected because rubber septa are known dispenser for pheromones and that the longer carbon chain of the principal component-dodecenyl acctate- is non polar. Polyvinyl pyrrolidone was preferred for blending with PVAc to add polar group and an amphyphilic character for increase the solubilization of the dodecenol molecule, other pheromone component. Cellulose acetate polymer was less efficient than PVAc and PCL the bioassays performed in this study, when assessed in vivo by electrophysiological responses of insects, in respect to pheromonal retention, and also for environment exposed durability. So these polymers were selected for the following testing.
The polyvinyl acetate blend (PVAc/ PVP), despite a drop in electrophysiological responses when the fibers were exposed to environment, the response remained throughout the five weeks at the same rate of release. The blend PVAc/ PVP is extensively studied in the pharmaceutical industry due to the efficiency of drug release, are ideal for this reason the healing drug delivery, requiring a release and prolonged linear (Jannesari et al. 2011). Such behavior is regarded as one of the properties assigned to this amphiphilic blend. Since the (Z)-8-dodecenyl acetate molecule has polar and non-polar groups, the amphiphilic character match each of these groups, and this could be one of the reasons of the controlled release achieved.
The polymer PCL showed the longest pheromone retention period even after exposure to environment, which is an important feature for agricultural purposes. This polymer is biodegradable polyester, aliphatic, what is also a desirable feature when applications to agriculture are in concern. Aliphatic polyesters have been employed as slow-release agents in agriculture for a long time (Sinclair 1973). They degrade into natural materials harmless to the environment while slowly releasing the encapsulated herbicides or pesticides. Because of these properties, two different formulations were tested, having two different solvent mixes, DCM or THF blended with DMF solvent. These two formulations have different impregnating efficiency when GC measured, as will be discussed below.
Analyzing the electronic microscope images, the PCL nanofibers were all beads-on-strings, although the fibers became smoother with the pheromone addiction. In the images 6A and 6B, it is observed that change. The reduction of the beads and reduction of fiber diameter are two morphologic characteristics that result from the increased polarity of the electrospinning solution (Tungprapa et al. 2007). The solvent used was tetrahydrofuran, an aprotic solvent with dielectric constant (ε) 7.58 to 20 °C. The nanofibers containing the active (0.835 ± 0.4 mM), had higher mean diameter greater than the control (0.370±0.2 mM). The increase in the average diameter of the nanofibers was statistically significant P < 0.05. The nanofibers in the form of bead-in-strings were becoming flat with the incorporation of the active ingredient (Fig. 5).
The same features were seen in figure 6 for the fiber made from polycaprolactone dissolved in DCM, although with more beads than the based on THF. The solvent dichloromethane is an aprotic solvent with a higher dielectric constant (ε) 9.08 to 20 °C. The nanofibers containing the active (0.681 ± 0.3 mM), showed larger diameter compared to control (0.442 ± 0.1 mM) (Table 1). The increase of average diameter of nanofibers was statistically significant (P<0.05).
In the third case, the nanofibers produced with PVAc/ PVP were smoother (Fig. 8). The fibers containing the active (1.217 ± 0.5 mM) had greater average diameter compared to control (1.630 ± 0.7 mM). This increase in diameter was also statistically significant (P < 0.05). Smoother, or uniform, fibers with fewer beads are desirable, since they indicate that they are more resistant and more compatible (Doshi and Reneker 1995). One way to reduce the amount of beads is to increase solution conductivity (Ramakrishna et al. 2005), which is obtained by selection of more polar solvents by the addition of salts to the solution, or by adding an active ingredient with polarity, which seems to have been the case under discussion. The smoother fiber should have occurred due to the high dielectric constant of the solution, higher than in the previous cases cited solvents (dielectric constant of 80 and 24, at 20 °C, for the cases of water and ethanol).
In the PVAc/ PVP fibers, there was also interweaving of the fibers, which is desirable because the increased resistance of the resulting film, but it is noted fusion of the fibers, which may indicate a case of wet-spinning, when the solvent is not evaporated completely in route to the collector metal (Tang et al. 2009). That signifies that the solvent has boiling point higher than other solvents used and not evaporated completely until reaching the metal collector. This high boiling point of the solvent used is the reason for the case of wet-spinning. In this case, increasing the distance collector or a change in concentration of each solvent can optimize the solution. This was not done because it standardized the distance between the needle and collector for 3 samples and their respective controls.
Yang et al. (2004) investigated the effects of solvents, including DCM, EtOH, DMF and mixtures of solvents, the morphology and diameter of PVP nanofibers obtained by ES. In solvent systems simple to 4% by weight of PVP, the nanofibers electrospining from ethanol were smooth, whereas the nanofibers from DMF and DCM exhibited beads-on-strings structure. In systems solvent mixtures, a mixture of ethanol/ DMF, with a mass ratio of 50/50 was obtained as a good solvent for PVP producing nanofibers with diameters as small as 20 nm. In a solvent mixture such as PVP concentration was increased from 4 to 8% by weight, the fiber diameter increased from 20 to 50 nm, no significant change in size distribution.
It is well known that physicochemical properties such as dielectric constant, boiling point, surface tension and viscosity of the polymer solution are important factors to define the morphological characteristics of the nanofibers generated. The polymer concentration and molecular weight of the polymer is also related to the latter property. Thin and uniform nanofibers were produced by electrospinning by authors from PVP solutions in solvents with a dielectric constant significantly high, low surfoce tcnsion and low viscosity (Chuangchote et al. 2009).
The field exposurc experimcnt demonstrated that although the PVAc/ PVP and tested polycster 10% PCL with THF had statistically superior capture than their rcspcct.ivc controls, it was lower than that obtaincd with thc commcrcial rubbcr scptum. Thc rcsults also showcd thal highcr phcromonc contcnt is rclatcd to highcr capture. Thc rcason may be lhal thc mass of thc commcrcial scplum is much highcr lhan thc nanofiber.
l11us, thcsc rcsults indicatc lhat adjustmcnls are nccdcd to maximizc thc ability to capture thc ficld wilh the use oflhcse polymcrs, as well as frnthcr investigation of othcr polymcrs cnpabl e of use in thc manufacture of nanofibers used as discpcrsing phcromones.
Thc res u lts of chromatographic analysis showcd a grealer phcromone impregnation of PCL with TJ-1 F nanofibcrs (Table 1). Thcsc nanofibcrs had thc greater pheromone persistence in thc environmcnt. The phcromone content was also higher than that found in PVAc/ PVP blcnd. The validation of the technique for pheromone dosage in nanofibers was obtained after the fie ld trials that was the reason we could not use fibers with similar content of pheromone. For future studies the pheromone content should be nonnalized in order to detennine what are the predominant factors involved in the controlled release of pheromone.
The results oflable 1 and SEM images shows that there is a relationship between the appearance of the nanofibers, the pheromone content in each fiber and the controlled delivery of the nanofibers. Other studies should be done to assess the relationship between the morphology of the fibers, the polymer utilized and the pheromone release. The polymer, solvent and dosage incorporated in the nanofibers are importanl to the controlled delivery. Understand these factors is importa.nt in order to develop better pheromone dispensers.
Footnotes
Acknowledgements
The authors are grateful for the financia! support provided by Brazil's National Council for Scientific and Technological Development (CNPq), FAPERGS (Fundai;:ao de Amparo a Pesquisa do Estado do Río Grande do Sul) and FINEP (Financiadora de Estudo e Projetos).
