Abstract
Telenomus remus and Trichogramma spp. (Hymenoptera: Platygastridae, Trichogrammatidae) are known egg parasitoids of the fall armyworm (FAW), Spodoptera frugiperda (Lepidoptera: Noctuidae) in Brazil. Parasitism of trichogrammatid on eggs of S. frugiperda has been relatively common, whereas the occurrence of T. remus in Brazilian crops has rarely been reported. Consequently, literature that gives insights about the interspecific competition among individuals of T. remus and Trichogramma spp. in the field is very scarce. Here we report the simultaneous occurrence of T. remus. Trichogramma pretiosum, and Tr: atopovirilia on eggs of the FAW in maize (Piracicaba, São Paulo, Brazil). Our findings imply that these three egg parasitoid species were engaged in exploitative competition (indirect interactions in which the depletion of resources by one species prevents the other access to them) toward eggs of S. frugiperda. Moreover. given the emergence of combinations of two and three of these parasitoid species from a single egg mass, our data strongly suggest that T. remus, Tr: pretiosum and Tr: atopovirilia were also engaged in interference competition (direct interactions among adults and/or immature individuals resulting in behavioral changes or even death of individuals). Both exploitative and interference competition may affect the population dynamics of parasitoids, with consequences for the biological control of pests. Hence, the present findings highlight the importance of determining the outcome of competition among T. remus, Tr. pretiosum and Tr: atopovirilia toward eggs of S. frugiperda in laboratory and field studies.
Introduction
The fall armyworm (FAW), Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae), is a key pest of maize in Brazil, as well as the greater Americas. Although its control is based mostly on pesticide sprayings, its biological control by means of augmentative releases of egg parasitoids is promising (Cave 2000; Beserra et al. 2002; Parra and Zucchi 2004; Beserra and Parra 2005). The goal of this work was to collect and identify egg parasitoids of S. frugiperda in maize in order to start laboratory cultures and perform studies to assess their competitiveness and potential as biological controllers of the FAW.
Material and methods
We conducted a field survey on May 29th (late autumn) 2010. in a maize plot (1 ha; plants with ca. 90 days after emergence) located at Piracicaba, São Paulo, Brazil (22°42'18.57"S 47°38'6.37"W). Fifty-two egg masses (< 18h, ca. 300 eggs) of S. frugiperda were randomly scattered in the plot, with a minimal distance of 10 m from each other. Egg masses were produced in laboratory according to Da Silva and Parra (2013). In an attempt to protect the eggs from predators and direct sunlight, we taped the egg masses inside a collection device, which consisted of a white disposable cup (50 mL) with a punctured bottom and a cotton string (50 cm) passed through it. A knot was made at one extremity of the string in order to prevent the detachment of the cup, while the other extremity was tied to a maize leaf (Fig. 1). The collection devices were recovered two days later and the egg masses were introduced individually into glass tubes (2 x 8 cm) and maintained in incubators (24 ± 1 °C, 70% RH, 14L:10D photoperiod) until parasitoid emergence. In the meantime, caterpillars from non-parasitized eggs were eliminated on a daily basis to prevent injury to the parasitized eggs. Adult females were then individualized, multiplied on eggs of S. frugiperda, and had some adult individuals of their offspring killed in 70% alcohol. The male individuals were set on glass slides with Hoyer's medium and identified based on the keys developed by Querino and Zucchi (2005), Johnson (1984), Nixon (1937), Nixon (1940) and Chou (1987).

Method used for collecting parasitized eggs of Spodoptera frugiperda in field.
Results and discussion
None of the fifty-two S. frugiperda egg masses exposed to parasitism in field showed any signal of predation (missing eggs or residues of predated eggs). Three species of egg parasitoids were identified: (1) Trichogramma atopovirilia Oatman & Platner, (2) Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae), and (3) Telenomus remus Nixon (Hymenoptera: Platygastridae). The latter species emerged from all 52 egg masses exposed in field, implying that both trichogrammatids always had to compete with the platygastrid for the same egg masses. Trichogramma pretiosum and Tr: atopovirilia emerged from 19 and 3 egg masses, respectively. Surprisingly, a combination of the three egg parasitoid species was found in one out of the 52 exposed egg masses (Fig. 2). The fact that T. remus emerged from every egg mass exposed in field suggests a competitive dominance in relation to both trichogrammatids. Facing such a high dominance, Tr: atopovirilia and Tr. pretiosum simply may have not been able to emerge from T. remus-free egg masses. Alternatively, the fact that the emergences of Ti: atopovirilia and Tr. pretiosum were invariably associated with the emergence of T. remus might be an indication that both trichogrammatids practice hyperparasitism on T. remus. It has been demonstrated that Tr: pretiosum can develop as facultative hyperparasitoid of T. sphingi (Strand and Vinson, 1984), although Carneiro and Fernandes (2012) found no evidence of emergence of Tr: pretiosum from S. frugiperda eggs previously parasitized by T. remus.
Telenomus remus, Tr. atopovirilia and Tr: pretiosum have been the most cited egg parasitoids of the FAW in Latin America (Cave 2000; Beserra et al. 2002; Parra and Zucchi 2004; Beserra and Parra 2005). Although Telenomus remus was introduced in Brazil 29 years ago, reports on its natural occurrence in field have been very rare, while wild populations of Tr: pretiosum and/or Tr. atopovirilia are often reported (Beserra et al. 2002; Bueno et al. 2009; Bueno et al. 2010). Out of these three species, T. remus has been considered the best candidate for augmentative biological control programs due to their large size, robustness, and ability to reach the inner layers of the FAW egg masses (Cave 2000). Indeed, laboratory studies have demonstrated that T. remus parasitizes eggs of S. frugiperda faster than Tr: pretiosum and, when both species are put together in an arena containing FAW eggs, the majority of the adults emerged are of T. remus (Carneiro and Fernandes 2012). Nevertheless, there are no actual studies exploring the interference and the exploitative competitions among T. remus, Tr. atoporivilia and Tr. pretiosum in field conditions.
According to Park (1954), a serious study of interspecific competition should start with proof that competition exists. Our report shows the coexistence of these three egg parasitoid species in the same environment and using the same host species, which implies that they are engaged in a competitive interaction with each other. Other species of Telenomus and Trichogramma have been found sharing host species at the same time/space in Japan and Spain (Cabello 1989; Hirose et al. 1968a; Hirose et al. 1968b). However, we are unaware of previous reports of the coexistence of T. remus, Tr: atopovirilia and Tr: pretiosum in the same environment and especially on the same egg mass. These results are not only evidence that T. remus, Tr. atopovirilia and Tr. pretiosum are engaged in exploitative competition (indirect interaction, where the depletion of resources by one species prevents the other access to it) towards S. frugiperda eggs, but our findings also strongly suggest that the three parasitoids are also displaying interference competition (direct interactions among adults and/or immature individuals resulting in behavioral changes or even death of individuals). Both kinds of competition, combined or not, may have profound implications in the population dynamics (Ricklefs and Miller 2000) of these egg parasitoids and, must be matter of further investigations. Additionally, recent studies (Penaflor et al. 2011a; b) showed that oviposition by S. frugiperda on maize leaves changes the volatile emissions of maize plants, and the authors hypothesize that such changes might constitute chemical signals that attract egg parasitoids of FAW. Our study showed that such signals are not critical for T. remus, Tr. atopovirilia and Tr: pretiosum to successfully find eggs of S. frugiperda in the field, since we attached the egg masses to the collection devices instead of inducing female moths to lay eggs directly on the maize leaves, and even so the parasitoids were able to find the hosts despite the lack of chemical cues released from the maize plants.
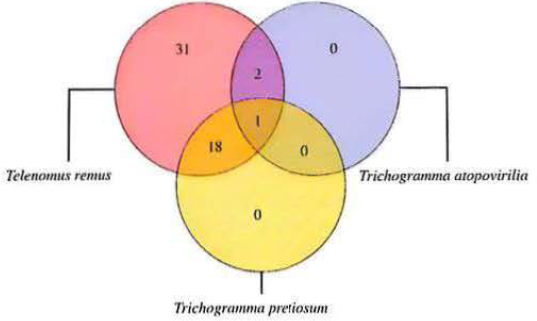
Numbcr of Spodoptera frugiperda egg masses parusitizcd by Telenomus remus, Trichogramma atopovirilia and Tr: pretiosum, in a maize field located in Piracicaba, São Paulo. Brazil.
Here we reported the spontaneous occurrence of T. remus, Tr. pretiosum and Tr: atopovirilia, three egg parasitoids of S. frugiperda, in maize, implying that these wasp species were engaged in exploitative competition and likely involved in interference competition towards eggs of S. frugiperda in field. Since both kinds of competition may affect the population dynamics of parasitoids with consequences to the biological control of pests, our study highlights the importance of determining the outcome of competition among T. remus, Tr. pretiosum and Tr: atopovirilia towards eggs of S. frugiperda in laboratory and field studies.
Footnotes
Acknowledgements
We are grateful to Arodi Prado for collaborating with the parasitoid collection in the field; to Mariana Graciano Zério for collaborating with the maintenance of insect colonies in the laboratory; and to Alyssa De La Rosa for the English revision. The first author received a scholarship from Fundação de Amparo à Pesquisa do Estado de São Paulo (process 2007/07054-8). This study was also supported by Secretaría de Ciencia y Técnica de la Universidad Nacional de La Plata, Argentina, processes 11N744 and 11A263 (Dras. Loiacono and Margaria).
