Abstract
Caryocar brasiliense (Caryocaraceae), a tree characteristic of e Cerrado, is widely distributed and can reach a height of 10 meters with a six-meter-wide canopy. The objective of this study was to study the distribution of sucking insects (Hemiptera) and their natural enemies in the canopies of C. brasiliense trees in the Brazilian Cerrado. One rare, nine common, and one constant species of sucking insects and three rare, seven common, and four constant species of natural enemies were observed on C. brasiliense trees. The diversity, number of individuals, and species of Hemiptera and their natural enemies were similar in all four cardinal directions of branches of host trees. Abundance of the natural predators Crematogaster sp. (Hymenoptera: Formicidae) and Zelus armillatus (Hemiptera: Reduviidae) was highest on leaves of the east and north sides of the C. brasiliense trees, respectively. A large number of Crematogaster sp. was observed on C. brasiliense, with a predominance of Dikrella caryocar n. sp. (Hemiptera: Cicadellidae) and Pseudoccocus sp. (Hemiptera: Pseudococcidae) also being observed. The predators Trybonia sp. (Thysanoptera: Phlaeothripidae) and Chrysoperla sp. (Neuroptera: Chrysopidae) showed the highest numbers, while the number of D. caryocar n. sp. and Aphis gossypii (Aphididae) decreased, respectively, in comparison to the control. An increase in the number of sucking insects also increased the number of their natural enemies, and this differential distribution negatively influenced sucking insects. The speed and direction of wind may have affected the distribution of sucking insects on different sides of C. brasiliense trees, as higher populations were found on the sides without prevailing winds.
Introduction
The Brazilian savanna Cerrado, which is characterized by a high diversity and endemism of plants and insects (Bridgewater et al. 2004), occupies approximately 23% of the Brazilian territory (Da Silva and Bates 2002). As a result of increasing threats to its biodiversity, this biome has been designated a biodiversity hotspot (Myers et al. 2000). The Cerrado is used primarily for grain and cattle production (Aguiar and Camargo 2004), in addition to forest cultivation with exotic species, primarily eucalyptus (Zanuncio et al. 2002). Government incentives have facilitated the deforestation of the Cerrado, and its vegetation has been reduced to 20% of the original amount (Klink and Machado 2005). It has a complex mosaic of phytophysiognomies that range from open savanna ("campo limpo") to tall and woody forests of 10-15 meters high, called "cerradão" (Oliveira and Marquis 2002). Large patches of rich Cerrado in southeastern Brazil are used for agriculture (primarily soybean and sugar cane), cattle farms and cities (urbanization), with similar uses to those of the Montes Claros region in northern Minas Gerais State.
Caryocar brasiliense Camb. (Caryocaraceae) or "pequí" is a characteristic tree of the Cerrado. It has a wide distribution (Brandão and Gavilanes 1992; Bridgewater et al. 2004; Leite et al. 2006a) and can reach a height of 10 meters with a six- meter-wide canopy (Leite et al. 2006a, 2011a, 2012a). Its fruits have an internal mesocarp that is rich in oil, vitamins, and proteins as well as compounds of medicinal importance. The fruits are used as food, in production of cosmetics and lubricants, and in the pharmaceutical industry (Segall et al. 2005; Ferreira and Junqueira 2007; Garcia et al. 2007; Khouri et al. 2007). Besides, they represent the main income source of many communities that remain in the deforested areas of the Brazilian Cerrado (Leite et al. 2006a). Fruit collectors favor leaves, flowers and fruits, and sucking insects (Hemiptera) with a high number of species incur higher damage on isolated trees (Freitas and Oliveira 1996; Oliveira 1997; Boiça et al. 2004; Leite et al. 2009, 2011a,b,c,d,e, 2012a) and in tree seedlings (Leite et al. 2006b). It is necessary to understand the ecology of the insects on this valuable tree, which both occurs naturally and is planted in the Brazilian Cerrado.
The position of branches on a tree affects the abundance of insects due to 1) the wind direction (Feng et al. 2005; Leite et al. 2011c, d, e), which causes leaf desiccation and the fall of flowers and fruits (Leite et al. 2006a); 2) the sun exposure, which may influence the quality of the host plant tissues (Unsicker and Mody 2005); 3) the creation of microclimates, and 4) the impact of herbivores, which prefers the parts of plants that have the smallest presence of natural enemies (enemy-free space) (Unsicker and Mody 2005). The identification of potential pests and their spatial distribution on the crown of forest and agriculture plants is important for sampling plans (Nichols-Orians 1991; Casas and Aiuja 1997; Villanueva and Childers 2005; Evans and Gregoire 2007). Sucking insects are abundant among the fauna in the Cerrado (Pinheiro et al. 2002). For the present study, they were reared on C. brasiliense to support management programs for pests of this plant and to study their spatial distribution at tree level. The objective was to study the distribution of sucking insects (Hemiptera) and their natural enemies in the canopy of C. brasiliense trees in the Brazilian Cerrado.
Material and methods
The insects were collected from the leaves, flowers and fruits with tweezers, brushes and aspirators and were preserved for identification in vials with 70% alcohol and 30% water.
A total of 900 leaves (samples), 225 flowers (samples) from July to September and 240 fruits (samples) from Sep- tember to January of C. brasiliense were evaluated per cardinal orientation.
The correlations between the diversity index, number of individuals, and species of sucking insects and the abundance and species richness of natural enemies (ants, predator thrips, bugs, spiders, ladybirds, and green lacewings) on the numbers of sucking insects were submitted to analysis of variance (ANOVA) (P < 0.05), using a simple regression analysis (P < 0.05), and the curves were adjusted for the quadratic function, as required. The effect of the cardinal orientation of the branches on the ecological indices, the number of individuals per species of sucking insects and the natural enemies was tested with ANOVA (P < 0.05) and Tukey's test (P < 0.05).
Results
One rare, nine commons, and one constant species of sucking insects and three rare, seven common, and four constant species of natural enemies were found on the C. brasiliense trees (Table 1). Using Hill's diversity index, the number of individuals and species of sucking insects and their natural enemies were similar (P > 0.05) among the cardinal sides of C. brasiliense trees (Table 2). There were collected 988 and 1191 total individuals of natural enemies and sucking insects, respectively, and 13 species of natural enemies plus spiders (19 species) and 10 species of sucking insects.
The abundance of most of the sucking species on leaves, flowers, and fruits of C. brasiliense trees were similar between the cardinal points (Table 3). The predators, Crematogaster sp. (Hymenoptera: Formicidae) (df = 72, F = 4.269, P = 0.00785) and Zelus armillatus (Lep. and Servi, 1825) (Hemiptera: Reduviidae) (df = 72, F = 2.794, P = 0.04633) presented the highest abundance on leaves from the east and north sides of C. brasiliense trees, respectively (Table 3).
The ant Crematogaster sp. was more frequent on C. brasi- liense trees that had larger numbers of Dikrella caryocar n. sp. (Coelho, Leite and Da Silva, 2014) (Hemiptera: Cicadel- lidae) and Pseudoccocus sp. (Hemiptera: Pseudococcidae). The numbers of the predators Trybonia sp. (Thysanoptera: Phlaeothripidae) and Chrysoperla sp. (Neuroptera: Chrysopi- dae) was inversely correlated to those of D. caryocar n. sp. and Aphis gossypii (Glover, 1877) (Aphididae), respectively. This pattern was also observed for the number of individuals of sucking insects and those of their natural enemies (Fig. 1).
Order, family, species feeding behavior and abundance of insects during the day-time on Caryocar brasiliense trees. Montes Claros, Minas Gerais State, Brazil.
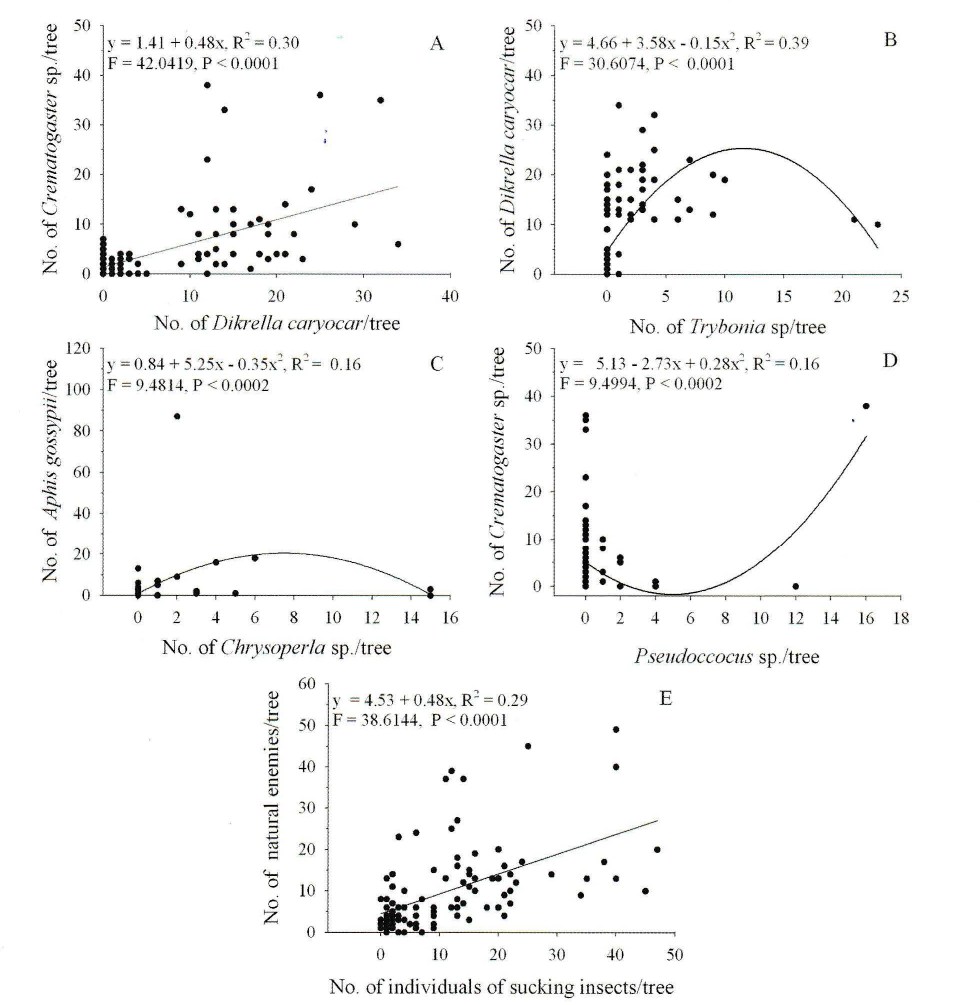
Effect of numbers (mean)/tree of Dikrella caryocar n. sp. (A) and Pseudoccocus sp. (D) on those of Crematogaster sp. B. Numbers of Trybonia sp. on those of Dikrella caryocar n. sp. C. Numbers of Chrysoperla sp. on those of Aphis gossy- pii. E. Numbers of individuals of sucking insects on those of natural enemies. Montes Claros, Minas Gerais State, Brazil.
Hill's diversity index, abundance, and richness of Hemiptera and natural enemies/tree (average + standard error) on different cardinal points of Caryocar brasiliense. Montes Claros, Minas Gerais State, Brazil.
Number of sucking insects (Hemiptera) and natural enemies (mean ± standard error) on leaves, flowers and fruits/tree in different sides of Caryocar brasiliense. Montes Claros, Minas Gerais State, Brazil.
Means fo llowed by thc same letter (± standard error) per line did not differ by the test of Tukey (P < 0.05). n.s. = non significant by ANOVA. L = leaves, Fl = flowers, and Fr = fruits.
Discussion
The similar diversity index and number of individuals and species of Hemiptera and their natural enemies on the different sides of C. brasiliense may be explained by the low number of constant species of sucking insects (9.10%) and their natural enemies (28.6%) on this tree. In other words, most of the species were found at low population densities. It was not possible to detect significant differences among the number of individuals and diversity on different sides in this plant, which may have been due to the higher floristic diversity of the cerrado area. This varies with the insect group considered because the environmental complexity and host plants can influence the diversity of the herbivore arthropods and their natural enemies (Coyle et al. 2005; Espírito Santo et al. 2007; Lazo et al. 2007; Leite et al. 2011a, 2012a). In less complex environments, the number of species associated with a given host species may be lower and the abundance of each species is generally higher, thereby increasing the likelihood that herbivores that feed on economically valuable plants will become significant pest (Gratton and Denno 2003; Coyle et al. 2005; Lazo et al. 2007).
Althrough we did not detect significant differences, the tendency (P > 0.05) for a higher diversity index value and more individuals and species of Hemiptera on the west side of C. brasiliense trees may be explained by 1) a prevailing wind from the northeast and eastern (Leite et al. 2006a, 2009, 2011c, d, e), 2) more sunlight on the north side in the Southern Hemisphere (Vianello and Alves 2000) and 3) avoiding ants and predators, which presented higher numbers in the east and north sides of C. brasiliense trees, respectively. The direction of the wind may have influenced the dispersion of insects (Pathak et al. 1999; Tixier et al. 2000; Auslander et al. 2003; Schooley and Wiens 2003; Feng et al. 2004, 2005; Leite et al. 2009, 2011c, d, e) and pollination. A wind speed greater 2.0 m/seconds reduced the visits of bees to flowers (Dutra and Machado 2001). The desiccant effect of wind is higher in regions that have a low relative humidity and high temperature, which is typical of the cerrado vegetation in the semi-arid north of Minas Gerais State, Brazil, and can reduce the fruit production and photosynthesis and cause the malformation or fall of flowers and fruits (Leite et al. 2006a), both of which influence insect populations. The higher desiccant effect of the wind on the east and north sides of C. brasiliense trees may be explained by the sun exposure influencing the quality of the host plants for insects (Fernandes 1990; Unsicker and Mody 2005). The number of species and individuals of insects was lower on the sunny side of the Australian Melaleuca trees (Richardson et al. 1999).
The lower populations of ants and predators may explain the tendency (P > 0.05) of higher ecological parameters of sucking insects on the west sides of C. brasiliense trees as spaces that are free of natural enemies. This could make the colonization by sucking insects on north and east sides of the C. brasiliense canopy difficult. Predators can respond to a local increase in vegetation complexity and alternative prey with higher efficiency against herbivores (Auslander et al. 2003).
The greater number of Trybonia sp. and Chrysoperla sp. reduced the abundance of D. caryocar n. sp. and A. gossypii, respectively, on the C. brasiliense trees, but the higher populations of Crematogaster sp. increased the abundance of D. caryocar n. sp. and Pseudoccocus sp. Ants and other predators reduced the infestation of Eunica bechina Talbot 1928 (Lepidoptera: Nymphalidae) and Edessa rufomarginata (De Geer, 1773) (Hemiptera: Pentatomidae), Prodiplosis floricola Felt 1907 (Diptera: Cecidomyiidae), petiole gall insects (Hymenoptera: Chalcidoidea) (Freitas and Oliveira 1996; Oliveira 1997), and beetle defoliators (Leite et al. 2012b) on this plant. Green lacewings are important predators of aphids as A. gossypii (Leite et al., 2006b, 2012c). The positive associations among ants, leafhoppers and aphids are common food-for-protection mutualisms (Moya-Raygoza and Larsen, 2014; Shik et al. 2014). The east side of the trees was apparently more unfavorable to insect herbivores in the African Savanna, as greater leaf damage occurred on the west and north sides. This finding can be explained by the species-specific reactions (plants and herbivores) and the biotic environment conditions (Unsicker and Mody 2005). The distribution of herbivores could also reflect the avoidance of predators in addition to reactions to the chemical composition of host plants or the microclimate (Unsicker and Mody 2005).
The sucking insect species that have greater potential to become pests in commercial C. brasiliense plantations under natural conditions are Aethalium reticulatum L., 1767 (Aetalionidae), E. rufomarginata, D. caryocar n. sp. and A. gossypii due to of their high abundance and status as a common species. Aethalium reticulatum and E. rufomarginata are pests in C. brasiliense trees (Leite et al. 2012c), and D. caryocar n. sp. and A. gossypii are pests in seedling in this culture (Leite et al. 2006b). Predators affected these insects on this plant. The high diversity of sucking insects shows the necessity of studying the population dynamics of these organisms in the arboreal systems of the Brazilian savanna.
Footnotes
Acknowledgements
We thank A.D. Brescovit (Instituto Butantã) (Aracnidae), A.M. Bello (Coleoptera), I.C. Nascimento (EMBRAPA- ILHÉUS-Centro de Pesquisas do Cacau, CEPLAC, Itabuna, BA) (Formicidae), and R.C. Monteiro (Thysanoptera) for the identification of the specimens, and C. Barbosa, O.M. da Silva and F.M. Ruas for supplying climate data. We also thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado de Minas Gerais and Secretaria de Ciência e Tecnologia do Estado de Minas Gerais for financial support.
