Abstract
Among the pests that cause reduction on common bean yield, the whitefly Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) stands out. The insect causes direct damages due to its feeding on plants and indirect damages by the sugary excretion of honeydew, providing the development of the sooty mould. In addition this species is an important vector of virus such as the golden bean mosaic virus. Thus, this work aimed to study the non-preference for oviposition and antibiosis in bean cultivars to the whitefly. Experiments were carried out in a greenhouse at Jaboticabal, SP, Brazil, from January to March 2012. The following cultivars were used in the assays: IAC-Centauro, IAC-Una, IAC-Formoso, IAPAR-81, IPR-Eldorado, IPR-Siriri (all resistant); and Pérola and IAC-Harmonia (both susceptible), previously screened from field experiments. Cultivars IAC-Harmonia, IPR-Eldorado, IAPAR-81 and IPR-Siriri were the least preferred for oviposition; and the cultivar IAC-Harmonia extended the whitefly life cycle, expressing non-preference for feeding and or/antibiosis-type resistance.
Introduction
Among the pest insects that cause economical losses to com-mon bean (Phaseolus vulgaris L.), the whitefly Bemisia tabaci (Genn., 1889) biotype B (Hemiptera: Aleyrodidae) stands out due to its direct damage by feeding on the phloem, weakening the plant by sucking nutrients, in addition to in-jection of toxins, resulting in physiological problems in bean plants, as well as indirect damages through the excretion of honeydew. This excretion serves as substrate for the growth of saprophyte fungi from genus Capnodium (sooty mould) on leaves, flowers and fruits, preventing gas exchange, such as respiration and photosynthesis and hence decreasing yield. It also complicates pesticide action, resulting in higher produc-tion costs (Lima 2001).
However, the most serious damage caused by B. tabaci biotype B is concerning to virus transmission, such as the golden bean mosaic virus (GBMV), one of the major pro-blems for common bean crop in Latin America. This disease causes economical losses that may range from 30 to 100% depending on the cultivation, plant growth stage, population level of the vector, presence of alternative hosts and environ-mental conditions (Salguero 1993).
Use of pesticides is the main control method adopted by bean growers. However, Horowitz and Ishaaya (1995) repor-ted that in several cases, treatment with conventional insecti-cides is not efficient mainly because adults are located in the lower surface of leaves and due to fast resistance deployment against the active ingredients. Moreover, successive utiliza-tion of pesticides may unbalance the environment and elimi-nate beneficial arthropods (Prabhaker et al. 1985).
The intrinsic ability that some genotypes possess in com-parison to others from the same species, in order to obtain higher yield and/or quality under the same attack of pest po-pulation in equal conditions is known as host plant resistance (HPR) (Lara 1991). Screening of plants resistant to B. tabaci biotype B, transmitting geminivirus or causing physiologi-cal disorders in cultivated plants, represents an important research tool aiming to diminish injuries and losses caused by this insect (Mcauslane et al. 1994). For HPR use, it is necessary to know morphological and physiological traits of the plant and the insect behavior and biology as well as its relationship with the plant host. These factors are indispen-sable to the host response to pest action, determining plant resistance or susceptibility (Campos 2003).
Resistance categories described by Painter (1951) were related as non-preference, antibiosis and tolerance. Non-preference or antixenosis refers to insect behavioral aspects on the plant, which may be non-preference for feeding, ovi-position or shelter, and the two formers are the most studied. Regarding antibiosis, direct lethal effects are verified on the different stages of the insect, whereas tolerance is defined as the plant ability to withstand insect attack without significant yield reduction.
Several studies have pointed out resistant cultivars that can be utilized by the growers, thus minimizing B. tabaci biotype B attack on bean crop, especially non-preference for oviposition- and antibiosis-type resistance (Toscano et al. 2002; Campos et al. 2005; Paron and Lara 2005; Jesus et al. 2010; Rodrigues et al. 2012a). In addition, HPR can be as-sociated harmoniously with other control methods, accord-ing to studies conducted by Jesus et al. (2009), Costa et al. (2010) and Janini et al. (2011).
In this context, HPR should be used as one more control method within the integrated pest management (IPM) con-cepts, aiming to attenuate injuries caused by B. tabaci biotype B. Among the main benefits provided by this method adop-tion, the following are highlighted: reduction of insect popula-tion to uninjured levels; non-interference on agroecosystem; is non-pollutant; provides cumulative and persistent effects; and does not demand specific knowledge by the producer (Boiça Júnior et al. 2013). Given the importance of bean crop to Bra-zilian population and HPR as a control method, the present re-search aimed to study the non-preference for oviposition and antibiosis in bean cultivars to the whitefly.
Materials and methods
Non-preference for oviposition and antibiosis experiments were carried out in a greenhouse of the Departamento de Fitossanidade at the Faculdade de Ciências Agrárias e Vete-rinárias - FCAV/UNESP, Jaboticabal, SP, Brazil, from Ja-nuary to March 2012. The following cultivars were used for the tests: IAC-Centauro, IAC-Una, IAC-Formoso, IAPAR-81, IPR-Eldorado, IPR-Siriri (all resistant), Pérola and IAC-Harmonia (all susceptible). These cultivars were previously screened from field experiments in winter, water and dry seasons (Silva 2012).
Non-preference for oviposition
The cultivars were sown in 5 L pots, containing soil and manure in the proportion of 3:1, and 20 days after the emer-gence one plant per plot was kept after thinning, which re-ceived cultural practices and fertilization as recommended for the crop. Twenty-five days after emergence (V4 growth stage – three opened trifoliates), the pots with bean cultivars were distributed at random and circularly inside screened cages (2.0 m base x 1.8 m height) in which 100 non-sexed adults of the whiteflies per plot (treatment) were released in the center (total of 800 adults). Adults were from the mainte-nance colony, and were reared according to the methodology by Toscano et al. (2002). The number of adults of B. tabaci biotype B released per treatment followed the methodology recommended by Jesus et al. (2011), who reported this den-sity provides adequate oviposition for plant resistance studies on bean cultivars. Five replicates were used for each cultivar.
After 24 hours of the insects release, plants were removed and taken to the Laboratório de Resistência de Plantas a In-setos at FCAV/UNESP. In the laboratory, eggs were counted per cmβ on the abaxial surface of one trifoliate from the upper, middle and lower thirds of the cultivars through a stereos-cope. Leaf area of the plants thirds was measured using an electronic leaf area meter, model LI-COR 3100A®.
Mean number of eggs (± SE) of Bemisia tabaci biotype B per cmβ obtained from the upper, middle and lower thirds on eight bean cultivars in free-choice and no-choice tests, in greenhouse. Jaboticabal, SP, Brazil, 2012.
Means followed by the same letter in column are not significantly different by Scott-Knott's test, at 5% probability. For analysis, data were transformed in (x + 1)12. (± SE) Standard error of mean.
The leaflets were uncovered until the end of the nymphal stage, when the cages were attached to leaves again in order to avoid adults to escape. Observations were done daily and at the same time, evaluating the duration of the incubation period, duration of period and viability of nymphs, dura-tion of development (from egg to adult) and adult longe-vity (without food). For longevity observation, 25 recently-emerged adults were collected randomly (five adults from each leaflet) from the cages and placed into glass tubes (2.5 cm diameter x 8.5 cm height), and the number of dead in-sects was reported daily.
Data recorded from duration of incubation period, duration of the nymphal period, duration from egg to adult, and longe-vity of adults were transformed in (x + 0.5), and data recor-ded from nymphal viability in arcsine (x + 0.5/100) for nor-malization and then were subjected to the one-way ANOVA. Tukey's post-hoc test was used at 5% probability for means separation, using the software SAS (Sas Insitute 1994).
In order to discriminate the bean cultivars regarding the resistance degrees to B. tabaci biotype B, the biological pa-rameters duration of incubation period, duration of period and viability of nymphs, duration of development from egg to adult and adult longevity were evaluated through the Prin-cipal Component Analysis (PCA) (Jackson 1991) to classify the cultivars that showed the maximum similarity and mini-mum dissimilarity among groups. Results from multivariate (PCA) and univariate (ANOVA) analyses were adopted to discriminate the bean cultivars regarding the resistance de-grees to B. tabaci biotype B. For multivariate analysis and graph preparation, the software Statistica version 7.0 (Stat-soft 2012) was used.
Results and discussion
Non-preference for oviposition
Means (± SE) ofthe deployment ofthe interaction between bean cultivars versus plan! third ofthe mean number of eggs of Bemisia tabaci biotype B per cm’, obtained on eight cultivars in free-choice test in greenhouse. Jaboticabal, SP, Brazil, 2012.
1 Means followed by the same lower-case letter in column and upper-case in row are not significantly different by Scott-Knott's test, at 5 % probability. For analysis, data were transformed in (x+1)1/2.(± SE) Standard error of mean.
In the no-choice test, no significant differences were ob-served among cultivars. However, there was a trend of higher number of eggs on the cultivar IAC-Centauro (2.80 per cmβ), and numerically the lowest mean of eggs were observed on IAPAR-81 (0.49 cmβ) (Table 1).
It is reported in literature that the presence of trichomes acts as a stimulant factor for the B. tabaci biotype B oviposi-tion, and the most pilose cultivars are the most infested since these cultivars may provide a more suitable microclimate for oviposition and better protection for the nymphs (But-ter and Vir 1989). In addition, females prefer to lay eggs on the base of trichome insertion (Omram and El-khidir 1978). Three types of trichomes are found in bean plants: acicular trichomes (needle-shaped), which are long, erect, distally narrowed and formed by two basal cells and one terminal cell; unciform trichomes (hook-shaped), which are smaller and also possess two basal cells and one terminal cell; and glandular trichomes, which are short. These characteristics were first described by Moutt (1932) cited by Dahlin et al. (1992). Costa et al. (2004) reported that non-preference for oviposition of B. tabaci biotype B on cowpea cultivars may be related to lower content of attractant substances or higher content of repellents, which influence the insect behavior on host selection. These factors may also be associated to the relationship whitefly-common bean.
Significant differences were found for plants thirds in both free-choice and no-choice tests (Table 1). In the free-choice test, the upper and middle thirds were equally pre-ferred for oviposition, with means of 1.08 and 1.07 eggs per cmβ, respectively, differing from the lower third (0.16 eggs per cmβ). In the no-choice test, the highest number of eggs was observed on the upper third (3.43 eggs per cmβ), differ-ing from the middle third (1.22 eggs per cmβ), which on the other hand, differed from the lower third (0.04 eggs per cmβ) (Table 1). Similar results were obtained by Rodrigues et al. (2012b), while studying the life history and non-preference for oviposition of B. tabaci biotype B on cowpea cultivars. The authors reported the whitefly prefers to lay eggs on the abaxial surface of leaves from the upper part of plant canopy.
The whitefly probably estimates the age and quality of the host plant through stylet insertion into the plant tissue before selecting the oviposition site (Vendramim et al. 2009), nevertheless, without ingesting sap in order to find a favo-rable chemical or morphological constitution depending on the plant age (Walker and Perring 1994) and stimuli involved between the insect and plant (Lara 1991). Likewise, Campos et al. (2005) reported the highest number of eggs laid by the whitefly on the apex leaf on cotton. According to Van Len-teren and Noldus (1990), preference for the youngest parts of the bean plant may be related to the highest concentration of nutrients (amino acids), which are promptly available for the sucking insects. In addition, younger leaves have thinner and softer cuticle, as well as a higher amount of water. Therefore, these characteristics may facilitate the whitefly oviposition (Eichelkraut and Cardona 1989) and eggs hydration (Gill 1990), providing a higher survivorship of the nymphs.
For the interaction of cultivar x plant third, significant difference was observed only in the free-choice test (Table 2). Although numerically, means of number of eggs per cmβ were higher on the upper third of bean plants of most culti-vars. We observed through the deployment of the interaction significant differences only in the cultivars that had over 3 eggs per cmβ. The cultivar Pérola stood out with 3.16 eggs per cmβ on the upper third, differing from the middle and lower thirds. On the cultivar IAC-Centauro, preference was observed for the middle third (3.35 eggs per cmβ) and differed significantly from the other plant thirds (Table 2).
Because they were the most preferred for oviposition (over 3 eggs per cmβ), the cultivars IAC-Centauro and Pérola also differed significantly from the other cultivars regarding the plant thirds separately. As results, Pérola exhibited the higher number of eggs on the upper third, and the cultivar IAC-Centauro on the middle third. There were no differences for the lower third among the bean cultivars, which had low infestation (Table 2). Berlinger (1986) underlines that physi-cal traits of leaf surfaces, such as pilosity, presence of adhe-rent glandular trichomes and leaf format are aspects affecting oviposition preference by the whitefly. In addition, pubes-cence is one of the factors that allows B. tabaci biotype B oviposition preference on the lower surface of leaves, but, other traits, e.g. number of leaves and foliar area, are also important for host selection (Simmons 1994).
For beans, specifically, feeding and oviposition non-pre-ference are the most prevalent resistance-types, in addition to antibiosis. There are few reports of tolerance in common beans against the whitefly. A study conducted by Oriani et al. (2005) showed that, among other factors, resistance in common bean against B. tabaci biotype B is related to the presence of arcelin or acicular trichomes. The presence of trichomes acts as stimulant for the oviposition of B. tabaci biotype B on beans, and the pilose cultivars were more in-fested as previously reported by Peña et al. (1992), Peña et al. (1993), Oriani and Lara (2000) and Oriani et al. (2005). Also, trichomes are stimulant in other crops such as soybean (Valle and Lourenção 2002), tomato (Toscano et al. 2002), and cotton (Chu et al. 2001). Therefore, this feature should be avoided in breeding programs of common bean against the whitefly.
Further studies are needed to unveil the resistance mecha-nisms present in the least preferred cultivars IPR-Siriri, IAPAR-81, IPR-Eldorado, IAC-Formoso, IAC-Una and IAC-Harmonia, especially in free-choice test, in order to head investigations to transfer or increase these resistance-related traits to new cultivars.
Index (± SE) and classification of oviposition preference of Bemisia tabaci biotype B, on eight bean cultivars, in free-choice and no-choice tests. Jaboticabal, SP, Brazil, 2012.
1 Means followed by the same letter in column are not significantly different by Tukey's test, at 5% probability. For analysis, data were transformed in (x + 1)1/2. OPI = [(T-P)/(T+P)] x 100. OPI ranges from +100 (very stimulant) until -100 (total deterrence), and value 0 indicates neutrality. (± SE) Standard error of mean.
In both assays, there was an increase of the OPI when the condition was switched from the no-choice to free-choice test (Table 3). The expressive augment of oviposition deterrence on Pérola in the no-choice test is highlighted as this cultivar was neutral in the free-choice test. Similar results were also reported for IAC-Formoso. This cultivar was classified as de-terrent in the free-choice test, since the expectation was an increase on deterrence (OPI = -52.45), and in the no-choice test it behaved as neutral (OPI = -2.56) (Table 3). According to Blua et al. (1995), behavioral changes of the whitefly is attributed to several factors that modify insect preference as the confinement conditions are different for free-choice and no-choice tests.
Baldin et al. (2005), while evaluating the resistance in tomato genotypes against B. tabaci biotype B, found that the OPI calculated in free-choice and no-choice tests clas-sified all genotypes as oviposition deterrent to the whitefly when compared to the susceptible pattern IAC-Santa Clara. The same authors still emphasized that results obtained with the genotypes LA-716, PI 134418 and PI 134417 are similar from those found by Toscano et al. (2002) and Fancelli et al. (2003), reporting expression of non-preference for oviposi-tion-type resistance in these genotypes against the whitefly.
For most experiments, we underline that the genotypes with deterrence traits were not commercial cultivars, but wild entries or breeding lines with agronomical characteristics of low yield, whereas for the present work oviposition deter-rence was reported for high yielding cultivars.
For the nymphal period, we observed that cultivar IAC-Harmonia extended the biological development of the white-fly (23.41 days), differing significantly from the results ob-tained for IAC-Centauro, IAC-Formoso, IAPAR-81 and IPR-Siriri, which had duration of the biological development around 21 days (Table 4). These data suggest the expression of non-preference for feeding and/or antibiosis in IAC-Har-monia, which, according to Lara (1991), is characterized by the longer period nymphs require to complete the immature stage comparatively to a susceptible plant (Table 4). The other cultivars were intermediate.
Shorter duration of nymphal periods than those obtained in the present study were found by Oriani and Lara (2000) while assessing the antibiotic effects of bean genotypes pos-sessing arcelin on B. tabaci biotype B in Jaboticabal, SP, Bra-zil, who observed duration varying from 11.0 to 15.4 days. It is important to mention that the study of the authors was car-ried out in the dry season, i.e. the hottest period of the year, which may have favored the insect development. In the same work, when the water season was evaluated the results were similar to our study, with means of 23 days for the duration of nymphal period.
Nymphal viability had amplitude between 69.30% on the cultivar IPR-Eldorado and 100% on IAC-Centauro, however, there was no significant difference among cultivars (Table 4). These percentages are considered high when compared to other studies with common bean or other plant species in the literature. Fancelli et al. (2003) obtained 31.2 to 86.9% nymphal viability on tomato genotypes, Campos (2003) found 30.7 to 64.2% on cotton and Rodrigues et al. (2012b) observed from 50.0 and 90.0% nymphal viability on cow-pea cultivars. These results prove this biological parameter is variable depending on the host plant.
With respect to the total period (duration from egg to adult), results were influenced by the nymphal period, with significant differences. The cultivar IAC-Harmonia highligh-ted as the most harmful to B. tabaci biotype B development, evidenced by the elongation of its life cycle (29.41 days), and differed from IPR-Siriri (27.09 days), IAC-Centauro (27.01 days) and IAC-Formoso (26.71 days), which were the most suitable for the whitefly development. Duration of the total period on these cultivars was about 2.5 days shorter than on IAC-Harmonia (Table 4).
Mean (± SE) duration of incubation, nymphal and total periods (days), nymphal viability (%) and adult longevity (days) of Bemisia tabaci biotype B obtained on eight bean cultivars in greenhouse. Jaboticabal, SP, Brazil, 2012.
(± SE) Standard error of mean.
Means followed by the same letter in column are not significantly different by Tukey's test, at 5% probability.
For analysis data were transformed in (x + 0.5)12.
For analysis data were transformed in arcsine [(x+0.5)/100]1/2.
For this parameter, Oriani and Lara (2000), while study-ing the antibiotic effects of nine bean cultivars with or with-out arcelin in greenhouse conditions, observed that B. tabaci biotype B total cycle was between 21 and 37 days in experi-ments conducted in the water and dry seasons in Jaboticabal, SP, Brazil, respectively. Studies conducted by Rodrigues et al. (2012b) on cowpea showed that the cultivar Sempre Verde was the most adequate for the whitefly development, and the cultivars BRS Urubuquara and IPA-206 expressed non-pre-ference for feeding and or/antibiosis-type resistance.
Adults of the whitefly lived less when their nymphs were fed the cultivars IAC-Una (1.0 day), IPR-Eldorado (1.02 days) and IAC-Harmonia (1.02 days), differing significan-tly from IAC-Formoso (1.28 days) and IAC-Centauro (1.26 days) (Table 4). Most studies in literature do not report diffe-rences among treatments for adults longevity, mainly because experiments were evaluated without adults feeding (Oriani et al. 2008; Rodrigues et al. 2012b).
It is important to note that the cultivar IAC-Harmonia, which stood out as the least suitable for whitefly develop-ment by extending its life cycle, was considered susceptible in the water season, with the highest infestation in compari-son to other 18 cultivars. In addition, this cultivar exhibited the highest incidence of the BGMV in field conditions (Silva 2012).
Considering the biological parameters concomitantly through the multivariate Principal Component Analysis, the first principal component (PC1) concentrated 61.39% of the entire variability from the original variables, and the para-meters that most influenced this factor were nymphal period (–0.93), total period (–0.91) and adults longevity (0.90). The second principal component (PC2) concentrated 16.08% of the variability presented in the original biological para-meters, and the most influent variable was nymphal viability (–0.67) (Fig. 1).
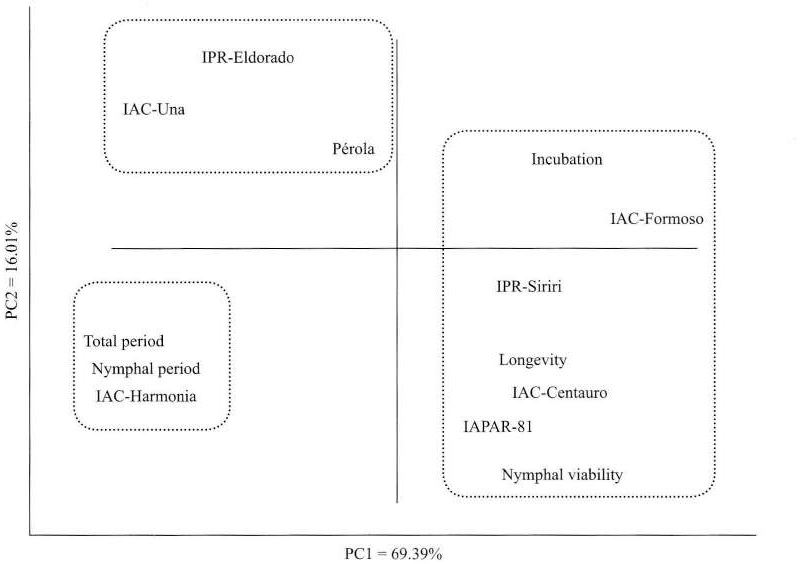
Distribution of bean cultivars and biological parameters, according to principal components analysis (PCA), obtained from Bemisia tabaci biotype B fed on bean plants.
Analyzing the distribution of the bean cultivars following the PCA obtained from the B. tabaci biotype B biological parameters, values of duration of nymphal period and total period were the factors that most influenced the analysis by isolating the cultivar IAC-Harmonia in the third quadrant (Fig. 1). Thus, based on the distribution of the cultivars from the whitefly biological aspects in univariate (ANOVA) and multivariate (PCA) analyses, and according to criteria esta-blished by Kaiser (1960), it is possible to classify the cul-tivars regarding the resistance degrees: IAC-Harmonia was classified as moderately resistant; IAC-Una, IPR-Eldorado and Pérola, as susceptible; and IAC-Centauro, IAPAR-81 and IPR-Siriri, are highly susceptible.
The biological parameters period of incubation and nymphal viability were the factors that least influenced the differentiation and classification of the bean cultivars for the resistance degrees to B. tabaci biotype B (Fig. 1), and these parameters also did not differ significantly in the univariate (ANOVA) analysis (Table 4).
It is important to emphasize that multivariate analyses, amongst them the Principal Component Analysis, have been used as an important tool for Entomology, especially Host Plant Resistance, as they are methods that take into account the assessment of the insect biological parameters altogether. Principal Component Analysis provides the formation of groups with genotypes that behaved similarly under the pest attack, allowing the researcher differentiate the genotypes concerning the resistance degrees.
Conclusions
The cultivars IAC-Harmonia, IPR-Eldorado, IAPAR-81, and IPR-Siriri were the least preferred when adults of the whitefly were given a choice for oviposition. In addition, the cultivar-IAC-Harmonia extended the life cycle of the whitefly, and holds moderate non-preference for feeding and/or antibiosis-type resistance. These cultivars, especially IAC-Harmonia, can potentially be grown in the field aiming to suppress po-pulations of the whitefly.
Footnotes
Acknowledgements
To Coordenação de Aperfeiçoamento de Pessoal de Nível Su-perior (CAPES) for the Doctorate scholarship granted to the first author and to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the scholarships granted to the second, third and seventh authors. Appreciation is also given to Instituto Agronômico de Campinas (IAC) for supply-ing the seeds of bean cultivars assessed in the present work and to Prof. Dr. Gener Tadeu Pereira from Departamento de Ciências Exatas of FCAV/UNESP for the assistance with the statistical analysis.
