Abstract
The aim of the study is to investigate barrier properties of a nanocomposite material against organic solvents. Molecular transport phenomena of three alcohols, namely methanol, ethanol and isopropanol, through a nitrile rubber-based nanocomposite, are carried out in temperatures ranging from 23 to 70°C. For each configuration, mass transfer kinetics are investigated for three different types of clay nanoparticles using mass uptake experiments. Maleated nanocomposites with enhanced dispersion morphology are also examined. Results show that all molecular transport parameters are generally susceptible to temperature variations. Sorption and diffusion coefficients noticeably increased as temperatures increased. Polymer/solvent interaction seems to be similarly affected. Molecular volume of the penetrant is observed to have an influence on molecule migration. Diffusion coefficients are likewise affected and decrease with the linear increase of molecular volume. The diffusion mechanism is slightly altered by this factor and the Fickian mode is maintained. When filling the rubbery matrix with layered silicates, sorption decreases at equilibrium. Its level drops even lower with the maleation of the nanocomposite. However, the diffusion coefficient exhibits a less systematic trend. Randomly filled nanocomposites appear to have higher diffusivity than neat rubber, but diffusion parameter considerably decreases after maleation, which emphasizes the nanoclay’s dispersion effect.
Introduction
Molecular transport phenomena through polymeric systems play an important role in several areas of engineering and have a considerable impact in many industrial sectors. 1 Structural integrity, chemical stability and dimensional invariance in the presence of aggressive substances are qualities of great importance when applications such as food packaging, controlled drug release, filtration and electronic encapsulation are concerned. 2 The sorption and transport properties of small organic molecules in a wide range of polymers have been extensively investigated. For instance, there are several studies on the molecular transport of most of the thermoplastic polymers, thermosets, elastomers, and geomembranes.3–5 The barrier properties of these materials were probed in the presence of the majority of commercial chemicals, such as n-alkanes, halo-alkanes, aliphatic esters and aromatic hydrocarbons.6,7 The molecular transport behavior was found to be deeply affected by the structural properties of the penetrant/polymer system. In fact, studies demonstrate the influence of the segmental mobility of the polymeric matrix, the size and shape of the penetrant as well as the chemical affinity in the diffusion system. 8 It was also found that the mass transfer phenomena closely depend on the environmental conditions surrounding the sorption process, namely temperature and humidity.9,10
Polymers reinforced with inorganic nanostructured and layered silicates have attracted substantial attention due to their unique and attractive characteristics even at low concentrations. 11 These materials, also called nanoclays, combine the simplicity of polymer processing with the resistance of the platelet-like nanoparticles. In addition, clay nanoparticles possess two characteristics. First, the constitutive platelets can be dispersed on individual layers. Second, they can functionalize the surface of these layers and apply chemical modifications that allow better interactions with the hosting polymers. 12 In previous works, their high aspect ratio was stated to contribute to the reduction of fluid permeability by creating a tortuous pathway to the penetrant substance.13,14 The ideal state would be having a complete separation of the nano-platelets leading to a maximum of interfacial surface in the rubber/clay system. However, total delamination is technically difficult to achieve due to the non-compatibility between the components and the hydrophilic nature of the bare clay nanoparticle surface. Therefore, the successful synthesis of nanocomposites depends on the chemical affinity between the polymeric matrix and the hosted nanoparticles that ensure the thermodynamic stability of the nanocomposite system. Over the years, new techniques are continually being developed to enhance the nanoclay/polymer compatibility. The most commonly adopted method consists of modifying the surface chemistry of the nano-platelets with aliphatic organic structures to reduce the surface tension and facilitate polymeric chain intercalation. 15 Rather than achieving intercalation via ion exchange, other methods to increase the interfacial adhesion were adopted, such as ion dipole bonding of bulk molecules to the metallic cation on the nanoclay surface and nano-platelet edge treatment with silane coupling agents. 16 It was also found that some studies used maleic anhydride monomers as compatibilizers due to their polarity and their highly reactive nature.17,18
Both in its solid and latex states, acrylonitrile butadiene rubber is considered to be one of the most mass produced and widely commercialized elastomers, also known as nitrile rubber. Mainly manufactured using the emulsion polymerization method, this elastomer exhibits low mechanical and barrier properties without reinforcing fillers. Attempts were made to incorporate clay nanoparticles to enhance its barrier properties, but this yielded a low state of dispersion of the nanoclays within the elastomeric matrix, resulting in poor chemical resistance quality. In our previous work, a clay nanocomposite based on nitrile rubber was developed. Combining the grafting of nitrile rubber backbones with maleic anhydride and the organic modification of the nanoclay surfaces led to a successful synthesis of the nanocomposite with a satisfactory state of nanoclay dispersion and orientation of the nano-platelets within the elastomeric structure. Thus, the aim of the present study is to investigate the barrier properties and better understand the sorption behavior of this material against organic solvents by probing the kinetic molecular transport parameters of the penetrant/nanocomposite system.
Reagents and materials
Baymod N34.52 NBR powder was used, complements of Matexion, ON, Canada. This nitrile rubber elastomer (NBR) is 34% acrylonitrile. Its Mooney viscosity is 45±5 at 100°C and its specific gravity is 0.98. For the grafted nanocomposite samples, maleic anhydride (MA) was used as a reactive monomer and benzoyl peroxide (BPO) as an initiator of the grafting process. Both substances were purchased from Aldrich Chemical with a purity level of over 98%.
The nanocomposites were filled with three different commercial-grade clay nanoparticles at a mass concentraton of 5 wt% (Sigma-Aldrich, Oakville, ON, Canada), one unmodified and two organically treated. The nClays were labeled as follows: nClay/0, a natural inorganic hydrophilic sodium montmorillonite (Na+MMT) with no surface modification. nClay/1, surface modified MMT containing 35–45 wt. % of dimethyl dialkyl (C14-C18) amine. nClay/2, Surface modified MMT containing 25–30 wt. % methyl dihydroxyethyl hydrogenated tallow ammonium.
High purity grade (over 98%) methanol, ethanol and isopropanol (propan-2-ol) from Fisher scientific (Hampton, NH, U.S.A.) were used as penetrant solvents in the sorption experiments. The molecular volume of the solvents are 40.45, 58.37 and 76.46 cm 3 ·mol−1, respectively.
Experimental
Preparation of the nanocomposites
Nitrile rubber-based nanocomposites containing the formerly described nanoclays were prepared. For every batch, a virgin nitrile rubber sample was prepared using the same processing protocol as for the nanocomposites for comparison purposes. A HAAKE Polylab OS—RheoDrive 4 co-rotating twin screw extruder (Rheomex OS PTW16) was used to prepare the conventional and maleated nanocomposites. After this, all the obtained compounds were press-molded into sheets for mass transfer experiments.
The preparation of maleated nanocomposite rubber was carried out in two stages; grafted nitrile rubber was firstly prepared by grafting NBR with maleic anhydride in the presence of benzoyl peroxide in a Brabender mixer. One part per hundred rubber (phr) was pre-mixed with nitrile powder for 5 min at 80°C and 100 rpm. Two phr of MA was then added for an additional 5 min under the same mixing conditions. The resulting mixture was then extruded in accordance with the procedure described in the previous section.
The nomenclature adopted to label the nanocomposites under study was as follows. Each nanocomposite was appointed as RCN/x where x stood for the nature of the nClay (0, 1 and 2 for nClay/0, nClay/1 and nClay/2 respectively). The same terminology was used for the maleated sample with an extra g letter referring to grafting (RCNg/x). NBR/P stood for pure nitrile rubber used as reference.
Mass uptake experiment
Mass uptake experiments were performed at 23, 40, 55 and 70°C. Rectangular samples (60 × 10 mm) were cut from the press-molded nanocomposite sheets. Screw-tight test bottles containing about 10 mL of the penetrant solvent were placed in a bath of silicone oil heated to the desired temperature within an accuracy of ± 1.0°C. Dry weights of the cut samples were then placed in the bottles. At predefined time intervals, the samples were withdrawn from the solvent containers. The surface-adhered liquid was removed by gently pressing the samples between filter paper wraps and the samples were weighed on a digital balance (accuracy ± 0.01 mg). The overall process was performed within a maximum of 20 seconds to minimize errors. The sorption tests were performed until equilibrium was reached, i.e. solvent was no longer being absorbed despite continued immersion. Each test was replicated three times to obtain statistically valid data.
The percent mass uptake during solvent sorption was given by:
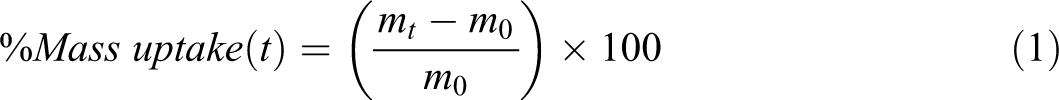
Theory
Sorption behavior
Even though nitrile nanocomposites are dense and nonporous materials, they can be permeated to some extent by liquids, gases and vapors of liquids at a molecular level. 19 An effective way to predict the diffusion mechanism in a given system is to establish a relationship between the sorption data from mass uptake experiments and the analytical approach. 4

Where,
Kinetics of mass transfer
Molecular transport through polymeric systems is admitted to be a diffusion process. 20 The diffusion coefficient of the molecular flow through a given polymeric membrane can be calculated using the thin-film solution with Fick’s second law of mass transfer.
Resolution of the second law of Fick’s equation leads to an expression of the mass of the penetrant absorbed by the film as a function of time, previously defined as the dynamic mass variation. A detailed development of the resolution of Fick’s second law can be consulted in our previous works 21 :

Therefore, the diffusion coefficient, D, can be graphically deduced from the slope of the reduced sorption plot overlaying the experimental data of sorption.
The solubility,

Results and discussion
Mass uptake
Mass uptake data are interpreted using sorption plots. In these plots, the mass variation of the immersed sample is portrayed as a function of time. Figures 1 and 2 shows the effect of four temperatures and three different molecular sizes of the penetrant on mass uptake of NBR/P and RCN for different nClays. The standard deviation of all sorption measurements is less than 1.8%.
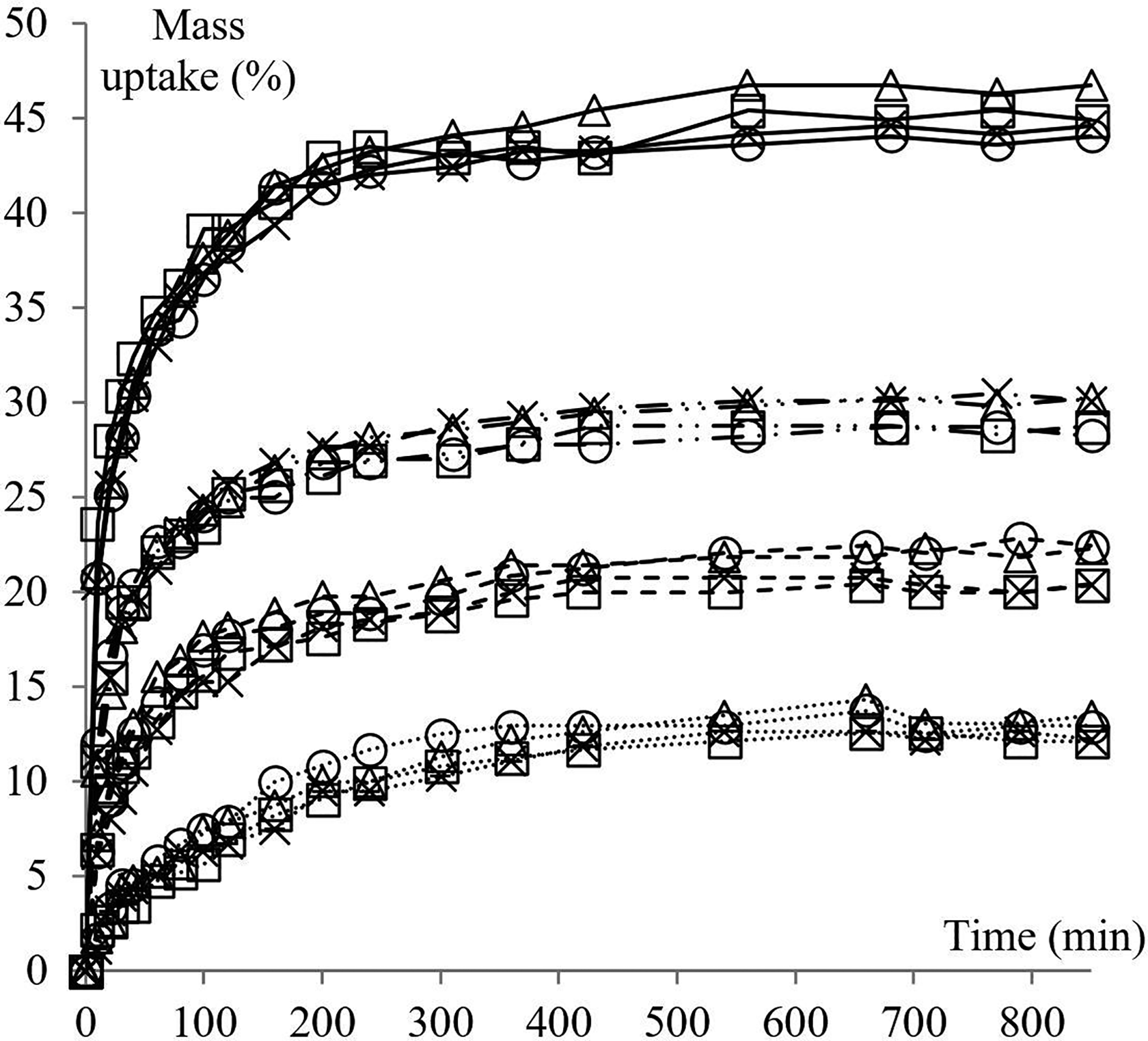
Mass uptake of NBR/P and RCN for different nClays in isopropanol at different temperatures. Line styles refer to Temp.: (···) 23°C, (
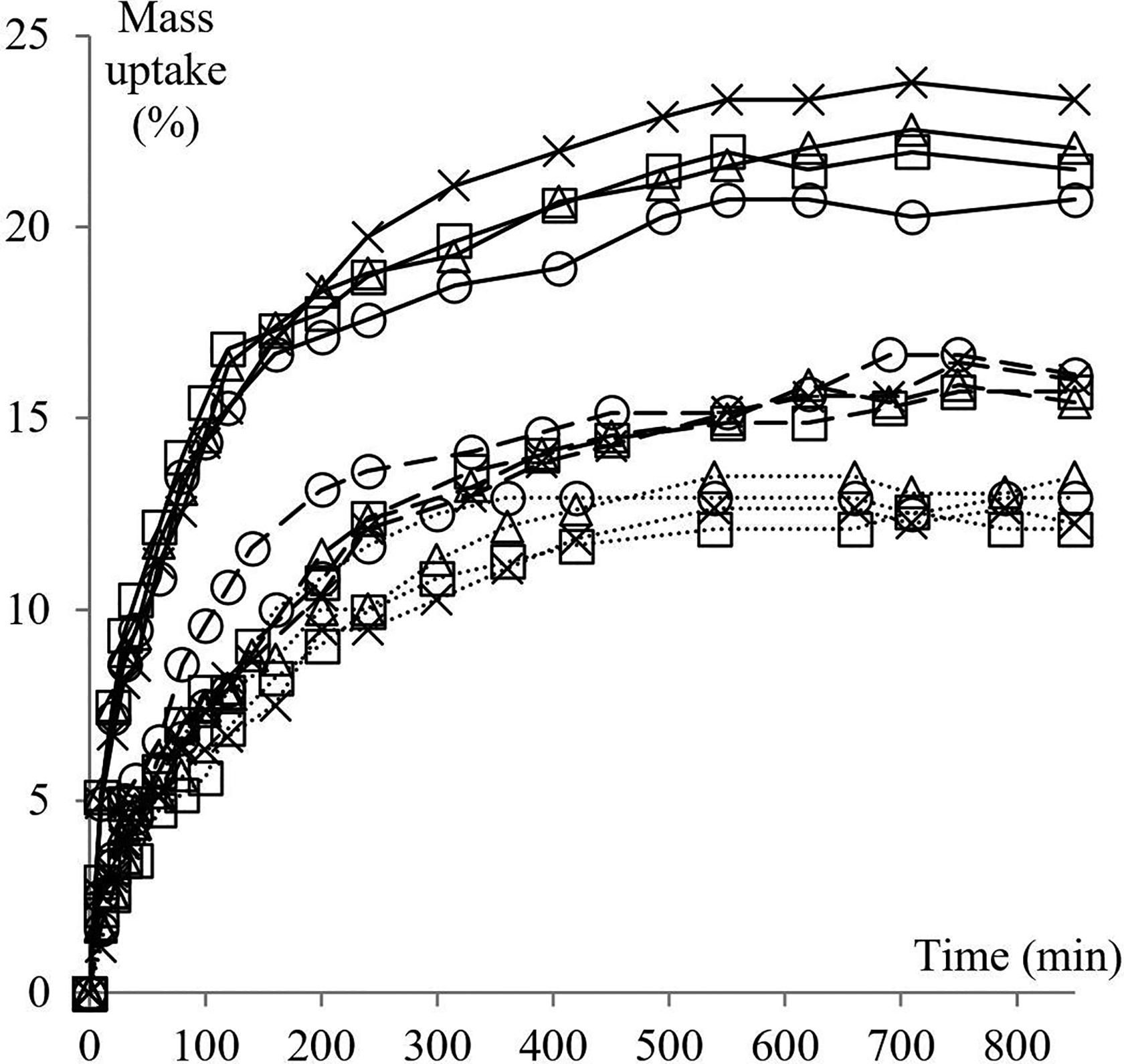
Mass uptake of NBR/P and RCN for different nClays in different solvents at 23°C. Line styles refer to: (···) isopropanol, (
As a general observation, one can notice that all curves exhibit the similar characteristic behavior of polymeric materials. The resulting curves start off showing rapid mass uptake after which they exhibit concave behavior toward the x-axis until approaching the sorption equilibrium values. 22
Figure 1 illustrates the sorption data of NBR/P, RCN/0, RCN/1 and RCN/2 in isopropanol at 23, 40, 55 and 70°C. Sorption attains the equilibrium state more quickly as the temperature increases. In fact, steady state is not achieved before 540 min of immersion at 23°C, while it is reached in 310 min at 70°C. The same trend occurs with intermediate temperatures therefore not all data is graphically represented to avoid redundancy (Table 1). It is also noted that the higher the temperature, the steeper the slope. These findings are directly related to the kinetic of mass transfer that will be discussed in the following sections. From another perspective, the proportion of sorption at saturation exhibits a systematic increase as the temperature regularly increases. For all temperatures, the nature of the nanoclay does not seem to have a substantial effect on the value of sorption at saturation. In what follows, RCN/1 is considered to illustrate the effect of temperature. From 23 to 55°C, the maximum of solubility rises from around (12.5 ± 1.1) % at regular intervals of about 6 to 7%. At 70°C, the maximum sorption value rises even higher (around 15% per 15°C) and sorption at equilibrium was reached at (43.6 ± 1.3) %. The temperature effect is also investigated with methanol and ethanol (Table 1). The same trend is observed with both alcohols and the regular increase of temperature results in an augmentation of the sorbed amount of solvent at equilibrium. Yet interestingly, temperature has a lesser effect on the sorption of methanol. The values of sorption at saturation vary more systematically with the increase of temperature, but with an interval of mass uptake barely exceeding 5%. No jump of sorption was noted at 70°C as with isopropanol.
Solubility coefficients (S) of NBR/P and RCNs in methanol, ethanol and isopropanol at four different temperatures (23, 40, 55 and 70°C).
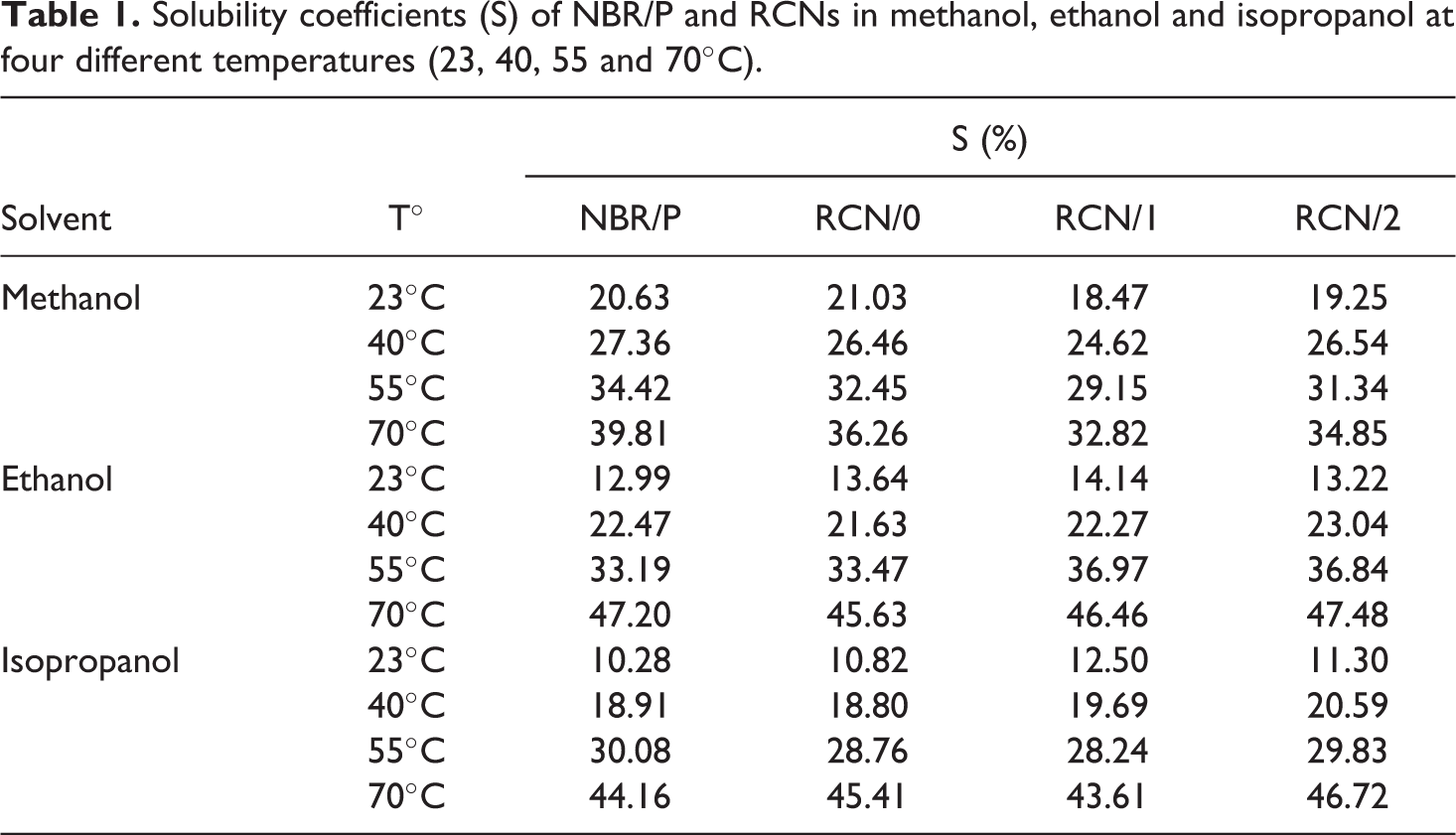
Figure 2 portrays the mass percent uptake data of NBR/P, RCN/0, RCN/1 and RCN/2, at 23°C, in methanol, ethanol and isopropanol. Homologous results at 40, 55 and 70°C are also examined although not graphically represented (Table 1). Here again, sorption plots of all solvents follow the same tendency as the typical mass uptake curve of a liquid/polymer system. However, the nature of the penetrant seems to have a substantial influence on the extent of sorption at equilibrium. Indeed, the methanol pattern of mass gain starts with a high rate then levels off at about 495 min. The same behavior is noted for the isopropanol curve except that equilibrium state is reached at around 540 min of immersion time. A more significant effect is observed on the solubility coefficient. Indeed, while methanol attains saturation at (20.7 ± 1.7) % of mass uptake in RCN/1, isopropanol does so at (13.4 ± 0.9) %, for the same nanocomposite and same immersion time of 850 min. An intermediate value is exhibited by ethanol. Similar conclusions are reported in a study involving solubility and diffusion of a number of hydrocarbons through poly(dimethyl-siloxane). 23 The authors state that the molar solubility is inversely proportional to the penetrant’s chain length. And as the solubility is governed by entropic effects, polymeric chain segments exhibit higher affinity to smaller molecules. A related point to consider is that the solubility is clearly affected by the presence of nanoclays, namely in the case of methanol. Further inspection of this behavior is provided in upcoming sections. Let us consider the evolution of the gap of sorption between the solvents as a function of temperature. Isopropanol molecules are less taken up by about 4% by the RCN/1 sample than the ethanol molecules at room temperature. This gap lessens as temperature increases until almost vanishing at 70°C within the interval of measurement error. Surprisingly, methanol seems to exhibit a less systematic behavior. The associated solubility coefficient increases as temperature increases as expected, but at a lower rate (around 5% per 15°C) compared to the other solvents. As a result, the sorption of methanol attains saturation at 32.8%, which is more than 10% lower than isopropanol.
The permeability of low molecular weight substances through a membrane structure involves matter migrating from one side to the other. For organic dense elastomeric matrices, a conceptual understanding of the phenomena is to say that this transport process combines the sorption of the small penetrants into the membrane’s surface followed by the diffusion by concentration gradient through the structure. Permeability is thus intrinsically linked to the ratio of the total path length traveled by the penetrant molecules over the thickness of the specimen. Diffusion and solubility are expected to be affected by the addition of inorganic and impermeable fillers in the polymeric matrix, such as clay nanoparticles. In effect, fillers act as obstacles forcing the penetrant molecules to work around them, increasing the tortuousness of available pathways. In addition, the presence of lamellar structure fillers, as is the case with nanoclays, is believed to modify the alignment of the adjacent polymeric macromolecules. This morphological modification results in an indirect alteration of the permeation mechanism.24,25 From a phenomenological perspective, and for a given size and concentration of the filler, a high degree of tortuosity is reached by increasing the specific surface of the filler capable of creating a maze path that slows down molecular penetration. At low level of filler loading, enhancing specific surface of nanoclays is only possible by increasing the extent of their dispersion. RCNg/1 is a maleated nanocomposite that was synthetized in our previous work. It exhibited a higher state of dispersion compared to conventional RCN/1. In what follows, molecular transport characteristics are investigated according to the structural properties of the nanocomposite. In every case, results are compared to the unfilled rubber matrix.
Figure 3 displays percent mass uptake curves of NBR/P, RCN/1 and RCNg/1 in methanol at 40°C and 70°C. For both temperatures, a consistent trend can be observed; solubility coefficient in methanol decreases with the presence of layered silicate fillers (RCN/1), the coefficient is even lower with maleated nanocomposite (RCNg/1). At 40°C, sorption of methanol is reduced from 30.2% to 26.6% with the addition of nClay/1 in the conventional nanocomposite. Mass uptake at saturation drops by an additional 4% after maleation of the nanocomposite. At 70°C, the effect of the nanoclay is more pronounced. The difference in mass percent uptake between NBR/P and RCN/1 decreases twofold compared to sorption data at 40°C. However, sorption is slightly reduced during maleation.
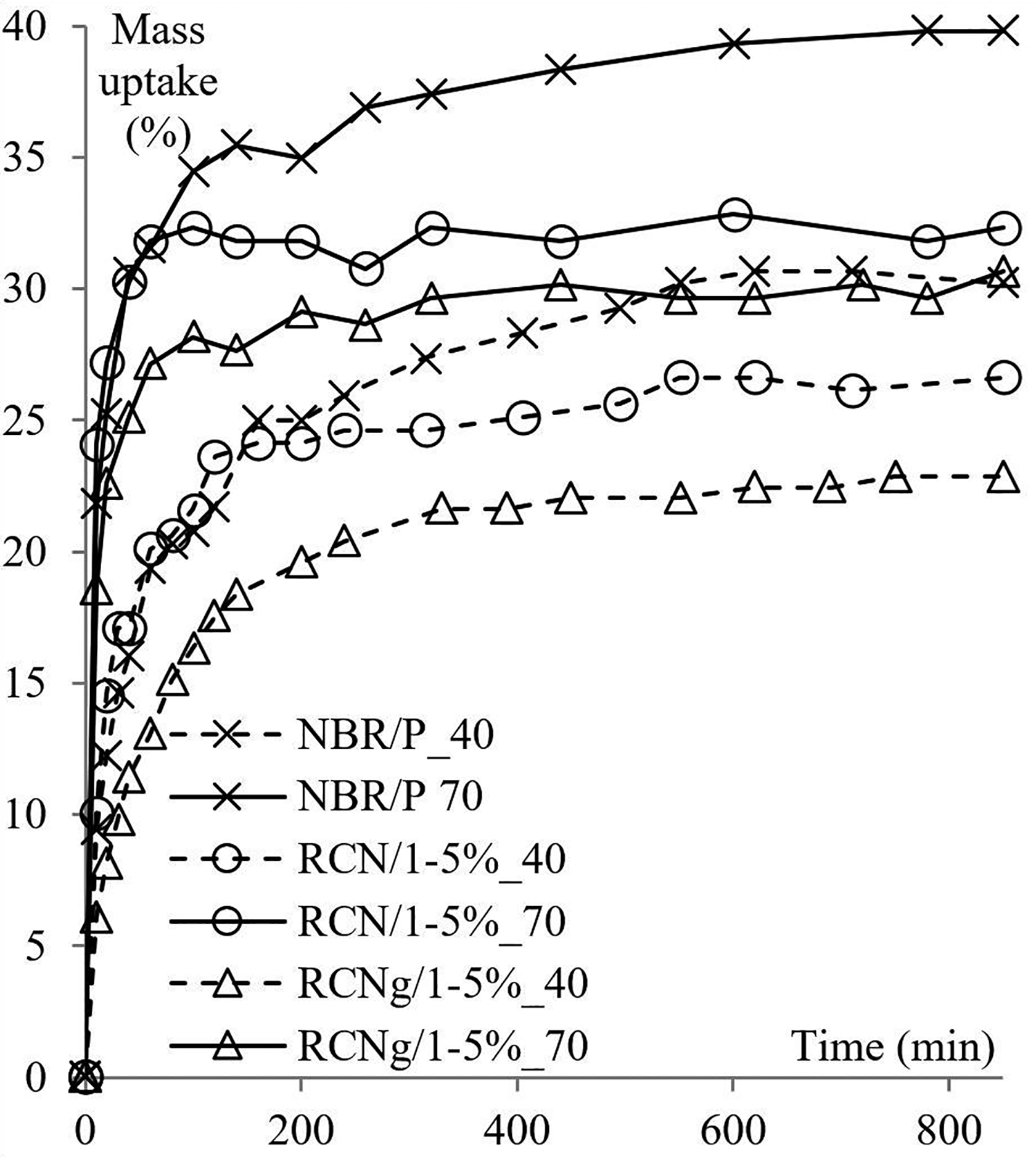
Comparison of mass uptake of NBR/P, RCN/1 and RCNg/1 in methanol at 40°C and 70°C.
For all three materials, the effect of the penetrant’s nature is also examined and the associated sorption data in methanol and isopropanol at 55°C are displayed in Figure 4. The same previously observed tendency appears to be preserved. For methanol, sorption data for the three materials follow the same behavior observed at 40 and 70°C. It can even be observed that the sorption pattern follows the trend of high temperatures and is closer to that examined at 70°C. The solubility coefficient values lay in the intermediate zone between the coefficients associated to 40 and 70°C. Interestingly, higher molecular volume penetrants deviate from the previous bias. In fact, sorption at equilibrium in isopropanol barely decreased while loading with nClay/1. Nevertheless, a drop of about 5% is clearly spotted with the maleated nanocomposite. Not shown in the figure but worth mentioning, ethanol sorption data exhibit intermediate values.
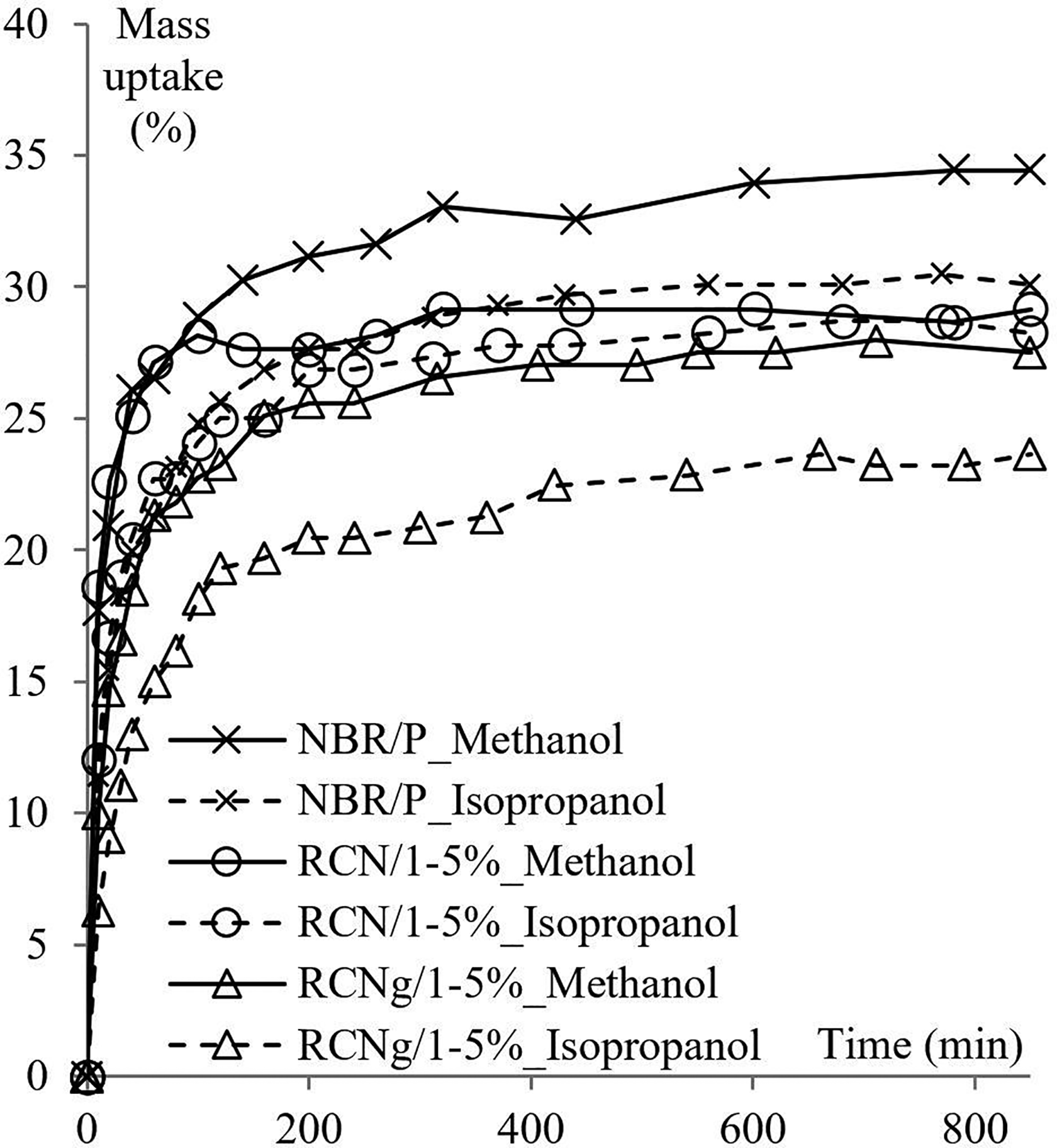
Comparison of mass uptake of NBR/P, RCN/1 and RCNg/1 in methanol and isopropanol at 55°C.
Mass transfer mechanism
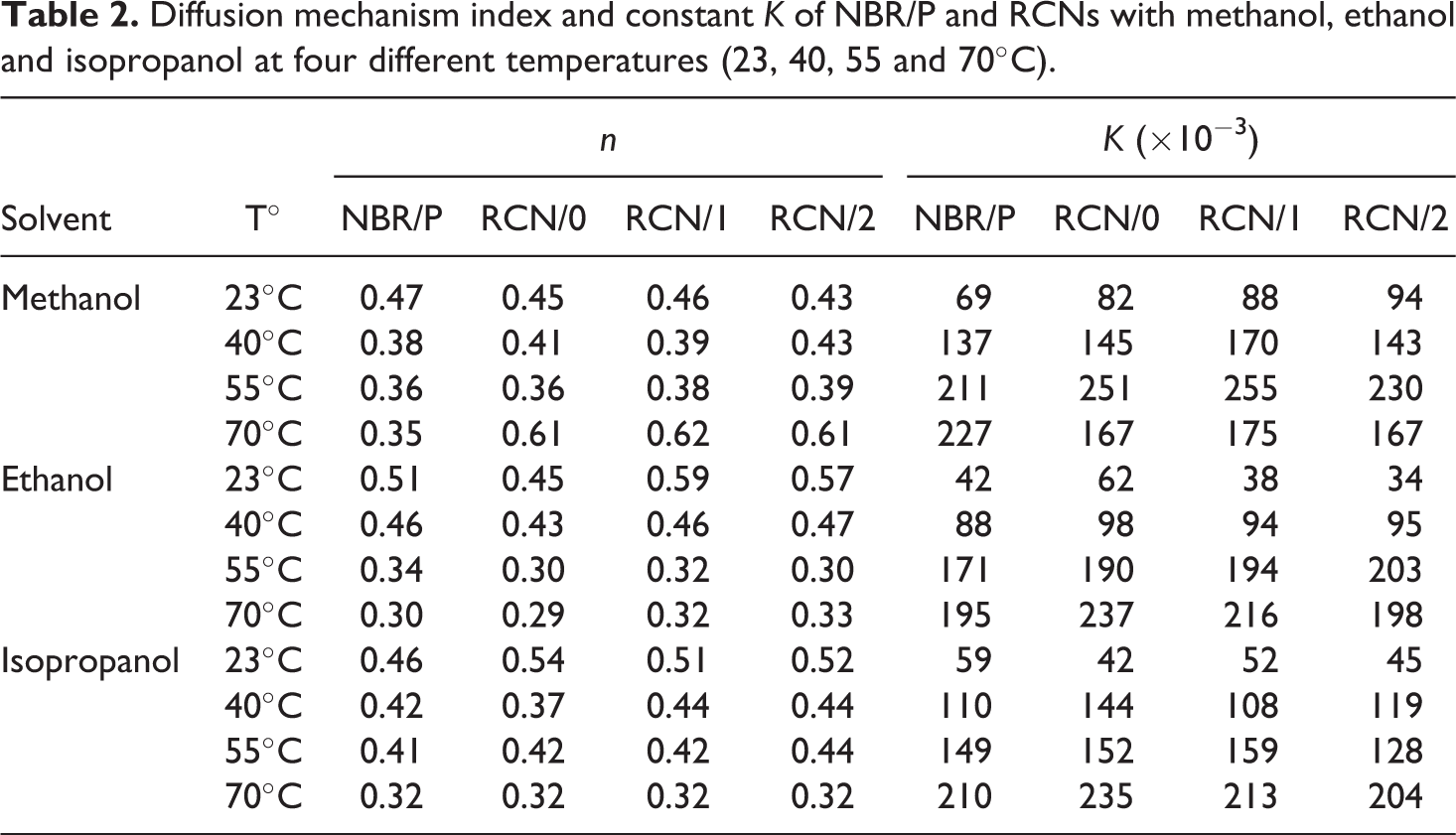
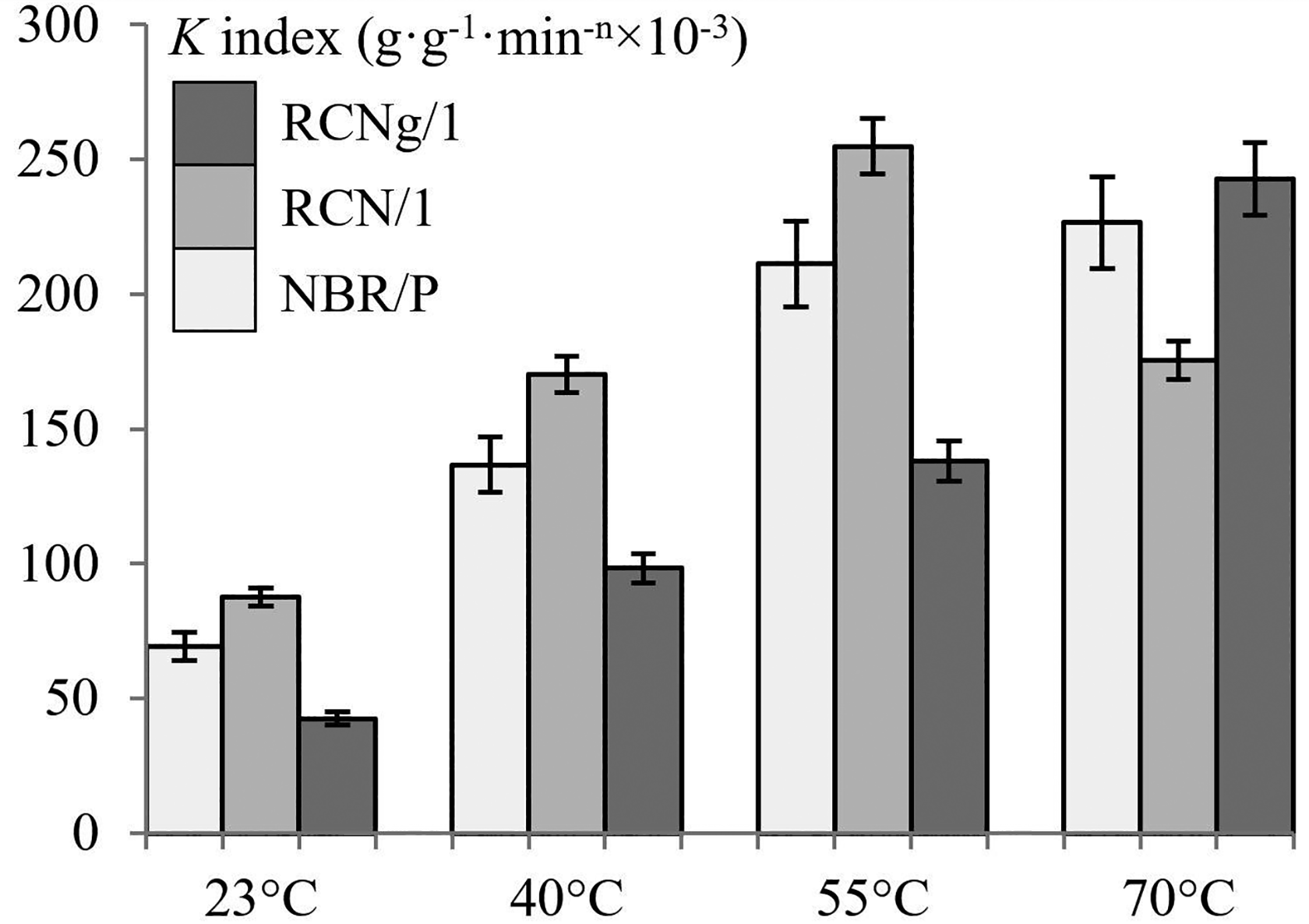
The empirical relation expressed by equation (2) is chosen to numerically estimate the diffusion mode of the systems investigated in this study. A very good correlation is obtained for all regressions (R
2
> 0.98). Table 2 summarizes the entire set of diffusion mechanism index
Diffusion mechanism index and constant
Constant
Let us shed light on the diffusion mechanism index under the described considerations. Still from data presented in Table 2 and starting with the effect of temperature, a general remark can be made. Factor
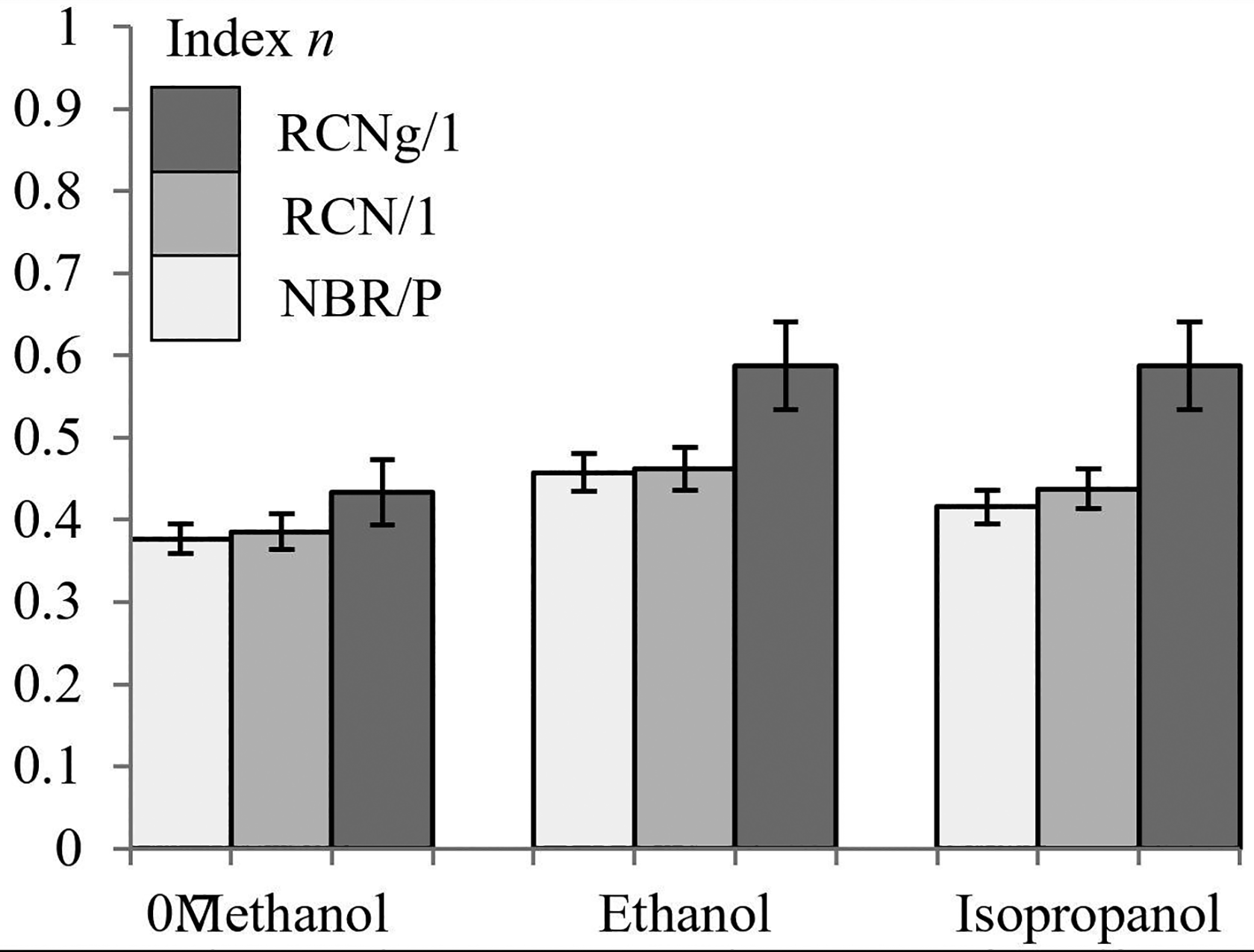
Diffusion index of NBR/P, RCN/1 and RCNg/1 with all solvents at 40°C.
Diffusion coefficient and permeability
The diffusion mechanisms of the systems under investigation are demonstrated to be more or less Fickian. Dynamic solvent uptake is therefore expected to exhibit linear dependence, for short durations, to the normalized square root of time (Equation 3). Hence, a fine approximation of the diffusion coefficient can be estimated based on Fick’s second law of mass transfer. Figure 7 shows histograms of the overall diffusion coefficients of NBR/R and the three RCNs with methanol, ethanol and isopropanol at the four formerly designated temperatures. Several comments can be made based on the presented data. Firstly, a clean and concise augmentation of the diffusion coefficients appears as the temperature increases from 23 to 70°C. The trend is observed with all polymer/solvent systems. Temperature seems to intensify every aspect related to the molecular migration process. The most pronounced variation is associated to the smallest molecular penetrant. For instance, the
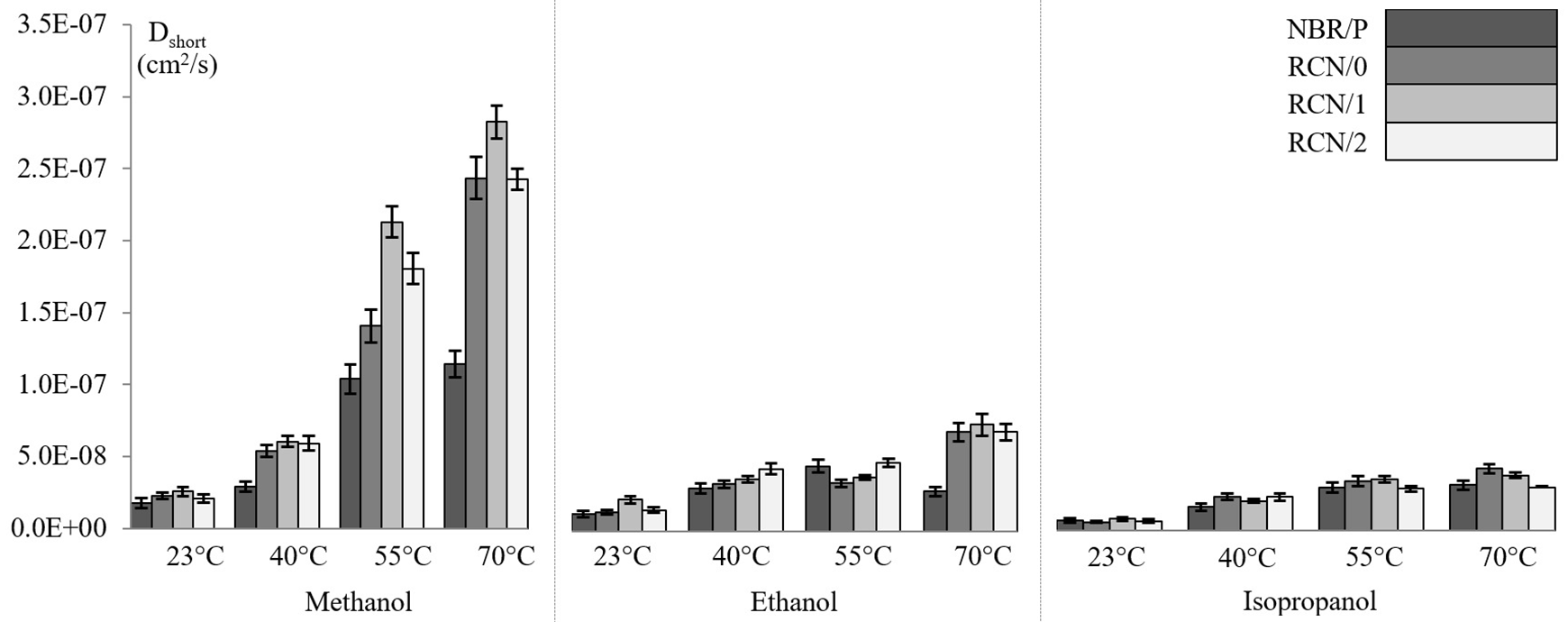
Diffusion coefficients of methanol, ethanol and isopropanol in NBR/P, RCN/0, RCN/1 and RCN/2 at 23°C, 40°C, 55°C and 70°C.
The following section investigates the effect of the presence of nanoclays on the diffusion coefficients. Since the early works of Nielsen, filled polymeric matrices with inorganic layered silicates are suggested to increase the tortuous path of the diffusing molecules. The proposed model is supposed to result in filled materials having enhanced barrier properties.
31
This theoretical approach is only valid when considering nanoclay fillers as impermeable and as having separated nano-platelets that are totally aligned perpendicularly to the diffusion direction. In Figure 8 (a), the diffusion coefficients of NBR/P, RCN/1 and RCNg/1 with methanol are compared at four temperatures. The diffusion coefficients of the same materials in methanol, ethanol and isopropanol at 23°C are presented in Figure 8 (b). In general, temperature and molecular volume have the same previously-described effects on all three materials.
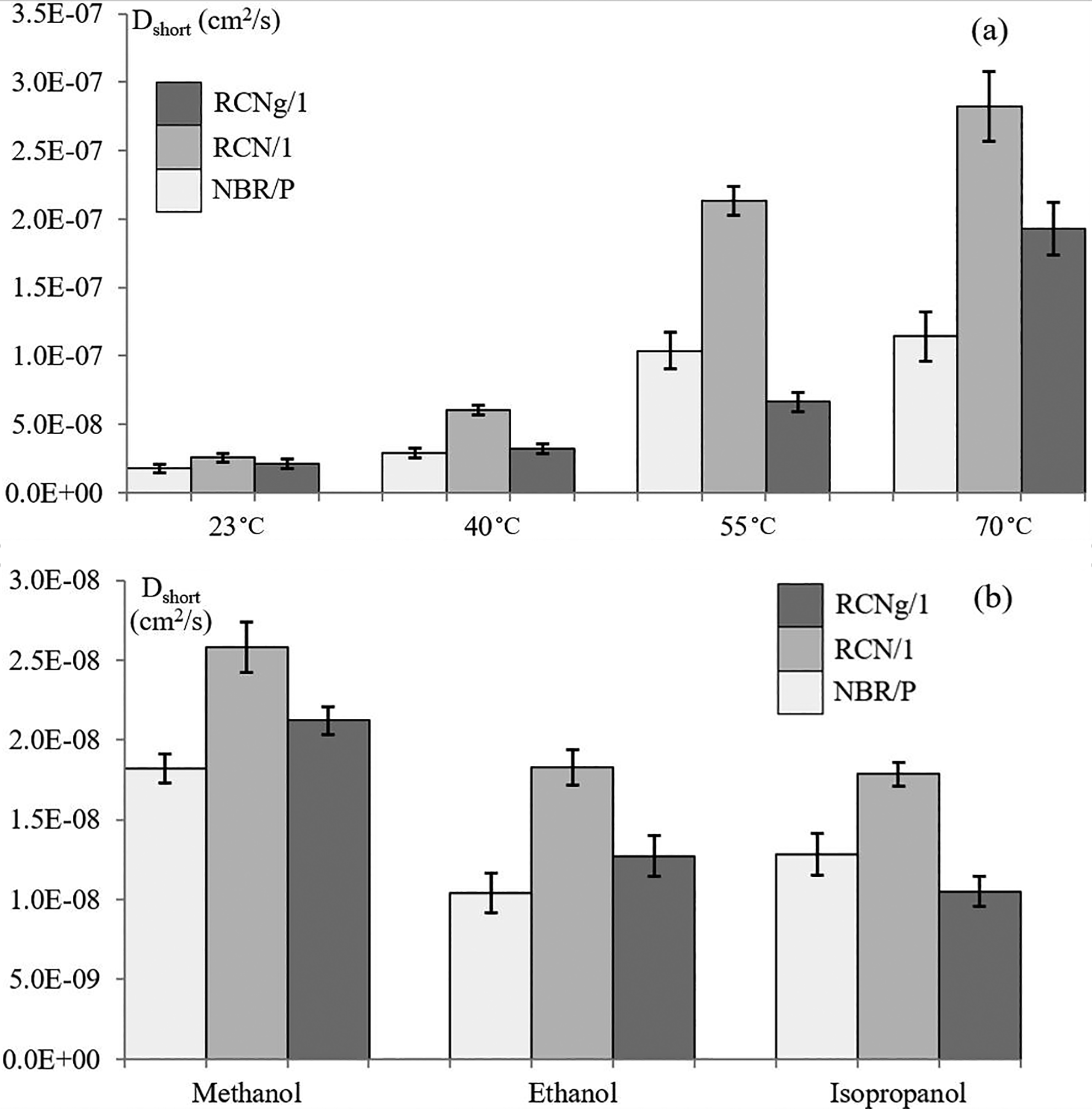
Diffusion coefficient of NBR/P, RCN/1 and RCNg/1: (a) with methanol at 23, 40, 55 and 70°C; (b) with all three solvents at room temperature (23°C).
When the diffusion mechanism obeys Fick’s law and the sorption process is governed by Henry’s rule, the overall permeability of the solvent/membrane system can be determined by multiplying the coefficients of diffusion and solubility. As previously seen, diffusion slightly deviates from the Fickian mode under certain circumstances. Therefore, permeation coefficients calculated in this study are only considered as estimations. An obvious observation is that
Conclusions
Achieving a high level of layered silicate dispersion in a nitrile rubber-based nanocomposite is quite challenging. Physical mixing of the nanometric fillers with the elastomeric matrix does not guarantee enhanced barrier properties. In our previous work, a specially made nanocomposite with a high degree of exfoliation was developed with maleation. In the present study, the chemical resistance of this maleated nanocomposite was compared to that of a conventional filled-material at four different temperatures ranging from 23 to 70°C. Mass uptake experiments based on a gravimetric technique were adopted to assess mass transport characteristics in three alcohols: methanol, ethanol and isopropanol. With all materials and regardless of the immersion medium, findings show a substantial increase of molecular transport parameters with the increase in temperature. These parameters were also found to be inversely proportional to the molecular volume of the penetrant, the only exception being methanol at 70°C. When it came to filling the rubber matrix with nanoclays, the sorption at equilibrium decreased proportionally with the concentration of filler. The decrease was seen to be intensified with the nanoclays’ enhanced state of dispersion. Conversely, diffusivity was clearly noticed to be more sensitive to the morphological properties of the nanocomposite. The presence of bulky fillers within the elastomeric system seemed to negatively affect the diffusion coefficient, which was observed to increase with the addition of 5% of nanoclays, after which, a substantial decrease in diffusivity was witnessed during maleation. The permeation coefficient behaved in the same way and followed the
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
