Abstract
The compatibility of key infrastructure elastomers with five ketone molecules was assessed via solubility studies and volume swell measurements. The elastomer materials included two fluorocarbons, six acrylonitrile butadiene rubbers (NBRs), and one each of fluorosilicone, neoprene, polyurethane, styrene butadiene rubber (SBR), and silicone. The ketone molecules included acetone, 2-butanone, 2-pentanone, 2-nonanone, and cyclopentanone. The ketones were added to gasoline containing 10% ethanol (E10) and a blendstock for oxygenate blending (BOB) in levels ranging from 0% to 30% by volume. The elastomers were exposed for 4 weeks in each test fluid. The solubility was modeled using Hansen solubility parameters and the volume change was determined for each material and test fuel. In general, the volume swell increased with ketone content and corresponded well to the predicted solubilities. In most cases, the highest level of swelling occurred with added cyclopentanone and acetone, while 2-nonanone produced the lowest levels of volume expansion. The chain length of the straight ketones was found to affect the volume swell behavior as volume expansion decreased with increasing chain length. This behavior is attributed to the reduction in polarity and hydrogen bonding with chain length. Neoprene, SBR, and silicone exhibited poor compatibility with the ketone molecules at all blend levels. Fluorocarbon and fluorosilicone also showed poor compatibility but may be suitable for use as static seals in very low blend levels with 2-nonanone. The results were more mixed for polyurethane and the NBRs. In general, better compatibility (low volume swell) was observed for mixtures containing BOB than for E10. This is due to the lower polarity and hydrogen bonding of the BOB.
Introduction
The storage and transfer of gasoline fuels necessitates the use of elastomers as seals and flexible hoses to facilitate operation and minimize cost. When used as a hose material, an elastomer must contain the fuel while at the same time exhibit the proper flexibility and strength. As seals, their purpose is to provide a leak-tight connection between two or more components or systems. 1 In order to prevent leakages and failures, these materials must be compatible with gasoline. However, the chemistry of gasoline has never been constant and has undergone changes in response to consumer and regulatory demands for improved performance and safety. Examples include the removal of tetraethyl lead (beginning in the 1970s) and the more recent addition of 10% ethanol to gasoline as E10. 2 For metallic components (steel, aluminum, copper, etc.), these compositional changes were not an issue since they did not contain acids, moisture, or other corrosive media. 3,4 These metals are inherently compatible with hydrocarbon fuels. However, for polymeric materials, the fuel chemistry can significantly influence the solubility with the polymer. Increased solubility translates to fluid ingress into the polymer structure, which is manifested as volume expansion, or swell. The ingress of one or more fuel components into the polymer structure also affects the polymer mechanical and physical properties. 5 –8
One of the more dramatic changes in gasoline chemistry occurred with the addition of ethanol. The polar nature of ethanol increased the solubility of gasoline with some fuel system elastomers causing unacceptably high levels of swelling. Elastomers that have been shown to be susceptible to high swell with ethanol-blended gasoline include acrylonitrile butadiene rubbers (NBRs), styrene butadiene rubber (SBR), neoprene, and polyurethane. 8 It is important to note, however, that high volume expansion, by itself, does not necessary define compatibility. The application also needs to be considered. Some nominal level of swelling is desirable and helps to provide a tight seal at the interface. However, excessive swelling may cause extrusion and the potential for structural damage. Parker Hannifin states that a good guideline is to consider volume swell of less than 30% as acceptable for static applications while swelling less than 15% is acceptable for most dynamic applications. 9 Because of the critical role that elastomers play in fueling systems, it is of paramount importance to know their compatibility to fuel chemistries before they are introduced in fueling applications.
In 2015, the United States Department of Energy initiated a multi-laboratory and multi-year effort to co-optimize fuels and engines known as the Co-Optimization of Fuels and Engines Initiative (Co-Optima). 10,11 Early activities included identifying potential fuel candidate molecules to be evaluated for their combustion, chemistry, and compatibility properties with current and advanced engine concepts. One of these early studies identified over 35 biomass-derived blendstock compounds, which included several ketones. Ketone molecules have good combustion characteristics and can be readily derived from biomass feedstocks. Therefore, they are of interest as potential carbon-neutral fuels or intermediate fuel feedstock. However, certain ketones have known incompatibilities (i.e. cause high volume expansion) with many elastomers, especially fluorocarbons due the highly polar carbonyl bond. Other structural features, such as molecular chain length, also influence the weak bonding forces which govern solubility.
This article describes results of a solubility analysis which was conducted to better understand the mutual solubility between the fuel chemistry and the elastomers, and subsequent volume swell measurement results. Selected elastomers include two fluorocarbons, six NBRs, and one each of fluorosilicone, neoprene, polyurethane, SBR, and silicone. Materials were exposed to five ketone structures (acetone, 2-butanone, 2-pentanone, 2-nonanone, and cyclopentanone) as blends with E10 and a blendstock for oxygenate blending (BOB). E10 was chosen since it is the current gasoline formulation and the BOB was chosen since it represents the gasoline formulation to which the oxygenate (ethanol) is added to obtain the desired octane number and other specifications Since solubility is manifested as polymer swell, solubility analysis may also be useful to predict relative swelling that may occur between elastomers and fuel types.
Solubility and solubility analysis
For polymer materials (such as elastomers), solubility is the ability of the fluid (solvent) to adsorb and dissolve into the polymer itself. The factors governing solubility are best described by the Flory–Huggins theory of solutions and are related to the forces governing intermolecular attraction. 12 Materials having similar intermolecular forces (such as London dispersion forces, polarity, and hydrogen bonding) will have high solubility. Solubility theory assigns parameters to substances (based on their intermolecular forces). The closer the parameter (or parameters) of one substance is to another, the higher the solubility. In practice, solubility is empirically assessed by measuring the volume increase of the polymer after it has been exposed to the fuel or fluid of interest. However, in lieu of volume measurement, the solubility potential between a polymer and a fuel can be assessed by calculating the difference in the solubility parameters that are characteristic for the polymer and fuel.
There are several approaches used to assess solubility that are based on Flory–Huggins solution theory, but the two most common are the Hildebrand and Hansen methods. The Hildebrand method is one dimensional and uses a single parameter to represent the total attractive force (van der Waals forces). In contrast, the Hansen method employs three separate parameters representing forces not only associated with dispersion cohesive energy, but also polarity (or dipole moment), and hydrogen bonding. Even though the Hansen method has better applicability than single parameter methods, the results from this approach are still approximations. The Hansen approach is the most widely used and parameters have been determined for many organic materials and solvents. Many of these parameters can be located readily from the tables provided in the Hansen Solubility Parameters Handbook. 12 Solubility-based analysis studies are important because they can be used to quickly identify fuel blend chemistries and concentrations which are potentially damaging to certain polymer materials. In this study, a Hansen solubility analysis was performed for common fuel system elastomers and for the ketone blends with E10 and dodecane (which was used to represent the BOB).
Experiment
Test fuels
Reagent grade acetone, 2-butanone, 2-pentanone, 2-nonanone, and cyclopentanone were procured from Sigma-Aldrich, St. Louis, Missouri. The chemical structures showing the location of the carbonyl bonds for each ketone is shown in Figure 1. For the paraffinic ketones, the ketone ranking in order of increasing chain length is acetone < 2-butanone < 2-pentanone < 2-nonanone. In contrast, cyclopentanone has a ring structure and would be expected to exhibit very different behavior.
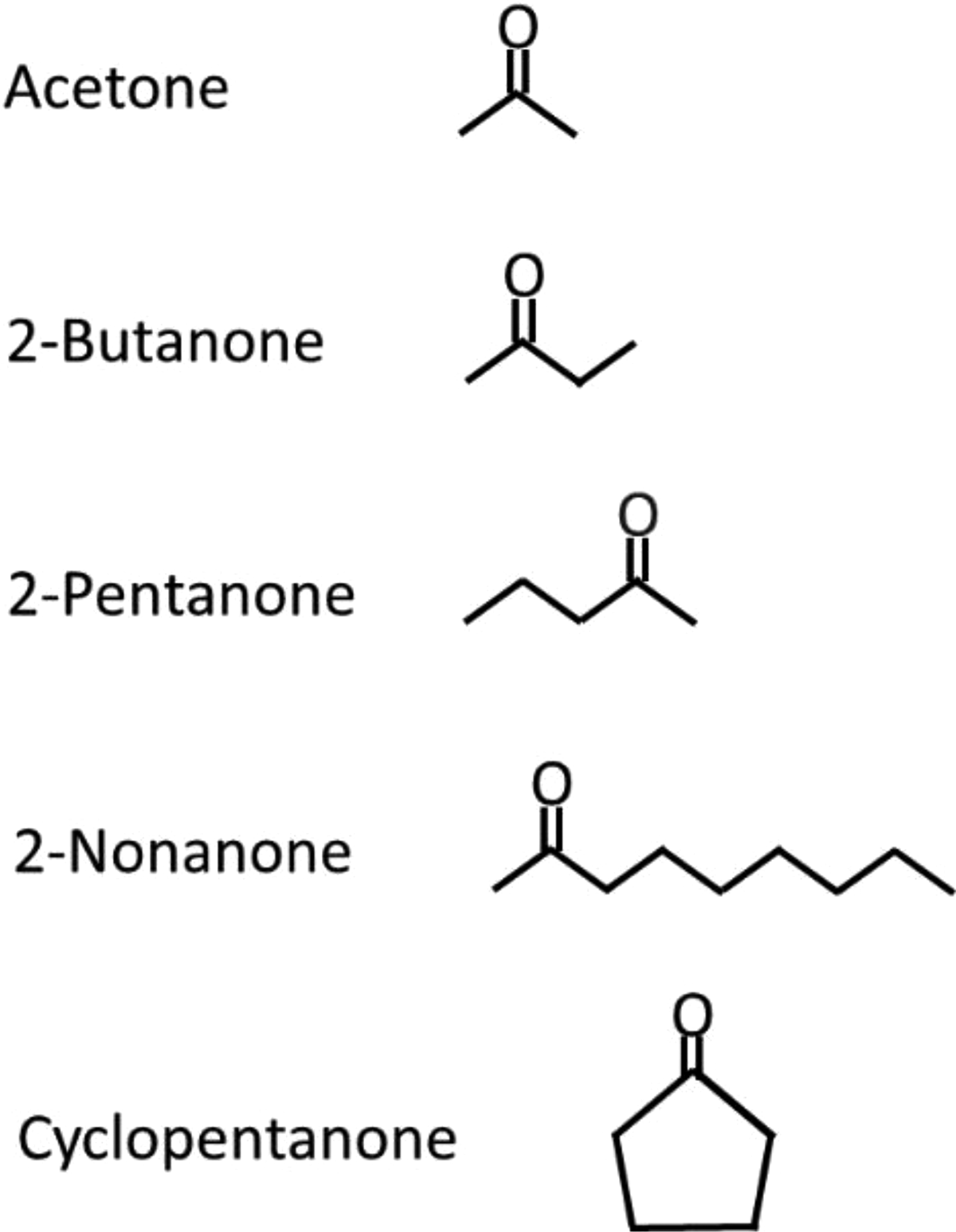
Chemical structures for each of the ketone molecules.
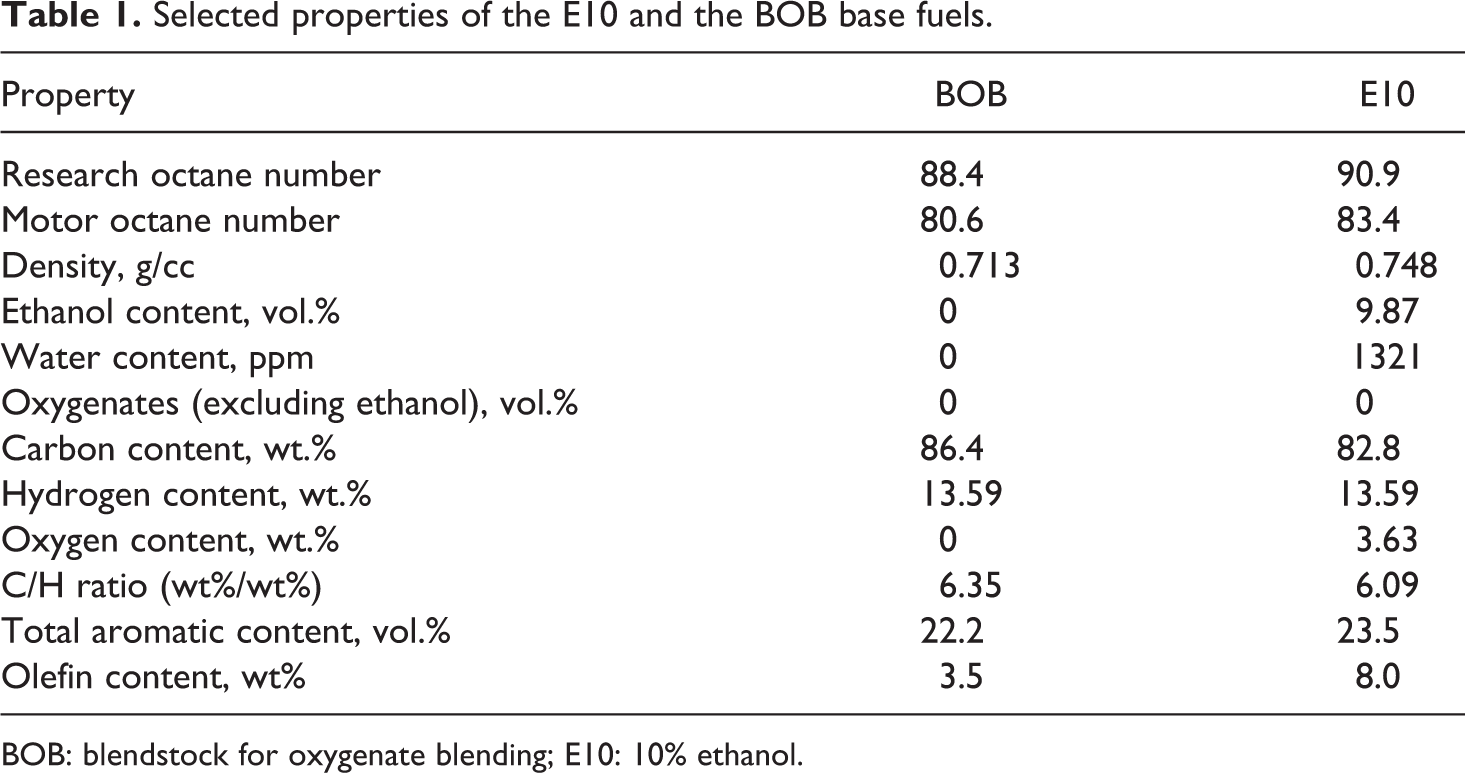
Each ketone was added to the two base fuels (E10 and BOB) at concentrations ranging from 0 to 30 volume percent. The two baseline test fuels (E10 and the BOB) were both procured from Gage Products Company, Ferndale, Michigan. The BOB used in this study was designed to mimic a regular-grade BOB from the marketplace that is used to achieve an antiknock index or pump octane number of 87 with the addition of 10% ethanol. A list of selected properties for these fuels are presented in Table 1. The E10 differs significantly from the BOB since it contains ethanol, which lowers the C/H ratio. Interestingly, the total aromatic content of the E10 roughly matches that of the BOB, while the concentration of olefins is significantly higher.
Selected properties of the E10 and the BOB base fuels.
BOB: blendstock for oxygenate blending; E10: 10% ethanol.
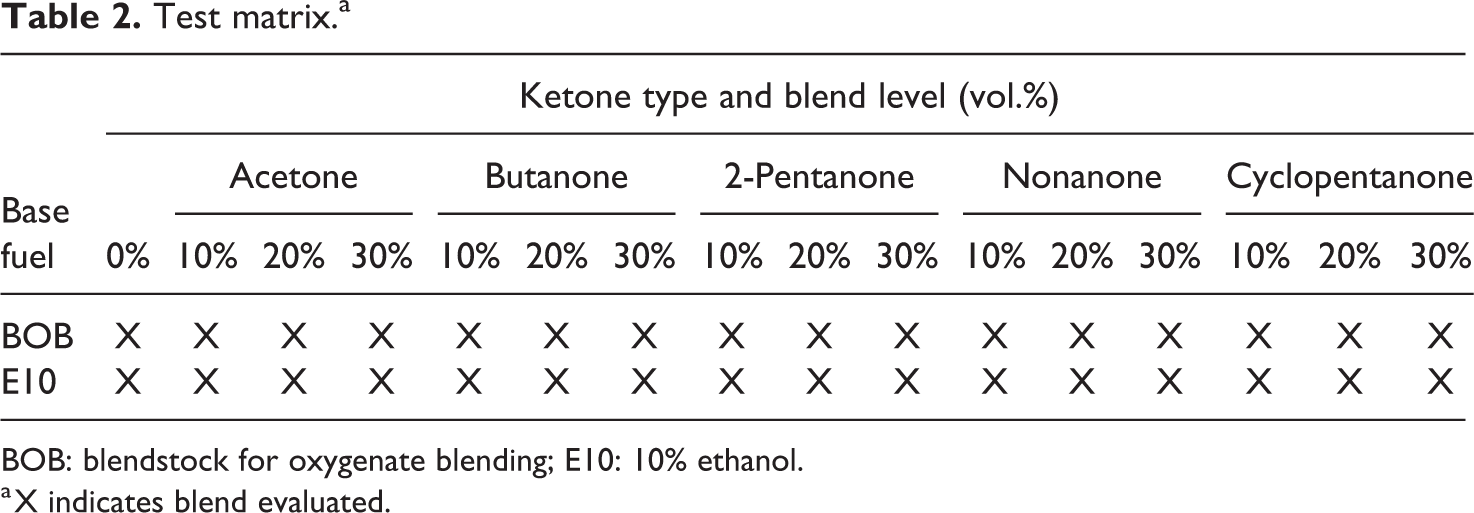
Each of the five ketones was splash-blended into the two base fuels, E10 and BOB, to create blends levels of 0, 10, 20, and 30 vol.% (of each candidate) as shown in the test matrix depicted in Table 2.
Test matrix.a
BOB: blendstock for oxygenate blending; E10: 10% ethanol.
a X indicates blend evaluated.
Elastomer materials
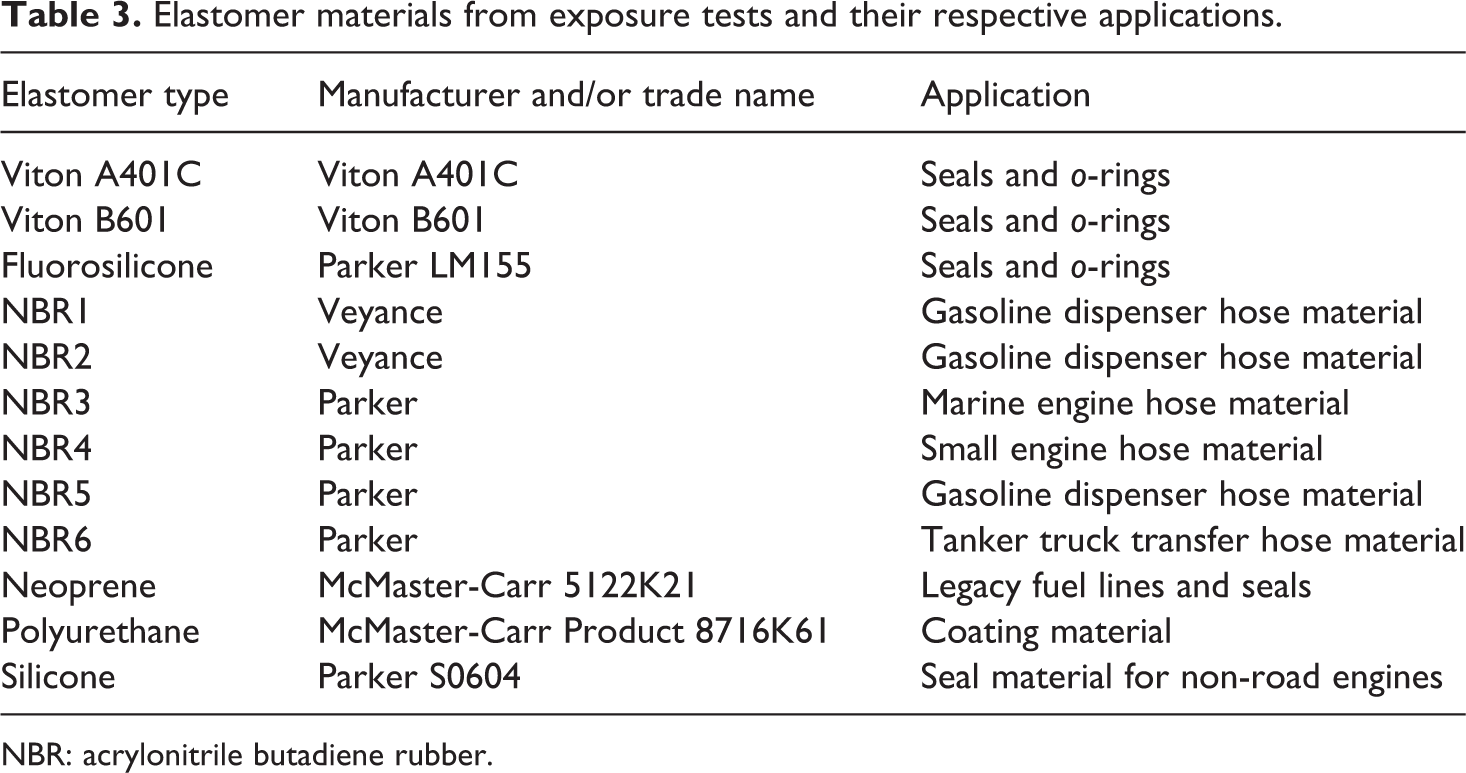
The elastomer materials evaluated in this study included two fluorocarbons (Viton A401C and Viton B601), six NBRs, and one type of fluorosilicone, neoprene, polyurethane, and silicone. A list of these elastomers and their applications is presented in Table 3. Many of these materials are used in sealing applications, especially as o-rings, while the listed NBR grades are used primarily as hose materials. The NBR materials used in this study were heavily compounded with plasticizers, fillers, antioxidants, ultraviolet inhibitors, and other additives. In many cases, the NBR compositions contained over 40% of these additives. Fluorocarbon and fluorosilicone are considered advanced high performance elastomers having high compatibility to ethanol and ethanol–gasoline blends. While silicone is not common in vehicle fueling systems, it is frequently used in small engine fuel systems, which are often fueled with E10, and was therefore included. Polyurethane was also added to the list of exposed elastomers. Although it does not contact the fuel, however, it is a common coating material, used to protect underbodies from corrosion or other degradation mechanisms. For each material type, two specimens were evaluated. The length, width, and thickness for each were 3, 8, 1.3, and 0.2 cm (1.5, 0.5, and 0.08 in.), respectively.
Elastomer materials from exposure tests and their respective applications.
NBR: acrylonitrile butadiene rubber.
The test protocol consisted of immersing the elastomer specimens in each of test fuel blends depicted in Table 2 for 4 weeks at 21°C. The volume change was determined using the protocol described in ASTM D471-06. 13
Results
Solubility analysis
A Hansen solubility analysis was performed to assess the solubility of the blendstocks as a function of blend level and base fuel chemistry. This approach has been described in detail elsewhere. 14,15 The Hansen solubility parameters (HSPs) for the blendstock and base fuels are presented in Table 4, while the HSPs used for the polymers are listed in Table 5. 12 In this study, dodecane was used to represent gasoline and a blend of dodecane and ethanol (referred to as E10D) was used to represent E10. Dodecane was recommended by the Hansen solubility team as a good surrogate for nonoxygenated gasoline. 16 Note that in Table 5, an additional parameter, called the interaction radius, is included for the polymer materials (solutes).
HSPs used for the blendstocks and base fuels.a
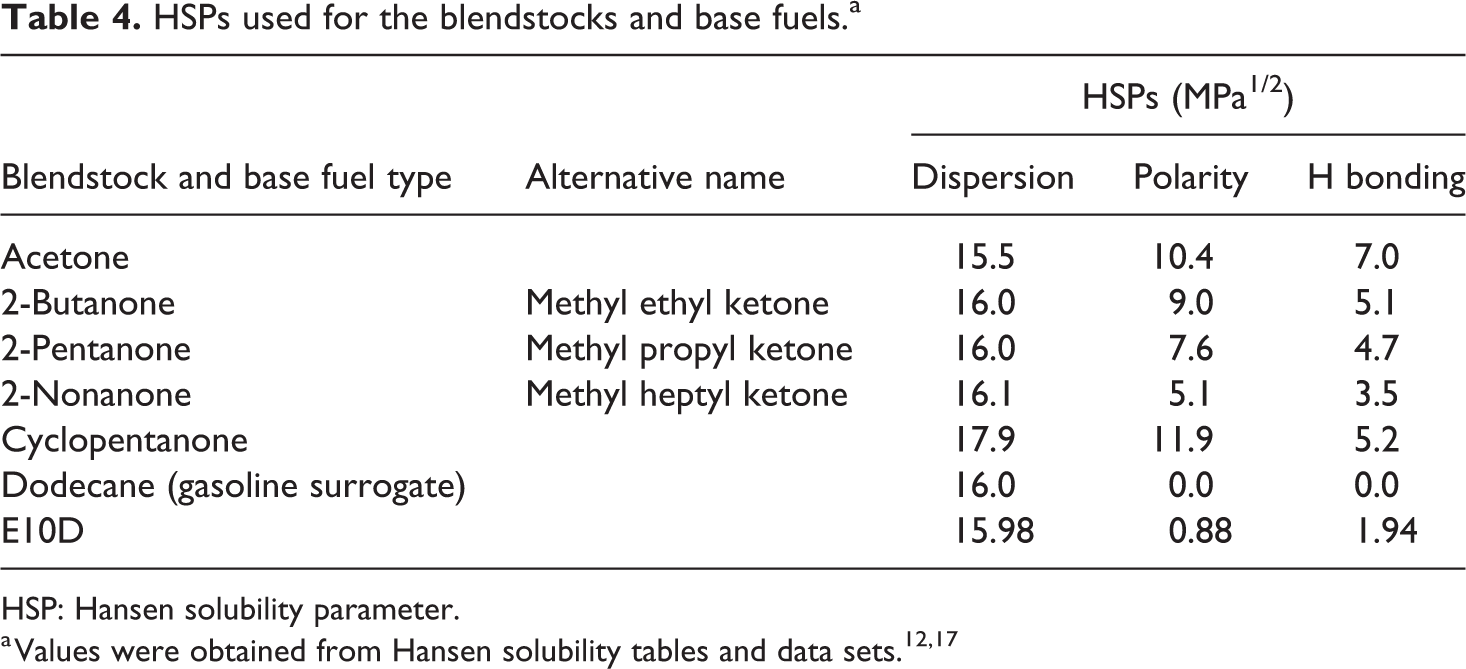
HSP: Hansen solubility parameter.
HSPs used for the elastomer materials.a
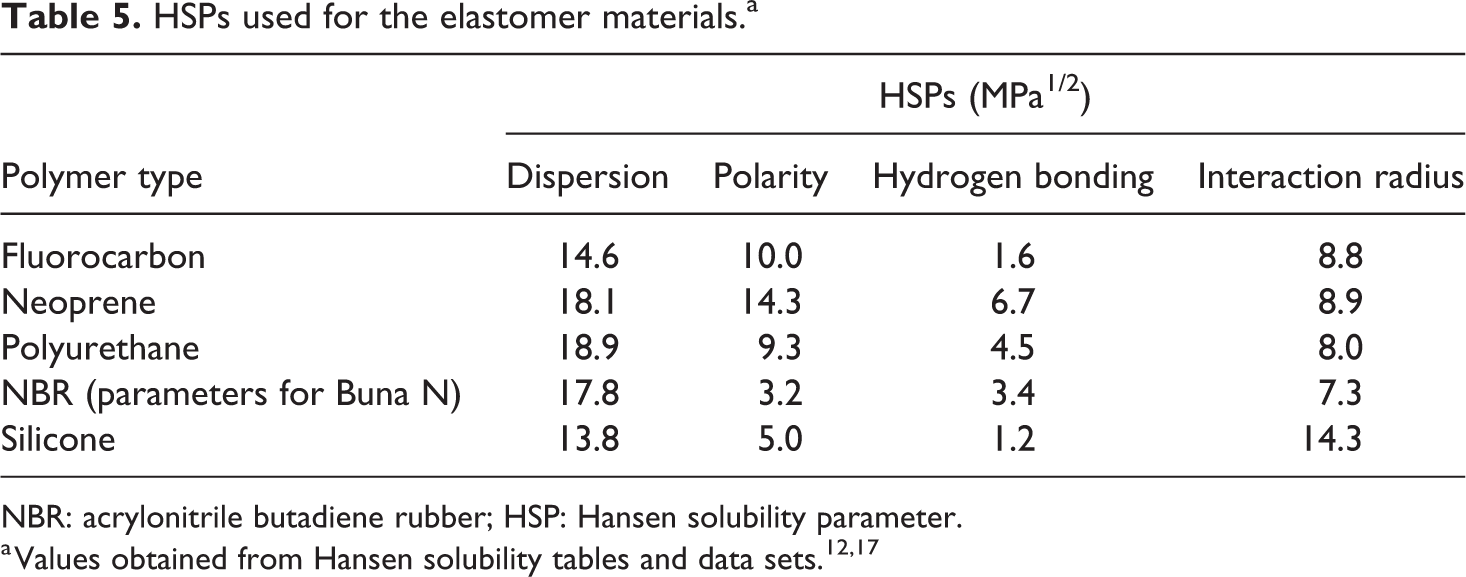
NBR: acrylonitrile butadiene rubber; HSP: Hansen solubility parameter.
A key observation in Table 4 is that the four straight chain ketones have similar dispersion values (though acetone is slightly lower), but the polar and hydrogen bonding parameters decrease with increasing chain length. The effect of increasing chain length is to reduce the overall strength of polarity and hydrogen bonding. Cyclopentanone differs from the straight chain ketones by having notably higher dispersion and polar forces. As presented in Table 5, fluorocarbon, neoprene, and polyurethane all feature high polarity and, as a result, high solubility and swelling would be expected with fuel chemistries that exhibited similar polarity values. In general, the hydrogen bonding values of the elastomers range from low to moderate. The dispersion values of the elastomer vary between types; fluorocarbon and silicone have parameter values less than the ketone molecules and base fuels, while neoprene, polyurethane, and NBR are notably higher than the ketone blendstocks and base fuels.
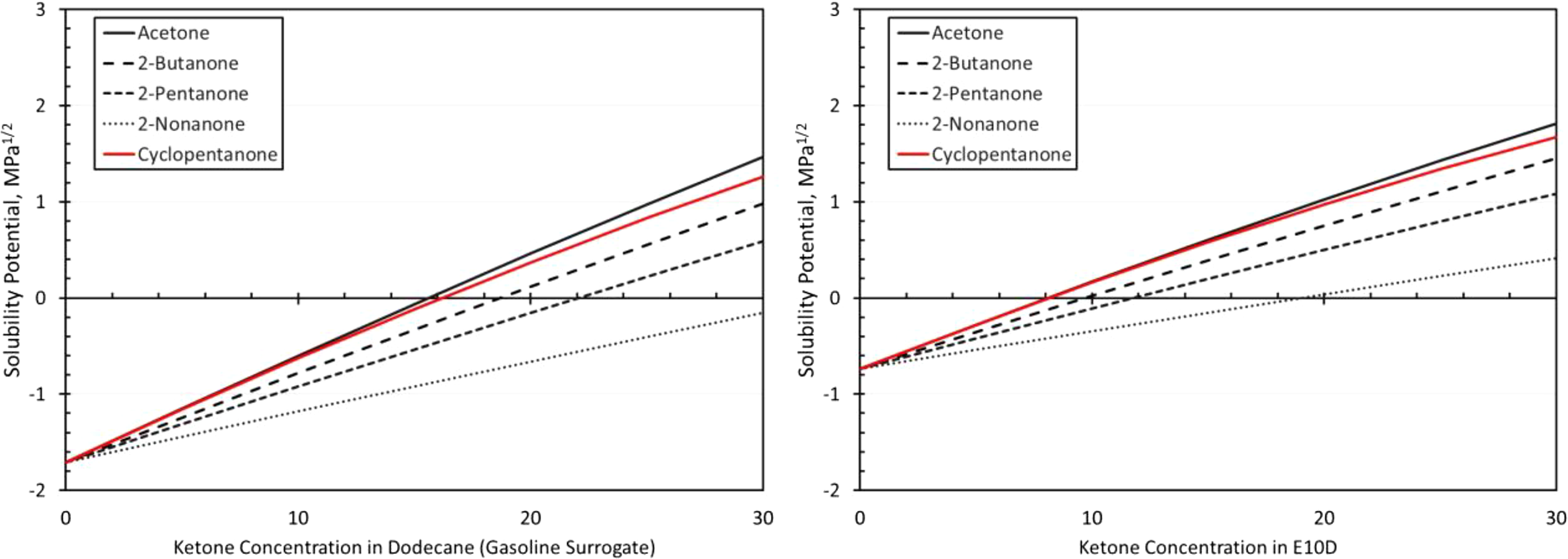
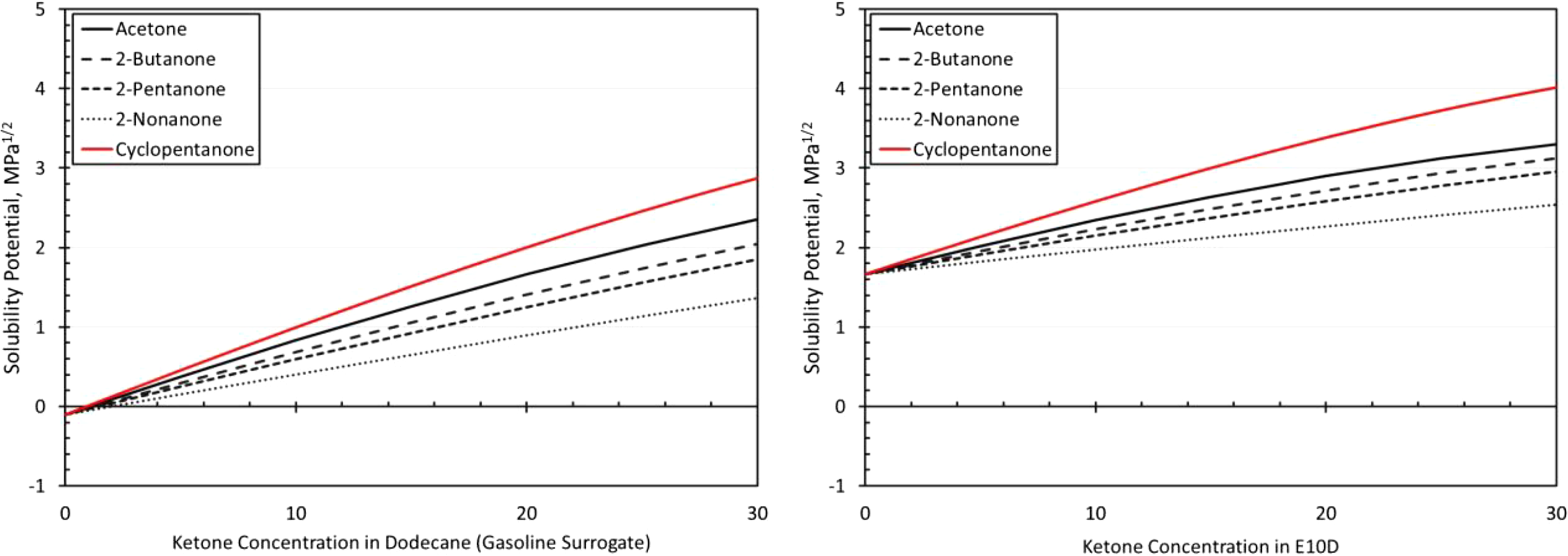
The resulting solubility curves are shown in Figures 2 to 7. Here the level of solubility is expressed as the solubility potential, which is a measure of mutual solubility (attraction) between the fuel and the polymer. Further details on the methodology for determination of the solubility potential have been discussed in previous studies. 14,15 Values much less than zero are considered to have negligible to low solubility, values approaching zero are moderately soluble, while values higher than zero are considered highly soluble. Since volume swell is dependent on solubility, the solubility potential is a useful means of predicting volume swell. The figures show that the solubility potential is observed to increase with ketone concentration, and this increase is due to the high polarity and hydrogen bonding forces associated with the ketones. An interesting deviation occurs for SBR and NBR, where the addition of acetone to the E10D base fuel resulted in maximum solubility for a 20% blend level rather than at 30%.
Solubility curves for fluorocarbon and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. E10: 10% ethanol.
Solubility curves for neoprene and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. E10: 10% ethanol.
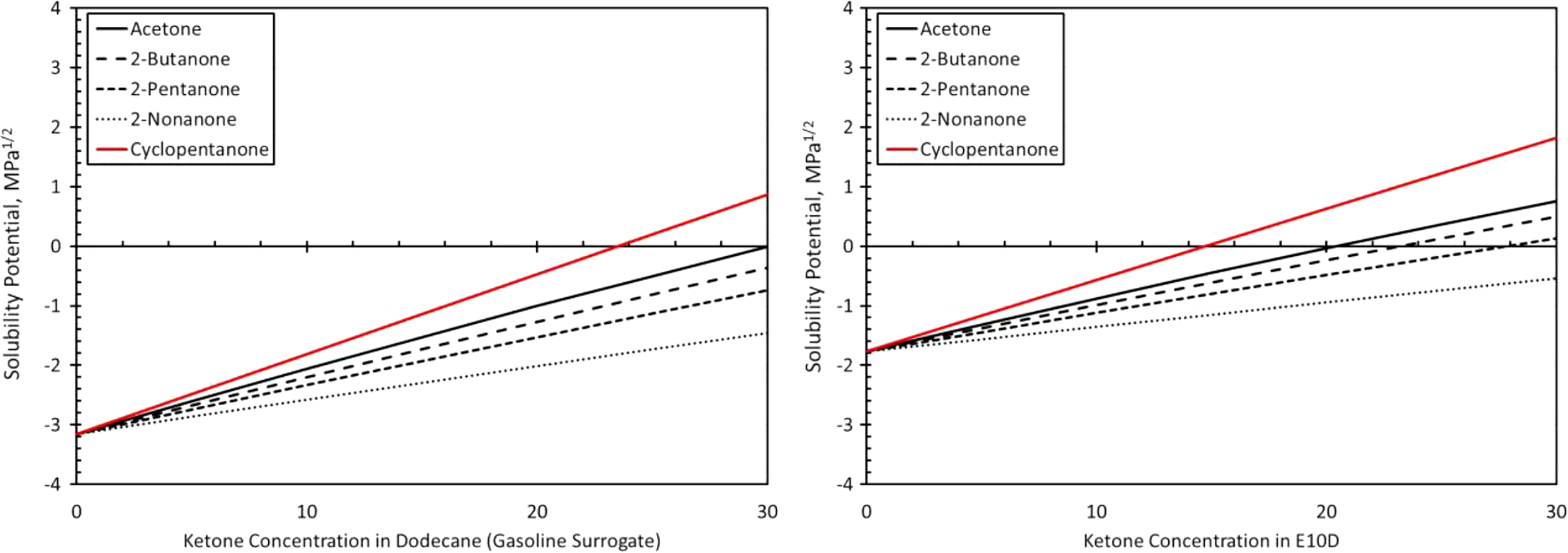
Solubility curves for polyurethane and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. E10: 10% ethanol.
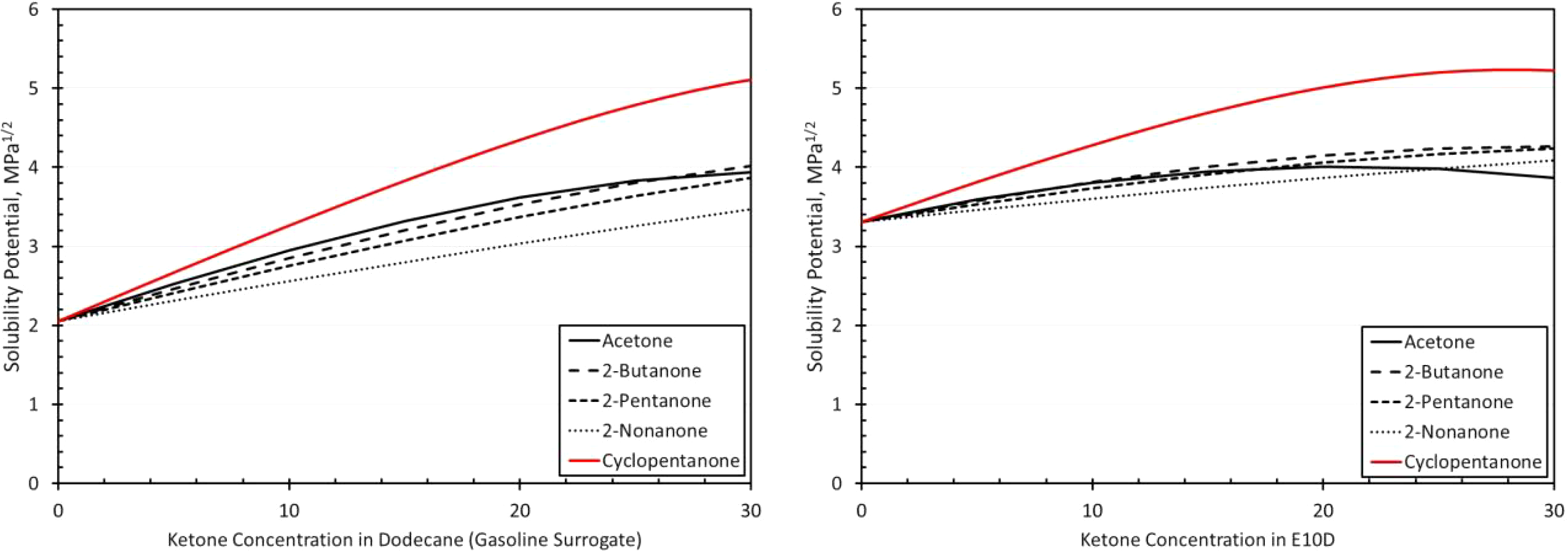
Solubility curves for SBR and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. SBR: styrene butadiene rubber; E10: 10% ethanol.
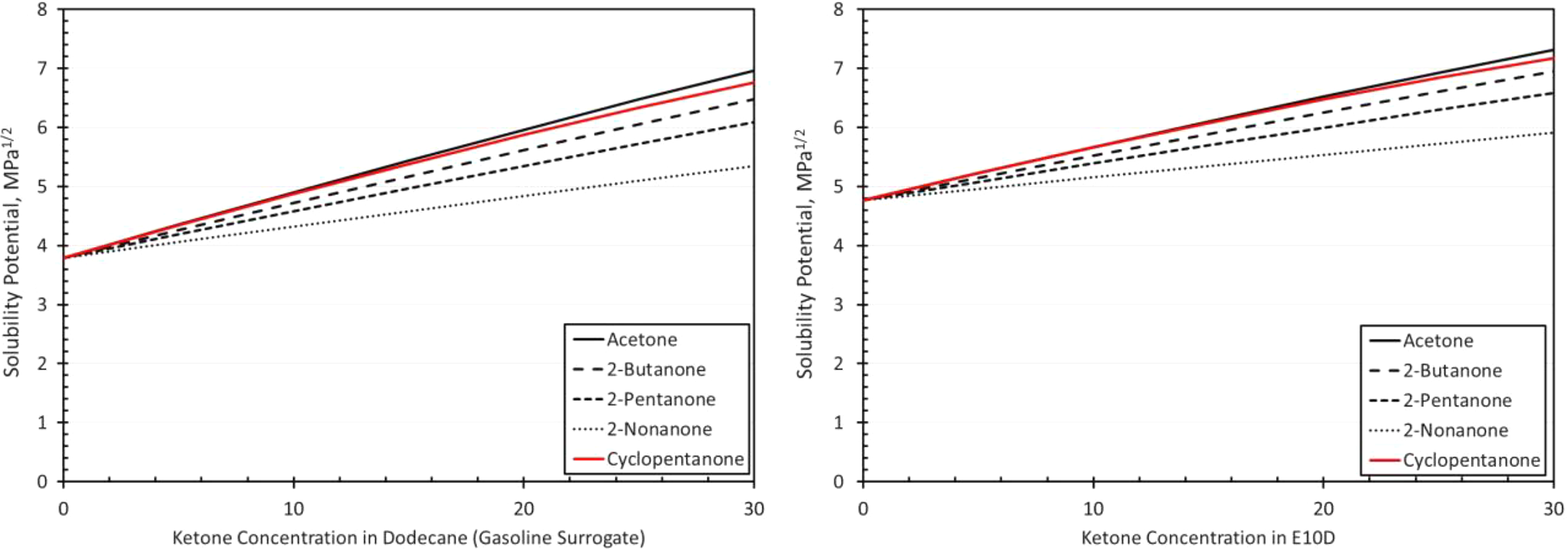
Solubility curves for silicone and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. E10: 10% ethanol.
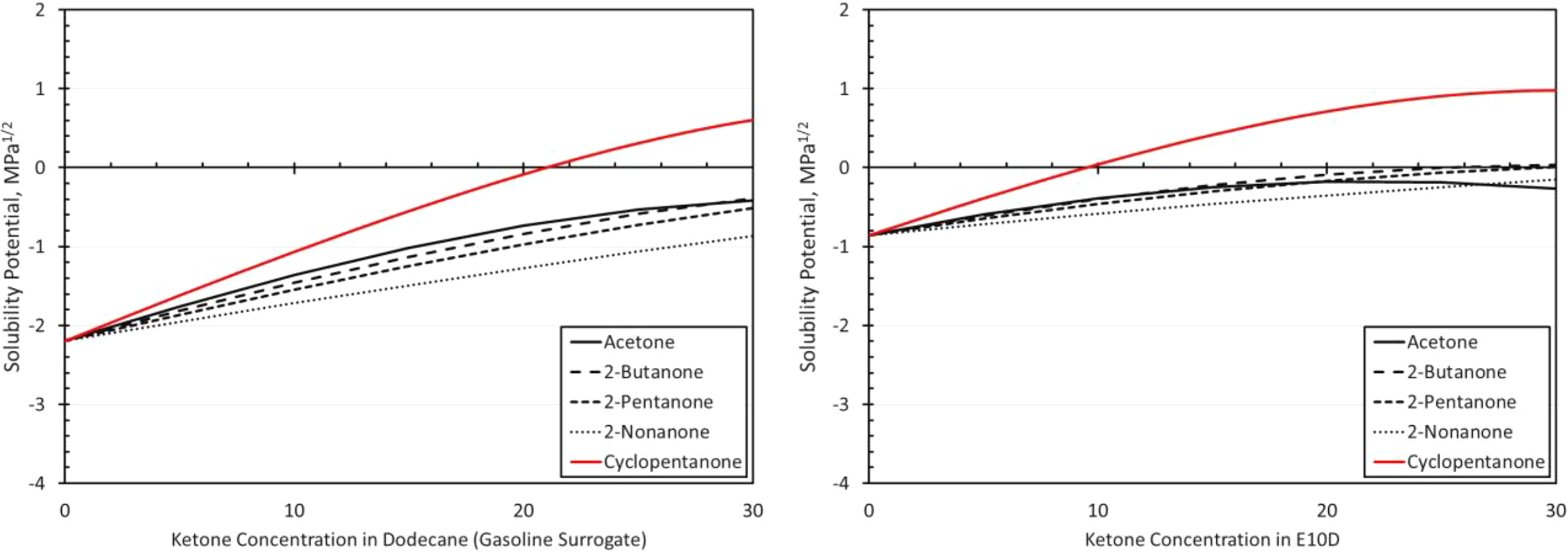
Solubility curves for NBR and each ketone as a function of blend level in dodecane (a gasoline surrogate) and E10D (an E10 surrogate) as depicted on the left and right graphs, respectively. NBR: acrylonitrile butadiene rubber; E10: 10% ethanol.
A key general observation is that the solubility is higher for E10D base fuel than for dodecane. This solubility increase is attributed to the higher polarity associated with the added ethanol of the E10D fuel. However, this difference becomes less pronounced with increasing ketone concentration, since the additional polarity of the ketone molecule becomes more dominant. The raised polarity (as indicated by the higher HSP for polarity) of this fuel (E10D) more closely matches the values of the elastomer materials, as presented in Tables 4 and 5.
Another general observation is that for each elastomer, the solubility decreases with increasing chain length of the ketone molecules. This effect is attributed to the decreasing polarity and hydrogen bonding associated with increasing chain length, and it is especially pronounced for fluorocarbon and silicone. In most instances, cyclopentanone, which has the highest polarity and hydrogen bonding of the ketones, produced the highest solubility potential. However, for fluorocarbon and silicone, as shown in Figures 2 and 3, the solubility potential curves of cyclopentanone roughly matched those of acetone.
Measured volume change results
The measured volume change results for fluorocarbon, fluorosilicone, neoprene, polyurethane, SBR, and silicone are shown in Figures 8 to 13, while the results for the six NBRs are shown in Figures 14 to 19. For each elastomer, higher swelling was measured in E10 than for the BOB base fuel. This result is consistent with the predicted solubilities shown in Figures 2 to 7 for these materials.
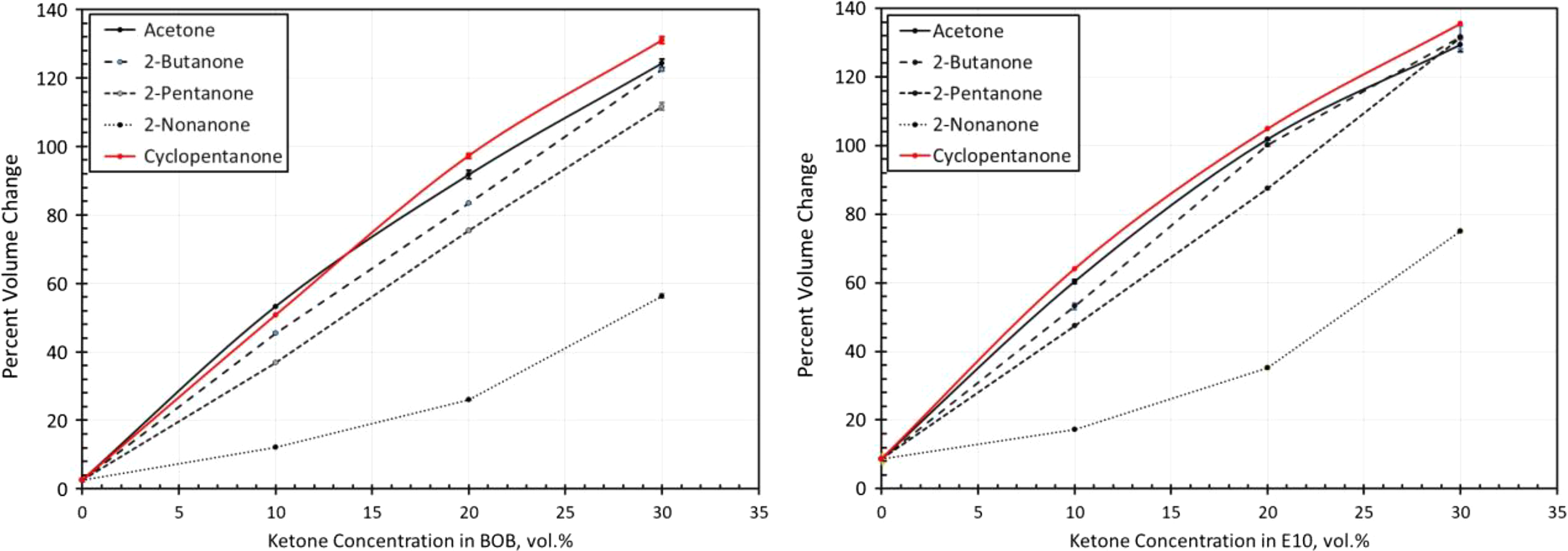
Volume change results for fluorocarbon exposed to each ketone blended with BOB and E10. BOB: blendstock for oxygenate blending; E10: 10% ethanol.
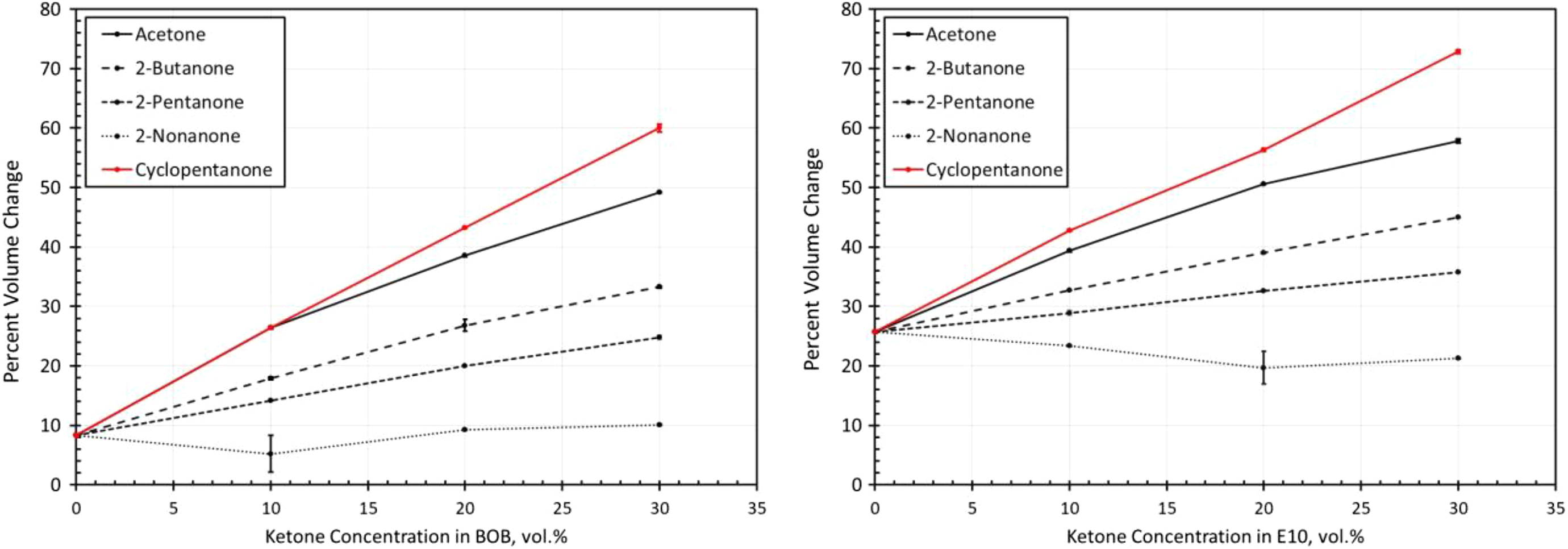
Volume change results for fluorosilicone exposed to each ketone blended with BOB and E10. BOB: blendstock for oxygenate blending; E10: 10% ethanol.
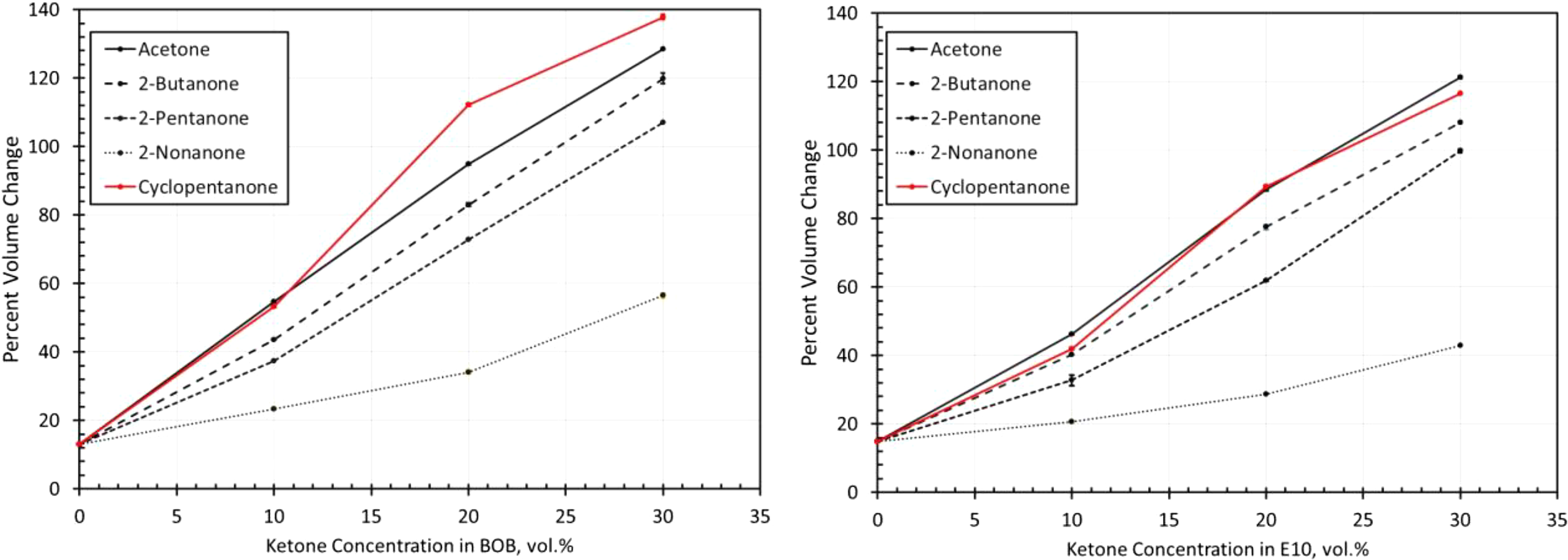
Volume change results for neoprene exposed to each ketone blended with BOB and E10. BOB: blendstock for oxygenate blending; E10: 10% ethanol.
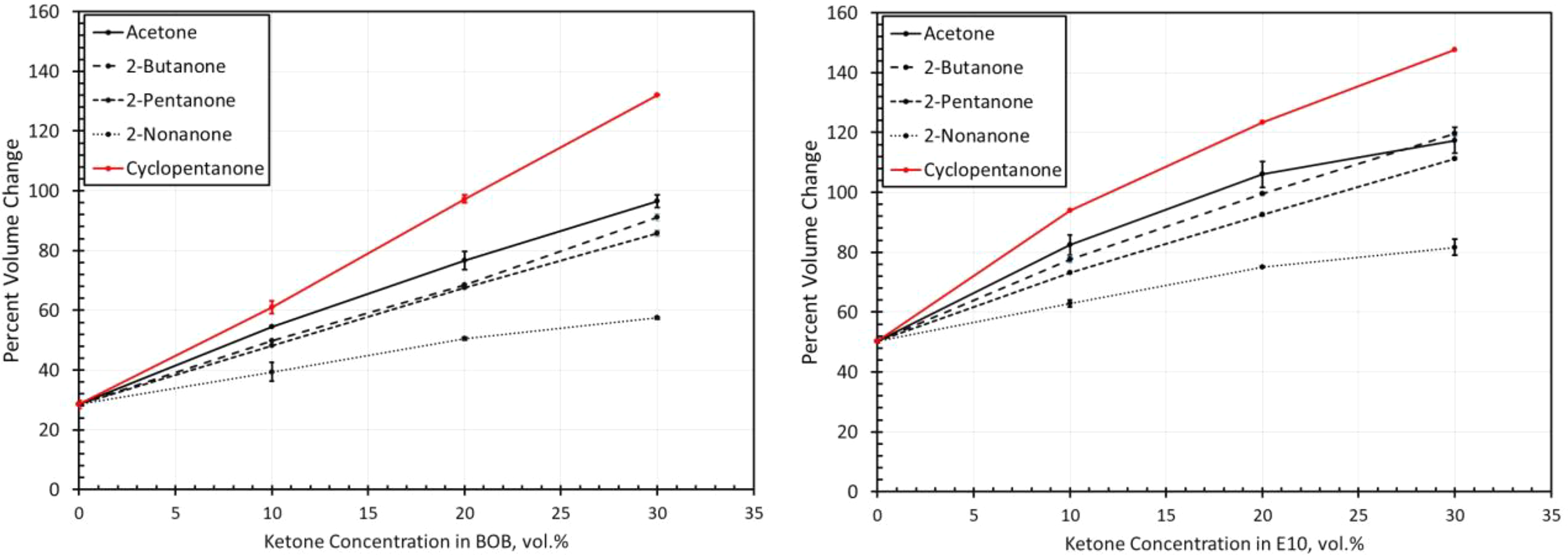
Volume change results for polyurethane exposed to each ketone blended with BOB and E10. BOB: blendstock for oxygenate blending; E10: 10% ethanol.
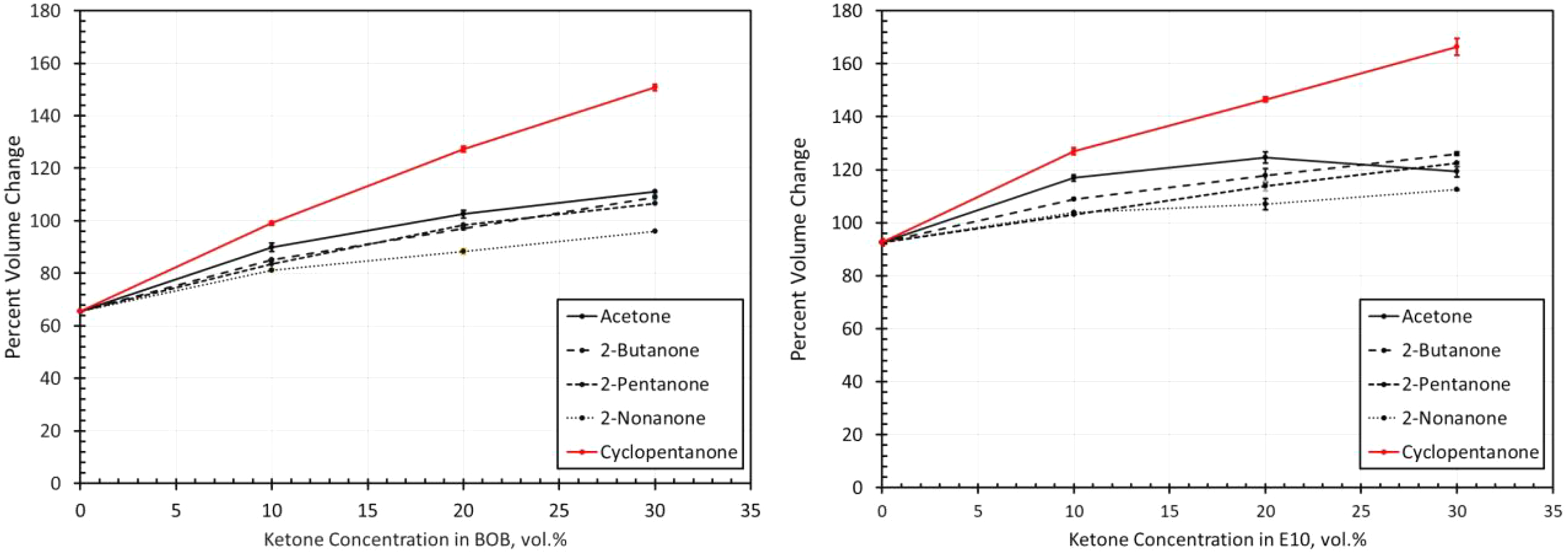
Volume change results for SBR exposed to each ketone molecule blended with BOB and E10. SBR: styrene butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
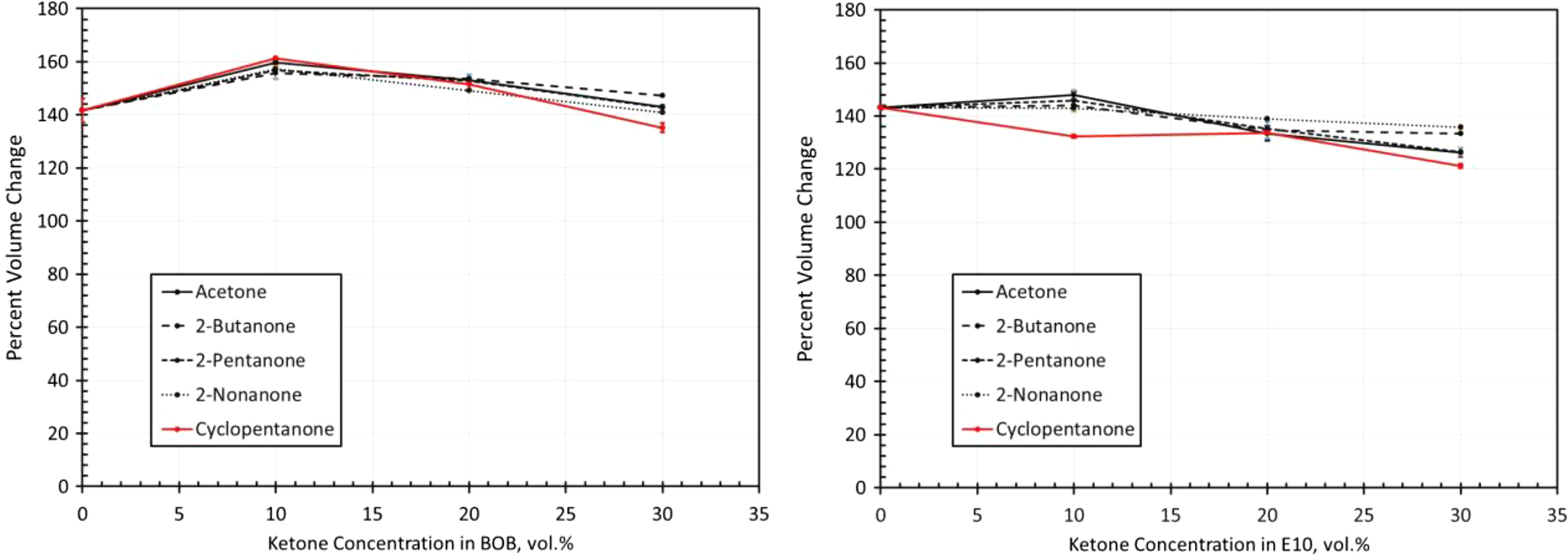
Volume change results for silicone exposed to each ketone blended with BOB and E10. BOB: blendstock for oxygenate blending; E10: 10% ethanol.
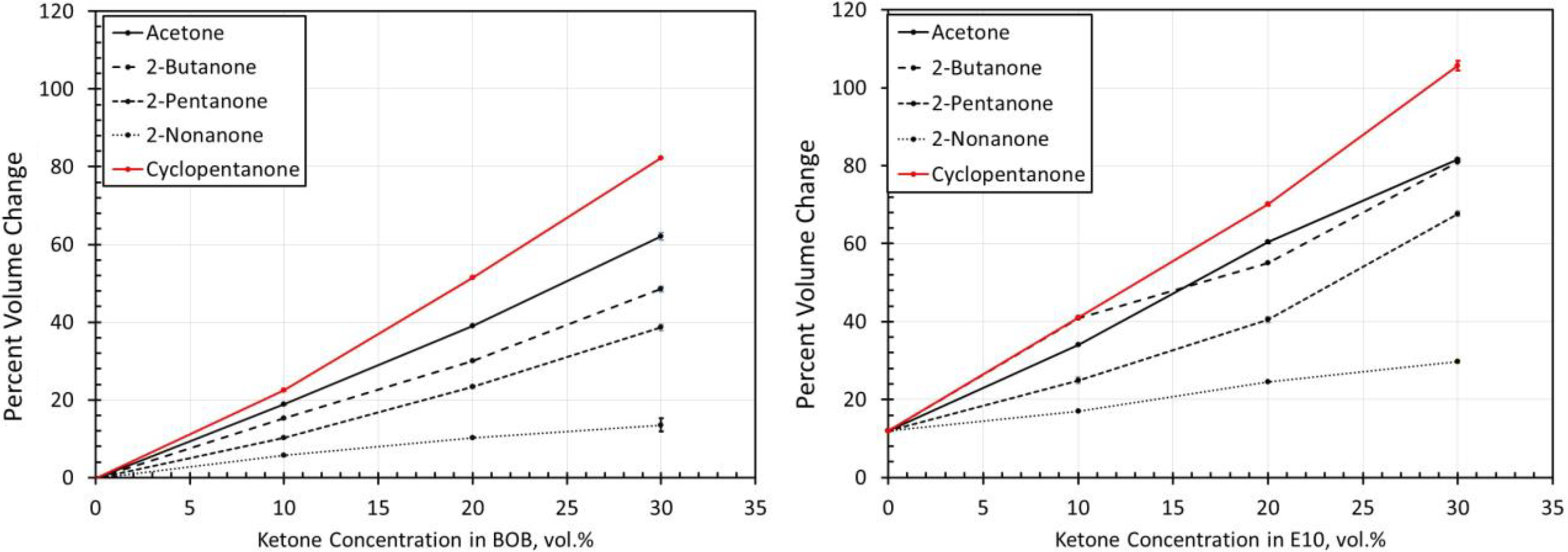
Volume change results for NBR1 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
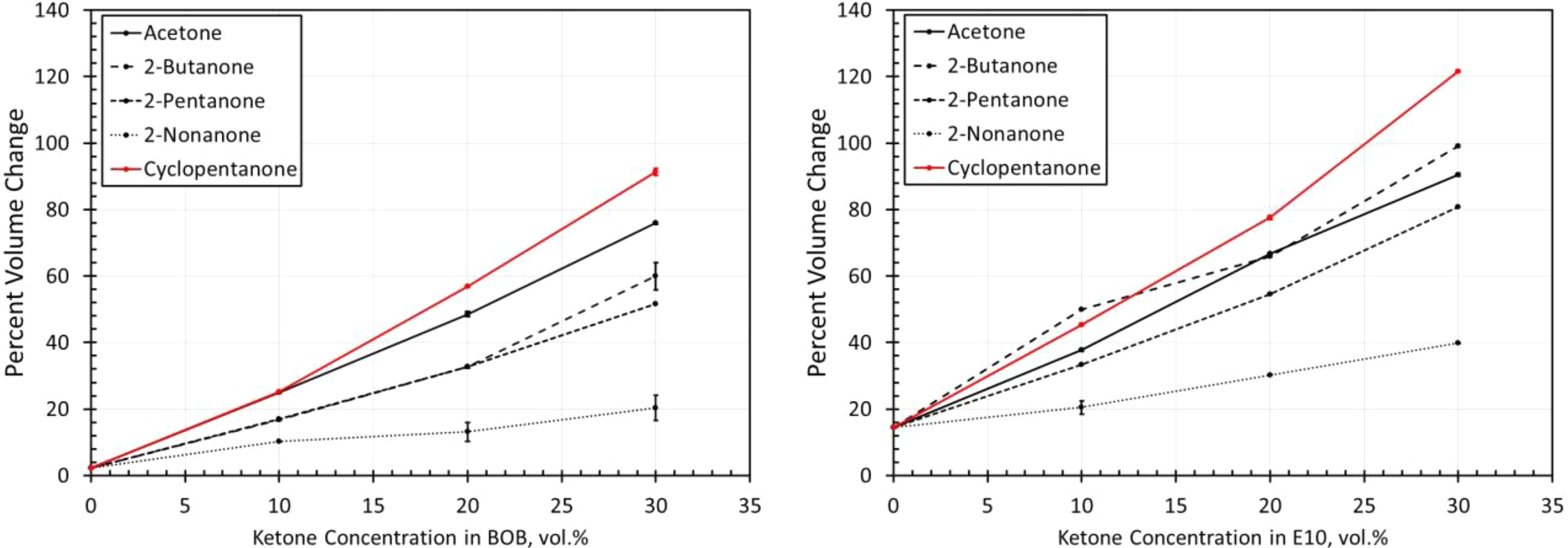
Volume change results for NBR2 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
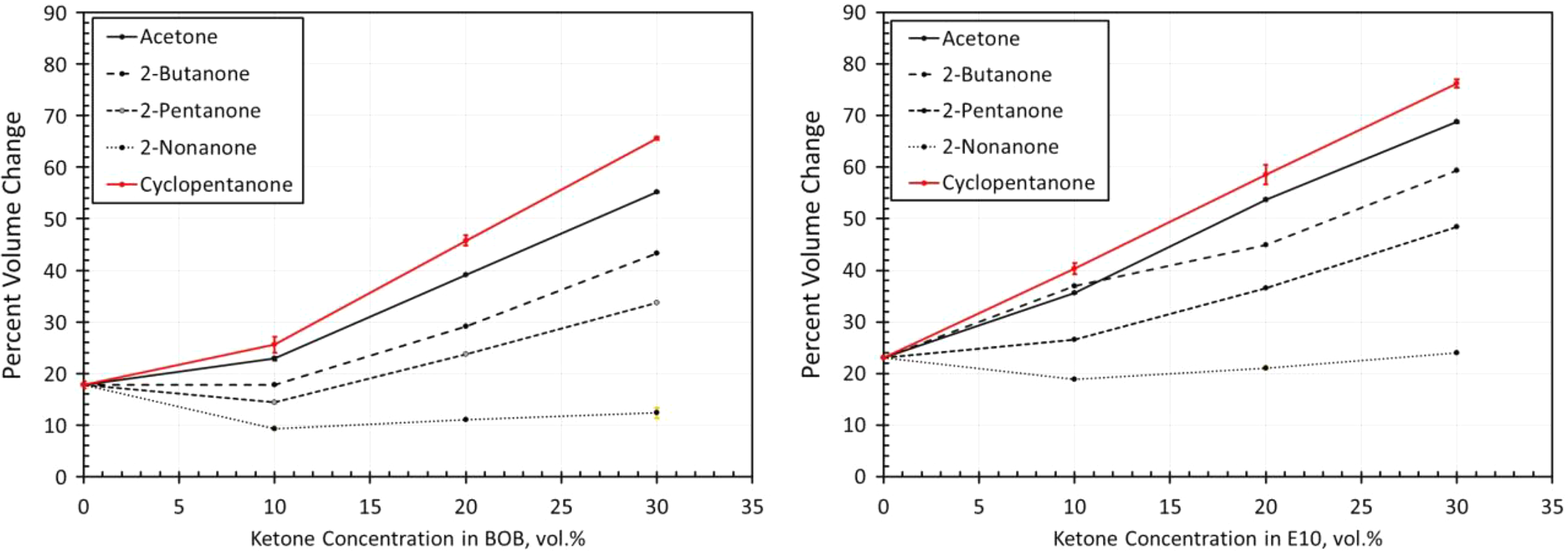
Volume change results for NBR3 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
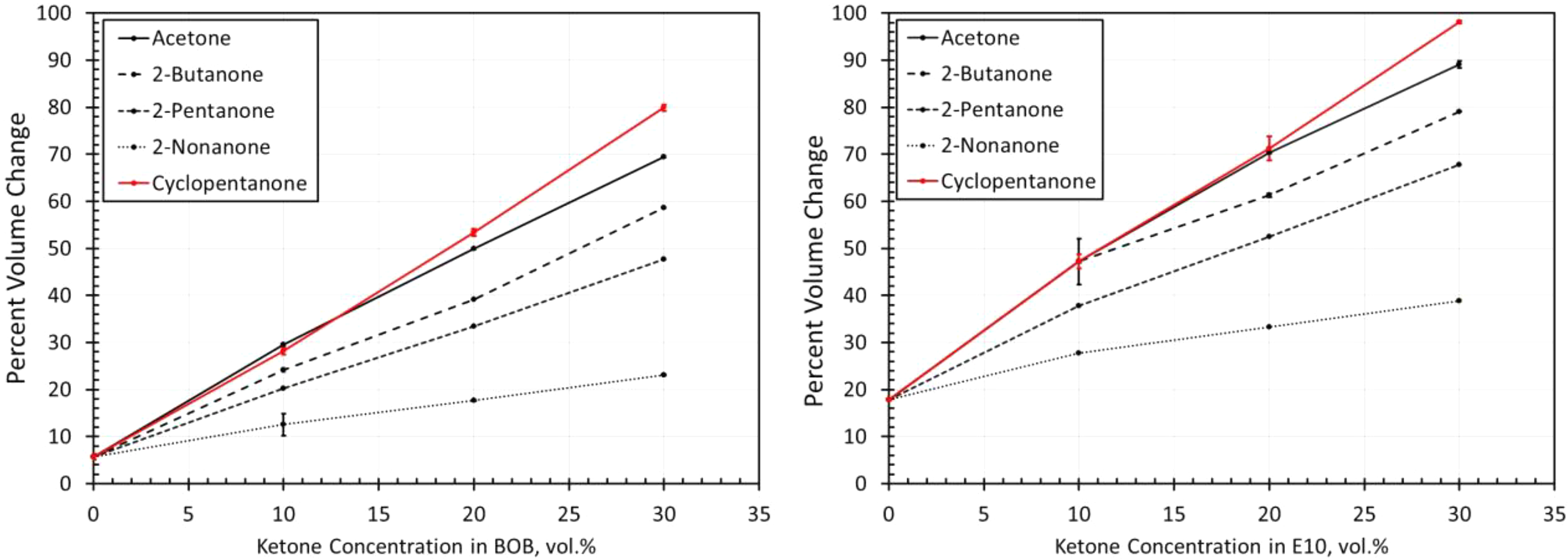
Volume change results for NBR4 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
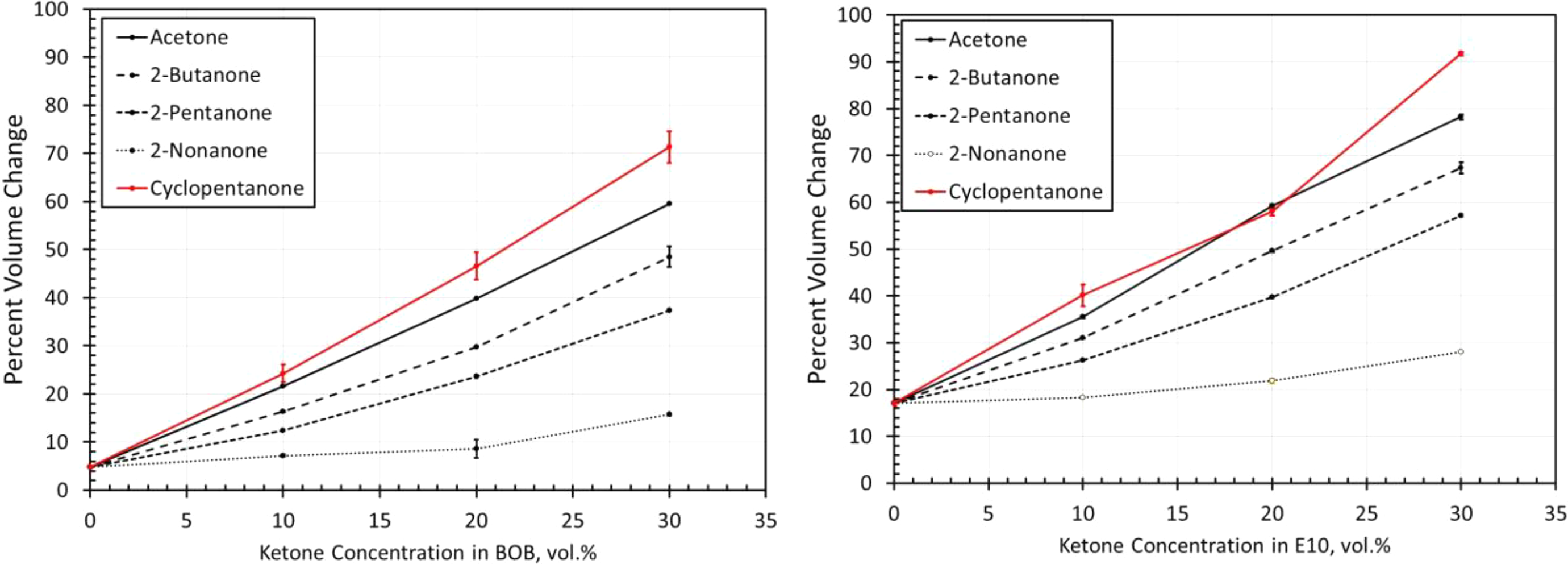
Volume change results for NBR5 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
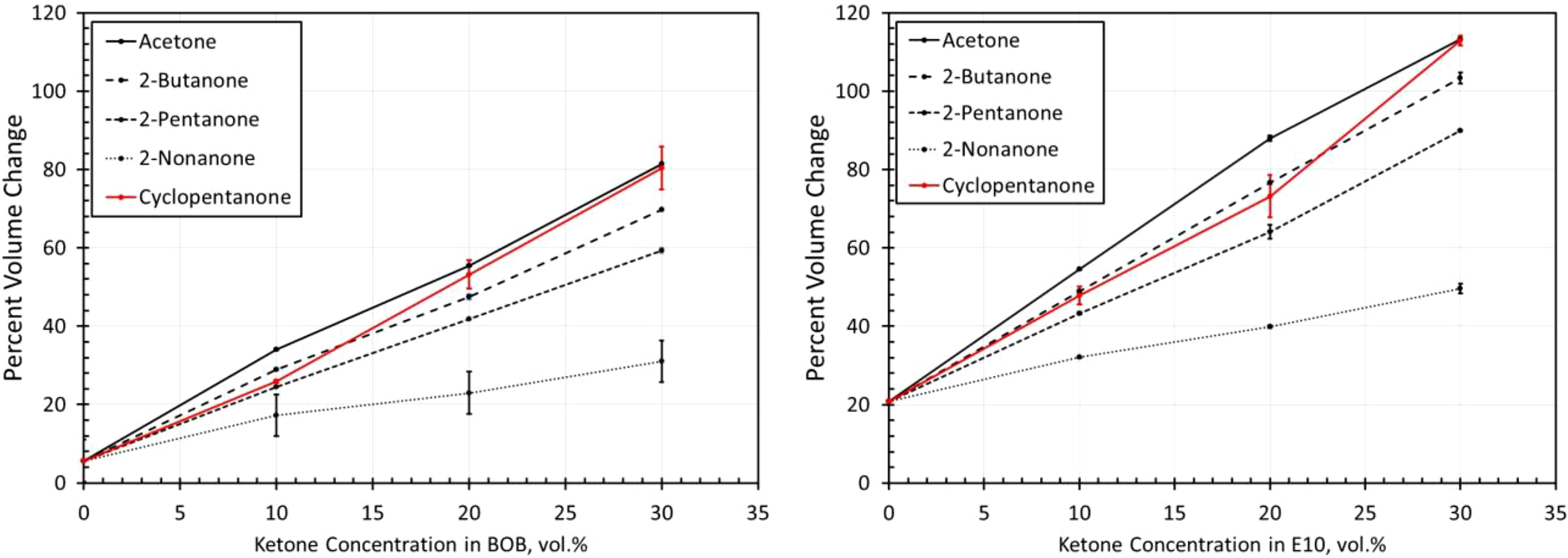
Volume change results for NBR6 exposed to each ketone blended with BOB and E10. NBR: acrylonitrile butadiene rubber; BOB: blendstock for oxygenate blending; E10: 10% ethanol.
Fluorocarbons
The two fluorocarbon materials (Viton A401C and Viton B601) gave very similar performance. As a result, the curves for Viton A401C are shown in Figure 8 to represent both materials. The relative differences in volume swell resulting from the different ketones roughly follow the solubility curves in Figure 2. As predicted in Figure 2, the observed volume swell behaviors for cyclopentanone and acetone were similar, especially when BOB is used as the base fuel. The relative level of swelling caused by the ketone molecules corresponds to the solubility curves (Figure 2). Acetone produced the highest swell, the next highest was 2-butanone, followed by 2-pentanone. 2-Nonanone produced the lowest level of swelling in the fluorocarbons and was significantly lower than the other ketones, as shown in Figure 8. The level of volume swell was observed to increase with decreasing chain length, which further indicates that polarity and hydrogen bonding were primarily responsible for the heightened solubility of fluorocarbon with ketone addition. The solubility analysis was useful in predicting the relative behaviors and trends of the ketones with the two Viton materials. However, the actual level of swelling in the two fluorocarbons was higher than would be expected from the solubility curves. For example, the solubility curves predict moderate solubility (and therefore swell) at 20% ketone content. However, at this point, the measured swell was over 60% (which is considered high) for cyclopentanone, acetone, 2-butanone, and 2-pentanone. The measured swell for 2-nonanone was around 25%, which is more in-line with the predicted solubility. 2-Nonanone showed good compatibility with the two fluorocarbons, but the other ketone molecules would not be considered suitable for use with these fluorocarbons.
Fluorosilicone
The volume change results for the fluorosilicone material are shown in Figure 9. These results were similar to the fluorocarbon materials, but the overall level of measured swelling was higher at the lower blend levels (<10%). Interestingly, at higher blend levels with E10, the volume swell was actually lower than for the fluorocarbons. The inability to obtain solubility parameters for fluorosilicone precluded the development of solubility curves. However, the resulting behavior showed strong similarities with the fluorocarbon curves in Figure 2.
Neoprene
The swelling behavior of neoprene with the ketones (in Figure 10) shows good correlation with the predicted solubilities in Figure 3. Exposure to the neat BOB fuel produced a volume increase of around 30%, which is considered acceptable for many static sealing applications, but not for dynamic applications. 9 The E10 baseline test fuel produced a significant (almost twofold) increase in volume expansion for neoprene compared to the BOB. The addition of cyclopentanone produced the highest level of swelling as predicted by the solubility curves in Figure 3. Note that in Figure 3, the curves for 2-butanone and 2-pentanone are similar, with 2-butanone being slightly higher on average. This closeness is manifested in the volume change measurements in Figure 10, especially when BOB is used as the base fuel blend. The lowest level of swelling was observed with the addition of 2-nonanone. However, even for 2-nonanone, the added volume expansion is considered too high for most sealing applications.
Polyurethane
As with neoprene, the polyurethane material also exhibited a significantly higher swell in E10 than for the BOB fuel as shown in Figure 11. While the relative placement or order of the curves (from highest to lowest) for each ketone molecule roughly follows the solubility patterns in Figure 4, there are several key differences. The solubility curves suggest very low solubilities at low blend levels. However, the results in Figure 11 show modest swelling for low blend levels in the BOB fuel. Moderate-to-high swelling occurs when E10 was used as the base fuel. Another important distinction is that the addition of 2-nonanone was not observed to increase swelling in either base fuel. In fact, a small decrease in swell with concentration was noted for E10 blends with 2-nonanone. Polyurethane shows good compatibility in BOB blended with 2-nonanone at blend levels up to 30%. 2-Pentanone shows good compatibility with polyurethane at blend levels less than 10%, but higher blends are not likely to be suitable for use with polyurethane as dynamic seals. The addition of 2-butanone causes polyurethane to swell to levels meeting the acceptability limit for static seal applications. However, polyurethane would not be suitable as dynamic seals as the swell exceeds the 30% rule of thumb guideline. Cyclopentanone and 2-butanone both caused similarly high swell behavior unsuitable for use with polyurethane. When E10 is used as the base fuel, the levels of swelling are higher than for equivalent levels of BOB. E10 and blends with 2-nonanone show acceptable swell levels for static, but not dynamic seals. A 10% blend with 2-pentanone would also meet the criteria for allowable static swell. Higher blend levels of 2-pentanone, along with additions of 2-butanone, produce relatively high swelling and preclude the use of polyurethane with these fuels.
Styrene butadiene rubber
Figure 12 shows that high levels of SBR swelling occurred in the test fuels, even for the two base fuels, BOB and E10. These results are consistent with the high solubilities predicted in Figure 5. Such high levels of swell preclude the use of SBR as a seal material in BOB, E10, and their blends with ketones. As with the other elastomer materials, the highest swelling occurred with cyclopentanone and the lowest with the 2-nonanone. However, for the acetone blended with E10, the volume swell drops markedly when moving from 20% to 30%. At 30%, the volume swell is lower than those for 2-butanone and 2-pentanone. This behavior shows some similarity with the solubility curve for acetone (Figure 5) which also shows a reduction in volume swell with acetone for the 30% blend in E10.
Silicone
As shown in Figure 13, silicone differed from the other elastomer materials in that the volume swell measurements for the different ketones (including cyclopentanone) were similar. Another key difference is the reduction in volume swell with ketone content for concentrations higher than 10%. The excessively high volume swell results and similar behaviors correspond to their predicted solubility results as shown in Figure 6. However, the solubility behavior shows an increase with ketone content over the blend range which contrasts with the measured results in Figure 13. The reason for this discrepancy is not clear. Even though the addition of ketones to E10 reduced the volume swell (in most instances), the overall high swell levels preclude the use of silicone with these fuel chemistries.
Results for NBRs
The volume change results for the NBR materials are shown in Figures 14 to 19. The results show some similarities in that the volume expansions of the NBR specimens were higher for the E10 fuel and its blends, than for the BOB. This result is consistent with many of the other elastomers investigated in this study. It is interesting to note that the extent of volume swell varied for each NBR grade. For instance, NBR1 showed no volume change in neat BOB fuel, while NBR3 swelled around 18% in the BOB fuel. In each case, the addition of cyclopentanone and acetone produced the highest level of volume swelling and 2-nonanone produced the lowest volume expansion. This result corresponds with the other elastomer materials in this study and follows the trend in which volume expansion decreases with increasing chain length of the ketone molecules. The measured results do not completely align with the solubility curves for NBR as shown in Figure 8. Whereas the solubility curves predict negligible volume change in the neat base fuels, appreciable swelling did, in fact, occur for many NBRs. The solubility curves also suggest that cyclopentane would produce notably higher swell than acetone and that the straight chain ketones would exhibit similar volume change performance. The reason for this discrepancy can be attributed to the high levels (up to 40%) of additive components of the NBRs. The solubility analysis does not account for these added unknown components which may dramatically influence the overall volume change behavior.
Conclusions
The compatibility of key infrastructure elastomers with cyclopentanone and four paraffinic ketone molecules was assessed via solubility studies and volume swell measurements. In general, the volume swell increased with ketone content and corresponded well to the predicted solubilities. However, there were several noteworthy inconsistencies between the volume swell and predicted solubility; these include high volume swelling of the NBRs in neat BOB and a reduction in volume expansion at high blend levels for silicone. In most cases, the highest level of swelling occurred for cyclopentanone and acetone, while 2-nonanone produced the lowest levels of volume expansion. The chain length of the paraffinic ketones was found to affect the volume swell behavior as volume expansion decreased with increasing chain length. This behavior is attributed to the reduction in polarity and hydrogen bonding with chain length. Neoprene, SBR, and silicone showed poor compatibility with the ketones at all blend levels. Fluorocarbon and fluorosilicone also showed poor compatibility but may be suitable for use in static seals at very low blend levels with 2-nonanone. Polyurethane showed good compatibility with the long chain ketone structures of 2-nonanone, 2-pentanone, and 2-butanone, when blended with BOB. However, when blended with E10, only the 2-nonanone was suitable for use with polyurethane as a static seal. The performance of the NBRs varied with grade. Typically, NBR showed suitable compatibility only when exposed to low concentrations of ketones blended with BOB.
Footnotes
Authors’ note
Oak Ridge National Laboratory is a multi-program laboratory operated by UT-Battelle for the US Department of Energy under contract DE-AC05-00OR22725.
Acknowledgements
The authors gratefully acknowledge the support and direction of Alicia Lindauer at BETO, Kevin Stork at VTO, and the Co-Optima leadership team. The authors are also very appreciative of cooperation and guidance received from Dr Steven Abbott of the Hansen Solubility Team.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article and the work described were sponsored by the US Department of Energy (DOE) Bioenergy Technologies Office (BETO) and Vehicle Technologies Office (VTO) under the DOE Co-Optimization of Fuels and Engines Initiative.
Data accessibility statement
This manuscript has been authored by UT-Battelle, LLC under Contract No. DE-AC05-00OR22725 with the US Department of Energy. The United States Government retains and the publisher, by accepting the article for publication, acknowledges that the United States Government retains a nonexclusive, paid-up, irrevocable, world-wide license to publish or reproduce the published form of this manuscript, or allow others to do so, for United States Government purposes. The Department of Energy will provide public access to these results of federally sponsored research in accordance with the DOE Public Access Plan (![]() ).
).
