Abstract
Because all aspects of social life have a mental component, sociology’s focus is not society alone but mind and society. Insofar as mind is an emergent level of brainwork, the description and measurement of mindwork amidst social interaction can be accomplished by neurometric measurement methodology. The authors’ topic, hyperscanning, involves the simultaneous recording of either hemodynamic or neuroelectric measurement of brain activity in two (or more) interacting individuals. The authors consider two hyperscanning methods, functional magnetic resonance imaging and electroencephalography (EEG). Although functional magnetic resonance imaging provides excellent spatial resolution of brain-region activation, the temporal resolution of EEG is unmatched. EEG’s low spatial resolution has been overcome by low-resolution electromagnetic tomography. Hyperscanning studies show that interpersonal coordination of action includes mutual entrainment or synchronization of neural dynamics, flow of information between brains, and causal effects of one brain upon another with respect to social-signaling processes involving fairness, reciprocity, trust, competition, cooperation, and leadership.
Keywords
The scientific discipline of sociology studies social interactions, relationships, organizations, communities, cultures, and institutions. Because all aspects of social life have a mental component, sociology’s focus is not society, alone, but rather mind and society. Insofar as mind is an emergent level of brainwork, the description and measurement of mindwork can be accomplished by application of neurobiological scientific theory and neurometric measurement methodology considered alongside sociological theories of social interaction. The resulting three-level analysis—of social relations, of mind and information processing, and of nervous systems and brain—defines neurosociology, a field of inquiry that addresses social interactions between individuals, groups, communities, societies, and cultures (Brothers 1997; Franks 2010, 2019; Franks and Smith 1999; Franks and Turner 2013; Kalkhoff, Thye, and Lawler 2012; Kalkhoff, Thye, and Pollock 2016; TenHouten 1997, 1999, 2013).
Neurosociology and social neuroscience often address the same topics; at a minimum, they are distinguished by professional-identity management, with neurosociology viewed as a subdiscipline of sociology, and social neuroscience a subdiscipline of neurology. Sociology’s object of inquiry is social interactions and social relations in community and society, whereas the neurosciences focus on the brain: its cells, functions, and structures. In recent years, however, both disciplines have expanded into one another’s analytic space. Just as sociology has discovered the brain, neuroscience now examines the social environment in which brain networks are expressed (Brothers 1997; DeFelipe and Jones 2010). The result of this process goes beyond the establishment of neurosociology and social neuroscience as subdisciplines of their respective fields: it is a broadening of both, as they share a topic that spans, and interrelates, mind, brain, and society. The result has been a generalization of sociology to neurosociology, and of neuroscience to social, cognitive, and affective neuroscience.
Neuroscience and sociology share a focus on the mind, which is enabled by the neurologist’s brain, and is given vast capability by the social scientist’s culture, moral order, institutions, and civilization. Whereas phenomenology directly studies the mind, both social neuroscience and neurosociology access artifacts or correlates of mind-at-work. The social neuroscientist can study brain activity on the basis of patterns of blood-oxygen flow and electrical field activity of a person interacting with others; the neurosociologist can focus on how patterns of social structure and interaction affect brain function. For investigations that use both neurometric and social-relations data, the boundary between these two interdisciplinary sciences fades into an emergent transdisciplinary space (Tibbets 2016:37). A science that spans the inner and the outer is necessary because, while neurobiology has shaped the social environment, our social life influences our genes, brains, and bodies (Richerson and Boyd 2005). Human social life and human neurobiology are deeply entwined. Our social institutions, cultures, and languages comprise a pool of information stored in the brain and transmitted from one brain to another, from one mind to another, from one generation to the next (Boyd and Richerson 2005). In the neurosciences, there is growing appreciation that human brains are shaped and developed through embeddedness in social interaction, and that capability for communicating with the environment derives not so much from lower level cognitive processes but from social interaction that is central to human learning and brain development (Hari et al. 2015).
Parallel to the recognition of the centrality of social interaction to human brain function, a complementary neuroscientific “turn” has occurred in several disciplines of behavioral and social science. Economics and psychology have established extensive collaboration and exchange with neuroscience, but sociology is a latecomer to this interdisciplinary enterprise. Yet contemporary sociologists increasingly acknowledge that “the fact that we are social beings” should not “obscure the biological foundations upon which our behavior ultimately rests” (Massey 2002:1). Despite their reservations and reticence, sociologists have gradually gained from social, cognitive, cultural, and affective neuroscience many useful concepts and narratives concerning the functioning of mind (Cerulo 2010), including how the brain organizes and processes information, perceives, plans, and predicts (Kelly et al. 2019).
Neuroscience has likewise achieved an appreciation that much of what the brain does revolves around social interactions and social relations (Frith and Wolpert 2004; Hari and Kujala 2009), and that sociological concepts and theories enhance understanding of the “social brain” (Brothers 1997; Graziano 2013). The exchange of information between social actors is an essential aspect of social life. Yet despite the substantial contributions of social neuroscience to addressing such phenomena, and the very preliminary efforts of neurosociologists, little is known about how such processes are organized in the brain (Tognoli and Kelso 2015). The real-time dynamics of coordinated social interaction have remained largely unexplored. One important reason for this lack of progress is methodological, concerning problems of design and measurement, which we next consider.
Neurosociological Methodology
In social neuroscientific studies, memory, decision making, cognition, and emotion are often placed in a context of social interaction. During social interaction, participating individuals’ actions are at once a consequence of, and antecedent to, their interaction partners’ behavior. The negotiation of social exchanges involves a highly complex and dynamic process, where the actions of each individual are influenced by the prior, present, and predicted behavior of the other. During the course of a dyadic exchange, each interactant can alternate between, and negotiate about, different social roles and kinds of social relations.
Social neuroscience has developed research designs that are social-interactional insofar as experimental subjects are exposed to social stimuli through the use of electronic devices such as headphones, video, or written messages. This “spectator science” approach has revealed numerous brain systems involved in social-information processing, but it provides little insight into how these systems are modulated during actual social interaction. In such studies, brain activity is measured for the subject, but not for the other, who might be a person in a different room, the experimenter, or a confederate of the experimenter. The limitation of this research design is that interaction between brains is not measured but is inferred from “independent observations aggregated by cognitive models and assumptions that link behavior and neural activation” (Babiloni et al. 2006).
Over the past two decades, a new methodology has developed that makes it possible to measure the effects of one brain on another in the midst of in vivo social interaction. This methodology, hyperscanning, simultaneously records multiple subjects’ brain activities as they interact with one another. This method was proposed by Read Montague (Montague et al. 2002; see also Dumas et al. 2010) and has led to a proliferation of research in social, cognitive, and affective neuroscience (Hari and Kujala 2009; Nam et al. 2020). Using hyperscanning as an experimental paradigm, it has become possible to investigate functional dynamic connectivity between brains as they engage in everyday, real-world social interactions.
Hyperscanning involves various scanning methods. Electroencephalography (EEG) measures voltage fluctuations over time, as recorded by electrodes placed at various scalp locations, which measure voltage levels generated by neuronal information flow in the brain. Magnetoencephalography (MEG) is a functional imaging method of mapping magnetic fields produced by electrical current flows in the brain. Functional magnetic resonance imaging (fMRI) spatially locates brain activity by mapping changes in levels of blood-flow oxygenation (hemodynamic response), which provide the necessary energy required for neuronal activation in specific areas of the brain. Functional near-infrared spectroscopy (fNIRS) uses optical sensors measuring nonionizing, near-infrared light at various locations over the surface of the scalp to measure changes in cerebral blood flow, an indicator of blood oxygenation levels similar to fMRI (Hamilton 2021; Hari and Kujala 2009:470–74; King-Casas et al. 2005). The first 126 published hyperscanning studies predominantly used EEG (47 percent), fNIRS (35 percent), and fMRI or MRI (17 percent). These studies focused on cognition (48 percent), decision making (26 percent), and motor synchronization (23 percent) (Nam et al. 2020). We comment on fMRI, then turn to our main topic, hyperscanning using EEG.
Functional MRI provides excellent spatial location of brain activation but limited temporal resolution. 1 Because it requires the subject to lie isolated and still inside of a large and noisy scanner, the ecological reality of social interactions is severely limited. With electroencephalographic hyperscanning, in contrast, everyday conversation and related social-interactional activities can be studied in realistic ecological settings.
Given the limitations of fMRI—including its total immobilization of the subject, low temporal resolution, and the high costs of purchase, operation, housing, and maintenance—EEG and fNIRS are perhaps more viable tools for multisubject measurement. fNIRS offers a compromise between EEG and fMRI, as it provides better spatial resolution than EEG and better temporal resolution than fMRI; it is also less sensitive to movement artifacts and in some cases more mobile. However, it is impractical for larger setups using many devices, because of expenses related to research-grade equipment. We thus focus on electroencephalographic hyperscanning.
Given the complexity of the human brain, we should not be surprised to find similar complexity in the brain’s electrical activity. Sine wave oscillations are a prevalent feature of electroencephalographic recordings and play a key role in neural communication and computation. Neural oscillations are characterized by their amplitudes, phase synchronization properties, shapes, and clustering in canonical frequency bands, including delta (0.5–4 Hz), theta (4–8 Hz), alpha (8–14 Hz), beta (14–30 Hz), and gamma (30–90 Hz), as illustrated in Figure 1. 2
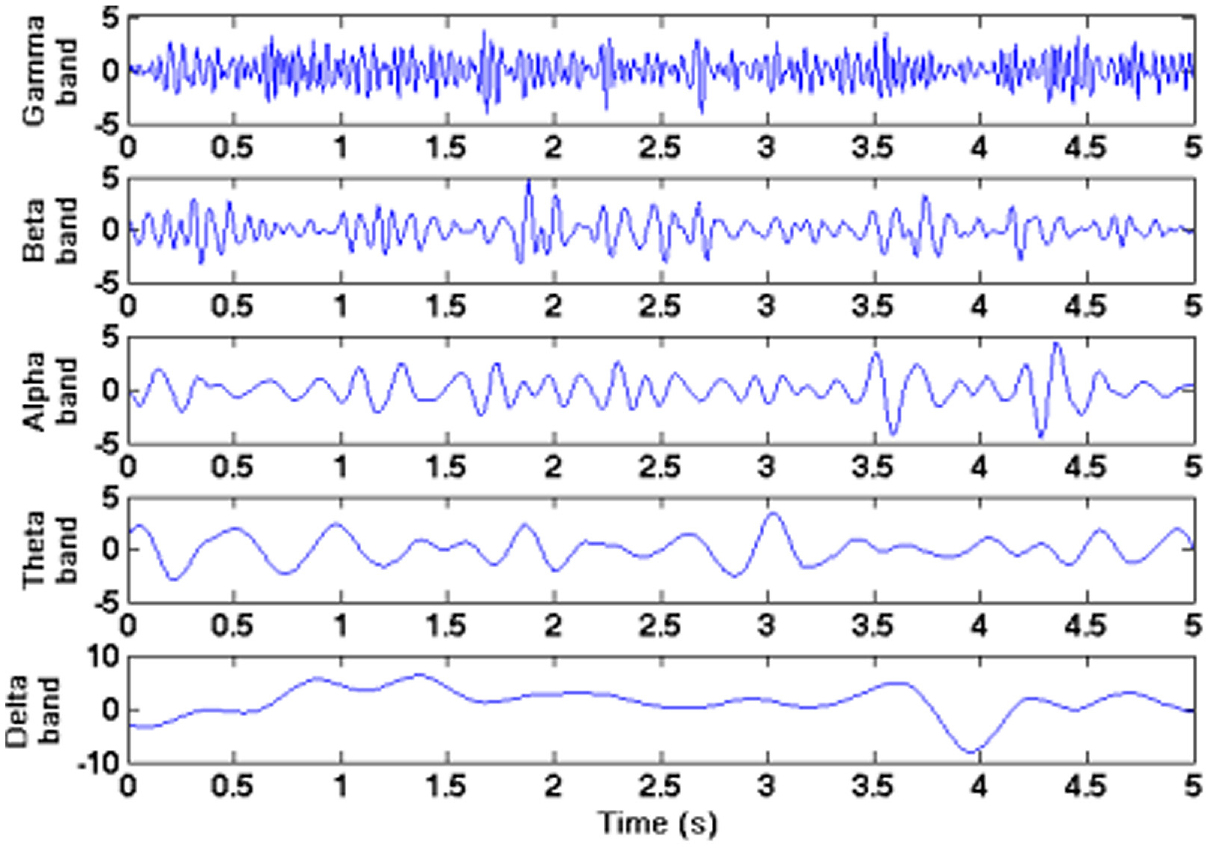
The five frequency bands of electroencephalographic signals.
Electroencephalographic hyperscanning examines the flow of information within and between the brains of dyads or larger groups during various tasks. Using the concepts of partial directed coherence (Baccalá and Sameshima 2001) or Granger causality (Granger 1969; Schippers et al. 2010), one can determine the direction of information flow, and of causality, between channels of waveform data within and between subjects. In the Granger causality–based method, directed causality between channels A and B exists if data from A are predictive of B but data from B are not predictive of A. Predictions are typically based on modeling the underlying cortical dynamics by vector autoregressive processes. 3
The most important advantage of EEG over fMRI, for social neuroscience and neurosociology alike, is its unmatched level of temporal resolution. The study of social interaction and its constituent mental activity demands methodology that is sensitive to rapid temporal unfolding. Everyday adaptive behavior requires rapid integration of complex, multimodal perception of information with previous knowledge, which is accomplished by widely distributed neurocognitive brain networks (Bressler and Tognoli 2006) that integrate information by coherent neuronal oscillations. Subsecond voltage fields, with durations of roughly 80 to 120 ms, are interpretable as brain microstates constituting the basic building blocks of human information processing (Koenig et al. 2002). In everyday conversation, speech is typically produced at a rate of 2.0 to 2.5 words per second. Facial expressions, which can be important cues in social interaction, can change dramatically in about 100 ms, even during the utterance of a single phoneme (Peräkylä and Ruusuvuori 2006). To track speech, mimicry, emotional expressions, gestures, and postures in real-time social interactions, scanning methods must provide temporal resolution in the order of 10 to 20 ms (Hari and Kujala 2009:467). In the brains of social actors listening to and producing speech, electrochemical impulses can be traveling along large-diameter myelinated neural pathways at more than 200 miles per hour. Given these facts, it follows that scanning methods with high temporal resolution, such as EEG and MEG, are required for analysis of face-to-face social interactions.
EEG provides unmatched temporal resolution, but it has disadvantages as well. Some research designs that call for 64 or more electrodes, meaning a long setup time, require acoustically shielded rooms and use sensitive electroencephalographic devices that cannot be easily moved. These problems of low transportability and longer setup times have been addressed by the advent of mobile wireless low-density electroencephalographic and neurofeedback systems, which became widely available after 2000 thanks to the Open EEG project, which made EEG an open-source technology (Griffith 2006). Low-cost but research-quality devices are becoming available for ever larger hyperscanning setups. Thanks to product design mechanics and mostly saline-based sensors, setup times have been substantially reduced. For example, the saline-based 14-channel Emotiv Epoch device takes only about four to seven minutes to set up per subject, and it produces a comparable result to other known systems (e.g., Neuroscan) (Badcock et al. 2015; de Lissa et al. 2015).
Because of the low cost and manageable setup times of many available electroencephalographic devices, it is now possible to target previously unmeasurable social interactions. Dikker et al. (2017), for example, measured 12 students over 11 class sessions with multiple electroencephalographic recordings in a classroom setting and found that larger group synchrony was predictive of social engagement and social dynamic changes in the classroom. Chabin et al. (2022) examined the effect of music and group dynamics of 12 simultaneous participants listening to orchestra and choir music; they found that, when the group was physically close, they experienced higher interbrain coherence and an enhanced subjective experience of pleasure. These electroencephalographic studies of close, social interaction in group settings have opened the door to the study of larger human social systems, their relative connectivity, and group-related neural morphology.
The quality of electroencephalographic data is limited by artifacts that manifest during measurement. These include slow artifacts such as the skin conductance response (where skin momentarily becomes more conductive of electricity because of physiological or mental arousal), facial muscle activity (swallowing and jaw movements), and ocular artifacts (eye blinking, eye movements). It is usual practice in electroencephalographic analysis to visually inspect electroencephalographic epochs or components and discard those distorted by excessive eye blinking, head movements, and other sources of distortion. Alternatively, a number of free programs are widely available for independent-component analysis (Hyvärinen and Oja 2000), along with other electroencephalographic standardized and automated preprocessing or “pipeline” artifact-removal operations such as EEG Lab (MATLAB’s freeware electroencephalographic program). These programs are not difficult to use (no coding is necessary) and have plug-ins such as ADJUST and Eye Subtract to deal with aberrant components related to eyeblinks, facial activity, and movement. ADJUST’s plug-in for independent-component analysis is automated and supplemented with a straightforward manual with pictorial examples of appropriate nonneural components that can be subtracted before carrying out spectral analysis of electroencephalographic signals.
Training programs are increasingly part of the purchase of new electroencephalographic systems. A basic EGI training can be completed in about four days, and advanced training in another three days, making such technology and its implementation available for sociologists looking to enter into this new line of inquiry. However, the best way to learn the techniques and methods of electroencephalographic research is to begin working collaboratively with experienced researchers and to learn from involvement in team efforts. It is not by chance that most published electroencephalographic studies have multiple authors.
Hyperscanning Studies
Electroencephalographic hyperscanning can provide important insights into the neural basis of decision making and other cognitive and affective functions that are at the foundation of social interaction. Neurosociology can follow neuroeconomics in using game playing (e.g., the investment game, the trust game, the prisoner’s dilemma game, the ultimatum game [UG]), which provides a natural beginning point for investigation of important social-signaling processes, including cooperation and noncooperation, selfishness and altruism, consensus and disagreement, fairness and unfairness, acceptance and rejection, reward and punishment, and deception and honesty. Prior findings suggest that cooperative social interactions activate core structures of the brain’s reward system, whereas noncooperative or competitive interactions do not. Interactive games typically activate the anterior insula (which, e.g., is stimulated by unexpectedly unfair monetary offers in decision games; Cheng et al. 2017) and the medial prefrontal cortex (shown in Figure 2), which is activated when subjects use a strategy of exchange deviating from the normative expectation that everyone should be treated equally when everything else is the same (Civai et al. 2012).
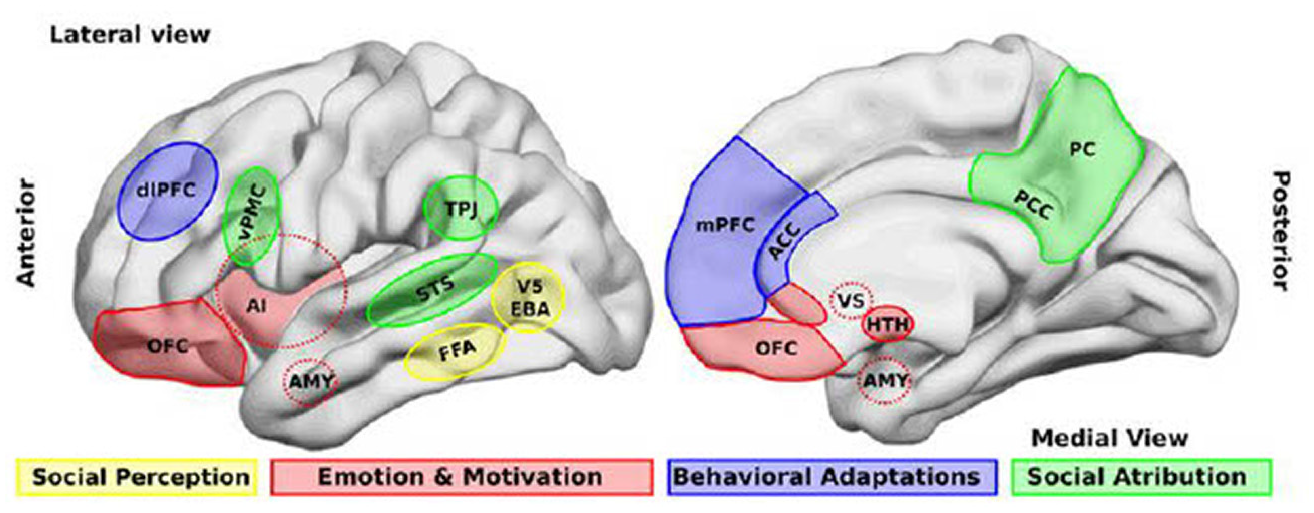
Brain areas that participate in four related processes. (1) Perception of social stimuli, such as biological motions (V5), part of the body (extrastriate body area [EBA]), and faces (fusiform face area [FFA]). (2) Emotional and motivational appraisal involves the amygdala (AMY), the anterior insula (AI), the anterior cingulate cortex (ACC), and the orbitofrontal cortex (OFC), which interact with subcortical structures, including the ventral striatum (VS) and the hypothalamus (HTH). (3) Goal-directed, adaptive behaviors and categorization processes involve the dorsolateral prefrontal cortex (dlPFC) and the medial prefrontal cortex (mPFC) and the ACC. (4) Social attribution, areas including the ventral premotor cortex (vPMC), the superior temporal sulcus (STS), the AI, the posterior cingulate cortex (PCC), and the precuneus (PC) participate in more automatic, bottom-up inferences of other people’s mental states, whereas structures such as the mPFC and the temporoparietal junction (TPJ) are involved in more cognitive theory of mind skills.
In a study exploring the mechanisms underlying analgesia related to social touch and interbrain connectivity, Goldstein et al. (2018) examined romantic couples in several different conditions while they experienced administered pain together and apart. During the condition where couples experienced pain together, they were asked to hold hands; hand holding during pain administration increased brain-to-brain coupling, especially in the alpha band. Shared pain through physical touch generated the largest networks of neural connectivity and more statistically significant coupling links between corresponding areas in the male and female brains (see Figure 3) (Goldstein et al. 2018).
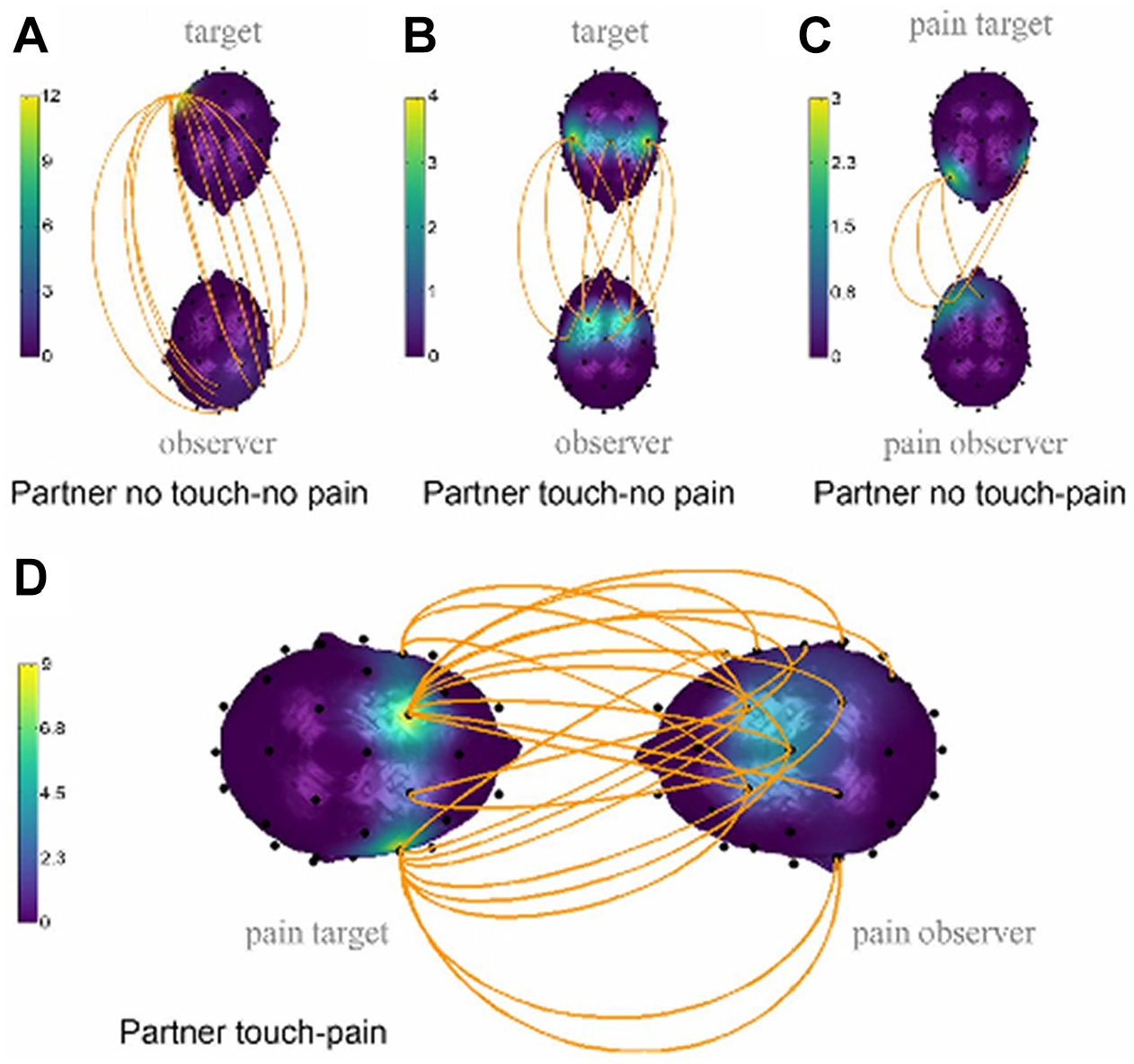
Interpartner electroencephalographic coupling in (A) partner no-touch/no-pain versus pain-alone condition (12 links), (B) partner touch/no-pain versus pain-alone condition (10 links), (C) partner no-touch/pain versus pain-alone conditions (5 links), and (D) partner-touch/pain versus all other conditions (22 links). The upper head represents the female partner (the pain target) and the lower head the male partner (the toucher). The orange lines represent statistically significant coupling links between corresponding areas in the two brains. The head color represents the number of links.
Lui et al. (2021) used EEG-based hyperscanning in a motion-sensing tennis game, finding that interbrain amplitude correlations differentiate cooperation from competition, two basic modes of human interaction. Using interbrain amplitude correlation and interbrain phase locking as measures of interbrain coupling, they found significant results only for the interbrain amplitude correlations. Cooperation elicited positive correlations in the delta and theta bands in extensive brain regions; competition, in contrast, was associated with negative interbrain correlations in the alpha and beta bands over the players’ occipital lobes.
In UG play, the decision to reject an unfair offer takes about five seconds, the time needed to resolve the conflict between an economically reasoned acceptance and an affect-laden, value-based rejection. The two-person UG (Gabay et al. 2014) has proved particularly useful in hyperscanning research. In this game, the experimenter gives player A a sum of money, a portion of which player A is asked to offer, on a take-it-or-leave-it basis, to player B. If the proposer, A, offers an even split, it is sure to be accepted. But if far less than a 50/50 split is offered, then the decider, B, is likely to decline, and both A and B receive nothing.
In the first study to use EEG to analyze temporal dynamics and functional connectivity in social decision making, Yun, Chung, and Jeong (2008) studied 13 pairs of UG players, using 54 scalp electrodes. The goals of the study were to locate the electrophysiological substrates of fairness and reason, and to explore the socioemotional interaction between the two players. They found that face-to-face interaction modulated UG play compared with other studies. The electroencephalographic data analysis revealed that high-frequency between-brain oscillations of frontocentral channels were stronger than that of other brain areas. Their finding of synchronized high oscillations within the right frontocentral regions of both players indicates that these regions are closely related to social decision making. These results are consistent with previous UG studies, which showed that the right dorsolateral prefrontal cortex is at the center of UG decision making (Koenigs and Tracy 2007; Sanfey et al. 2003).
Shaw et al. (2018) used fMRI hyperscanning to study brain activity during a modified, iterated UG. They identified numerous brain regions activated by the prospect of monetary gain. They calculated the signal time courses in the identified regions between players as a measure of interbrain synchronization, and they found synchrony in the right anterior insula and the anterior cingulate cortex. The anterior cingulate cortex, in turn, was involved in reciprocity, that is, adapting to the partner’s behavior in an effort to maximize reward.
Hyperscanning and Sociological Research and Theory
Hyperscanning enables the simultaneous measurements of brain activity of all individuals engaged in a social interaction, which is a fundamental aspect of everyday life (Dumas et al. 2010) and a key concern of sociology. During social interactions, information is transmitted between brains, and by inference, between minds. In this meeting of minds, synchronous behaviors play a central role in establishing social ties, social community, and collective social action. Interpersonal action coordination requires anticipation of a partner’s and one’s own likely actions; this can lead to the emergence of “an additional suprapersonal action representation,” a joint neural network that would likely engage the (putative) mirror neuron system, which resonates with the actions of others (Sänger, Lindenberger, and Müller 2011:656).
In everyday goal-directed decision making, interacting individuals endeavor to “mind-read” the feelings, thoughts, and intentions of their negotiating partners, using cognitive skills such as working memory and executive function. Executive functioning involves frontal lobe activity, with cognition either using, or repressing, emotions and affect. In negotiation and exchange, dynamic interaction occurs between cognition, affect, social perception, and norms of fairness, which can override rational calculation of self-interest. One early finding of hyperscanning research, on the basis of the UG, suggests that, in social decision making, it is primarily the frontal lobes that are activated in interpersonal interactions in which interactants’ brains become causally interdependent (Lui et al. 2021).
Micro-level Theory and Research
Social neuroscientists have realized their topic centrally involves social interaction (Kelly et al. 2019). They have perused sociological theory and research in search of useful conceptualizations in which social-interaction theory has been used and, at least preliminarily, linked to the brain: culture and learning theory (Vygotsky 1978) was given such interpretation by Luria (1979) and Vandervert (2017), conversation analysis (Schegloff 2009) by Dennman and Wilkenson (2011), ethnomethodology (Garfinkel 1967) by Lynch (2006), symbolic interactionism (Blumer 1969) by Franks (2003), and interaction-ritual chains theory (Collins 2004) by Bruvik and Liebst (2016).
Despite these beginning steps toward bringing the brain into approaches to social interaction, practitioners have yet to consider how the minds and brains of interacting individuals are structured, or how their mental processes affect one another through directed causality and other forms of interbrain entanglement. In the midst of social interaction, individuals’ experience of the other “is marked by a [mental] recognition of mutuality” in which “the unit of analysis is not the subject’s behavior, but the subject–object interaction” (Murray [1951] 1962:436).
Macro-level Theory and Research
On the macro-sociological level, neurosociology can potentially address not only the entanglement of interactants’ minds (and brains) but also the sharing of suprapersonal cognitive structures, ranging from normative belief systems, to religious systems, to sociopolitical ideologies, to class consciousness. This raises a key theoretical question: can brains influence each other at a distance beyond face-to-face social interaction? An accumulating body of evidence suggests that brain-state correlations can exist between human subjects under conditions excluding sensory communication via their environment. Wackermann et al. (2003) found that when one subject of an “empathically bound” pair of subjects was stimulated by light flashes, waveforms similar to evoked potentials were found in the nonstimulated other subject. Pairs of subjects who participated in a “tuning in” process to attain an empathic bond were measured by six electroencephalographic channels, with subjects seated in separate, shielded rooms. One subject was exposed to a pattern-reversing stimuli while the other relaxed; the nonstimulated subject showed a significant increase in average voltage of the six electroencephalographic channels.
These and related studies (e.g., Persinger et al. 2010) have shown correlations between brain activities of separated subjects. Orme-Johnson et al. (1982) found that individual subjects located in a nearby Faraday chamber (an enclosure used to block electromagnetic fields from entering or leaving) manifested an increase in interhemispheric coherence, as did a group of subjects engaged in a meditation session some distance away. These remarkable results evoke Durkheim’s ([1912] 1995) study of Australian Aborigines’ experience of a sense of collective effervescence as their minds are forcefully affected by ongoing rites and rituals. Durkheim saw religious systems and their ceremonial expressions of the sacred as socially constructed, but once so constructed, he argued, they exist as social facts and exert a real force upon participants’ minds, and, we can speculate, can do so nonlocally. 4
Durkheim conceptualized human capability for shared meaning, for culture, through the enactment of rite, ritual, and ceremony. When a member of a collectivity perceives a god, feels belief, or experiences collective intentionality, the person “actually perceives the accumulated energy of on-going creation and maintenance of objects and ideas by members of a collectivity” (Badia 2016:969). Durkheim insisted his linkage of thought and action occurred through a real, not metaphorical, framework of energy and social force. It is not difficult to envision this classical theoretical topic being investigated by means of electroencephalographic hyperscanning methodology.
Hyperscanning in Neurosociology: An Example Study
Hyperscanning can also be used for data collection in naturalistic settings to test or compare theoretically derived empirical hypotheses and models in a wide range of fields, including the sociology of education, small group theory, medical sociology, the sociology of aging, organizational sociology, and social psychology. It can assist sociologists in testing causal hypotheses, identifying the causal mechanisms linking social and mental phenomena, and illuminating the micro-foundations—the neural processes—underlying a wide variety of social relationships and interactions.
Given that hyperscanning methodology has yet to be exploited in neurosociology, we can expect that the first sociological hyperscanning studies will be conducted by social neuroscientists and published in neuroscience journals. We consider one example of this, an analysis of the emergence of leadership in small groups, a classic concern of sociology. In this study, social neuroscientists Jiang et al. (2015) used functional near-infrared spectroscopic hyperscanning to test two competing theories of leader emergence in small groups: the “frequency of communication” versus the “quality of communication” hypotheses. In this study, 11 three-person groups were arranged in face-to-face triangles, then asked to perform a leaderless group discussion as their brains were simultaneously measured; the principal focus was on activation of the left temporoparietal junction, a brain area centrally involved in social cognition and mentalizing (see Figure 2). Videotapes of these social interactions were used to measure leadership as well as verbal and nonverbal communication. During this process of leader emergence, hyperscanning enabled comparison of the interpersonal neural synchronization (INS) of group members in each of the three pairwise dyadic subgroups. Subjects were classified as leaders (L) or followers (F), and INS data were analyzed for leader-follower (LF) and follower-follower (FF) dyads. For each triad, even during the initial phase, after one minute of discussion, LF dyads displayed higher levels of INS than did FF dyads. Emerging leaders in the LF groups did not initiate more communications than followers, but INS levels were higher during leader-initiated communication than during follower-initiated communication and also higher when verbal (rather than nonverbal) communication was occurring. Granger causality analysis showed that, in LF pairs, INS was bidirectional but was stronger from emerging leader to followers. That is, emerging leaders’ communications modulate followers’ intragroup neural synchronization. In the LF dyads, INS levels in leader-initiated communications were significantly correlated with video-based measures of leaders’ communication skills and communicative competence, but not with communication frequency. These findings support the claim that the quality of communication and the accompanying rise in INS levels could predict leader emergence in small task-oriented groups. High-quality communication skills, in turn, reflect an individual’s ability to quickly and accurately interpret and respond to the interaction dynamics of a social situation.
An obvious limitation of Jiang et al.’s (2015) study of leadership and INS is that it measured only the left hemisphere, whereas for the emergence of leadership in LF dyads, we can hypothesize asynchronization of left and right temporoparietal junction channels. 5 Such within-brain synchrony should indicate integration of verbal and nonverbal information processing as a possible mechanism of social competence and leadership, the ability to become influential by communicating in speech articulated with nonverbal channels, and expressing the right thing at the right time. Electroencephalographic research shows that coordination or synchrony of homologous left and right hemisphere structures is indicative of the mental competence that characterizes cognitive development. This has been demonstrated by the gradual emergence of subsecond epochs with stable field topography, whose artifacts correspond to basic building blocks of human information processing, integrating information by coherent neural oscillations in the brain (Koenig et al. 2002). In the process of cognitive development, symmetric microstates indicate bilateral activation; this process is related to the development of the corpus callosum (which connects the two hemispheres of the brain) and continues into late adolescence (Giedd et al. 1999).
Discussion
Our behavior in groups depends on our social positions, our social roles, and our social relationships, all of which involve consideration of rights, responsibilities, beliefs, and norms. An individual participating in a social interaction is necessarily involved in social cognition, social emotions, and related mental processes. This mental activity is interior to all social interaction, as there are continuities and discontinuities of one’s and others’ intentions, beliefs, opinions, and norms. These exist on the level of culture and as suprapersonal cognitive structures that individuals bring to social interactions, where they can be invoked, challenged, and modified in the midst of social interactions. We claim that inhibition of the theoretical development of social-interaction theory has been the inevitable result of an inability to empirically study the mental processes of interacting individuals as they engage in social behavior. We cannot assess individuals in group interaction patterns without in situ measurement of ongoing mental processes. This can be accomplished, indirectly, by continuous measurement of physical artifacts of mental activity, which in turn can be accomplished through hyperscanning.
By recording the brainwork involved in mindwork, we potentially gain understanding, for example, of what it means for a normative order to exist as an aspect of culture in a larger society. Thus, the study of social norms, beliefs, and opinions, which are essentially mental and social behavioral, as they are manifested in human groups, can be advanced through hyperscanning methodology. This records not only expressed social behaviors—of speaking and hearing, gesturing and posturing—but also the ongoing mental processes that enable communication of thoughts, ideas, perceptions, intentions, and emotions.
Social communication centrally includes speech and language (including intonation, prosody, gesture, posture, and facial expression), all of which involve both brain and body. With the further development of methodology for bringing everyday social interactions into experimental settings, it will become necessary to scan other systems of the body, as well as the brain. As Hamilton (2021) pointed out, “interacting brains exist within interacting bodies. Visual, auditory, and motor processes mediate any coordination between two brains, so we must study brains and bodily coordination together” (p. 406). Such multimedia data will include the hands, eyes (gaze), breath, and heart. The eyes are important for social signaling, as they can entice, express disapproval, or reveal shame. It is not unusual for individuals engaged in conversation to develop a synchronization of respiration (McFarland 2001). In everyday social interactions, the communication of affect is a matter not only of the brain but also of the heart. Electroencephalographic rhythm can vary with heart rate (Dé Munck et al. 2007). Beyond the laboratory, social interactions that develop into positively valued and close social relationships likely have coherence as their embodied infrastructure and synchrony of the key organs of sociality, brains and hearts, on the levels of cognition and emotion. 6
Hyperscanning methodology has constraints and obstacles, but none is insurmountable. Hyperscanning emerges as an essential tool for social neuroscience and, we propose, is potentially of great value for neurosociology and for neuroanthropology, neuroeconomics, neuropolitics, and related fields of inquiry. Hyperscanning provides for neurosociology, and for general sociology, an ecologically valid way to study the mental phenomena generated by the workings of the social brain in social interactions, in the performance of social roles, and in engagement in social relations and social relationships. Given that all social behavior has a mental aspect, and that all mental phenomena emerge from brainwork, hyperscanning methodology uniquely enables empirical study, and theorization, of mind, brain, and society.
In the long run, the advent of hyperscanning methodology means that neurosociology is not to remain some quaint and curious lore, politely rapping at sociology’s chamber door. As Babiloni et al. (2006) state, “to study the concurrent activity in subjects interacting in cooperative or competitive activities . . . the simultaneous recording of their brain activity becomes mandatory [italics added]” (p. 3666). Indeed, hyperscanning is the necessary methodological foundation of a more general sociology, a sociology that, together with social neuroscience, addresses the dynamic interdependence of the social, the mental, and the biological in a context of social interaction.
