Abstract
Artificial intelligence (AI), like ChatGPT, Microsoft copilot, and Google Gemini, is transforming pharmaceutical research, production, and healthcare delivery. Despite global adoption, the level of AI awareness, readiness, and application among pharmaceutical professionals remains unclear. This study aimed to evaluate the awareness, knowledge, attitudes, and perceptions of AI among pharmaceutical professionals and to identify factors influencing its adoption. A cross-sectional survey was conducted among professionals from various pharmaceutical sectors using a structured online questionnaire. Descriptive statistics were applied to analyze demographic characteristics, AI familiarity, usage, and perceptions. Most respondents (81.8%) were aware of AI, but only 2.3% reported extensive familiarity. ChatGPT was the most frequently used AI tool (63.6%), while only 27.3% reported organizational adoption. Support for AI adoption was high (86.4%), yet only 22.7% considered the industry fully ready. Perceived benefits included improved efficiency (86.4%) and quality (70.4%), while major concerns involved automation dependency (36.4%) and implementation costs (34.1%). Pharmaceutical professionals show strong interest in AI despite limited organizational readiness and formal training. Structured education, regulatory guidance, and ethical frameworks are critical for effective AI integration in the sector.
Keywords
Introduction
Artificial intelligence (AI) refers to advanced computational architectures engineered to emulate human cognitive processes, enabling autonomous execution of complex tasks. When supplied with robust and well-curated foundational data, these systems demonstrate the capacity for adaptive learning, independent decision-making, and iterative problem-solving, thereby driving substantial advancements across diverse operational domains. 1 AI is a major development in many areas, especially in healthcare. 2 In the pharmaceutical field, AI has greatly cut down the time and money needed to find and develop new drugs. It helps with things like predicting outcomes and creating personalized treatment plans for patients.3 -5 For example, they have been used in medical research to predict bad outcomes and chances of death in healthcare settings.6 -8
Around the world, many pharmaceutical companies are looking to adopt newer technologies, including platforms such as AI and virtual platforms in this field. These new technologies may be helpful in the restart or recreation of these clinical trials, with minimal interaction for face-to-face types.9 -15 Combining AI with the Internet of Things (IoT) is changing pharmaceutical manufacturing processes, helping to make production faster and more reliable. This teamwork helps keep an eye on things in real time and predicts when equipment might need fixing, which is important for keeping drug quality up. 16 Pharmaceutical sector is very important for the healthcare system and helps both local and global markets. As more attention is given to digital changes, AI can help make drug production better, improve the quality of medicines, and create stronger and more efficient supply chains.16 -18 The pharmaceutical industry faces disruptions from pandemics, price changes, logistics delays, furthermore international trade issues worsen resource shortages and production challenges. AI can be a game changer by enhancing forecasting, automation, and regulatory compliance, making operations faster and more efficient. 19 A study discussed how artificial intelligence applications, such as AI-driven image analysis, telemedicine, and virtual health assistants, are enhancing patient care and operational efficiency. 20 This is particularly relevant for rural place, where access to healthcare services can be limited, and AI’s ability to facilitate remote consultations can bridge significant gaps. Furthermore, another research emphasizes the potential of AI in addressing supply and demand challenges through tools like pharmaceutical chatbots and automated assistance, which can enhance patient engagement and support healthcare professionals in making informed decisions. 21
This exploration is not merely about understanding the technology but also about comprehending and mitigating the associated ethical and regulatory concerns, which is increasingly critical as AI technologies become more pervasive in healthcare settings.22,23 The results of various studies indicate a promising trajectory where AI not only enhances operational workflows but also fosters innovation in drug design and patient interaction, thereby leading to improved health outcomes.24,25 In light of AI’s emerging role in the pharmaceutical industry, it remains unclear to what extent understand its potential, limitations, and practical applications. To explore this, the authors designed a survey to assess the level of familiarity, understanding, and perspectives among pharmaceutical professionals regarding the role and impact of artificial intelligence in pharmaceutical operations, research, and development. We hypothesize that, given the novelty of AI technologies and LLMs, many professionals may not yet be familiar with their use or have integrated them into routine practice.
Current literature and Studies indicate that AI technologies have led to reduced time frames for drug discovery processes, enhanced decision-making capabilities for clinicians, and improved patient engagement through personalized healthcare. 26 Furthermore, ongoing explorations in the ethical dimensions of AI, such as data privacy and algorithmic bias, underscore the necessity for a balanced approach as these technologies develop. To summarize, the incorporation of AI, into the pharmaceutical industry holds substantial promise. The exploration of its capabilities and applications has revealed a transformative potential that could redefine healthcare delivery. As this discourse evolves, continued collaboration among data scientists, healthcare professionals, and policymakers is paramount to fully realize AI’s benefits while maintaining ethical standards and regulatory. 26 By carefully navigating these dimensions, stakeholders can leverage AI technologies to foster innovation and enhance healthcare accessibility. AI is increasingly reshaping the pharmaceutical industry, influencing multiple stages from drug discovery to manufacturing, quality assurance, and regulatory processes. Its integration has revealed notable efficiencies, enhancing predictive modeling, optimizing workflows, and enabling more precise decision-making across complex operations. At the same time, AI adoption uncovers persistent challenges, including data quality constraints, ethical considerations, regulatory compliance, and the need for skilled human oversight. This study explores both the tangible contributions and existing limitations of AI in pharmaceutical practice, highlighting areas where the technology is already demonstrating impact and identifying domains ripe for future innovation. Central questions guiding this exploration include, the specific areas within pharmaceutical operations where AI is applied, the measurable outcomes of its deployment on efficiency and quality, and the practical, ethical, and regulatory hurdles affecting its broader implementation.
Literature Review
The integration of AI into the pharmaceutical industry is an emerging field of study that addresses the significant potential of AI in enhancing industry practices and improving healthcare delivery. With the country’s pharmaceutical sector experiencing marked growth and increased innovation, AI plays a pivotal role in this transformation. This literature review outlines the current status, implications, challenges, and future prospects of AI within this context.
Purpose and Rationale of the Literature Review
This literature review is presented to establish the scientific foundation of the manuscript by critically synthesizing recent advancements in artificial intelligence (AI) relevant to cancer drug discovery and development. The purpose of this section is threefold: (i) to contextualize the evolving role of AI within modern pharmaceutical research, particularly oncology; (ii) to identify how AI-driven methodologies address long-standing challenges in cancer therapeutics, such as high attrition rates, prolonged development timelines, and molecular complexity; and (iii) to justify the relevance of the present study by highlighting existing research gaps and translational opportunities. By reviewing contemporary evidence, this section provides a logical framework linking technological innovation to therapeutic impact.
Current Status of AI Implementation
The integration of AI into pharmaceutical research has progressed rapidly, with oncology emerging as one of its most impactful application domains. AI-driven approaches and network-based modeling, are now routinely employed to analyze high-dimensional biological data such as genomics, transcriptomics, proteomics, and chemical libraries. These techniques enable the identification of novel cancer targets, prediction of drug–target interactions, and optimization of lead compounds with significantly greater efficiency than conventional methods. 27 Recent studies demonstrate that AI platforms can rapidly screen millions of chemical entities and prioritize candidate molecules based on predicted efficacy, toxicity, and pharmacokinetic profiles. This capability is particularly valuable in cancer drug discovery, where tumor heterogeneity and pathway redundancy complicate traditional experimental approaches. In regions with strong pharmaceutical manufacturing capacity, AI-assisted discovery pipelines also support cost-effective innovation and accelerate the transition from preclinical research to clinical evaluation. 28 Studies have reported that AI applications can streamline operations and enhance product management within pharmaceutical businesses, thereby improving overall organizational performance. 29
Implications for Drug Development and Patient Care
The implications of AI in drug development and patient care are profound. The incorporation of various AI methodologies, including deep learning, has resulted in significant innovations in drug discovery. For instance, AI’s ability to simulate and predict the interactions of drugs at the molecular level allows for faster identification of potential therapeutics. 30 Furthermore, AI enhances precision in clinical trials by enabling patient stratification based on genetic profiles, thus optimizing treatment plans and improving patient outcomes. 31 Moreover, AI can significantly impact post-market surveillance, ensuring that any emerging safety concerns related to pharmaceutical products are addressed promptly. The pharmaceutical industry has recognized the need for regulatory frameworks that can support the safe integration of AI solutions. 32 As such, legislators are urged to develop guidelines that encompass the ethical use of AI while ensuring compliance with patient safety and data privacy standards. 33
Challenges and Regulatory Considerations
Despite its transformative potential, the application of AI in cancer drug discovery faces notable challenges. Data quality, interoperability, and algorithmic transparency remain critical concerns, particularly given the sensitive nature of oncological and genomic data. Ensuring data privacy, minimizing algorithmic bias, and maintaining explainability of AI models are essential for regulatory acceptance and clinical trust. 34 Moreover, the successful deployment of AI requires interdisciplinary collaboration between data scientists, oncologists, pharmacologists, and regulatory authorities, which remains uneven across research environments.
Future Prospects
Looking ahead, the future of AI in the pharmaceutical industry seems promising. The country is on the brink of a technological revolution, with AI poised to enhance the efficacy of pharmaceutical research and healthcare. By adopting AI broadly can strengthen its capacity for innovation and improve its competitiveness in the global pharmaceutical market. 35
Methodology
Study Design
The primary objective of this study was to gather insights regarding the perceptions and attitudes of pharmaceutical professionals toward AI utilization in their industry. A comprehensive literature review was conducted to frame the research context and to justify the importance of understanding AI’s role within the expanding pharmaceutical landscape. A cross-sectional survey-based study was conducted between August 06, 2025 and November 12, 2025, among pharmaceutical professionals. An online questionnaire was administered via email using Google Forms to professionals working in different sectors of the pharmaceutical industry. The study employed an exploratory design to assess current awareness, attitudes, and perceptions regarding AI adoption in pharmaceutical practice.
Sampling Techniques
This study aimed to engage a diverse group of professionals across the pharmaceutical sector, including personnel from manufacturing facilities, regulatory bodies, retail pharmacies, and hospital pharmacies. To ensure comprehensive representation, a stratified approach was employed, segmenting organizations based on key characteristics such as organizational type and geographic location. Within each stratum, representative samples were selected through a combination of random and convenience-based strategies. Random selection facilitated equitable representation of individuals within each organizational category, while convenience sampling accounted for practical considerations, including accessibility and availability of participants. Data collection was conducted from 6th to 29th August 2025, with follow-up reminders sent 1 week after initial contact to maximize participation. This deliberate sampling strategy ensured a heterogeneous and balanced inclusion of participants, thereby enhancing the reliability and generalizability of findings regarding the applicability of artificial intelligence in pharmaceutical operations across multiple organizational settings.
Participants Eligibility Criteria
The survey targeted professionals across diverse roles within the pharmaceutical sector. This population was chosen due to their direct involvement with pharmaceutical practices and potential interactions with AI technologies. 36 A total of 200 professionals were invited to participate via email and professional social media platforms (eg, Facebook, LinkedIn, Gmail). Participants were requested to complete the structured questionnaire focusing on AI awareness, adoption, and perception in the pharmaceutical context. To enhance participation, personalized invitation messages highlighting the relevance of the study to their professional practice were sent, followed by a reminder notification 1 week later.
The inclusion criteria required participants to be actively employed pharmaceutical professionals with a minimum of 6 months to 1 year of professional experience in any pharmaceutical division. Respondents needed sufficient English literacy to complete the questionnaire and had to be willing to provide informed consent. Conversely, the exclusion criteria eliminated students, interns, unemployed graduates, and professionals working outside, as well as any respondents who submitted incomplete survey forms, defined as less than 80% completion.
Questionnaire Development
To ensure the survey instrument was relevant and original, 2 independent members of the research team conducted a systematic search on August 2, 2025, in the PubMed and Scopus databases. The search strategy employed was: [((Artificial Intelligence) OR (ChatGPT) OR (Chatbot)) AND ((scientific production) OR (scientific writing)) AND (survey)]. No existing validated survey tools were identified that specifically addressed AI perceptions in the pharmaceutical sector within context. Consequently, a novel questionnaire was developed with a systematic, multi-step process, consultations with pharmaceutical professionals (n = 5) and AI specialists (n = 2). Content validity was ensured through review by a panel of 3 experts in pharmaceutical sciences and 2 survey methodology experts. Face validity was assessed through pilot testing with 10 pharmaceutical professionals, leading to refinements in clarity, readability, and item sequencing. The final instrument consisted of 22 items, including single-choice (n = 15), Likert-scale (n = 4), multiple-choice (n = 1), and open-ended questions (n = 2), organized into 4 sections covering demographics (Section A), Knowledge and awareness of AI (Section B), attitude toward AI (Section C), and perceptions of AI (Section D).
Limitation Acknowledgment
The convenience sampling approach was chosen due to the absence of a comprehensive registry of pharmaceutical professionals and resource constraints typical of exploratory research. We acknowledge this introduces selection bias, potentially oversampling professionals with pre-existing interest in or awareness of AI technologies. To partially mitigate this, recruitment messages emphasized the importance of diverse perspectives, including those unfamiliar with AI, and assured anonymity to encourage candid responses.
Sample Size Calculation
Given the exploratory nature of this study, the required sample size was estimated using a modified form of Cochran’s (1977) formula, commonly employed in social and applied sciences research:
Where, N = required sample size, Z = z-value corresponding to the desired confidence level, P = estimated proportion of the population possessing the attribute of interest (0.5, assuming maximum variability), e = acceptable margin of error. For an exploratory study, a confidence level of 85% (Z = 1.44) 37 and a margin of error of 0.12 (12%) were considered appropriate, allowing for a practical balance between precision and feasibility. Substituting these values gives:
Thus, the estimated minimum sample size required for the study was approximately 44 respondents (N = 44).
Statistical Analysis
Variables were summarized as frequency and percentage (N%) and presented in 4 descriptive tables to provide a comprehensive overview of the dataset. Descriptive statistics summarized the distributions of participants’ demographics, knowledge, attitudes, and perceptions regarding AI in the pharmaceutical sector. Results for categorical variables are presented as percentages to clearly reflect response patterns. Data were also visualized using tables to distributions and highlight key trends. Analyses were structured to assess how awareness and experience influenced participants’ attitudes, perceptions, and support for AI integration. All statistical analyses were conducted using SPSS (version 26.0; IBM SPSS Corp).
Achieved Sample and Justification
Although the sample size is modest, it is appropriate for this study because the focus is descriptive rather than hypothesis-driven, making strict power requirements less critical. As the first study of its kind in the country, even limited data offer valuable preliminary insights. Open-ended responses add qualitative depth, and the findings will help guide power calculations and sampling strategies for future, larger studies.
Ethics Statement
This study was conducted in accordance with the principles of the Declaration of Helsinki, 38 and was approved on August 01, 2025 by the Institutional Review Board, Center for Research & Innovation (CRI) R. P. Shaha University, Narayanganj, Bangladesh, Reference Number: RPSU/EC/Phr/2025/07.
Bias
This study may contain minor sampling and response biases due to self-reported data and limited participant representation. Efforts were made to reduce these through anonymous responses and neutral question design.
Result
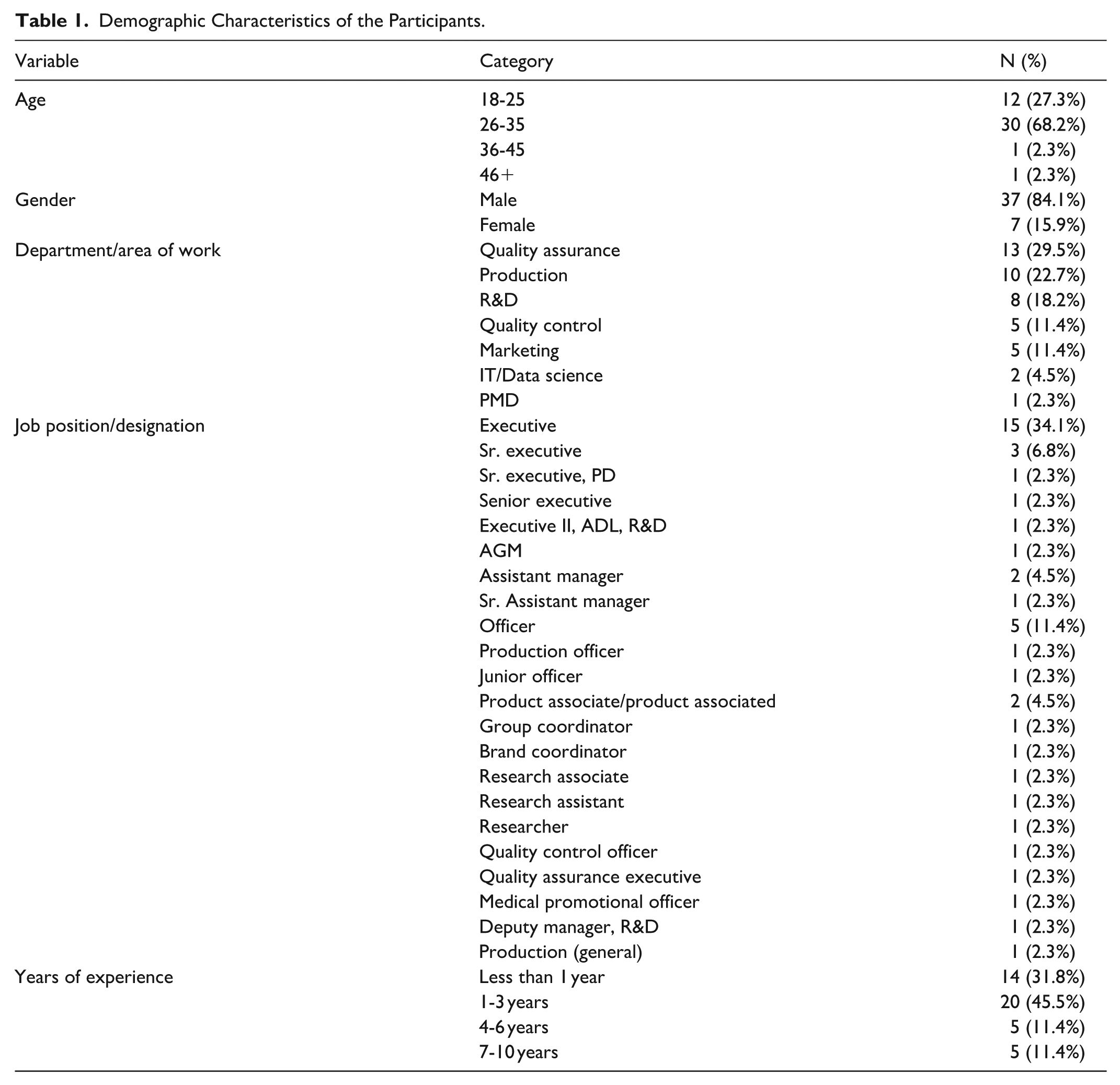
A total of 44 professionals participated in the survey. The majority were employed in Quality Assurance 29.5%, Production 22.7%, and R&D 18.2%, with smaller representation across Marketing, Quality Control, IT/Data Science, and PMD. Executive positions constituted the largest proportion of job roles 34.1%, followed by Officer-level posts 11.4% and a range of mid- to senior-level managerial designations. Regarding experience, most respondents had 1 to 3 years of industry exposure 45.5%, while 31.8% reported less than 1 year; participants with 4 to 6 years and 7 to 10 years of experience each accounted for 11.4% of the sample as described in Table 1.
Demographic Characteristics of the Participants.
In Table 2, substantial proportion of respondents 81.8% had heard about the use of art AI in the pharmaceutical domain, reflecting broad awareness. However, self-reported knowledge levels showed significant variation. Only 2.3% considered themselves extensively familiar with AI tools, while the majority reported being either moderately familiar 38.6% or only slightly familiar 31.8%. This suggests that while awareness is widespread, in-depth knowledge remains limited among many professionals. In terms of practical experience, 70.5% had used AI tools in their work. The most frequently used tool was ChatGPT 63.6%, with smaller proportions reporting use of Microsoft Copilot, Gemini (each 4.5%), or other large language models and domain-specific tools like meta-analysis assistants (each 2.3%). Notably, 18.2% had not used any AI tools, indicating a gap between awareness and application. Despite individual adoption, organizational implementation remains inconsistent. Only 27.3% confirmed that their organizations had adopted AI technologies, whereas 40.9% reported non-adoption, and 31.8% were unaware of their organization’s stance.
Knowledge and Awareness of Artificial Intelligence (AI).

A strong majority 86.4% either agreed or strongly agreed that AI can improve efficiency in pharmaceutical tasks, suggesting a high level of optimism regarding its operational benefits. Similarly, 70.4% believed that AI could contribute to improving the quality of work, although a notable minority 18.2% remained neutral, and 11.3% expressed disagreement, indicating some caution about AI’s consistency or reliability. When asked whether AI reduces errors or enhances decision-making, 61.4% responded affirmatively. However, a significant portion 25.0% remained neutral, and 13.6% expressed disagreement, reflecting some hesitation about fully trusting AI in critical decision-support roles. Concerns about professional displacement were clearly addressed in the responses. A majority 68.2% disagreed or strongly disagreed with the statement that AI would replace pharmacists, indicating a strong belief in the irreplaceable nature of human judgment and patient interaction in pharmaceutical care. Only a small proportion 20.4% agreed or strongly agreed with the idea of replacement, and 11.4% remained neutral which are shown in Table 3.
Attitude Toward Artificial Intelligence (AI).

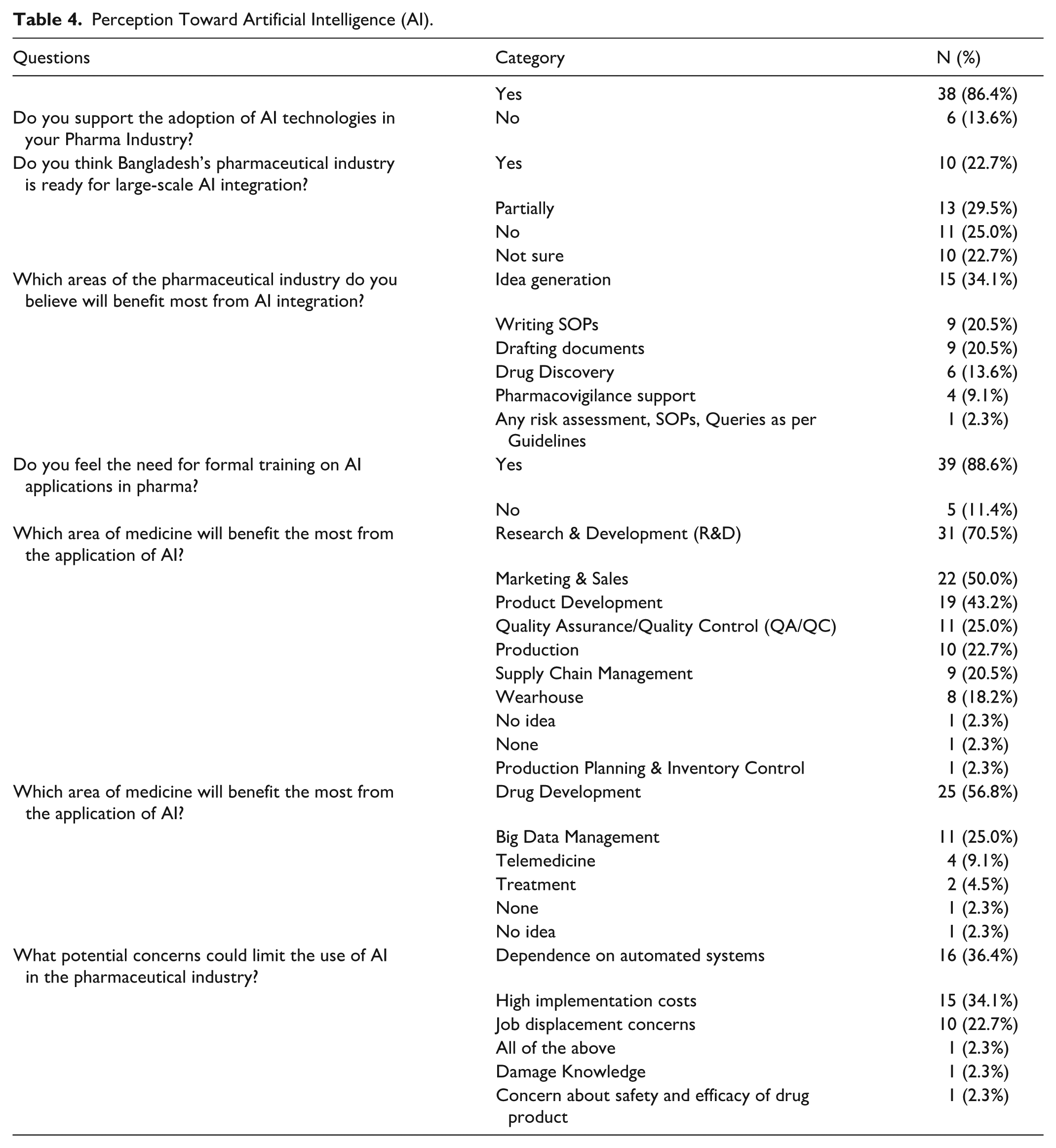
From Table 4, The majority of respondents 86.4% supported the adoption of AI in the pharmaceutical industry, though only 22.7% believed the industry is fully prepared. Many viewed the sector as partially ready 29.5% or not ready 25.0%, with 22.7% unsure. Preferred AI applications included idea generation 34.1%, SOP writing 20.5%, and document drafting 20.5%, reflecting its perceived utility in cognitive and administrative tasks. An overwhelming 88.6% recognized the need for formal AI training, highlighting a critical gap in workforce preparedness. R&D was identified as the area most likely to benefit from AI 70.5%, followed by marketing, product development, and QA/QC. In medicine, drug development 56.8% and big data management 25.0% were viewed as key beneficiaries. Despite the enthusiasm, concerns remain. Dependence on automated systems 36.4%, high implementation costs 34.1%, and job displacement 22.7% were cited as key barriers. These findings underscore strong interest in AI, tempered by readiness challenges and perceived risks.
Perception Toward Artificial Intelligence (AI).
Discussion
The integration of Artificial Intelligence (AI) within the pharmaceutical industry presents various implications for policy and regulatory frameworks. There is a notable interest among professionals in pharmaceutical sector toward adopting AI, along with a recognition of the necessity for robust governance structures to facilitate this integration effectively. Thus, several critical policy implications should be considered to enhance the uptake and successful deployment of AI technologies in the sector.
A foundational element is the establishment of regulatory pathways that ensure AI systems, such as those used in drug development and patient management, meet safety and efficacy standards. Existing literature emphasizes that appropriate regulatory frameworks are essential for overseeing AI devices, much like traditional medical devices, requiring clear guidelines regarding approval processes from relevant health authorities. 39 As proposed by Farah et al, Health Technology Assessments (HTA) can help categorize the necessary regulatory approvals that AI-based medical devices and applications must undergo, particularly to safeguard public health and ensure that these innovations are effective. 40 Given the fragile acceptance of AI in healthcare, it is paramount for HTA agencies to incorporate ethical considerations and regulatory implications into their assessments. Bélisle-Pipon et al underline that failure to comprehensively manage these factors could emerge as significant barriers to deploying AI technologies within healthcare systems. 41 Consequently, governance frameworks must also prioritize transparency and accountability, empowering healthcare professionals to make informed decisions regarding AI utilization.
Additionally, addressing local context and resource constraints is critical. Regulatory frameworks should enable the efficient evaluation and approval of AI technologies while remaining flexible to the unique needs and challenges faced in developing markets. Research conducted by Hassan et al indicates that identifying regulatory gaps within existing frameworks is vital for laying the groundwork for a supportive AI environment. 32 Another policy implication lies in the integration of reimbursement and financial support mechanisms for AI technologies within health system budgeting. As the pharmaceutical industry increasingly recognizes the benefits of AI—namely improved efficiency and decision-making—the implementation of reimbursement models tailored for AI-driven solutions can facilitate their wider acceptance and use. Massella et al suggest that aligning reimbursement policies with evidence on the effectiveness of AI interventions can enhance their integration into the healthcare delivery framework. 42
Organizational readiness for AI adoption must also be addressed by all stakeholders, including government, private sectors, and educational institutions. The readiness levels documented in various studies highlight the need for structured initiatives that build capacity and confidence among healthcare professionals. Initiatives could include partnerships for workforce training in AI technologies and data management, ensuring that talent is equipped to meet the emerging demands of AI’s evolving role in pharmaceuticals.43,44
Notably, while a high degree of awareness of AI, particularly generative AI like ChatGPT (63.6%), was reported, self-assessed familiarity levels were limited. Only 2.3% of respondents felt extensively knowledgeable, indicating a disparity between awareness and substantive understanding or ability to apply AI tools effectively. This aligns with previous studies emphasizing the need for targeted educational initiatives to bridge knowledge gaps and enhance practical engagement with AI technologies among health professionals.45,46 The 70.5% usage rate of AI tools in work settings further highlights the acceptance potential, yet also points to a concerning 29.5% who have not utilized AI despite awareness.47,48
This hesitation may be a consequence of the perceived risks associated with AI, including fears of job displacement, as 68.2% of respondents disagreed with the notion that AI would replace pharmacists. This resonates with behavioral theories about risk perception, indicating a careful balancing of trust and doubt toward automation in critical healthcare roles.49,50 The call for formal AI training (88.6%) is a profound indicator of the necessity for a structured approach to workforce readiness, ensuring that employees not only understand AI tools but can also navigate their influence on traditional workflows —fostering accountable use of AI that augments rather than replaces human expertise.51,52 This focus on AI’s utility in enhancing cognitive tasks rather than direct clinical decision-making echoes sentiments in existing literature that advocate for collaborative frameworks where AI supports, rather than supplants, human judgment.53,54 These findings resonate with the growing body of global literature endorsing AI’s transformative potential in streamlining drug discovery, pharmacovigilance, and quality assurance, aligning with studies that emphasize AI’s role in streamlining workflows and reducing errors in healthcare and pharma. 55 While only 20.4% believed that AI might replace pharmacists, a substantial 68.2% either disagreed or strongly disagreed. This suggests that, within this cohort, AI is viewed as a complementary rather than disruptive force, an assistant to human professionals rather than a substitute. 56 Despite the positive outlook reflected in Tables 2 to 4 reveals critical challenges to AI’s sustainable integration. While 86.4% supported AI adoption, only 22.7% believed the industry was truly ready, with 51.4% either partially convinced or uncertain. This dichotomy between support and readiness emphasizes the necessity of capacity-building, infrastructural investment, and policy standardization. The call for formal training was almost unanimous, In this study, while only 27.3% of respondents reported formal AI adoption by their organizations, a significantly higher 70.5% were already using AI tools individually, highlighting a notable gap between institutional implementation and personal adoption. Furthermore, 88.6% of participants expressed a need for formal AI training, underscoring the urgency of structured capacity-building efforts within the pharmaceutical sector. This highlights significant associations, greater familiarity correlated with higher AI use, while support for adoption was strongly linked to the demand for training. This reinforces evidence from recent surveys suggesting that workforce upskilling is critical for successful AI adoption in pharma.16,57 Moreover, the strong association between perceived efficiency and support for adoption underscores that acceptance is driven by tangible benefits, consistent with technology acceptance models.
Furthermore, the query about whether AI improves product quality produced a significant effect, where staff in organizations with AI adoption rated these technologies positively. This finding underscores that organizational readiness to adopt AI is crucial, as it has been demonstrated that infrastructures and policies can influence AI outcomes significantly. 58 The belief that AI improves product quality not only enhances support for adoption but is also intricately linked to perceived job security, where users of AI tools reported lesser fears of job displacement compared to non-users. 59
Finally, the expression of a need for formal AI training correlates with strong support for AI adoption, a relationship strongly suggested by the statistical analyses presented. This reflects the notion that comprehensive training initiatives can facilitate greater acceptance and utilization of AI technologies, ultimately aligning with previously published works advocating for organized training frameworks as essential for successful technology uptake in healthcare. 60 Fostering innovation through effective knowledge dissemination and collaborative approaches is crucial for driving AI advancements in the pharmaceutical landscape. This reflects a shift toward creating platforms for continuous education and training that promote knowledge sharing among professionals in the industry, thus enhancing their readiness and ability to engage successfully with AI tools. 61 A multi-faceted strategy integrating regulatory policies, governance frameworks, reimbursement mechanisms, organizational capacity building, and innovation facilitation will collectively bolster the adoption of AI pharmaceutical sector.
Conclusion
The study advances the view that the pharmaceutical industry is not simply preparing to “adopt” AI but is already engaged in a cultural negotiation with it. The future of pharma will not be defined by whether AI is integrated, but by the depth, intelligence, and intentionality with which this integration is orchestrated. For AI to transition from peripheral use to core integration, a multi-pronged strategy is essential, combining formal training programs, robust governance structures, and ethical safeguards. Only then can AI achieve its full potential in revolutionizing the pharmaceutical landscape without compromising professional integrity or patient safety.
Limitation of the Study
This cross-sectional design, although practical and cost-efficient, inherently restricts any ability to infer causal relationships. Furthermore, the modest response rate and the limited sample of 100 participants substantially weaken the study’s external validity and constrain the extent to which the findings can be generalized.
Footnotes
Acknowledgements
The authors express their sincere appreciation to all pharmacists who participated in the survey for their valuable time and insights. The authors also acknowledge the contributions of all researchers whose referenced works supported this study.
Ethical Considerations
Participation in this cross-sectional study was strictly voluntary, with all individuals retaining the right to discontinue at any stage. All data were collected anonymously, with no personally identifiable information recorded. Ethical approval for the investigation was granted by the Institutional Review Board, Center for Research & Innovation (CRI) of R. P. Shaha University, Narayanganj, Bangladesh, Reference Number: RPSU/EC/Phr/2025/07.
Consent to Participate
Informed consent was obtained from all participants prior to their inclusion in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Other Journal-Specific Statements
Not applicable.


