Abstract
Antibiotics are vital for treating infections, but their inappropriate use has driven widespread resistance, now a major global health threat. This systematic review examines antibiotic use patterns in low- and middle-income countries using WHO’s prescribing indicator, aiming to guide interventions that promote rational antibiotic use, reduce resistance, and improve public health outcomes. The review followed PRISMA guidelines, searching databases such as Scopus, Embase (Ovid), and Medline (Ovid) for publications from January 2010 to November 2025. Titles and abstracts were initially screened by the first reviewer and discussed with a second reviewer to assess eligibility. Full texts of eligible articles were then independently reviewed by the reviewers, with final inclusion determined through consensus. The methodological quality of selected studies was evaluated using the Kmet Critical Appraisal Checklist. Studies were categorized into 3 groups—public and private sector, public sector only, and private sector only—to examine antibiotic use patterns relative to WHO standards, alongside demographic and institutional data. Electronic searches identified 2912 studies, of which 20 cross-sectional studies were from 11 countries considered eligible for inclusion. Their methodological quality scores ranged from 0.50 to 0.78. Across all studies, the average “percentage of prescriptions with antibiotics” was 51.4%, with 52.7% in the private sector, 49.3% in the public sector, and 61.8% in studies covering both sectors. Amoxicillin, an “Access” antibiotic under the WHO AWaRe framework, was most frequently prescribed, while “Watch” antibiotics such as ceftriaxone and cefixime were also widely used. Some studies reported high Watch antibiotic use (47.4%-74.0%), exceeding WHO recommendation and raising significant concern. The types of antibiotics prescribed also varied across age groups. The percentage of prescriptions with antibiotics and prescribing Watch group antibiotics was found to be higher than the WHO recommendation across studies. Implementing effective antibiotic stewardship programs is recommended.
Introduction
Antibiotics play a vital role in modern medicine and remain among the most widely used therapeutic agents worldwide. They have significantly reduced the burden of infectious diseases and have been instrumental in saving millions of lives. 1 In many low- and middle-income countries (LMICs), where infectious diseases are still a major cause of morbidity and mortality, antibiotics are essential for both routine treatment and complex medical procedures. 2 However, despite their critical importance, the rapid emergence ad spread of antibiotic resistance has become a significant global health threat. 3 Resistant bacteria are now widespread in both human and animal populations globally. 4 Prominent examples include methicillin-resistant Staphylococcus aureus and carbapenem-resistant Enterobacteriaceae, both of which pose significant international public health threats.5,6 As the effectiveness of commonly used antibiotics continues to decline, high-income countries increasingly rely on more expensive, second-line treatments. In contrast, many LMICs face limited access to and the high cost of these medicines, 7 which contributes to greater morbidity and mortality associated with resistant infections. 8
Recent global estimates highlight the substantial burden of antimicrobial resistance (AMR), with studies indicating that approximately 8.9 million deaths in 2019 were associated with bacterial infections, a considerable portion of which were linked to AMR. More recent assessments from 2021 suggest that about 4.71 million deaths were related to AMR, including an estimated 1.14 million deaths directly attributable to drug-resistant infections. Over the past 3 decades, AMR-related mortality has shown wide variation across different age groups and geographical regions. 9 Although antibiotic resistance is a global challenge, its impact is disproportionately severe in LMICs, where rates of healthcare-associated infections are significantly higher than in high-income countries. 10 In fact, the incidence of such infections in LMICs has been reported to be nearly double that observed in the United States and Europe.11,12
To address this growing threat, the World Health Organization (WHO) launched the Global Action Plan on Antimicrobial Resistance (GAP-AMR) in 2015, which has since guided international efforts to optimize the use of antibiotics, strengthen surveillance, and promote research and innovation. This plan emphasizes multi-sectoral collaboration across human health, animal health, and environmental sectors (the One Health approach), aiming to curb the spread of AMR globally. 13
The primary driver of antibiotic resistance is the improper use of these drugs, which includes actions such as not completing the prescribed course, overuse, and in dosing or indication. 14 Evidence suggests that a large proportion of antibiotic prescriptions globally are inappropriate. 15 Such misuse is strongly associated with adverse clinical outcomes, including side effects, prolonged illness, longer treatment durations, and increased risk of hospital admissions. 14 Multiple studies indicate that both healthcare providers’ prescribing practices and patient behaviors play a substantial role in this inappropriate use.16,17
One of the key factors contributing to the misuse of antibiotics, frequently highlighted in academic studies, is the practice of incorrect or inappropriate prescribing by healthcare professionals. 18 In alignment with the WHO GAP on AMR, the WHO has defined the “percentage of prescriptions with antibiotics” as a core indicator to monitor and promote rational use of antibiotics. 19 According to WHO standards, this figure should ideally range between 20.0% and 26.8%. 20
Numerous studies have explored antibiotic misuse at the community level across both developed and developing countries. However, a systematic analysis of antibiotic usage patterns in LMICs, specifically comparing the public and private healthcare sectors, remains limited. This comparison is particularly important, as the 2 sectors often differ in regulatory oversight, resource availability, and adherence to clinical guidelines. In addition, private facilities are more common in LMICs and frequently serve as the first point of contact for many patients. Understanding these differences can reveal variations in prescribing behaviors that drive inappropriate antibiotic use and inform the design of targeted interventions. Existing evidence consistently highlights widespread inappropriate antibiotic use in LMICs, emphasizing the urgent need for a thorough evaluation to promote more rational prescribing practices. To address this gap, we conducted a systematic review focused on LMICs, examining the indicator “percentage of prescriptions with antibiotics” and comparing findings between the public and private sectors.
Methods
The review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 21 The review protocol was registered in the PROSPERO database (CRD42024534259).
Searches in Databases
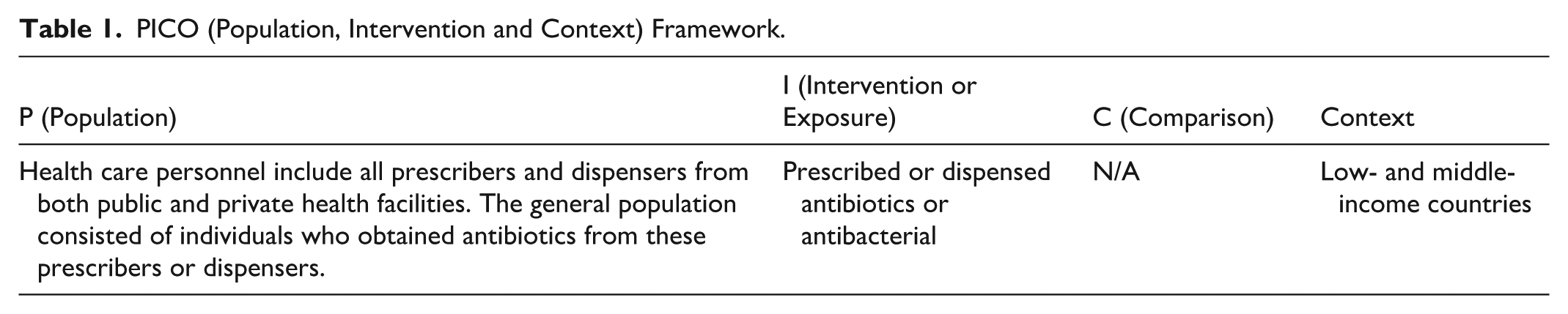
We applied PICo tool—Population (health personnel or general population), Intervention or Exposure (inappropriate antibiotic use) and Context (low- and middle-income countries)—to formulate the systematic review question. The aim of this systematic review was to assess antibiotic-prescribing practices and inform interventions to promote rational antibiotic use. Because antibiotics are used across all age groups, the review included studies involving the general population to examine overall prescribing patterns. Based on this, we developed a search strategy to find relevant publications in cooperation with an experienced librarian of author’s institute. We searched Scopus, Embase (Ovid) and Medline (Ovid) electronic databases using combinations of key terms such as (i) “health care personnel or general population” (ii) “antibiotic use,” and (iii) “low- and middle-income countries or developing countries” along with other alternative terms (Table 1). Boolean operators were used to combine search terms in specific ways to broaden or narrow the results (Supplemental Annex A. Search Strategy).
PICO (Population, Intervention and Context) Framework.
Inclusion Criteria
In order to assess whether the references found were relevant, we formulated the following criteria concerning types of studies, target groups and variables:
Studies: Peer-reviewed with quantitative analysis were eligible for inclusion. We included quantitative randomized controlled (trials), quantitative non-randomized (non-randomized controlled trials, cohort study, case-control study, cross-sectional analytic study), and quantitative descriptive studies (incidence or prevalence study without comparison group). The articles published from January 2010 to November 2025 were included in the review, providing a comprehensive overview of relevant studies while minimizing the inclusion of outdated data. The WHO Global Action Plan to reduce AMR was launched in 2015, and by 2023, 178 countries had developed and endorsed multisectoral AMR National Action Plans (NAPs). Therefore, this review captures current and past evidence that may support countries in planning and implementing antimicrobial stewardship programs.
Target groups and variables: Antibiotic use in the general population, dispensed from public and private facilities with or without prescription, was included. “Without prescription” covered the use of antibiotics through self-medication or over the counter purchases.
Exclusion Criteria
Studies were excluded via search limits if they were: qualitative study; studies focusing on specific antibiotics use; studies on antibiotics for specific conditions or specific groups, studies antibiotic use in specific hospital units (eg, intensive care units [ICUs] or critical care units [CCUs], maternity wards, or in-patient department); studies on antibiotics used for surgery, prophylaxis or antibiotic resistance. The articles with gray literature, case series, and case reports were also excluded.
Inclusion Process
Step 1—Identification
Records identified through data-based searches (n = 2912) were collected and initially reviewed by the first reviewer, who also removed duplicates entries.
Step 2—Screening of Titles and Abstracts
Titles and abstracts of the references found (n = 2835) were screened by a first reviewer and discussed with second reviewer to determine whether these publications satisfied the inclusion criteria. During the title review, articles were excluded, if they were not relevant to the subject matter. Any article with an ambiguous title or title suggestive of the topic was evaluated in the abstract review for details that indicated the article might meet the inclusion criteria. In this phase, both reviewers agreed in virtually 100% of the cases.
Step 3—Screening Based on Full Texts
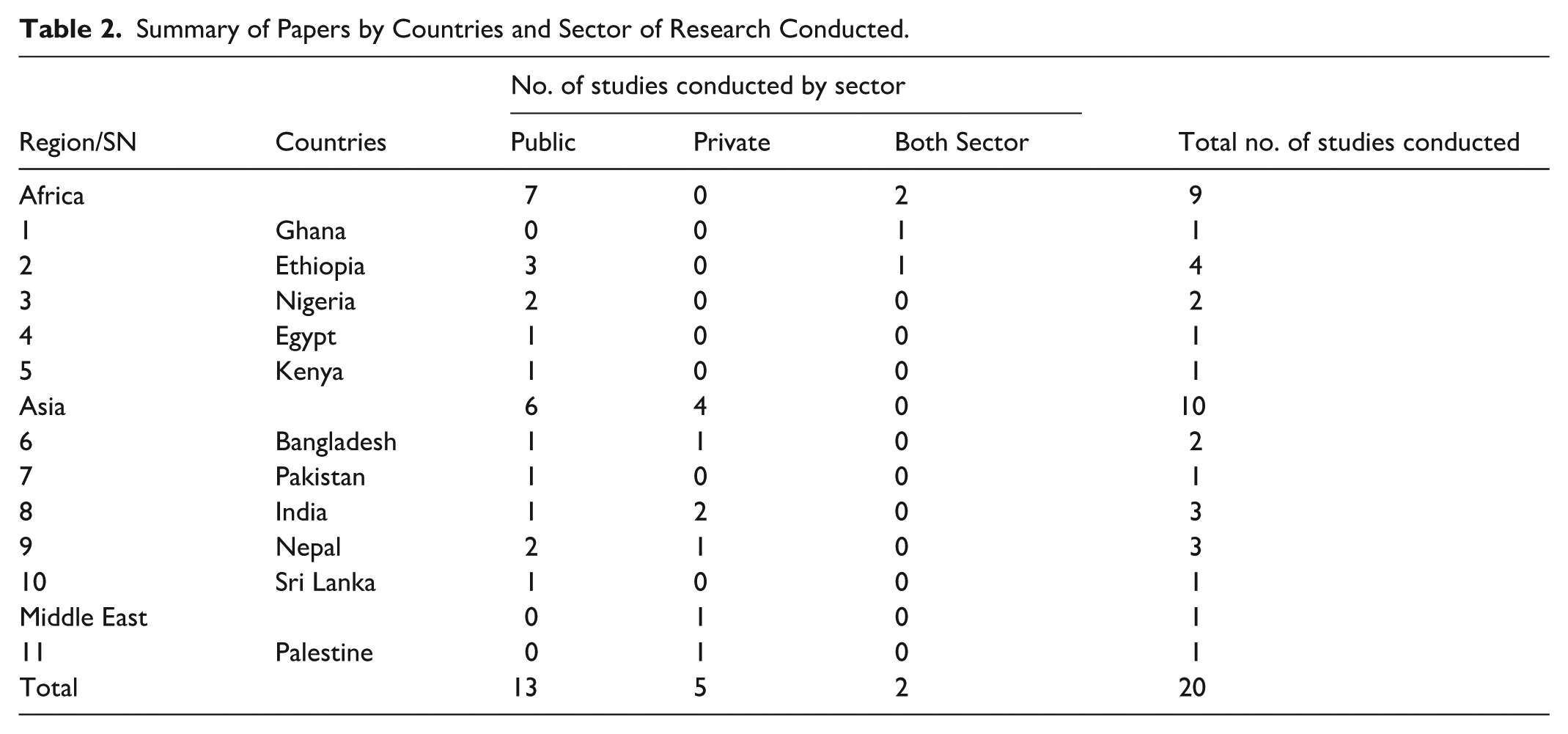
Full texts of the selected articles (n = 240) were independently assessed by the first and second reviewers to determine whether the inclusion criteria were met. Final inclusion decisions were made through discussion and consensus among all 4 reviewers. With consensus, a total of 20 studies, all displayed in Table 2, appeared to be eligible for inclusion.
Summary of Papers by Countries and Sector of Research Conducted.
Critical Appraisal
The methodological quality of 21 studies was assessed by 2 reviewers (AN and DH) independently, using the Critical Appraisal Checklist for Reviews (Supplemental Annex B. Critical Appraisal Checklist for Reviews). One study was excluded because its mean quality score was below 50%. Consequently, a total of 20 studies were included in the review. This checklist is an adopted version of Standard Quality Assessment Criteria for Evaluating Primary Research Papers from a Variety of Fields (Kmet et al). 22 The Kmet guideline is widely used for health science review, providing a standard, reproducible criterion for critically appraising the quality of a broad range of studies.
Nine items were scored depending on the degree to which the specific criteria were met (“yes” = 2, “partial” = 1, “no” = 0). Items not applicable to a particular study design were marked “n/a” and were excluded from the calculation of the summary score. A summary score was calculated for each paper by summing the total score obtained across relevant items and dividing by the total possible score [ie,: 18 − (number of “n/a” × 2)]. Assigning “n/a” was not permitted for any of the items, and the summary score for each paper was calculated by summing the total score obtained across the 9 items and dividing by 18 (the total possible score). The final scores presented by summing the average scores of both reviewers, and the mean quality scores are reported in Table 5.
Data Extraction
A data extraction spreadsheet using Excel version 2010 was adopted from Joanna Briggs Institute Reviewers’ Manual. 23 Relevant information were extracted from articles into an adapted data extraction tool (Supplemental Annex C: Data Extraction File). Headings included in the data extraction tool are lead author’s name, country name where the research was conducted, study type, sample, objectives of the research, statistical analysis, research results/outcomes, author’s specific comments, reviewer’s comments and conclusion.
Data Analysis and Synthesis
Based on the information covered by sectors, included studies were divided into 3 groups: public and private sector, public sector only, and private sector only for analysis. The antibiotic use pattern is presented by the ratio of prescriptions with antibiotics, analyzed with the target of WHO standard. 20 The number of samples, along with the included health institution surveyed and the ratio of prescriptions with antibiotics and antibiotics use under WHO’s AWaRe (Access, Watch, and Reserve) classification is the major information presented in the analysis. The extend of antibiotic use in the general population, male and female ratio, groups of people using antibiotics, and other related information are also presented where applicable.
Results
Description of Included Studies
Our initial search identified 2912 records, of which 2835 non-duplicate titles were screened for inclusion. After reviewing the title and then abstract, 240 were included for full-text review. Of these, 220 were excluded for various reasons: totally irrelevant (n = 130), qualitative studies (n = 05), studies with specific groups (n = 11), studies with specific units/department (n = 28), studies on specific disease conditions (n = 30), studies on specific antibiotics (n = 04), documents not available (n = 11) and one study excluded after assessing the quality (Figure 1). Finally, 20 included articles were considered to be relevant with good or high quality.24 -43

PRISMA flow diagram of study selection.
A total of 20 studies from 11 different countries were included (Table 2). When grouping the countries into United Nation’s country grouping system, 9 studies were conducted in Africa, 10 in Asia and the remaining one in the Middle east. No studies from LMICs in the European or American regions were included, as none met the predefined search criteria. By country, 4 studies were conducted in Ethiopia; 3 studies each were conducted in both India and Nepal; 2 studies each were conducted in Nigeria, and Bangladesh; and 1 study each was conducted in Ghana, Egypt, Kenya, Pakistan, Sri Lanka, and Palestine.
While analyzing the sector, 13 studies24 -28,36 -43 covered data collection in public sector, however 5 studies29 -33 covered the private sector only, and 2 studies34,35 covered data collection in both public and private sectors. Among the included studies, 7 from Africa, and 6 from the Asia conducted research in the public sector. Four studies from Asia and one from the Middle East were conducted in the private sector. Similarly, the 2 studies that conducted research in both sectors were from the African region. Unfortunately, no studies from Asia or the Middle East conducted research in both sectors.
Methodological Quality of Studies
All 20 studies utilized a cross-sectional design. In addition to descriptive analysis using by all studies, 2 studies each used ANOVA and non-parametric tests, and 4 studies used regression to determine the association or effect of the variables. The reasons for a study to receive an overall lower quality rating were the use of convenient sampling (which may have compromised the results), 29 lack of socio-demographic data24,28,30 -32,36,38,39,42,43 lack of clear presentation of the sampling method, 31 and conducting the study in a referral or tertiary care hospital only,25 -27,32,37 which may limit the generalizability of the results.
Despite all studies discussing the “percentage of prescriptions with antibiotics,” many did not adequately account for the clustering of respondents with different socio-demographic characteristics. Failing to account for this clustering tends to overlook the association and effects of prescribing behavior with socio-demographic characteristics.
While assessing the methodological quality of included studies using Kmet et al forms and guideline, 22 the mean quality scores reported in Table 4 ranged from 0.50 to 0.78.
Characteristics of Studies
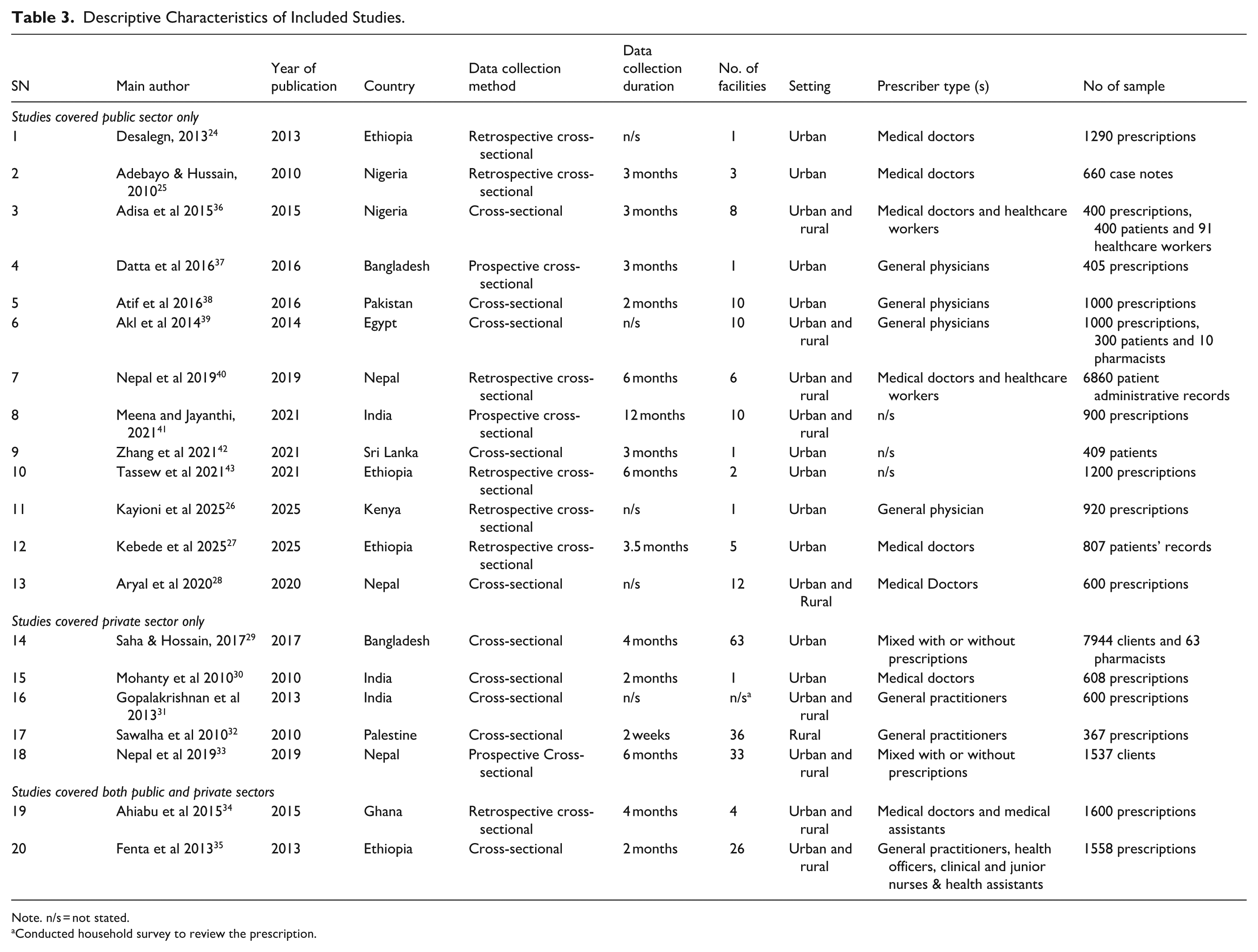
Characteristics of the included studies are summarized in Table 3. Although, all studies utilized a cross-sectional design, 7 studies24 -27,34,40,43 were retrospective, and three33,37,41 were prospective. The duration of data collection varied among the studies, ranging from 2 weeks to maximum of 1 year; however, 5 studies24,26,28,31,39 did not clearly mention their duration.
Descriptive Characteristics of Included Studies.
Note. n/s = not stated.
Conducted household survey to review the prescription.
In the survey, a minimum of one to maximum of 680 health facilities were included. Nevertheless, one study, 31 which was household survey, did not mention the total number of health facility included in the study. Ten studies24 -27,29,30,37,38,42,43 were conducted in urban areas, 9 studies28,31,33 -36,39 -41 in both urban and rural areas, whereas one study 32 was conducted in rural areas only. The number of samples in each study was extremely dissimilar with a minimum of 367 to maximum of 1600 prescriptions reviewed. Some studies, however, reviewed case notes, 25 medical records27,40 and interviewed clients.29,33 Additionally, patients or consumers,36,39,42 health workers 36 and pharmacists29,39 were interviewed.
The studies included different prescriber types such as doctors or general practitioners, nurses, health or medical assistants and health assistants. Eleven studies24 -28,30 -32,37 -39 included medical doctor only or under other name such as general physicians, village doctors, or general practitioners. Six studies29,33 -36,40 included mixed prescribers, whereas 3 studies41 -43 did not mention the prescribers’ name.
Appropriateness of Antibiotics Prescribing
The “percentage of prescriptions with antibiotics” is one of the standard indicators established by the WHO to monitor appropriate prescribing, and the target recommended to be between 20.0% and 26.8%. 20 This target may require modification over time and between countries but is currently recommended to be below 30%. 44
While analyzing this indicator across all 20 studies, the finding show considerable variation. Two studies25,36 fall within the WHO’s recommended range of 20% to 30%. Four studies33,39,41,42 fall within the 30-<40% range and 3 studies32,38,40 fall within the 40-<50% range. Seven studies24,28,30,31,34,37,43 fall within the 50-<60% range, while another 3 studies27,29,35 fall within the 60-<70% range. The remaining study 26 fall extremely high value, exceeding 70%.
Within the sector-wise analysis, among the 13 studies conducted in the public sector, only 2 studies25,36 fall within the WHO’s recommended range. However, none of the studies conducted in the private sector or in both sectors combined fall within this recommended range. In summary, the average “percentage of prescriptions with antibiotics” is highest in studies covering both sectors (61.8%), compared to the private sector (52.7%) and the public sector (49.3%). Overall, the average across all studies is 51.4% (Table 4).
Percentage of Prescription With Antibiotics Found by Number of Studies With Sector.

Factors Related to Antibiotics Prescribing and Commonly Used Antibiotics
Table 5 presents the factors associated with antibiotic prescribing, along with information on the most prescribed antibiotics. Ten studies did not provide information on the age group and/or gender of individuals to whom antibiotics were prescribed. However, 3 studies34,35,41 presented the male and female ratio of antibiotic prescriptions. One study 40 stated that males (P = .005) were more likely to receive antibiotics than female. Two studies33,40 presented results showing that prescribed antibiotics were significantly more common in younger age group (p =< .001). Two studies showed that more than two-thirds of paediatric 35 and child 37 patients received antibiotics. Adebayo and Hussain 25 stated that adults aged 19 to 29 years received more antibiotics (36.2%) than other age groups. However, Meena and Jayanthi 41 stated that antibiotics prescribing was highest among 25 to 64 years age group, whereas a study 29 conducted in Bangladesh stated that antibiotics were the most commonly used drug in all age groups.
Summary of Statistical Methods Used, Results and Kmet Quality Rating of Included Studies.

Note. PHC = Primary Health Centre; IRDS = Index of Rational Drug Supply; IRDP = Index of Rational Drug Prescribing; COPD = Chronic Obstructive Pulmonary Disease.
Mean quality rating: the scores by each assessor were calculated as (actual score/potential maximum score).
Eight studies did not report data on the group or specific name of the most commonly prescribed antibiotics. Another, 8 studies24,26,28,31,32,36,42,43 identified amoxicillin, a first-line antibiotic categorized under the “Access” group of AWaRe classification, as the most frequently prescribed antibiotic, with prescribing rates ranging from 16.4% to 49.2%. Additionally, 3 studies reported that Ceftriaxone27,40 and Cefixime 33 —both classified under the “Watch” category of the WHO AWaRe classification—were the most commonly prescribed antibiotic classes.
Among these studies, three27,33,40 reported particularly high prescribing rates of “Watch” group antibiotics—49.8%, 47.4%, and 74.0%, respectively. Such higher use of “Watch” antibiotics compare with “Access” antibiotics suggests potentially inappropriate prescribing practices that may contribute to an increased risk of antimicrobial resistance. This pattern contrasts with WHO recommendation, which states that at least 60% of total antibiotic consumption should come from the “Access” group, not the “Watch” group.
Discussion
This review is among the first to comprehensively examine patterns of antibiotic use in LMICs. The analysis focused on the percentage of prescriptions with antibiotics, a key indicator recommended by the WHO to monitor rational antibiotic use. Across the 20 included studies, the prescription percentage exceeding WHO’s recommended threshold of 30% 44 was persistently higher in most studies. Although this review includes studies conducted in Asia, Africa, and the Middle East, evidence from broader global analyses suggests similar patterns of elevated antibiotic prescribing in European LMICs; for example, systematic reviews indicate that antibiotic prescription rates, while somewhat lower than those observed in this review (47.0%), still exceed recommended levels. 45
In this review, consistent patterns of prescribing were obtained in both public and private health sectors, with notable overprescribing regardless of prescriber type, level of health care facility, or urban-rural setting. Despite variations in study designs and sample sizes, all the studies employed a cross-sectional methodology and included diverse exposure and outcome measures. However, the consistency in antibiotic prescribing trends across these studies indicates a widespread and systemic issue in LMIC health systems.
Among the studies reviewed, 2 studies34,35 that included both public and private sectors reported antibiotic prescription rates of 59% and 64.5%, respectively, clearly exceeding the WHO recommendation. These studies shared similarities in prescriber profile and study setting. Within the public sector alone, eleven,24,26 -28,37 -43 out of 13 studies documented similarly high prescription rates ranging from 35.7% to 84.1%, while only 2 studies25,36 met or remained below the threshold of 30%. Consistent with this pattern, none of the studies conducted in the private sector fell within the recommended range. A significant finding obtained from the review is the consistent overuse of antibiotics, often in the absence of clear clinical justification. Several factors may drive this trend. One of the study found that limited access to updated clinical guidelines and a lack of training in antimicrobial stewardship contribute to inappropriate prescribing, especially for viral infections where antibiotics are ineffective. 46 Similarly, another identified diagnostic limitations in LMICs lead clinicians to rely on empirical judgment rather than evidence-based decisions. 47 In addition, a study explored patient demand and expectations exert pressure on providers to prescribe antibiotics unnecessarily to maintain satisfaction or avoid confrontation. 14 Besides, weak regulatory environments in many LMICs permit over-the-counter sales of antibiotics without prescription, further promoting misuse and self-medication. 48
The consequences of inappropriate prescribing practices—both under prescribing such as antibiotics withheld when needed, and over-prescribing such as antibiotics giving without appropriate indication—are substantial. 49 These practices accelerate the development of antimicrobial resistance (AMR), a growing global health threat that can lead to prolonged illness, increased treatment costs, and higher mortality.49,50 Even when bacterial infections are present, misuse through incorrect drug selection or incomplete treatment courses can result in therapeutic failure. 51 In this context, the widespread over prescription of antibiotics identified in the review suggests the urgent need to educate prescribers, strengthen prescription regulation, and raise public awareness about the consequences of inappropriate antibiotic use in LMICs.
This review found that amoxicillin, an “Access” antibiotic under the WHO AWaRe framework, was most frequently prescribed, while “Watch” antibiotics such as ceftriaxone and cefixime were also commonly used. Although many studies reported high use of Access antibiotics, some showed disproportionately high prescribing of Watch antibiotics (range from 47.4% to 74%), deviating from WHO recommendation that at least 60% of national antibiotic consumption should come from the Access group. 52 Antibiotic consumption under the AWaRe framework varied across European LMICs: Albania experienced a post-pandemic rise with Watch antibiotics comprising 82.0% of the most used agents, whereas Armenia moved closer to WHO targets with Access antibiotics reaching 62.7% by 2023. Azerbaijan reported high parenteral use of ampicillin and amoxicillin, and Kosovo showed frequent ceftriaxone use alongside widespread self-medication. 53 These patterns highlight the persistent overuse of Watch antibiotics, which should be reserved for specific indications, such as severe infections or when pathogens are resistant to first-line agents. 54 Frequent empirical use of Watch antibiotics without diagnostic confirmation increases the risk of antimicrobial resistance and undermines the effectiveness of critical antibiotic classes, 55 driven by factors including over-the-counter availability, limited laboratory services, and reliance on clinical judgment and insufficient stewardship support. 46
These prescribing trends highlight the urgent need to strengthen antimicrobial stewardship (AMS) programs across all levels of the health system, including capacity-building for prescribers, improved access to rapid and affordable diagnostics, and stronger adherence to national treatment guidelines and WHO-recommended protocols. 56 Community-level strategies, such as public education on appropriate antibiotic use, can further help reduce pressure on prescribers and limit self-medication. 57 Importantly, AMS initiatives must be context-sensitive and aligned with global efforts, including the WHO Global Action Plan on AMR 13 and guidance from the Quadripartite Joint AMR Secretariat, 58 ensuring that efforts to reduce misuse do not compromise equitable access to essential, life-saving antibiotics, particularly in LMICs settings.
Prescriber behavior also reflects broader systemic challenges. Misuse of antibiotics due to incorrect selection, dosage, or treatment duration is one of the recognized forms of irrational medicine use. 59 Though, antibiotic selection should ideally be based on confirmed diagnosis and infection severity, 60 inappropriate choices still occur due to limited training, lack of protocol awareness, or patient pressure. 61 Supporting prescribers with ongoing education and enforcing the use of standardized treatment protocols can help mitigate these issues. 62 However, further research is needed to evaluate the degree of guideline adherence in practice, as this review was based on prescription data alone and may not capture clinical rationale.
Diagnostic gaps further exacerbate inappropriate antibiotic use. In many LMICs, bacterial culture and sensitivity testing are either unavailable or inaccessible due to cost or infrastructure constraints. 63 For example, studies in Nepal highlight that many health facilities operate without basic laboratory services and regularly face antibiotic stockouts.64,65 Similar challenges have been reported in hospitals in Kosovo, where over 91% of patient receive antibiotics empirically due to limited microbiological testing and delayed results, leading clinicians to rely on clinical presentation rather than laboratory confirmation. 66 Across these contexts, prescribers often make decisions based on drug availability rather than clinical necessity, which can lead to suboptimal treatment and increased resistance risk.
Another critical issue is antibiotic use without prescription. In Nepal, over one-third of patients were found to obtain antibiotics from private pharmacies without a prescription. 40 Similar practices have been documented in several LMICs in Eastern and Southern Europe, even though antibiotic dispensing legally requires a prescription in all European countries. 67 Such non-prescription use is common in LMICs, largely due to weak enforcement of pharmaceutical regulations.68,69 Studies indicate a strong link between community-level antibiotic misuse and rising AMR rates.70,71 While such practices are problematic, interventions must avoid restricting access for patients in urgent need of antibiotics. In many settings, over-the-counter purchases may represent the only feasible treatment option. Thus, policy solutions must consider health system constraints while promoting responsible use.
Although research on antibiotic use in LMICs has expanded, significant knowledge gaps remain—especially concerning the volume and patterns of antibiotics obtained through informal channels, such as self-medication, unregulated pharmacies, or unlicensed drug vendors. In the absence of reliable and comprehensive data, it becomes difficult for policymakers to design, implement, and evaluate effective antibiotic stewardship programs. To bridge this gap, future studies should focus on conducting nationally representative surveys, including pharmacy exit interviews and household-level assessments, to capture both formal and informal antibiotic use. Furthermore, using standardized definitions and methodologies—particularly for measuring antibiotic misuse, consumption patterns, and prescribing behaviors—would improve cross-country comparability and strengthen global strategies to combat AMR.
In summary, this review highlights the widespread and often inappropriate use of antibiotics in LMICs, driven by systemic challenges in regulation, training, diagnostics, and patient behavior. Addressing this issue requires coordinated action at all levels of the health system, including strong stewardship frameworks, improved education for provider and communities, and sustained investment in diagnostic capacity. Global initiatives—such as the WHO Global Action Plan on Antimicrobial Resistance (GAP-AMR) and its translation into National Action Plans (NAPs)—provide an essential foundation for guiding countries in implementing evidence-based interventions. 72 Without urgent and sustained efforts aligned with these frameworks, inappropriate antibiotic use will continue to erode treatment efficacy and intensify the global threat of antimicrobial resistance.
Limitations
The review lacked adequate evidence to comprehensively describe antibiotic use patterns within the general population. Most of the available studies relied on prescription records or data from healthcare facilities, with only a few incorporating patient exit interviews at private pharmacies. This presents a major limitation, particularly in LMICs, where private pharmacies often serve as the initial point of care and a common source for medicine procurement. 68 Moreover, the widespread practice of obtaining antibiotics without a prescription further complicates the picture. Consequently, to accurately understand antibiotic usage trends, research must encompass all potential sources—including non-documented use among the general population and informal healthcare providers.
A key limitation in the current body of literature is the lack of comparative analysis to assess whether antibiotic prescriptions were unnecessary, excessively costly, or aligned with established clinical guidelines. While meeting the clinical needs of patients is essential for rational antibiotic use, the broader economic burden on communities is often overlooked and deserves equal consideration.
Another notable constraint is the imbalance in sample sizes across the studies reviewed, which may have introduced bias and affected the representativeness of findings. The variability in study design and methodology also led to inconsistencies, limiting the ability to draw uniform conclusions. Furthermore, the absence of robust evidence to explain these inconsistencies presents a challenge in developing clear, evidence-based policy recommendations.
This review is not intended to guide specific national policies or enforce practices but rather to offer a general understanding of antibiotic use trends in LMICs. To strengthen the evidence base, future research should adopt standardized methodologies, consistent definitions, and representative sampling across diverse settings. In-depth studies focusing on individual prescribing behaviors and patient practices are particularly important to capture real-world use, especially in regions where gaps between policy and practice are significant.
Another limitation of this review is the inability to fully analyze antibiotic use by the WHO AWaRe classification, as this review primarily focused on prescription percentages. Available data were limited and incomplete, so the review presented what could be extracted. A dedicated review of AWaRe-based antibiotic consumption is recommended, especially given recent WHO and other agency initiatives to monitor and evaluate stewardship outcomes.
Conclusion
This systematic review examined the “percentage of prescriptions with antibiotics,” a core prescribing indicator outlined by the WHO. Findings indicated that in the majority of studies conducted across LMICs, antibiotic prescribing rates surpassed WHO-recommended thresholds. Notably, prescribing patterns showed minimal variation by prescriber type, healthcare setting (urban or rural), or facility level. However, differences were observed in patient age groups and the specific antibiotics most frequently prescribed. Some studies reported disproportionately high use of Watch antibiotics under the AWaRe classification, deviating from WHO recommendation. These findings underscore the critical need for antibiotic stewardship initiatives to encourage appropriate prescribing practices, curb the rise of antibiotic resistance, and improve public health outcomes.
Supplemental Material
sj-docx-1-inq-10.1177_00469580261420724 – Supplemental material for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis
Supplemental material, sj-docx-1-inq-10.1177_00469580261420724 for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis by Anant Nepal, Nilam Bhandari, Ranjeeta Subedi and Delia Hendrie in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-4-inq-10.1177_00469580261420724 – Supplemental material for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis
Supplemental material, sj-docx-4-inq-10.1177_00469580261420724 for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis by Anant Nepal, Nilam Bhandari, Ranjeeta Subedi and Delia Hendrie in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-xlsx-2-inq-10.1177_00469580261420724 – Supplemental material for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis
Supplemental material, sj-xlsx-2-inq-10.1177_00469580261420724 for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis by Anant Nepal, Nilam Bhandari, Ranjeeta Subedi and Delia Hendrie in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-xlsx-3-inq-10.1177_00469580261420724 – Supplemental material for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis
Supplemental material, sj-xlsx-3-inq-10.1177_00469580261420724 for Antibiotics Use Patterns in Low- and Middle-Income Countries: A Systematic Review Analysis by Anant Nepal, Nilam Bhandari, Ranjeeta Subedi and Delia Hendrie in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
We thank the researchers whose work was included in this review and acknowledge Mr. Umesh Bhusal, PhD scholar at the University of New South Wales, Australia, for supporting the additional searches for this revision.
Ethical Considerations
Ethical approval was not required for this review, as it included only previously published, publicly available studies.
Consent to Participate
No human subjects were involved; therefore, informed consent was not applicable.
Author Contributions
AN and DH designed the study. AN, NB, RS, and DH cleaned and analyzed the data. AN and NB drafted the manuscript. AN, NB, RS, and DH revised the manuscript. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
As this article is a systematic review, all pertinent data are contained within the manuscript, and there are no raw data available. However, the data that supports the findings of this study are available on request from the corresponding author.*
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
