Abstract
Commercial interests have increasingly engaged in scientific research, particularly as the European Health Data Space (EHDS) allows access to electronic health records for such studies. Normative legal research was conducted to examine EU Member States’ effective medical research legislation and landmark cases. Eleven EU Member States’ legislation do not reference commercial medical research. Regarding the others, some adopt stricter rules for commercial research. Others prioritize incentives for non-commercial research. Related legislation primarily regulates research based on funding sources, diverging from the multi-actor and multi-interest nature of health data research. Both the GDPR and ethical guidelines generally exempt anonymized data from oversight, creating a governance gap. Low marginal costs and the black-box nature of data-driven approaches further intensify funding biases, potentially undermining public trust. Member States also differ in data subjects’ right to opt out of scientific research. Such regulatory fragmentation undermines EHDS implementation and creates opportunities for regulatory arbitrage. Commercial scientific research should be understood as a type of research conducted by profit-driven actors through ongoing business operations. Special measures should be applied to commercial scientific research within the EHDS, including mandatory consideration reflecting data market pricing, tiered data dividends, compulsory health data access body ethical review, conflicts of interest declaration, and opt-out rights for data subjects. Rules for providing data access are tailored to the stage of research and require a balancing test between research interests and trade secrets. Raw data should be made accessible. Processed data should be made available when classified as essential. Research results should always be provided. A centralized EU platform is recommended to reduce administrative burdens.
Keywords
There is funding bias in industry-funded research.
This study proposes special regulatory measures for the access to and provision of electronic health records within the European Health Data Space (EHDS).
This research helps prevent potential funding bias in scientific research on health data and promotes harmonized implementation and improvement of the EHDS.
Introduction
The involvement of commercial actors in the research landscape has increased in recent decades. More than half of the research and development in Europe is funded by the private sector. 1 Commercial scientific research is a paradox, although it is a key driver of scientific progress, its commercial motivations can give rise to distinct ethical and legal challenges. Essentially, the nature of scientific research is inherently in conflict with commercial activities. The nature of science calls for the open reuse of health data to serve public health, whereas commercial logic tends toward closed and exclusive use to sustain profitability. Science requires researchers to remain impartial in their work, free from the influence of private interests. 2 Conversely, commercial activities—driven by economic interests and the pursuit of private gain—often seek to privatize or monopolize research outcomes. Therefore, as a mixed blessing, commercial scientific research warrants special attention within regulatory frameworks.
The European Union’s (EU) approach to commercial scientific research reflects a gradual shift from caution to conditional acceptance. Traditionally, the EU has maintained a cautious position regarding its commercial involvement in scientific research. For instance, Directive (EU) 2019/790 defines a “research organization” as one that operates on a nonprofit basis or reinvests all profits into its scientific research activities. 3 Similarly, the Digital Services Act requires “vetted researchers” to be independent from commercial interests. 4 However, recently, the EU has acknowledged privately funded research within its health data governance frameworks. The General Data Protection Regulation (GDPR) allows exemptions for privately funded scientific research from the general prohibition on processing health data. 5 More recently, the European Health Data Space (EHDS) obliges health data access bodies to grant data access when the stated purpose is scientific research in the health or care sectors. 6 Furthermore, it confirms in Recital 61 that such research includes applied, fundamental, technological, and privately funded activities.
Although funding bias in commercial scientific research is widely recognized, it has received limited attention in EHDS-related studies. Moreover, legislative responses remain limited, with current regulations falling short in providing clear rules for commercial scientific research. Thus, tailored legislation is required to strike a balance between scientific and commercial interests in the EHDS.
Method
We adopted a primarily normative research methodology. Relevant legislation and policy documents from all EU Member States were collected up to 31 July 2025. We consulted official national legal repositories (such as Legifrance), as well as the websites of health authorities (such as The State Institute for Drug Control of the Czech Republic), and national ethics committees(such as Cyprus National Bioethics Committee). These sources were identified through systematic web searches and were verified as official national platforms before inclusion.
Within these databases and websites, we conducted structured searches using the keywords “(medical OR clinical OR health) AND (research) AND (standard OR legislation).” We then further refined the search scope with the terms “commercial,” “non-commercial,” “profit,” “non-profit,” to isolate provisions relevant to the commercial dimension of research. As these queries initially yielded limited results, we broadened the strategy to include additional commercial and industry-related concepts, such as “sponsor,” “industry-sponsored research,” and “economic interests.”
To ensure comprehensive coverage, we retained all documents on medical or health research that contained any reference to commercial factors or industry involvement. (The detailed search procedure is illustrated in Figure 1) During the collection of legislation, some documents were not available in English. These were first translated using Google Translate and then manually cross-checked by 3 researchers to ensure accuracy. Based on the collected materials, we then compared the legislative approaches of the Member States toward commercially oriented scientific research and classified the regulatory approaches into 4 categories.
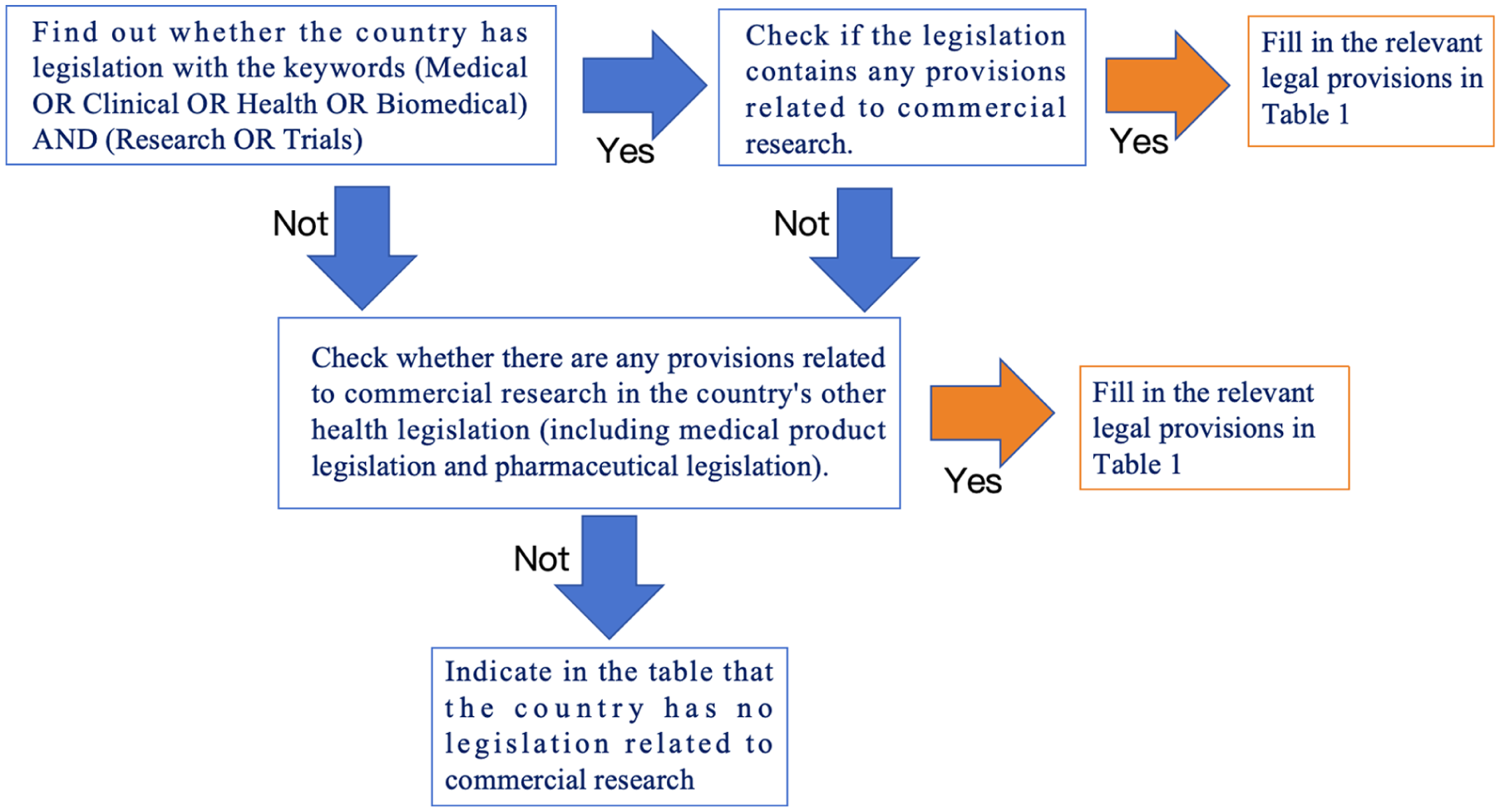
Methods to find out if there are commercial medical research legislation in the EU countries.
There were a few research limitations. Non-English legal texts were translated via Google Translate, which may give rise to misunderstandings. The reliance on keyword-based searches and manual reviews may have overlooked relevant regulations. Finally, the study focuses solely on statutory texts without empirical investigation.
Results
Most frameworks focus on clinical research and they rarely regulate health data. And Research involving commercial elements, such as private funding, is distinguished from other studies in 14 countries by several sector-specific restrictions. The results are presented in Table 1. To enhance the visualization of these findings, we have colored the EU map differently based on the regulatory approaches adopted by EU Member States toward commercial research (Figure 2).
Detailed Legislative Provisions on (Non-)Commercial Research in EU Member States.


Overview of regulatory approaches to (non-)commercial research in EU member states.
Direct Restrictions on Commercial Research
France and Italy have explicitly restricted their commercial research. France defines the concept of “noncommercial research” as research whose outcomes are not for profit and aims to achieve public health goals. When research initially classified as noncommercial no longer meets the classification, sponsors are obligated to transfer any generated funds to the health insurance. 7 Additionally, when research projects are conducted for commercial purposes, sponsors must bear the additional costs arising from supplies or examinations explicitly required by the research protocol. 8
Similarly, Italy defines “trials conducted for industrial or commercial purposes” as studies promoted by the pharmaceutical industry, corporate entities, or in any case by private institutions for profit-making purposes. Moreover, private scientific hospitals and treatment facilities are excluded from this definition. The outcomes of such trials may be utilized for the industrial development of pharmaceuticals, as well as for regulatory or commercial purposes. 9 Additionally, Italy shows a clear preference for noncommercial research. For instance, in nonprofit clinical trials, relevant data and trial results may be submitted for registration purposes, even in studies involving low-risk interventions. 10
Special Incentive for “Noncommercial Research”
Some countries focus on promoting noncommercial research rather than tightening regulations for commercial research. “Noncommercial research” is defined by its conducting entities—typically academic institutions or public hospitals—and the absence of profit motives. For instance, Slovakia defines noncommercial clinical trials as those with sponsors including higher education institutions and other specific entities; no contracts permit third-party commercial use of trial data. 11 Furthermore, Czech and Polish legislation forbid using data from noncommercial trials for drug marketing authorization.12,13 These countries often offer incentives for such trials. For example, Hungary streamlines authorization procedures. 14 Belgium provides full/partial exemptions from specific experimental requirements and waives related fees. 15 Estonia waives professional assessment fees. 16
These countries encourage noncommercial research for several reasons. “Noncommercial research” is technically easier to define due to its consistency. Conversely, commercial scientific research is more diverse regarding both the purposes and types of entities involved, making its regulation more complex. Furthermore, biomedical research is cold and complex, 17 therefore, it requires incentives.
Regulation of Research Purposes in Specific Fields
This approach limits the research to critical areas for noncommercial purposes. For example, Spain prohibits the use of personal genetic data for commercial purposes. 18 Such restrictions exist because domains such as genetics are directly correlated to human health, safety, the interests of specific ethnic groups, and national or ethnic security.
Similar to many other countries, Denmark defines the concept of “sponsor” in its legislation. However, Denmark’s Act on the Ethical Treatment of Health Science Research Projects and Health Data Science Research Projects is distinctive. It stipulates that, for the approval of health sciences research projects, the financial support researchers receive from private companies, foundations, and other entities—along with any other financial ties—must be clearly identifiable 19 ; moreover, it sets forth special rules for health data science research projects with commercial sponsors. 20 If a commercial sponsor is involved, the relevant scientific ethics committee must review key aspects, such as contract terms and remuneration, particularly for projects involving diagnostic imaging data from patients’ medical records.
Lack of Specific Provisions for “commercial” or “Noncommercial” Research
However, some countries—such as the Netherlands, Finland, Portugal, Greece, Bulgaria, Cyprus, Lithuania, Sweden, and Luxembourg—neither define “noncommercial” nor “commercial” trials nor provide detailed rules governing entities such as sponsors. This gap may reflect either unawareness of the issue or a deliberate reluctance to address it. Some are concerned that imposing stricter regulations on commercial scientific research may lead to over-regulation, creating unnecessary administrative burdens. Since all scientific research requires funding, singling out commercial scientific research may stigmatize it. 21 Additionally, imposing restrictions on “commercial research” may reduce enterprises’ willingness to invest.
Discussion
Typological Definition of Commercial Scientific Research in the European Health Data Space Framework
The EHDS provides no clear definition of “scientific research.” Current legislation defines commercial scientific research based primarily on funding sources. However, scientific research often entails collaborative efforts and overlapping interests, making it difficult to strictly define commercial scientific research using funding sources alone.
Commercial activities are too diverse and complex to be adequately captured by a fixed definition, and are further challenged by ever-evolving business practices. When real-life situations are too diverse to fit an abstract concept, typology can serve as a supplementary. 22 Typologies, with open and fluid boundaries, are well suited to capture the dynamic nature of commercial scientific research.
Thus, commercial scientific research is proposed as a type generally characterized by actors such as merchants, motivated by the pursuit of profit, and engaged in sustained and continuous business operations (see Figure 3). In commercial scientific research, the actors are usually merchants such as companies. The purpose of this study can be operationalized by examining the historical allocation of profits derived from similar activities if more than 50% of prior profits are distributed to the researchers themselves, other members of the organization, or shareholders. Continuous business operations can be assessed based on their duration, frequency, and actor dependence. Duration can be measured in fiscal years, and company finances and tax obligations are calculated on this basis. Frequency refers to activities that occur three or more times. Actor dependence reflects whether or not research constitutes a core business activity and can be indicated by a company’s registered scope, articles of association, and corporate resolutions.

A comparison of the characteristics of commercial research (internal) and noncommercial research (external).
The typology emphasizes the consistency of overall values rather than the completeness of elements. In other words, scientific research is only considered commercial for the purposes of this analysis if it is the primary means for an entity’s long-term continuous profit generation.
For example, the Arc Institute—a nonprofit biomedical research organization affiliated with Stanford University—has received private funding. Under traditional funding-based definitions, this study can be considered commercial. However, the Arc Institute focuses on “high-risk, high-reward” science, with the goal of advancing public health. This illustrates that private funding alone does not render research commercial. Similarly, a start-up may conduct one-off research to explore its market potential. Although it is profit-driven, research should not be subject to special regulations for commercial research if it is a single activity and not the primary source of revenue. Although profit-driven purposes are important, research that does not immediately yield financial gains may nevertheless facilitate commercial objectives. For example, a health product company may conduct functional claims research that supports marketing and boosts product sales without generating direct revenue. This is considered a commercial research.
Commercial Health Data Research Deserves Special Regulation
Empirical evidence has confirmed that commercial interests may pose significant risks to the quality and integrity of scientific research. 23 Such risks include practices such as skewing the publication record, data manipulation, research misconduct, selective data presentation, and inappropriate authorship. 24 Due to the non-competing and reusable nature, health data may exacerbate this paradox. Compared to the vast economic benefits already generated, the social benefits of commercial scientific research are still lagging. 25 This study argues that tailored regulation of commercial scientific research is warranted.
Big Data Amplifies Undue Commercial Influence in Research
While all commercial activities inherently pursue economic profits, commercial scientific research often intersects with public interest objectives. This overlap creates opportunities for private entities to frame profit-oriented initiatives as beneficial to the public, which could result in the misuse of scientific research exemptions. Compared with traditional biomedical research, the non-competitive nature of health data, the “black-box” of data processing, low marginal cost, and high reusability across contexts collectively heighten the vulnerability of the research process to commercial influence.
Conventional biomedical research incurs high costs. Conversely, once collected, health data can be reused across multiple studies by different commercial entities; this could reduce the marginal cost of reuse and makes it easier for companies to participate in scientific research.
Besides, big data has a magnifying-glass effect: when the underlying sampling model is flawed, a large sample size can amplify rather than eliminate bias. For instance, during the early stages of the 2013 influenza outbreak in the United States, analyses based on Internet search queries significantly overestimated the peak of flu activity compared with traditional public health surveillance. 26
Furthermore, health data research has a black-box nature, undermining both the transparency of the research process and the interpretability of its results. This makes tactics of unlawful profiteering covert and difficult to identify, posing significant challenges to external oversight and critical scrutiny. 2
At the same time, intellectual property can turn the outcomes of scientific research in the public interest into private assets; data accelerates this process. Consequently, the scientific community will decline and the collaborative innovation capacity of the EHDS will be undermined.
Regulatory Vacuum in Health Data Research
Existing legislation has paid little attention to the specific challenges posed by health data research. Thus, health data research often falls through the cracks in the existing oversight regimes. Under the GDPR, anonymized data fall outside the scope of data protection obligations, whereas scientific research is granted exemptions that may limit its oversight.
Ethical reviews play a crucial role in scientific research. Article 67 of the EHDS states that if an applicant intends to access personal electronic health data in a pseudonymised format, an ethical assessment may be required in accordance with national law. Core ethical principles are widely agreed upon; however, case-specific confidential reviews hinder uniform oversight across the EU Member States.
Under the CIOMS International Ethical Guidelines, studies using anonymized or properly coded health records may be exempt from ethical review. 27 Many EU members follow this approach. However, anonymization is not a panacea. Although this prevents the identification of specific individuals, it does not eliminate group-level ethical risks, rendering entire communities vulnerable to harm through negative labeling, stereotyping, or biased algorithmic outcomes. For example, pulse oximeters estimate blood-oxygen saturation by emitting infrared light through the skin. However, pulse oximetry measurements may be less accurate in patients with dark skin pigmentation, which may increase the risk of undetected hypoxemia. 28
Commercial Interests Undermining Public Trust
Many surveys have indicated that public attitudes toward the use of health data are significantly influenced by commercial interests. For example, a survey in Germany established that less than 20% of participants supported consent-free data donation for private medical research (66.4%) compared to publicly funded research (85.1%). 29 Similarly, the public displayed less approval for data access by commercial entities in Canada. 30
Variations in Opt-Out Rights for Scientific Research in the European Union
Although scientific research constitutes a legal basis for processing health data, it does not always take precedence. 31 For example, the Dutch Civil Code allows medical data to be used for scientific research only when obtaining consent is impossible or unreasonable. 32 In other words, EU Member States apply data subjects’ opt-out rights inconsistently in the context of scientific research. These differences are exacerbated by the inevitable influence of commercial interests on subjects’ exercise of their opt-out rights. Consequently, reconsidering if the opt-out scheme should be applied differently in commercial and noncommercial research is necessary.
Inconsistent Rules Intensify the European Health Data Space Fragmentation
The divergent attitudes and regulations regarding commercial scientific research pose significant obstacles to the cross-border flow of health data. Particularly, the varying distinctions between “commercial research” and “noncommercial research” create inherent barriers during the ethical review of clinical studies. In jurisdictions in which research categories and specific restrictions are not clearly defined, the absence of detailed provisions within the review processes may result in differential treatment and regulatory arbitrage. Furthermore, certain countries implement specific regulations on sensitive data types, such as genetic data, which often prohibit their use for commercial scientific research purposes. These legislative constraints can restrict access to such data for researchers from other countries, resulting in unidirectional data flow within these fields. These limitations undermine data sharing within the EHDS.
Legislative Recommendations: Special Rules for Commercial Research
Commercial scientific research requires special attention. Thus, this study proposes the following suggestions for the EHDS.
Rules for Data Access in Commercial Scientific Research
Mandatory Consideration
To prevent abuse of research exemptions, commercial scientific research should be subject to mandatory compensation. Article 62 of the EHDS allows optional fees for health data access. When providing electronic health data for secondary use, access bodies and data holders may primarily charge for covering of data collection and storage expenses. Within the health data space, such data can be understood as a form of commons in which each actor seeks to maximize their individual benefits. Since researchers often bear little cost for accessing or using such data, the resource may be subject to overuse. 33 It is recommended that access to health data for commercial scientific research be subject to compulsory consideration, reflecting both cost and reasonable market value. This is because data hold substantial economic value for the marketplace. Accordingly, the fee should reflect the market dynamics and the value generated. Mandatory consideration requirements may serve as a credible signal, 34 helping distinguish researchers genuinely committed to advancing scientific knowledge from those who merely exploit the label of research as a façade for pursuing commercial gains.
Mandatory consideration does not deviate from the legislative intent, which aims to remove barriers to data flow and promote data utilization. Article 62 acknowledges that data holders should be compensated for the costs incurred in data collection, and further stipulates that certain entities may have their fees reduced proportionately to their size or budget. 35 Since proportionate fees are recognized, commercial scientific researchers should likewise bear data access costs in proportion to the substantial commercial benefits they may derive.
Mandatory Data Dividends
Compared to the vast economic benefits already generated, the social benefits of commercial scientific research are still lagging. 25 Due to the communal nature of scientific research, its outcomes must benefit everyone. Thus, mandatory data dividends are recommended to ensure fair profit-sharing and counterbalance the influence of commercial interests on scientific research. Although there is no consensus on the concept of data dividends, they generally refer to the idea of allowing data subjects to share the profits generated from data processing. 36 Data dividends can help correct the inequitable allocation of health resources caused by funding biases. However, empirical evidence suggests that heavy tax burdens hinder innovation. 37 Similarly, limited financial capacity makes smaller institutions vulnerable to the impact of high dividends on innovation. Thus, a progressive, tiered “data dividend” should be implemented, with higher rates applied to larger payouts.
The design of data dividends has been considered along 6 dimensions: funding source, implementer, tasks, time period, disbursement, and observation and valuation. 36 In commercial scientific research, health data dividends can be distributed at the individual, collective, or public level.
At the individual level, data subjects should benefit from priority access and discounted or free health services, including health monitoring, management programs, education, and incentive schemes. For homogeneous and interconnected data, benefit sharing can be achieved at the group level. For example, patients with the same disease may differ in genetic profiles, physical conditions, and lifestyles; although their data are not identical, associative analyses can collectively advance diagnosis and drug development, generating substantial benefits. Data flow across the EU cannot be realized without a robust infrastructure, which requires continuous funding to ensure its long-term functionality. At the public level, a data dividend tax can be established with provisions directing a portion of the revenue to the EHDS infrastructure.
Declaration of Conflict of Interest
Although the declaration of conflicts of interest has been widely adopted in ethical guidelines, such as the International Ethical Guidelines for Health-related Research Involving Humans, disclosure of interests that may affect the research should be submitted to a research ethics committee. 27 However, some research may be exempt from ethical review according to the EHDS and national law. Research ethics committees are often affiliated with data controllers 38 and follow an internal review model that may be influenced by institutional interests. Given the potential for funding bias in research, it is recommended that the health data access body—as a third party—review the declaration of interest. Currently, data access applications under the EHDS primarily focus on describing the type of data requested and its intended use. 39 The sources of data and any related interests or conflicts of interest must be disclosed in detail. Such related interests should not be limited to funding sources but should be interpreted broadly to include financial, personal, and professional interests that may affect the impartiality of the research review or conduct. If a study violates ethical or legal requirements owing to undisclosed conflicts of interest, it may be barred from submitting future access requests.
Mandatory Ethical Review by Health Data Access Body
Ethical reviews play a crucial role in scientific research. However, as noted above, studies involving anonymized health data often fall outside the scope of the existing ethical review frameworks. It has been proposed that commercial scientific research should not be exempt from ethical oversight. Such a requirement does not stigmatize commercial research; rather, it represents a legitimate differentiation based on varying levels of risk. Commercial scientific research is inherently exposed to higher ethical risks owing to the structural tension between commercial interests and the norms of scientific inquiry. The influence of commercial incentives objectively increases the likelihood of ethical risks; this does not imply that commercial scientific research is inherently unethical; rather, it warrants proportionate safeguards. Moreover, public skepticism toward commercial scientific research stems mainly from weak oversight. Stricter ethical reviews can enhance trust and reduce stigmatization.
Further, in response to the increasing difficulty of oversight caused by the “black box” nature of algorithms, embedded ethical review is recommended, which refers to ethicists and researchers together addressing ethical issues through an iterative and continuous process from the outset of development. 40 It enables research to incorporate ethical requirements from the outset of algorithm design, addressing ethical risks at their source.
Finally, a shared ethical consensus regarding commercial scientific research is required. Such a consensus could help establish a relatively harmonized standard across jurisdictions, and to enhance consistency and oversight, the establishment of a centralized EU-level Data Ethics Review Board could be considered. Such a body is responsible for reviewing and supervising national ethical review decisions.
Data Subject Opt-Out in Commercial Research
Concerning the role of consent in commercial scientific research, we argue that such research should proceed only when the subjects do not explicitly object to the use of their data. More specifically, in countries where consent is prioritized, commercial scientific research should follow the same consent requirements. Conversely, in countries where scientific research takes precedence over consent, commercial scientific research may proceed based on the absence of an explicit objection from the data subjects. This approach will decrease the consent-related costs of commercial research. This approach aligns with the spirit of the GDPR and EHDS. It is grounded in the principle that public interest in scientific research takes precedence over individual autonomy, aiming to advance the greater public good while placing only restrictions on data subjects.
Rules for Providing Data Access in Commercial Research
The normative logic underlying data-sharing frameworks and trade secret protection regimes is fundamentally different. In commercial research, data sharing must be carefully balanced with the protection of trade secrets. However, uncertainty persists regarding where the boundary lies between safeguarding commercial interests and advancing scientific research goals.
To prevent such overclaiming, identifying the boundaries of trade secrets in commercial scientific research is necessary. Article 51 of the EHDS stipulates that researchers acting as data holders are required to generate electronic health data for secondary use in clinical trials. When such data contain intellectual property or trade secrets, this obligation is not waived; instead, appropriate safeguards must be implemented to ensure confidentiality. However, the EHDS does not explicitly state whether data holders are exempted from the obligation to provide data on the grounds of trade secret protection. Nevertheless, data holders may reject access requests if the safeguards proposed by the applicant are insufficient to ensure adequate protection of commercially sensitive information. Further clarification is required to determine the conditions under which health data containing trade secrets should be accessible in commercial scientific research.
The Balance Test Approach
Under the Data Act, if a data holder demonstrates that the disclosure of trade secrets, despite the application of protective measures, is highly likely to result in serious economic harm, the data holder may be denied access. 41 Should the EHDS adopt the same exemption standard for data provision as the Data Act? The Data Act primarily targets private-sector data, whereas the EHDS does not limit data utilization to the private sector and specifically focuses on health data. Consequently, there are notable differences in regulatory objectives and value orientations. The EHDS does not provide detailed provisions regarding the protection measures for trade secrets. Encryption technologies and confidentiality agreements are the most commonly used protection measures. However, data access requirements may conflict with protective measures, the primary purpose of which is to preserve information confidentiality. Additionally, encryption technologies may undermine the research value of the data, that is, while data with genuine research value remain inaccessible, only data without research value can be disclosed. It is recommended that the EHDS apply a balancing test when considering such exemptions to ensure that commercial interests are appropriately weighed against the advancement of scientific research. Furthermore, “all necessary measures” should be understood as all appropriate safeguards, not sufficient ones.
The balance test can be conducted using the factors presented in Figure 4, including accessibility, scientific criticality, substitutability, public health significance, and commercial value. Accessibility refers to the practical feasibility of obtaining the relevant data, including the effort and cost involved in acquisition, the scarcity of available samples, the range of collection channels, and the technical complexity of the collection process. Scientific criticality captures the indispensability of the data for the research, such as the extent to which the study depends on it and whether it constitutes core variables or unique empirical findings necessary for advancing the field. Substitutability assesses the ease with which the target data can be replaced, taking into account the availability, relevance, quality, completeness, and accessibility of potential substitute datasets. Public health significance evaluates the broader societal relevance of the research, including the urgency and importance of the problem addressed and its relation to rare diseases, severe conditions, or public health emergencies. Finally, commercial value concerns the financial stakes for the data holder, particularly the potential economic losses, competitive disadvantages, or significant commercial harm that could result from disclosure or third-party use.

Evaluation dimensions for the balance test.
In summary, if the data are easily accessible, highly substitutable, of low research value, and disclosure causes significant economic harm to the data holder, the holder may refuse the access request. However, if the data are non-substitutable, of high scientific importance, and qualify as essential data for other research, access should be granted. Although doing so entails significant economic harm, this study examines this issue using a phased approach. Commercial scientific research can be divided into 3 distinct phases: data collection, processing, and conclusion. Health data can be categorized into 3 types: raw data, processed data, and research results.
Raw Data
The technical inputs involved in the raw data collection are relatively modest. Thus, some argue that it is less likely to qualify as a trade secret. 42 Additionally, raw data typically have broad applicability and can support a wide range of medical research purposes, making them less likely to qualify as a trade secret. Therefore, during the collection stage, balance tests should focus primarily on accessibility and substitutability. For example, in Sasqua Group, Inc. v. Courtney, Sasqua collected extensive information about its clients, including contact details, personal information, recruitment preferences, records of previous collaborations, resumes, and other data and claimed that these constituted trade secrets. The court ruled that the client list did not qualify as a trade secret because the related information was readily accessible through social media and other public channels. 43 However, where the collection of data reflects a unique design and considerable endeavor, such data may be of secret and commercial value. For example, by collaborating with hundreds of thousands of platforms and developing diagnostic criteria for a disease, a company can collect large-scale data on how different population groups respond to a particular drug.
In principle, the raw data should be made accessible. However, the data holder may be entitled to request appropriate compensation or impose reasonable conditions such as restricting the applicant from conducting identical or substantially similar research within a specified period.
Processed Data
Processed data often have a significant commercial value. Access must be granted when health data are essential. This follows the essential facilities doctrine: a company controlling infrastructure crucial for competition cannot deny access without justification. 44 Zachary Abrahamson defines essential data according to 5 factors. First, the data holder refuses to provide access to the data. Second, competition cannot occur effectively without data access. Third, it is difficult to duplicate the data. Fourth, a feasible method exists for sharing the data. Fifth, the data holder must have a monopoly power over the antitrust market. 45
EHDS research exemptions aim to promote scientific research rather than address competition issues. In commercial research, essential data should be determined by research needs rather than by the data holder’s market power, with an emphasis on data usability. Health data may be considered essential if they can be reliably reused in future studies, are critical to work, or are closely related to significant public health interests. For example, Chylomicron Retention Disease is a rare disorder of fat absorption with only approximately 40 cases reported worldwide. 46 Each individual case is highly valuable for the research. To illustrate an extreme scenario, assume that all 40 known cases are controlled by a single data holder who refuses to share information; it would be impossible for other researchers to conduct studies on the disease, effectively halting any progress toward understanding or treating it.
Research Results
Although the research results might match the criteria for trade secret protection, this kind of protection is incompatible with the publicly oriented nature of scientific research, which demands that the knowledge it generates be openly shared for the benefit of society. As previously noted, selective reporting is a common form of funding bias. Ensuring the comprehensive publication of research findings is an important measure for addressing this problem. Accordingly, all resulting data must be made available. Data holders may resort to alternative protection mechanisms such as patent law, which grants a time-limited legal monopoly in return for public disclosure. Alternatively, establishing a protection period during which the disclosure of results may be delayed is possible. However, if the data holder demonstrates that granting access would cause substantial economic harm, the data access body may exempt the holder from the obligation to provide data at its discretion.
Centralized European Union Platform to Reduce Administrative Burdens
However, stricter regulations on commercial scientific research may increase administrative burden. To address this, we propose a centralized EU electronic system for commercial health research, where researchers self-declare their operational and commercial status, ethical review outcomes are mutually recognized, and data subjects’ objections are recorded. Such a platform would streamline data access and reduce administrative burden across Member States.
Conclusions
This study collected and analyzed the attitudes and approaches of EU Member States toward regulating commercial scientific research. The EHDS and existing EU legislation did not adequately address the specific challenges posed by commercial scientific research. To ensure the EHDS functions consistently across Member States, and to enable commercial scientific research to foster sustainable innovation while preventing misuse, we propose stricter, harmonized rules to govern such research. However, as the EHDS has only recently been implemented, our study relied on legal texts and lacked empirical evidence. Future research should strengthen the empirical investigations of commercial scientific research under the EHDS.
Footnotes
Acknowledgements
We extend our gratitude to Professor Thierry Vansweevelt (University of Antwerp), and the Health Law and Policy Research Institute of East China University of Political Science and Law. We also wish to thank the Polish Office for Personal Data Protection, Swedish Authority for Privacy Protection, The Office for Personal Data Protection of the Slovak Republic, Health Data Agency of Belgium, Centre for Diseases Prevention and Control of Latvia and General Secretariat of the Council of the European Union for their responses to our inquiry emails.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
I have confirmed the above published content, understand the scope and impact of its public release, and agree to publish it in accordance with the requirements of the journal.
Author Contributions
First Author, Xuxin Yao: Drafted the manuscript (excluding the Results). Corresponding Author, Hongjie Man: Conceived the study and provided supervisory guidance. Second Author, Zheyuan Sun: Collected EU regulatory legislation, drafted the Results.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Visiting Scholar Funding Program for PhD Students at East China University of Political Science and Law.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
