Abstract
Advanced therapy medicinal products (ATMPs) are ground-breaking genetic and cell/tissue-based therapies with the potential to treat and even cure myriad health conditions. However, they have faced multiple challenges in decision making with relatively low success in achieving reimbursement. The aim of this study was to review the published preference-based research on ATMPs to identify value attributes as a starting point in a research workstream aimed at understanding the true value of these innovative therapies. A literature review was used to identify studies across MedLine, Embase, Allied and Complementary Medicine Database, Global Health, ProQuest Central, Social Sciences Premium Collection, Applied Social Sciences Index and Abstracts, Econ Lit, Web of Science and SCOPUS from inception to 3rd March 2024. Search terms for genetic and cell/tissue-based therapies were conducted separately. Studies were included if they were a peer-reviewed primary research study or systematic literature review using stated preference measures to elicit preferences for ATMPs. Thirteen studies were included with 8 pertaining to genetic therapies and 5 to cell/tissue therapies. Attributes of value were similar across the studies and ATMP categories. Attributes were grouped into the following for genetic therapies: clinical benefits, uncertainty, risk, treatment burden, and quality of life; for cell/tissue therapies clinical benefit, uncertainty, risk and treatment burden. There is limited research on the preferences for ATMPs; but these findings suggest that the value attributes of ATMPs are consistent across gene and cell/tissue therapies with clinical benefit, uncertainty, risk, treatment burden, and quality of life being important.
Highlights
● This literature review aimed to identify the published literature on preference studies of ATMPs to understand the value attributes of ATMPs and how they are quantified, and to contribute to research to determine whether current HTA methods capture the relevant attributes of value.
● This review has found that ‘clinical benefit’, ‘uncertainty/risk’, and ‘treatment burden’, and ‘quality of life’ are important value attributes to stakeholders – although this conclusion must account for the limited published evidence in this space in that the findings will require validation.
● Although the studies identified suggest that value attributes between the categories of ATMP are broadly similar, in light of the relatively low number of studies identified, further research is needed to explore this and to confirm any similarities and dissimilarities to ensure fair assessment of these therapies and to understand whether current HTA methods suffice.
● Additional research aimed at understanding the broader value of ATMPs and the attributes of value associated with these innovative therapies is necessary. This should include additional stakeholders beyond those of clinicians and patients to ensure all relevant value attributes are considered in HTA to ensure appropriate value assessment of ATMPs.
Introduction
Advanced therapy medicinal products (ATMPs) are ground-breaking treatment options which include gene therapies, somatic cell therapies or tissue engineered products with the potential to cure a myriad of health conditions.1,2 These range from rare diseases with limited treatment options to chronic conditions such as in oncology. 3 To date, 21 ATMPs have been approved by the European Medicines Agency (EMA) 4 while the United States (US) Food and Drug Administration (FDA) has approved 46. 5 However, despite their curative potential, their adoption into mainstream healthcare has been limited with pricing and reimbursement challenges being key factors. A reason for this is that there is uncertainty surrounding the value that these innovative medicines bring to patients and society and whether they warrant the relatively high prices.6 -10
Many countries undertake an assessment of healthcare interventions, including ATMPs, through a process known as health technology assessment (HTA) which is used to inform decision making in healthcare. 11 This interdisciplinary process assesses the clinical, economic, legal and societal impacts of an intervention as well as its price. 12 In many countries, the outcome of this assessment determines patient access to health interventions with ATMPs having a relatively low success rate in achieving full reimbursement. 13
One of the reasons for this may be that current HTA methods are not suited for value assessment of ATMPs. 14 As such, there may be a disconnect between existing value assessment frameworks which HTA agencies employ and the potential value ATMPs bring to patients and society.
Thus, understanding the attributes of value of ATMPs from a multi-stakeholder perspective may provide information which would help economists to understand whether current HTA processes capture the full value of ATMPs.
This literature review aimed to identify the published literature on preference studies of ATMPs to understand the value attributes of ATMPs and how they are quantified, and to contribute to research to determine whether current HTA methods capture the relevant attributes of value. This study is part of a larger research workstream with its findings intended to contribute to understanding the value these innovative therapies may bring to patients and wider society. The findings will help to inform whether a review of HTA methodologies specific to ATMPs is needed.
Methods
This literature review was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement to ensure study robustness. 15 The definition of advanced therapy medicinal products applied was that of the European Medicines Agency (EMA). 16 In summary, ATMPs are classed into 3 categories: ‘gene therapy medicines which contain genes that lead to a therapeutic, prophylactic or diagnostic effect’, ‘somatic cell therapy medicines contain cells or tissues that have been manipulated to change their biological characteristics or cells or tissues not intended to be used for the same essential functions in the body’, and ‘tissue-engineered medicines which contain cells or tissues that have been modified so they can be used to repair, regenerate or replace human tissue’. Combined ATMPs consisting of 1 of the above 3 categories in combination with a medical device is the final category of ATMP recognised by the EMA. 16
Review Aims
The aims of this literature review were twofold:
To identify the available research on quantitative stated preference studies for ATMPs.
To identify the available research on qualitative development studies which informed quantitative preference studies (ie, these were studies which informed the development of stated preference studies but contained no quantitative assessment themselves).
This review included quantitative preference studies as this enables an understanding of the quantification of value attributes while qualitative studies provide evidence on potential attributes of value.
The review was not limited to any specific stakeholder, disease area or geography.
The review intended to narratively synthesise the findings of the identified studies with findings separated in line with the type of study – quantitative or qualitative – regardless of methodology used to study preferences or qualitative methodology applied, respectively. This was done in order to amalgamate the findings from the studies.
Search Strategy
The searches were conducted separately for genetic therapies and cell- and tissue-based therapies. The search strings contained a combination of MeSH terms, title/abstract and free text terms and were designed to be comprehensive to ensure that all relevant studies would be identified. For genetic therapies, terms specific to genetic therapies were used: ‘gene therapy’, ‘personalised medicine’, ‘genetic’, and ‘genomic’. For cell- and tissue-based therapies, terms specific to these therapies included: ‘stem cells’, ‘tissue engineering’, and ‘regeneration’. All search strategies contained terms related to preference elicitation such as the methodologies used to elicit preferences (time trade-off, willingness-to-pay etc.) and methods in which they might be incorporated for example, economic evaluation. The full search terms used in each database as given below are provided in Appendix 1.
The search for relevant literature was conducted in 2021 and 2024 as an update to the original search of 2021 was required due to a delay in the research, as described below.
Initial Search
MedLine, Embase, Allied and Complementary Medicine Database, ProQuest Central, Social Sciences Premium Collection, Applied Social Sciences Index and Abstracts, Econ Lit, Web of Science, and SCOPUS from inception to 28th May 2021 were searched.
Updated Search
The search for MedLine, Embase, Allied and Complementary Medicine Database, ProQuest Central, Social Sciences Premium Collection, Applied Social Sciences Index and Abstracts, Econ Lit, Web of Science, and SCOPUS was re-run to include publications listed in the database from 1st January 2021 to 3rd March 2024. It was noted that there had been a change in the thesauri for Allied and Complementary Medicine Database and Applied Social Sciences Index and Abstracts thus requiring updated search terms.
The searches were also run in Global Health. For Global Health, it was found that when running the search from inception to March 2024, with the updated search terms the same results as previously identified were not present. This led to Global Health only being included in the updated search covering inception to March 2024.
For any records identified in the overlapping period (ie, 1st January 2021 and 28th May 2021), the records were included in the initial search. That is, they were considered duplicates and excluded.
Selection Criteria
All studies were screened against the eligibility criteria as given in Table 1.
PICOS Criteria.

The Population, Intervention, Comparison, Outcomes, and Study design (PICOS) criteria as given in Table 1 were applied to both treatment types to identify relevant studies. In summary, all peer-reviewed primary research study or literature review using stated preference measures to elicit preferences for advanced therapy medicinal products or qualitative studies informing quantitative preference studies were included. Studies were limited to the English language.
Screening and Data Extraction
Due to the high volume of records resulting from the initial search, 1 investigator (NFe) initially screened all titles for eligibility. This has been shown to have no impact on the findings of reviews. 17 All eligible abstracts were then screened by 2 investigators (NFe and WA) with the resulting eligible full texts screened by NFe.
For the updated search, as there were fewer records for screening, NFe screened the titles and abstracts simultaneously followed by all eligible full texts. Texts were screened against the PICOS criteria (Table 1) with articles not meeting these criteria being excluded. All articles meeting the criteria at full-text review were included in a pre-developed extraction grid used to extract relevant information: author, year, study type, geographical location/setting, sampling and sample size, disease area, methodology, analysis methods, attributes, preferences elicited.
Hand-searching
Reference lists of identified relevant systematic literature reviews were hand-searched with relevant references pearl-grown, until no new records were identified. Additionally, we searched Google Scholar using terms synonymous with ‘preferences for advanced therapies’ including ‘preferences for gene therapy/cell therapy/tissue therapy’ to identify relevant studies which may not have been indexed in the databased included in the search strategy.
Further, the International Society for Pharmacoeconomics and Outcomes Research conference proceedings were searched for studies not already identified in the database search or the hand-searching.
Quality Review of Included Studies
There are limited quality assessment tools available for stated preference studies with the exception of a checklist developed for conjoint analysis studies. 18 The results of a summary of compliance against this pre-defined checklist in light of limited methods for assessing preference studies is provided in Appendix 2.
Results
Search Results – Genetic Therapies
The initial search identified a total of 6010 records; 391 duplicates were removed. Due to the volume of studies identified, an initial title screen was conducted by NFe to improve the efficiency of the review with no loss of robustness. 17 About 5619 titles were screened leading to the exclusion of 5577 due to lack of relevance. Subsequently, 42 abstracts were screened leading to the exclusion of 15. A total of 27 full texts were then screened. Twenty-five were excluded giving 2 records for data extraction and evidence synthesis.
From the March 2024 updates, 704 records were identified with 43 duplicates removed. This resulted in 661 titles and abstracts being screened. For the updated search, it was not necessary to assess titles only as a first step due to the lower number of identified studies. Six hundred forty-eight were excluded with 13 eligible for full text screening. Thirteen full-text articles were excluded at full-text screening with no records included for data extraction.
Overall, from the initial and updated database-based searches, 2 records were included in the data extraction and evidence synthesis.
A further 6 records were identified from the hand-search. The authors sought to understand why these 6 records were not identified in the database searches. Following investigation, it was found that the 6 studies were not indexed in the databases at the time the searches were run.
The PRISMA for genetic therapies is presented in Figure 1.

PRISMA: genetic therapies.
Search Results – Cell and Tissue-Based Therapies
The initial database searches for cell and tissue-based therapies gave a total of 8846 records. Three hundred six of these were duplicates which were removed. Similar to the original search for genetic therapies, the titles were initially screened by NFe. 17 Following removal of duplicates, 8540 titles were screeded with 63 proceeding into abstract sifting meaning that 8477 were excluded due to not meeting the PICOS inclusion criteria. Of the 63 abstracts screened, 22 were excluded with 41 entering full-text screening. Forty were excluded resulting in 1 record included for data extraction and evidence synthesis.
From the March 2024 database updates, similar to that of genetic therapies, an initial title screen was not conducted as 1838 records were identified. Fifteen of these were duplicates resulting in 1823 records eligible for title and abstract screening. This resulted in 20 full texts being screened as 1803 records were excluded for ineligibility at the title and abstract screening stage. Nineteen full-text articles were excluded. One record was included for data extraction.
Overall, from the database-based searches, 2 records pertaining to cell and tissue-based therapies were identified.
A further 3 studies were identified through hand-searching. Similarly, the authors investigated why more studies were identified from the hand-search compared to the database searches. The 3 studies identified from the hand-search were not indexed in the databases at the time the searches were run.
The PRISMA for cell and tissue-based therapies is presented in Figure 2.
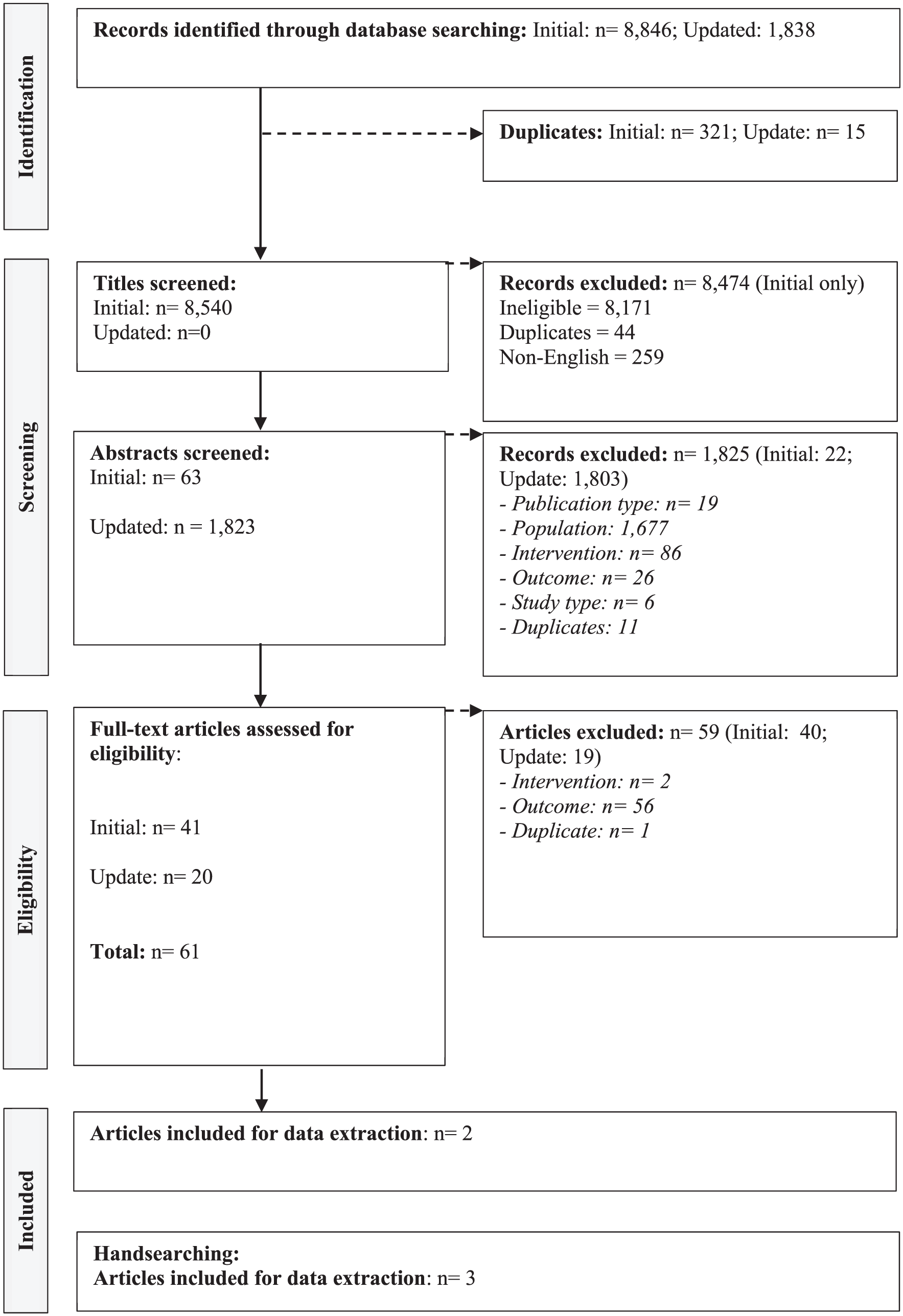
PRISMA: cell and tissue-based therapies.
Summary of the Evidence
The 2 ATMP category-specific searches and hand-searches identified a total of 13 relevant records. Eight of these related to studies for genetic therapies19 -26 and 5 to studies for cell and tissue-based therapies.27 -31 For genetic therapies, 6 of the 8 studies19 -22,25,26 examined preferences or conducted qualitative research ahead of preference studies for gene therapies in haemophilia with the remaining 2 focussing on sickle cell disease 23 and Duchenne muscular dystrophy, 24 respectively. For cell and tissue-based therapies, all identified studies were focussed on oncology with 4 explicitly studying chimeric antigen receptor T-cell (CAR T) therapies in B-cell lymphoma.28 -31
There was a limited geographical spread with studies being conducted in either Europe or the United States only. Stakeholders considered in the identified studies were limited to clinicians or patients.
The authors would have liked to have conducted a quality assessment of the included studies, but this was not possible due to the lack of assessment tools for stated preference studies. The DCEs were assessed against a conjoint analysis checklist 18 – this is presented in Appendix 2. Most of the discrete choice experiments were largely compliant but with a lack of description on the methods around how the preferences were elicited.
The results are presented in line with the aims of this literature review. The authors defined categories for value attributes (‘treatment attribute themes’) following data extraction to enable cross-comparison between studies so that findings could be synthesised. The study findings were amalgamated for 2 reasons. Firstly, the low number of studies identified and limited types of preference study conducted precluded the authors from synthesising methodologies separately; secondly, the ultimate aim of the review was to understand the value attributes of importance to stakeholders of ATMPs. This meant that study findings needed to be amalgamated.
What is the Available Research on Quantitative Stated Preference Studies for ATMPs?
A summary of the characteristics and findings from the quantitative studies is presented in Table 2.
Characteristics and Findings of Quantitative Studies.

Note. ABR = Annual bleeding rate; CAR T = Chimeric antigen receptor therapy.
Categories defined by author so that studies could be cross-compared.
Regarding the genetic therapy studies, 3 of the studies were discrete choice experiments21,23,25 with the final conducting a threshold analysis. 22 Three of the studies elicited preferences for gene therapy in haemophilia21,25,26 and one in sickle cell disease. 23
In haemophilia, Witkop et al 21 conducted a discrete choice experiment with adult haemophilia patients in the United States. The analysis found that patients attached the greatest importance to ‘effect on overall bleeding rate’ followed by ‘dose frequency and durability’. Similarly, Sun et al 25 presented patients with 14 choice cards based on attributes developed through targeted literature reviews and telephone interviews. ‘Administration-related measures’ (‘route and frequency of administration’, ‘out-of-pocket cost’, ‘place of administration’) were the most important class of attribute with ‘risk of serious side effects’ the least important. Overall, these studies indicate that in haemophilia, patients value ‘clinical benefits’, ‘treatment burden’, ‘uncertainty/risk’, and ‘quality of life’.
Finally, in haemophilia, van Overbeeke et al 22 conducted a threshold analysis in Belgium with 117 patients to determine the point at which patients would prefer a gene therapy over the current standard of care (SoC). These findings are not entirely relevant to this literature review as they do not identify relative importance, but they do suggest that patients value ‘clinical benefits’, ‘treatment burden’, ‘quality of life’, and ‘uncertainty/risk’.
In sickle cell disease, Gonzalez Sepulveda et al 23 aimed to quantify benefit/risk trade-offs in adult patients and parents/caregivers of paediatric patients. The analysis found that patients preferred improved efficacy, including greater improvement in life expectancy and no sickle cell disease symptoms thus suggesting that ‘clinical benefits’, and ‘uncertainty/risk’ are important to patients and their caregivers.
Regarding the cell/tissue-based therapies, all studies examined preferences in oncology with 2 of the studies utilising a time trade-off approach27,28 while 3 conducted a discrete choice experiment.29 -31
Leuthold et al 27 studied patient preferences using time trade-off among 184 patients at a Switzerland-based hospital who had all received stem cell transplants as part of their oncology treatment. The study authors found that patients preferred a survival length of at least 5 or 10 years and a cure rate of 50% to legitimate haematopoietic stem cell transplant. Interestingly, Howell et al 28 examined the impact of adverse events of CAR T-cell therapy in large B-cell lymphoma with the valuation using time trade-off from a UK societal perspective. Unsurprisingly, preference was for a health state with no adverse events with the health states with the highest grades of adverse events having the lowest preference. This indicates that with increased severity of adverse events from CAR T-cell therapy, respondents would prefer to trade time in perfect health to avoid living in these burdensome health states. These 2 studies which employed similar methods indicate that ‘clinical benefits’ (including the potential for a cure) and ‘uncertainty/risk’ are key attribute themes.
Similar findings were reported by Boeri et al, 29 Birch et al, 30 and Liu et al 31 who all conducted discrete choice experiments in diffuse large B-cell lymphoma to understand the trade-offs between risks and benefits of CAR T-cell therapies. The latter 2 studies sought the preferences of patients and found that treatment success was the most important attribute.30,31 Specifically, patients in both the United States and Europe placed the highest importance on 1-year survival followed by reducing the risk of serious CRS or neurological events. This was followed by 3-year survival for patients in the United States and time to return to the pre-treatment level of functioning in Europe. 30 This indicates that preferences differ by geography albeit patients in both regions would require at least a 13.2% increase in probability of 1-year survival to accept a 13% chance of serious neurological event or 28% chance of cytokine release syndrome (CRS). 30 This is supported by Liu et al 31 who found that patients would accept a 69.5% increase in risk of serious infections to increase chance of treatment success from 5% to 45%. There was no evidence of differences between patients and physicians with Boeri et al 29 highlighting that physicians also preferred improved outcomes, reduced adverse events and less time to infusion. Additionally, physicians were willing to accept increases in adverse events for improved outcomes. 29
Overall, these studies highlight that ‘clinical benefit’, ‘uncertainty/risk’, ‘quality of life’, and ‘treatment burden’ are important to stakeholders.
What is the Available Research on Qualitative Development Studies Which Informed Quantitative Preference Studies?
Table 3 summarises the characteristics and findings of the qualitative studies. The 4 identified studies were limited to genetic therapies. Three of these were focussed on haemophilia19,20,26 and were used to inform discrete choice experiments. It is possible these are linked studies, but different results are presented meaning that the studies are described separately.
Characteristics and Findings of Qualitative studies.

Categories defined by author so that studies could be cross-compared.
This study does not make reference to a subsequent preference-based study but was included as the title suggests findings may be used in a quantitative study.
Morgan et al 19 and Woollacott et al 26 employed similar methods and techniques to identify values of attribute for gene therapy in haemophilia including semi-structured interviews with around 20 patients and clinicians each. Ranking exercises were used to determine the relative importance of the attributes. Across the 2 studies the following attribute themes were found to be important: ‘therapeutic option’, ‘clinical benefit’, ‘uncertainty/risk’, ‘treatment burden’, ‘informed decision making’, and ‘quality of life’. These findings are supported by Munetsi et al, 20 in a likely linked study which described a literature review and interviews to identify value attributes in haemophilia.
Finally, Landrum Peay et al 24 described qualitative research with parents of paediatric patients and adult patients to identify factors influencing treatment choice in Duchenne muscular dystrophy. A thematic analysis indicated that participants place value on treatments which impact on the skeletal muscle, cardiac and pulmonary functioning with the value increasing with disease progression. Risk tolerance was highest in the later disease stages. Thus, ‘clinical benefits’ and ‘uncertainty/risk’ are important in Duchenne with an indication that patient age impacts value attributes.
Comparison of Value Attributes for Gene Therapies and Cell and Tissue-Based Therapies
Figure 3 presents a comparison of the value attribute themes identified in the published quantitative studies. Eight studies in total (4 genetic therapies and 4 cell and tissue-based therapies) identified that clinical benefits are important to patients and clinicians.21 -23,25,27,29 -31 Uncertainty/risk was also important for both categories of ATMP with 3 and 4 studies of genetic and cell/tissue-based therapies, respectively, highlighting that uncertainty/risk play a role in healthcare decision making.21,23,25,28 -31 Similarly, the burden to patients associated with receiving ATMP treatment was described in an equal number of studies (2 studies each) examining preferences for cell and tissue-based therapies and genetic therapies.25,29 -31 However, quality of life was only noted in genetic therapy studies (n = 2).21,22

Value attributes by ATMP category.
Discussion
This literature review aimed to identify the published literature on preference studies of ATMPs to understand the value attributes of ATMPs and how they are quantified, and to contribute to research to determine whether current HTA methods capture the relevant attributes of value. The review forms part of a larger body of research being conducted to review and inform the development of HTA processes for ATMPs.
This literature review identified 13 quantitative and qualitative studies in total with 8 focussing on genetic therapies19 -26 and 5 on cell and tissue-based therapies.27 -31 The quantitative genetic therapy studies were focussed on haemophilia (3 studies; 75%)21,22,25 and sickle cell disease (1 study; 25%) 23 and were limited to discrete choice experiments21,23,25 and a threshold analysis. 22 These studies identified that ‘clinical benefits’, ‘treatment burden’, ‘uncertainty/risk’, ‘quality of life’ were important considerations for stakeholders. The quantitative cell and tissue-based therapy studies were all focussed on oncology.28 -31 Discrete choice experiments were the preference measure of choice in 3 of the studies29 -31 with 2 utilising a time trade-off approach.27,28 These studies identified that ‘clinical benefit’ (including the potential for a cure), ‘uncertainty/risk’, and ‘treatment burden’ are the most important considerations for stakeholders.27,29 -31 A comparison between the 2 categories of ATMP found that attributes are widely similar between the categories of ATMP but there is an indication that there are differences between them, namely in the importance of quality of life; however, this may be as a result of the small number of studies identified and with the cell and tissue-based therapy studies focussing on understanding the trade-offs between clinical benefits and adverse events.28 -31
In terms of the qualitative studies identified, stakeholders expressed that ‘clinical benefit’, ‘quality of life’, ‘uncertainty/risk’ and ‘informed decision making’, ‘treatment burden’, and ‘therapeutic option’ are important considerations for stakeholders. These findings are largely consistent with the findings from the quantitative studies although they carry less weight in the findings of this review. ‘Informed decision making’ and ‘therapeutic option’ were not explored in the quantitative studies identified; this could be because these were not ranked as important attributes ahead of preference elicitation studies.
In summary, ‘clinical benefit’, ‘uncertainty/risk’, and ‘treatment burden’, and ‘quality of life’ were found to be important value attributes to stakeholders with suggestions that other elements such as having an additional treatment option and being informed as part of treatment decisions are also important to stakeholders.19 -31
These findings must be taken in the context of the studies identified which were of limited geographical spread, restricted to a small number of disease areas and considered a limited number of stakeholders. Specifically, only clinicians and patients were included in the determination of value attributes with studies limited to the United States and Europe.19 -31 This suggests that study authors considered these stakeholders to be the most important when considering the value of advanced therapies. Conversely, it means that other perspectives have not been considered meaning that the findings of this review are not as comprehensive as intended meaning that applicability is limited and further research is needed. The geographic bias of the identified studies also limits the generalisability of the findings to specific geographies. Finally, the focus on haemophilia, and oncology, limits the representative of the studies and their findings and suggests that further research is needed to decipher whether value attributes of advanced therapies differ by disease area.
This review suggests that current HTA methods capture attributes of value of ATMPs to stakeholders32 -34 but may not capture all relevant attributes including the potential for a cure and the value of having an alternative treatment option. There may also be additional value attributes which have not been captured in this review particularly in light of the stakeholders considered in the quantification of value being limited to clinicians and patients albeit 1 study utilised a societal perspective but the specific value attributes being valued were defined by clinicians. 28 It may be that the inclusion of additional stakeholder perspectives such as that of developers, payers, regulators, and the general public would bring to light yet more potentially relevant attributes of value of advanced therapies which are currently not being captured in HTA methods. Indeed, there is evidence that there are other value attributes14,35,36 but they have not been included in quantitative ATMP preference elicitation studies; a gap in the field that requires further research.
Although the studies identified as part of this review suggest that value attributes between the categories of ATMP are broadly similar, in light of the relatively low number of studies identified, further research is needed to explore this and to confirm any similarities and dissimilarities to ensure fair assessment of these therapies and to understand whether current HTA methods suffice. Decision makers should be instrumental in discussions around whether current HTA processes are sufficient to ensure that research findings are captured in their value assessment frameworks.
A key strength of this literature review is the comprehensive search strategy both in terms of the search terms and the breadth of databases searched. This ensured a thorough examination of the literature enabling all relevant studies to be identified. This thorough examination identified the lack of research in the understanding of the preferences for ATMPs. To this end, a further key strength of this study is the focus on quantitative preference studies and qualitative studies which contributed to preference studies as this enables all relevant research pertaining to the quantification of value in relation to ATMPs to be identified. Further, the consideration of non-preference-based studies would have meant a different, albeit interesting research question, something that is planned as part of subsequent research.
A drawback of the study is that the title screen for the initial searches was conducted by 1 researcher which may have led to the exclusion of relevant studies. However, the hand-searching will have limited the impact of this. An additional drawback is the lack of robust quality assessment of the included studies which is due to the lack of tools to conduct this assessment. The development of quality checklists for preference studies would help the field to develop robust methods and techniques. A further drawback of this study is that although a thorough assessment of published attributes of value was undertaken, linking these to cost to assess value for money was beyond the study’s scope.
Finally, the low number of studies identified (13 in total) makes it difficult to make firm conclusions from the findings meaning that the strongest conclusion of this study is the need for extensive further research. Further research building on the findings from this study would support the development of a value assessment framework for all ATMPs which could be used in economic evaluation of these health interventions. Indeed, this study forms part of a wider research workstream aiming to develop a value assessment framework for advanced therapies.
Conclusion
In conclusion, this review has identified the limited published evidence on preference studies in advanced therapies. It has found that ‘clinical benefit’, ‘uncertainty/risk’, and ‘treatment burden’, and ‘quality of life’ are important value attributes to stakeholders – although this conclusion must account for the limited published evidence in this space in that the findings will require validation. However, the review has also identified that there are key gaps in this research space. To ensure that methods used in HTA are relevant for ATMPs and allow their value to be demonstrated, additional research aimed at understanding their broader value and the attributes of value associated with these innovative therapies is necessary. This should include additional stakeholders beyond those of clinicians and patients to ensure all relevant value attributes are considered in HTA to ensure appropriate value assessment of ATMPs. Although there has been some work to date to expand further the attributes of value considered when health interventions are assessed, these are neither mainstream nor do they relate specifically to ATMPs. 36 There are pre-existing frameworks specifically aimed at advanced therapies although these focus on valuing potential cures and transformative treatments. 35 HTA agencies should ensure their processes are fair and appropriately assess these innovative therapies.
There is a lack of quality assessment tools for preference studies with this limited to conjoint analysis, 18 suggesting the strong need for the development of tools to help advance this space. It may be that preference elicitation methods may need to be developed which can be tailored to the complexity of advanced therapies although this will need to be confirmed. This will help to expand the research to emerging markets and diverse healthcare systems to help understand whether value attributes of advanced therapies, and their quantification, are subject to the setting as well as different disease areas. As there is expected to be a large number of advanced therapies entering the market in the next few years, addressing these gaps will need to be swift. 37
To summarise, there is limited research on the preferences for ATMPs; but these findings suggest that the value attributes of ATMPs are consistent across gene and cell/tissue therapies with clinical benefit, uncertainty, risk, treatment burden, and quality of life being important. However, additional research is needed to confirm and expand on these findings especially in light of the limited number of studies identified. Finally, in order to support the field to advance, new tools and methods will need to be developed.
Supplemental Material
sj-docx-1-inq-10.1177_00469580251390763 – Supplemental material for Preferences for Advanced Therapy Medicinal Products: Understanding the Published Literature on the Value of Innovative Health Interventions
Supplemental material, sj-docx-1-inq-10.1177_00469580251390763 for Preferences for Advanced Therapy Medicinal Products: Understanding the Published Literature on the Value of Innovative Health Interventions by N. Ferizović, P. Lorgelly, C. S. Clarke, R. M. Hunter, R. Plackett, W. Abbas and N. Freemantle in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-2-inq-10.1177_00469580251390763 – Supplemental material for Preferences for Advanced Therapy Medicinal Products: Understanding the Published Literature on the Value of Innovative Health Interventions
Supplemental material, sj-docx-2-inq-10.1177_00469580251390763 for Preferences for Advanced Therapy Medicinal Products: Understanding the Published Literature on the Value of Innovative Health Interventions by N. Ferizović, P. Lorgelly, C. S. Clarke, R. M. Hunter, R. Plackett, W. Abbas and N. Freemantle in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Author Contributions
NFe and PL conceptualised the study; NFe conducted all analyses with support from WA; NFe, PL, CC, RH, RP and NFr interpreted the data; NFe, PL, CC, RH, RP, and NFr contributed to the drafting of the manuscript. All authors reviewed and approved the manuscript for submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NFe has provided consulting support to many pharmaceutical companies through employment. RP is funded by the National Institute for Health and Care Research (NIHR) Three Schools’ Mental Health Programme (project reference MHF013) and supported by NIHR ARC North Thames. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. NFr has provided consulting support (methodological advice) to ALK, Sanofi-Aventis, and AstraZeneca. PL, CC, RH and WA have no disclosures to declare.
Data Availability Statement
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
