Abstract
While thousands of health systems have begun to implement the Age-Friendly Health System’s 4Ms Framework to improve care for older patients, an important phase of work is achieving consistent adherence to 4Ms care processes. Identifying mechanisms that may lead to higher versus lower adherence serves to guide efforts to achieve consistent, equitable adherence. Drawing from prior literature, we identified three mechanisms that may influence 4Ms adherence. We then conducted a 3-year retrospective, observational study of inpatient encounters (n = 28 833) at UCSF Health System with patients aged 65+. We used least squares regression models to assess associations between hospital encounter-level measures of 4Ms adherence and proxy measures of patient and encounter characteristics for each hypothesized mechanism along with control variables. Encounter-level adherence to the 4Ms was 65.5% (SD = 14.3%). We found support for all three mechanisms. Negative implicit biases were associated with lower adherence for patients who were obese [0.79 percentage points (PP) lower; P < .001] or on Medicaid (0.64 PP lower; P = .002). Positive implicit biases were associated with higher adherence for the oldest old (aged 85+; 2.85 PP higher; P < .001) or with reduced mobility (2.01 PP higher; P < .001). Patients with comprehensive geriatrics care contact (ACE unit and a geriatrics consult) had 5.33 PP higher adherence (P < .001). While most effects were modest in magnitude, our results suggest that both positive and negative implicit biases, as perceived by the provider, may influence the level of 4Ms adherence. Contact with comprehensive geriatrics care appeared most influential. These insights can be leveraged to develop strategies to achieve equitable delivery of 4Ms care.
The 4Ms Framework promotes delivery of comprehensive, evidence-based care to older adults, yet the complex, interprofessional nature of the underlying processes makes achieving consistent practice difficult.
Drawing on prior literature, we identify three specific mechanisms that could drive higher versus lower levels of 4Ms adherence and then empirically assess them using patient- and encounter-level measures.
Our work reveals variability in adherence to the 4Ms and identifies underlying mechanisms that could be addressed as part of ongoing implementation efforts to achieve equitable and reliable practice of the 4Ms.
Introduction
While thousands of health systems have begun to implement the Age-Friendly Health System’s 4Ms Framework designed to improve care for older patients, an important phase of work is achieving consistent adherence to 4Ms care for every older patient. Consistent adherence (also referred to as reliable practice) is challenging, particularly in the inpatient setting, because of the interprofessional nature of the care processes involved in 4Ms care, and the fact that many processes have to be repeated every shift or every 24 hours. While prior work has identified barriers to consistent 4Ms implementation at the organizational level, 1 these results do not address why a given patient encounter may feature higher or lower adherence to 4Ms care. Potential barriers span from those relevant to any new intervention (eg, limited time, limited resources) to those specific to the 4Ms (eg, how to achieve daily mobility goals using an interprofessional team). Understanding the underlying mechanisms through which higher versus lower 4Ms adherence occurs directly informs needed improvements to implementation to ensure consistent adherence as well as equitable delivery of the 4Ms for all older adults.
There are three broad mechanisms that prior literature and insights from 4Ms implementation suggest may influence the level of adherence to the 4Ms. First, given that 4Ms assessments and follow-up actions must be routinely carried out by an interprofessional care team that will likely need to use nuanced communication (including discussions about What Matters to the patient), it is possible that negative implicit biases based on characteristics of the patient that are observable to the provider may impact care and adherence. Prior work has shown that such biases among health care professionals in general result in lower adherence to care protocols and may impede patient-provider communication. 2 By extension, frontline clinicians may be less likely to adhere to 4Ms processes for patients for whom adherence is more challenging—such as those with limited English proficiency (due to communication challenges when discussing medications or care goals) or are obese (given equipment-limited mobilization challenges). Prior work has also shown that negative implicit biases by health care professionals result in patients from racial minorities and those who are on Medicaid receiving less evidence-based care,2,3 which could also manifest as lower adherence to a protocolized 4Ms implementation.
While negative implicit biases may be in play, it is also possible that positive implicit biases, also based on provider observation of patient characteristics, are a second mechanism influencing 4Ms adherence. Specifically, patients with visible signs of frailty—in particular the oldest old 4 and those with visible mobility limitations—may be more likely to receive higher levels of adherence because of perceived additional value from 4Ms care. For example, a patient who appears unstable while walking with a walker to the restroom may prompt the care team to investigate the underlying causes via 4Ms assessments of high-risk medications and possible contributions from active delirium. Lastly, because the 4Ms is an intervention requiring interprofessional care, patients who come into contact with care that is designed to cross disciplines may be more likely to have higher adherence. For example, a patient in an Acute Care for the Elderly (ACE) unit or who receives a geriatrics consult likely would get a comprehensive geriatrics assessment, 5 which is a multidimensional assessment of not only their medical problems, but also functional, social and environmental issues (which routinely and historically include assessments of the 4Ms). The findings from this type of complex assessment often initiate referrals to interprofessional disciplines, physical or occupational therapy, social workers, or pharmacists who will work together to form a comprehensive plan to support 4Ms care.
To the extent one or more of these mechanisms drives the level of 4Ms adherence, it suggests the opportunity to design strategies to achieve consistent, equitable adherence. For example, if we find evidence that contact with interprofessional care is associated with higher adherence, it suggests an imperative to focus the design of 4Ms assessment and intervention workflows to optimize expectations and expertise of each discipline by streamlining communication. There are also important insights to be gained from examining these relationships not only for the 4Ms together as a set but also by factors that may influence an individual M’s adherence. It may be that, for example, observable frailty (eg, reduced mobility) is associated with higher adherence to mobility and mentation care practices due to the more obvious ties between those Ms and improved mobility. However, given the important role medications play in addressing frailty-related risks, lower adherence to medication processes could point to an opportunity to design workflows that standardize medication assessments and interventions.
With the growing opportunities to measure 4Ms adherence in older hospitalized patients, it is now possible to generate novel insights into what factors are associated with higher or lower levels of adherence. In this study, we built upon prior measures of 4Ms adherence in an academic inpatient setting to examine the three potential mechanisms described above. Taken together, our results reveal where there are opportunities to modify approaches to 4Ms implementation to achieve consistent, equitable adherence—a core goal of the Age-Friendly Health System movement.
Methods
Setting and Sample
The University of California, San Francisco Health System (UCSF Health) began implementing the 4Ms in 2019 in the inpatient setting. The Ms were implemented sequentially beginning with Mentation (which focused on delirium assessment and action) followed by Medication then Mobility, first in the Acute Care for the Elderly (ACE) unit and then spreading to other inpatient units. Standardized care practices for What Matters have not yet been defined. Additional description of UCSF Health’s 4Ms implementation and level of maturity is reported by Adler-Milstein et al. 1
We identified a cohort of 19 175 UCSF Health patients, aged 65 or older with a completed inpatient encounter between 1/1/2019 and 12/31/2021 (n = 28 833 encounters). We limited encounters to those admitted to an acute care, non-psychiatric service, not on hospice care, and any discharge disposition other than the following: “elopement,” “left without being seen,” “left against medical advice,” “deceased,” or “unknown” in order to ensure we could accurately capture a full picture of care received. We excluded encounters with the discharge disposition “deceased” based on the premise that these patients did not have the full opportunity to receive 4Ms adherent care.
This study was approved by the UCSF IRB (IRB #20-31 337).
Measures of 4Ms Adherence
For this study we utilized a set of EHR-based, hospital encounter-level measures of 4Ms care process adherence that were defined and published by Thombley et al. 6 The 18 measures were designed around the Age Friendly care practices outlined in IHI’s “Age-Friendly Health Systems: Guide to Using the 4Ms in the Care of Older Adults” 7 and capture adherence to both assessment and action care processes within each M, based on the specified frequency of the process (eg, each shift, each day, each encounter). In our setting, we were able to implement 16 of the measures. To do so, we defined the set of all nursing shifts and days where the admitted patient had a status of “inpatient” for each encounter in the sample, excluding any days or shifts for which the patient was admitted for less than 8 hours or where the patient was in the hospital but had a status other than “inpatient” (eg, in the emergency department). While What Matters did not have standardized care practices at UCSF Health in the study timeframe, we used a proxy measures that capture: (1) delivery of whole-person care that included the existence of specific types of notes (eg, social work notes, advanced care planning notes, social history) as well as notes for care that does not typically occur unless requested by the patient (eg, chiropractors, music therapists, spiritual care) and (2) whether the notes were modified or viewed by someone other than the original author.
Given that our research questions focus on adherence to each M and the 4Ms as a whole, after calculating adherence to each individual care process measure, our five primary measures of adherence in this study were composites. The hospital encounter-level 4Ms Care Composite is the mean of all adherence measures recorded during an entire hospital encounter, calculated at the M-level and then averaged to generate a 4Ms composite. Specifically, since individual process measures that comprise the composites have different frequencies—shift, day, hospital encounter—the composite aggregates in a frequency-agnostic way, first at the M-level and then again across all Ms. Each frequency represents an opportunity to deliver 4 Ms-adherent care. For example, if a given measure was at the shift level, with 6 shifts making up an entire hospital encounter, its contribution to the composite was “six” toward the denominator and the number of shifts in which the process was followed (ie, a value between zero and 6) toward the numerator. Similarly, total performance was calculated for each M by taking the sum of total performance across all care processes within that M, and dividing by the count of total opportunities to practice those care processes. Finally, the average composite performance was calculated across all Ms, thereby equally weighting each M at 25%. Additional detail on all measures can be found in prior work. 6
Patient and Hospital Encounter Characteristics
To test our three focal mechanisms, we defined a set of seven characteristics that we measured for each encounter. (Detailed definitions are reported in Supplemental Appendix Table 1.) To assess for negative implicit biases based on observable patient characteristics, we measured whether the patient had limited English proficiency (LEP), whether the patient met the definition for obesity (BMI ≥ 30), the patient’s self-identified race/ethnicity (with “White” serving as the reference group), and whether the patient had Medicaid as their insurance. To assess for positive implicit biases based on observable patient characteristics, we measured whether the patient met the definition of oldest old 4 (85+) and whether they had reduced mobility (immobile, in a wheelchair, or aided). Finally, to assess for contact with comprehensive geriatrics care by a specialized geriatrics team, we determined whether the encounter included significant time in the ACE unit (either discharged from ACE unit or spent at least 50% of the encounter in the unit) and/or whether the encounter included at least one inpatient geriatric consult. The final coding of this categorical variable was: “ACE unit stay and geriatrics consult,” “ACE unit stay and no geriatrics consult,” “geriatrics consult and no ACE unit stay,” and “no geriatrics consult or ACE unit stay.”
Beyond our focal characteristics, we captured an additional set of 17 characteristics that served as control variables. These included additional patient demographics (eg, sex assigned at birth), prior utilization, clinical complexity (eg, comorbidities, MS-DRG), and encounter characteristics (eg, medical vs surgical admission, percentage of time spent in the ICU, and length of stay).
Analytic Approach
After calculating descriptive statistics for our sample, we ran five ordinary least squares regression models with multi-way clustering at the hospital encounter and patient levels. Each model included the focal as well as control characteristics. The dependent variables were: (1) the 4Ms composite and (2) each of the individual M composites. We then generated forest plots to support easy visualization of the relationships by mechanism. For example, the first figure plots the coefficients and 95% confidence intervals for the four characteristics related to the negative implicit biases hypothesis. The 4Ms composite is presented first, followed by each M composite. Lastly, we generated predicted marginal effects that we report to capture the magnitude of the differences in characteristics that we observe. This approach holds all other variables at their mean while comparing the predicted level of adherence for one category of a variable versus the other (eg, LEP versus non-LEP).
Results
Sample Characteristics
As reported in Table 1, our sample reflected diversity across all focal characteristics. For observable characteristics hypothesized to trigger negative implicit biases, 16.7% of patients had limited English proficiency, and 23.7% met the definition of obesity. By race and ethnicity, 58.7% of patients in our sample identified as White, 19.5% as Asian, 8.9% as Latinx, and 6.2% as Black. Across hospital encounters in our sample, 22.5% were for patients with Medicaid insurance. For observable characteristics hypothesized to trigger positive implicit biases, 13.8% of encounters were for patients who met the definition of oldest old, and 42.6% were for patients experiencing reduced mobility. Lastly, for characteristics of contact with comprehensive Geriatrics care, 5.8% of encounters included both an ACE unit stay and a geriatrics consult, 4.9% of encounters had a geriatrics consult only while 2.3% had an ACE unit stay only. The remaining 87% had neither an ACE unit stay nor a geriatrics consult. Hospital encounter-level and patient-level measures are included in Table 1, and Supplemental Appendix Table 2 contains the full set of sample characteristics.
Sample Characteristics. Individuals 65+ with a UCSF Health Hospital Admission 1/1/2019-12/31/21.

Overall, encounters in our sample had 65% adherence to the 4Ms with a standard deviation of 14%. This varied from Mentation adherence with a mean adherence of 77% (SD = 12%) to Medication adherence (mean = 67%; SD = 23%) to Mobility and What Matters adherence (both at 59% mean adherence and SD = 20% and 45%, respectively; Table 1).
Negative Implicit Biases Mechanism
For our first mechanism, we found that obesity and Medicaid were associated with lower encounter-level 4Ms provider adherence (Figure 1). Obesity was associated with a 0.79 percentage point (PP) lower level of adherence (P < .001), such that provider adherence for obese patients had a marginal effect of 64.8% compared to 65.6% provider adherence for non-obese patients (Supplemental Appendix Tables 3–4). Broken down by M, this relationship was primarily driven by lower adherence for the mobility composite, which had a 1.29 PP lower provider adherence (P< .001; Supplemental Appendix Table 3).

Associations between proxy variables for negative implicit biases and 4Ms adherence.
Similarly, the relationship for Medicaid was small in magnitude with a 0.64 PP lower level of provider adherence (P = .002) and a marginal effect of 65.0% provider adherence for Medicaid patients compared to 65.6% provider adherence for those not on Medicaid (Supplemental Appendix Tables 3–4). Broken down by M, Mentation, Mobility, and Medication all had lower provider adherence for Medicaid patients (P < .05 for all 3 Ms) while What Matters did not differ for Medicaid versus non-Medicaid (P = .15).
For the other two measures, we did observe some variation by M. For limited English proficiency (LEP), Medication adherence was higher (2.09 PP; P < .001) provider adherence for patients with LEP. Differences in race and ethnicity categories reflected a mixed picture by M. For example, compared to patients identifying as White, provider adherence for patients identifying as Asian, Latinx, or Native Hawaiian/Pacific Islander was higher for Medication (P < .001 for all 3 groups). Provider adherence for patients identifying as Black was lower for Mobility (P = .02) with no difference in provider adherence for the other 3 Ms (Figure 1).
Positive Implicit Biases/Visible Frailty Mechanism
Provider 4Ms adherence was higher for both the oldest old and those with reduced mobility (Figure 2). Provider adherence for the oldest old was 2.85 PP higher (P < .001), such that those in this age group had a marginal effect of 67.9% provider adherence compared to 65.1% for those under 85 (Supplemental Appendix Tables 3–4). Broken down by M, this relationship was driven by higher provider adherence for Medication (7.91 PP; P < .001) and What Matters (4.25 PP; P < .001) and lower provider adherence for Mentation (−0.90 PP; P < .001) while Mobility adherence (0.12 PP; P = .73) did not differ for the oldest old compared to those under 85 (Figure 2; Supplemental Appendix Table 3).

Associations between proxy variables for positive implicit biases (visible frailty) and 4Ms adherence.
Provider adherence for patients with reduced mobility was 2.01 PP higher (P < .001), with a marginal effect of 66.6% (compared to 64.6% for those for patients without reduced mobility, Supplemental Appendix Tables 3–4). This was driven by higher provider adherence for Mobility (2.08 PP; P < .001) and What Matters (10.07 PP; P < .001), while provider adherence for Mentation (−3.37 PP; P < .001) and Medication (−1.39 PP; P < .001) were significantly lower for those with reduced mobility (Figure 2; Supplemental Appendix Table 3).
Contact With Comprehensive Geriatrics Care Mechanism
Contact with both forms of a comprehensive geriatrics care—the ACE unit and a geriatrics consult—was associated with the highest hospital encounter-level 4Ms adherence—5.33 PP (P < .001)—with a marginal effect of 70.3 (compared with 65.0 for neither; Figure 3; Supplemental Appendix Tables 3–4). Broken down by M, this relationship was driven by higher adherence for Mentation (1.70 PP; P < .001), Mobility (5.18 PP; P < .001), and What Matters (12.10 PP; P < .001) while Medication (0.25 PP; P = .66) did not differ (Figure 3; Supplemental Appendix Table 3).
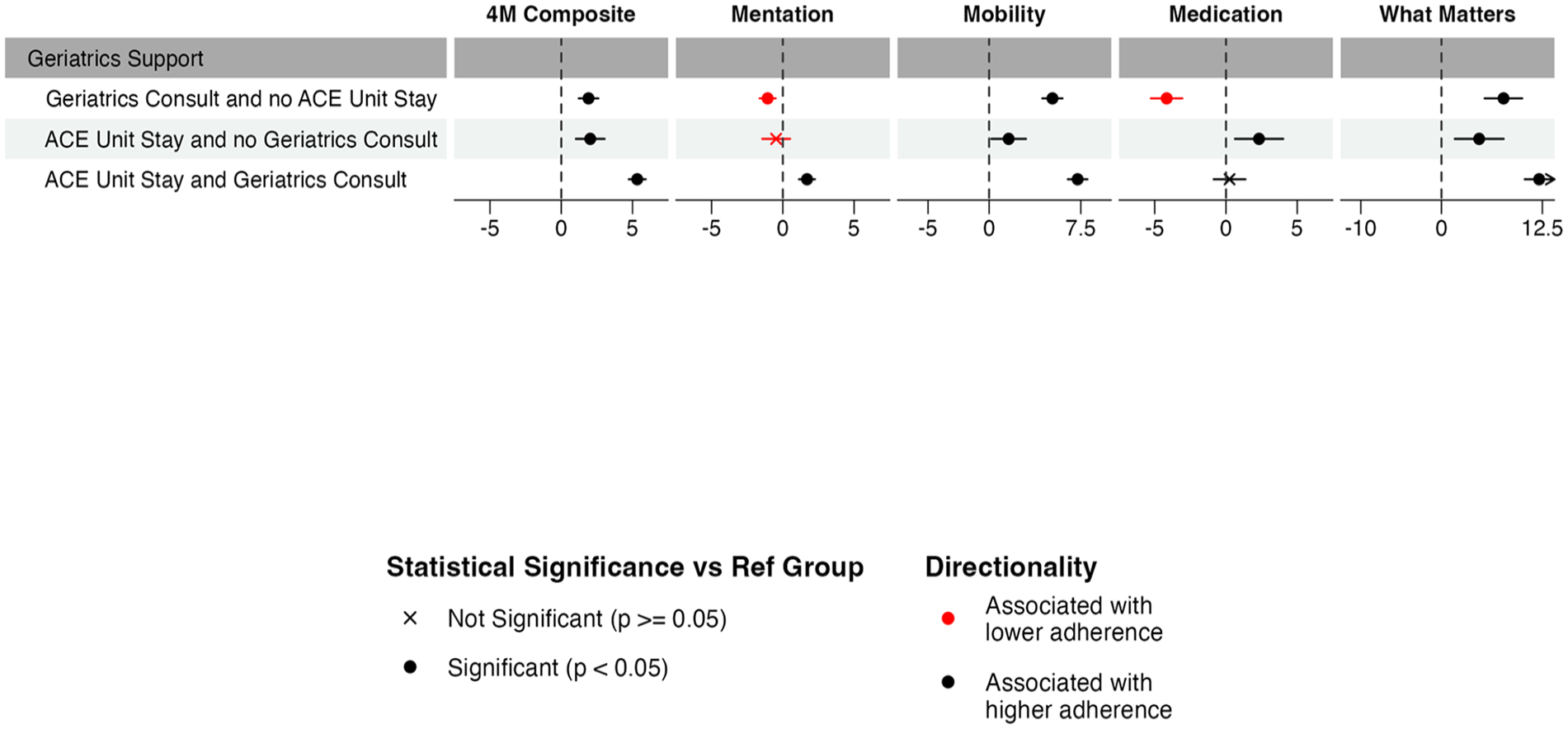
Associations between proxy variables for comprehensive geriatrics care and 4Ms adherence.
Each type of contact on its own was also associated with higher adherence [Geriatrics Consult alone: 1.91 PP (P < .001); ACE unit Stay alone: 2.03 PP (P < .001)]. However, M-specific relationships varied somewhat. A geriatrics consult with no ACE unit stay had higher Mobility (5.18 PP; P < .001) and What Matters (7.69 PP; P < .001) adherence but lower Mentation (-1.07 PP; P < .001) and Medication (-4.16 PP; P < .001) adherence. An ACE unit stay with no geriatrics consult had higher Mobility (1.60 PP; P = .03), Medication (2.32 PP; P = .01), and What Matters (4.67 PP; P < .001) adherence while Mentation adherence did not differ (−0.47 PP; P = .35; Figure 3; Supplemental Appendix Table 3).
Control Variables
Few control variables were associated with meaningfully higher or lower 4Ms adherence (Supplemental Appendix Table 3). Notably, provider adherence for patients who spent a greater portion of their encounter in the ICU had lower 4Ms adherence (−7.81 PP; P < .001). We also observed higher 4Ms adherence for each subsequent year of the study period. Relative to 2019, provider 4Ms adherence was 4.53 PP (P < .001) higher in 2020 and 7.91 PP (P < .001; Supplemental Appendix Table 3).
Discussion
In this study we extended the use of novel hospital encounter-level measures of 4Ms care process adherence to assess whether three hypothesized mechanisms were leading to disparities in receipt of evidence-based care for older adults. Overall, our sample had reasonable levels of adherence—with 65% of opportunities to deliver 4Ms care being fulfilled by providers. We did however find support for our three hypotheses—such that negative implicit biases based on observable characteristics (specifically for proxy measures of obesity and Medicaid insurance) may be driving lower levels of adherence while positive implicit biases and contact with comprehensive geriatrics care may be driving higher levels of provider adherence. In terms of overall magnitude of differences, our marginal effect estimates were small—such that no group was receiving markedly higher levels of 4Ms adherent care. This suggests that these mechanisms may be acting on the margin—increasing or decreasing a specific instance of a care process (such as a mentation screen during one shift) rather than entirely avoiding (or rigorously adhering to) a broad set of processes.
Interestingly, when we broke down adherence by an individual M, we did not observe a consistent pattern of higher adherence for one M or two Ms driving the overall relationship. This suggests that, when our hypothesized mechanisms are in play, it is not because a given M is the more challenging one (such that it would be the most likely to be avoided if the provider observed characteristics of the patient that may make doing so more complex) or the most beneficial (such that it would be the most likely to be followed, again based on provider response to observable characteristics). This makes sense given that our bias-related mechanisms are general and subconscious—and work through factors such as communications challenges that would make any 4Ms care process more difficult. Perhaps obesity and mobility are the exception to this as we did observe lower mobility adherence for older adults who met the definition of obesity. However, this was not true for our third hypothesis—where contact with comprehensive geriatrics care was associated higher mobility adherence—suggesting that understanding how to overcome challenges delivering 4Ms care as part of geriatrics training is at least part of the underlying mechanism driving higher adherence. The mechanisms underlying some of our other findings may require further work to understand. For example, patients with reduced mobility had lower levels of provider adherence to mentation and medication care processes. It could be that greater provider attention devoted to mobility came at the expense of focus on these two Ms.
Given that we did not find large magnitude differences, even where they were statistically significant, it raises the question of whether those leading 4Ms implementation efforts should focus on addressing these mechanisms versus broader efforts to promote better process adherence. Our results suggest that a combination might make sense. Specifically, we observed some of the largest magnitude increases in adherence for the oldest old, with reduced mobility, who were in the ACE unit and had a geriatrics consult. Together, these characteristics identify already known gaps in providing care for older adults: lack of ACE units for those most vulnerable (ie, the oldest old and those with reduced mobility), and lack of Geriatricians and geriatrics-trained interprofessional health team members. This is despite the fact that ACE units have over 25 years of robust evidence showing that they improve clinical and cost outcomes for older adults, yet only 43 ACE units exist in US hospitals. 8 20 000 geriatricians are currently needed to care for over 14 million older Americans but as of 2016, only 7293 geriatricians were practicing.9,10 In contrast, the nursing workforce is projected to increase from 3.6 million in 2021 to 4.2 million in 2030. However, only 8% of nurse practitioners specialize in gerontology (compared to 55% specializing in family practice), and fewer than 1% of registered nurses are certified in geriatrics. 11 Therefore, there is a clear need to recruit nurses to geriatrics and increase geriatrics training more broadly across the nursing workforce. The Partnership for Health in Aging has identified interprofessional geriatrics training as a priority area, but training has not yet been sufficiently responsive to needed demands. 12 Our findings therefore add to the already known gaps of caring for older adults, and they also suggest what is needed to ensure equity in 4Ms care for all older adults: (1) health system investments in ACE units for every hospital for those most vulnerable, (2) increases in the Geriatrician workforce, and (3) widespread interprofessional geriatrics education.13 -15 While not a focal variable, our finding that time in the ICU is associated with significantly lower adherence to 4Ms care also reveals opportunity to work on continuity of 4Ms care across units.16,17
Our results also build on previous literature on 4Ms adherence. Notably, evidence from the primary care setting also points to inequities in 4Ms adherence based on patient characteristics. 18 Specifically, one study found English as a preferred language to be associated with higher adherence to What Matters, Mobility, and Mentation (Depression). Given that we did not find LEP to be a predictor of lower adherence, it may be that setting-specific differences account for varied relationships. Translators may be more readily available in the inpatient setting. Alternatively, the primary care study was conducted in a primarily non-Hispanic White and English-speaking patient population, whereas our setting serves a more diverse population that may therefore be better equipped to support those patients. Taken together, these contrasting findings suggest the need to replicate analyses of adherence by patient characteristics in a wider array of settings in order to understand generalizable versus setting-and population-specific mechanisms.
Key limitations to our study include that it is a single site, and we are evaluating one academic health system’s efforts to implement the 4Ms framework. It is likely that overall levels of 4Ms adherence were shaped by UCSF Health’s priorities within the 4Ms (ie, starting with the goal of reducing the prevalence of delirium to improve overall lengths of stay) and how it approached education, EHR workflows, etc. However, none of these should explain the differential adherence assessed in this paper for the three mechanisms. Nonetheless, it will be critical to test these mechanisms in other health systems that have implemented the 4Ms. Two of our hypotheses were tested using proxy measures as we did not have direct measures of negative or positive implicit biases. In future work it would be valuable to interview frontline clinicians and understand their perceptions of what drives higher versus lower adherence. Our measures of 4Ms process adherence relied on structured EHR data and did not include natural language processing. As a result, some of the care delivered may not have been captured in our measures. In general, this should not have systematically biased our results but there may be specific instances, such as lower Medication adherence for encounters with a geriatrics consult, where the medication assessment done by the geriatrician may be in the note and not in a discrete field. Lastly, our analytic approach included five outcomes with multiple focal variables and a broad set of controls and yielded relatively small effect sizes. Given this, there is a possibility that random chance alone accounted for some of our statistically significant findings.
Conclusion
Our study assessed levels of adherence to 4Ms care processes in an academic inpatient setting to evaluate for factors that may be systematically driving lower versus higher adherence. We found support for the three mechanisms assessed but small magnitude differences. Moving forward, it will be important to understand whether these mechanisms contribute to variation in adherence within other health systems and how best to respond to this variation. In particular, whether more general strategies for increasing adherence versus targeted strategies in subpopulations best achieve the ultimate goal of reliable and equitable practice of the 4Ms, a vital goal of the Age-Friendly Health System movement.
Supplemental Material
sj-docx-1-inq-10.1177_00469580241285598 – Supplemental material for Assessing Equitable Adherence to the Age-Friendly Health System’s 4Ms Framework in an Academic Inpatient Setting
Supplemental material, sj-docx-1-inq-10.1177_00469580241285598 for Assessing Equitable Adherence to the Age-Friendly Health System’s 4Ms Framework in an Academic Inpatient Setting by Julia Adler-Milstein, Robert Thombley, Sarah Rosenthal, Benjamin Rosner and Stephanie Rogers in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
N/A
Data Availability Statement
Data cannot be shared due to institutional restrictions on identified data sharing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The John A. Hartford Foundation [Grant #2020-0056].
Ethical Considerations
This study was approved by the University of California, San Francisco IRB (IRB #20-31337).
Consent to Participate
Consent to participate was waived by the University of California, San Francisco IRB.
Consent for Publication
N/A
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
