Abstract
Index case Human Immunodeficiency Virus (HIV) testing is a voluntary procedure wherein service providers request index clients to enumerate all sexual partners, including biological children, before proceeding with Human Immunodeficiency Virus (HIV) testing. This approach has demonstrated efficacy as a case-finding strategy aimed at curbing future transmission. Through index testing, we can pinpoint partners and children with undiagnosed infections, thereby facilitating early identification of Human Immunodeficiency Virus (HIV) -positive children who are currently healthy. The study aimed to assess the acceptance of index-case Human Immunodeficiency Virus (HIV) testing and its associated factors among anti-retroviral therapy (ART) patients in public health facilities within the Gedeo zone, southern Ethiopia. The study employed a cross-sectional design with a document review. It involved 396 anti-retroviral therapy (ART) patients and utilized a pretested checklist to extract data from clients’ medical records. A simple random sampling technique was used to select anti-retroviral therapy (ART) facilities in the Gedeo zone. Data collection was done from anti-retroviral therapy (ART) patient documents by ODK-Kobo Collect form and data analysis was conducted using statistical package for social science (SPSS) version 25, with bivariate and multivariate analyses performed to identify associations between index case testing and associated factors. A significance level of
The absence of the index case HIV testing increases the transmission of Human Immunodeficiency Virus (HIV) and makes the prevention of the disease difficult.
This research will contribute to our understanding of index-case Human Immunodeficiency Virus (HIV) testing procedures in high-prevalence and densely inhabited locations.
Through raising community knowledge of the Human Immunodeficiency Virus (HIV), especially among those who have been exposed to the virus, this project will help develop index-case HIV testing methods.
Introduction
Index case (ICT) Human Immunodeficiency Virus (HIV) testing is a voluntary practice where the service provider asks index clients to list their entire sexual partner within the past year and biological children and offers and conducts HIV testing.1,2 It is a high-yield, targeted testing approach for identifying and linking new HIV-infected individuals at an early stage to care and treatment services and linking HIV-negative individuals at risk for HIV to prevention services, and it has made a significant contribution to increasing the number of people diagnosed with HIV. 3 This approach needs to be optimally utilized for case detection and to break the HIV transmission cycle by offering HIV testing to individuals who are exposed to HIV and linking them to care and treatment in the event of HIV-positive results.3,4
Since the onset of the HIV epidemic, an estimated 85.6 million people have been infected with the virus, resulting in approximately 40.4 million deaths globally. By the end of 2022, about 39.0 million people were living with HIV worldwide. In 2022, Africa had an estimated 25.6 million people living with HIV. Among these individuals, 90% were aware of their HIV status, 82% were receiving antiretroviral treatment (ART), and 76% had achieved viral suppression. Approximately 20.9 million people were on antiretroviral therapy during this period. The number of new HIV infections in Africa was estimated at 660 000, with an incidence rate of 0.57 per 1000 uninfected population. These figures indicate significant progress in HIV prevention, testing, and treatment across the continent. However, despite these achievements, challenges remain in ensuring universal access to HIV prevention, testing, and treatment services to further reduce the burden of HIV/AIDS in Africa.5,6
HIV partner notification helps identify individuals with undiagnosed HIV and connects them to antiretroviral therapy (ART), reducing the disease's impact on their health and interrupting transmission chains.7 -9 The UNAIDS has established the 95-95-95 target, a universal goal to curb HIV transmission and enhance the timeliness of ART treatment. Meeting these primary objectives is expected to pave the way for achieving subsequent targets.10,11
Index case HIV testing began in Africa around the early 2000s as a strategy to identify individuals who might not otherwise get tested and to reach those at higher risk of HIV infection. In Ethiopia, this program was launched in 2017 with a similar goal of reducing HIV transmission. 12 Index case HIV testing provides critical data for stakeholders, helping them to focus on and plan strategies to improve the acceptance of this testing method, thereby disrupting the chain of HIV transmission. 3 Index case HIV testing provides critical data for stakeholders, helping them to focus on and plan strategies to improve the acceptance of this testing method, thereby disrupting the chain of HIV transmission. 13 This study, therefore, aimed to determine the acceptance rate of index case testing and the factors influencing its acceptance among ART patients.
Objectives of the Study
General Objective
To determine the acceptance of index-case HIV testing and associated factors among HIV-positive patients among anti-retroviral therapy patients in public health facilities in the Gedeo zone, southern Ethiopia.
Specific Objectives
(1) To determine the magnitude of HIV index case testing among index cases on ART follow-up in ART facilities in Gedeo Zone public health facilities
(2) To identify factors associated with HIV index case testing on ART follow-up in Gedeo Zone public health facilities
Methodology
Study Area and Period
The research was carried out in the Gedeo zone of southern Ethiopia at public health facilities that offer anti-retroviral therapy (ART) services. The Gedeo Zone, situated approximately 360 km from Ethiopia’s capital, Addis Ababa, has a population of 1,166,163, consisting of 571 420 males and 594 743 females. ART services are available only at public health facilities within the zone, which includes Dilla Teaching Hospital, 3 primary hospitals (Gedeb, Bule, and Yirga Chefe), and 3 health centers (Haroresa, Wonago, and Chelelecktu). The study spanned from July 1 to August 30, 2023.
Study Design
For this study, we employed a cross-sectional study design coupled with a document review. This approach was chosen as it was the only viable method to obtain data on estimated study participants within the three-month collection period. Secondary data from document reviews of ART patients were utilized to fulfill this requirement.
Source Population
The source population was HIV-positive patients who had at least one contact and had enrolled in ART service for the last 5 years at ART facilities in the Gedeo zone, South Ethiopia region.
Study Population
The study population was sampled from patient cards who were elicited for HIV index contact testing during the study period at public health facilities, in the Gedeo zone, South Ethiopia region
Inclusion and Exclusion Criteria
Inclusion criteria
The inclusion criteria focused on HIV index cases with at least 1 index who is either a partner (married or non-spousal) or a biological child.
Exclusion criteria
Excluded from the study were patient medical records from the ART unit that did not contain information about index partners or children. Additionally, index clients not documented in the index testing registers were also excluded.
Sample Size Determination
The sample size for the initial objective was determined using the prevalence of HIV index client testing (P), which was sourced from a cross-sectional study on disclosure conducted at Jimma Specialized Hospital. This study reported a prevalence of 37.7%. Based on this prevalence, the sample size was calculated using the single population proportion formula:


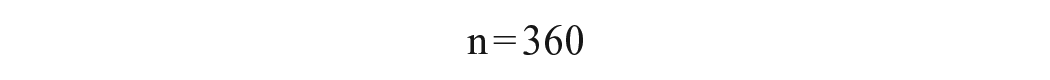
Where, n = the desired sample size, Z = percentiles of the standard normal distribution corresponding to 95% of the confidence level
P = there is research conducted in Jimma (0.377) 14 and d = marginal error between sample and population was taken as 5%. Hence, the calculated total sample size was 360 with adding a 10% non-response rate, a total required sample size was 360 + 36 = 396.
Sample Size Calculation for the Second Objective
The sample size for assessing factors associated with index case HIV testing among adult ART clinic attendees was determined based on several considerations, including significant factors associated with outcome variables, a two-sided confidence level of 95%, a margin of error of 5%, and a power of 80%. This calculation was performed using Open Epi Info version 7. The possible calculated sample sizes for selected factors are depicted below (Table 1).
Sample Size Calculation for Different Factors Associated with Acceptance of Index Case HIV Testing among Adult ART Clinic Attendants at Gedeo Zone, Southern Ethiopia, 2023.

Sampling Procedure
For this study, public health facilities offering Antiretroviral Therapy (ART) services in the Gedeo zone were included. This comprised 4 hospitals (1 General Hospital and 3 primary hospitals) and 1 health center, selected purposively due to their high HIV patient load. The documents of study participants from all selected ART facilities in Gedeo were proportionally allocated based on the history of the case. Subsequently, a simple random sampling technique was employed to select 7 health facilities with ART services from the Gedeo zone (Figure 1).

Proportionate allocation of the sample size among public health facilities with ART service in Gedeo Zone, Southern Ethiopia 2023.
Variably of the Study
Dependent variable
Acceptance of HIV index testing
Independent variables
The study collected data on various socio-demographic and family-related factors, including (age, residence, educational status, marital status distance to health facility), family-related factors (number of children and family size), disclosure status, duration on ART, and clinical stage of disease.
Operational Definitions
Index case
The index case in this study refers to an individual who tested HIV positive during HIV Testing and Counseling (HTC) service at the health facility.15,16
Acceptance
Acceptance is defined as when an indexing client agrees to the testing of their family members (household members) and consent from the index client is obtained, indicating approval for sharing the test results of their partner and/or children, which are then documented in the individual’s folder. 17
Index case HIV testing
Index case HIV testing encompasses offering HIV testing to all individuals who have been exposed to HIV by the index case.
Data Collection Tool and Procedure
The data extraction checklist, developed by the principal investigator, was meticulously crafted after a comprehensive review of relevant literature and an assessment of various ART follow-up cards and registration books. Subsequently, this checklist was integrated into ODK-Kobo software 18 for streamlined data collection. The tool comprised 4 distinct sections: Socio-demographic factors (including age, residence, educational status, marital status, and distance to a health facility), family-related factors (such as the number of biological children and overall family size), care and treatment-related factors (encompassing duration on ART and disclosure status), and service delivery-related factors (including the WHO clinical stage of the disease and adherence status).
Data extraction was conducted through a systematic review of documents, with the collected information inputted directly into the ODK-Kobo Collect form. The team of data collectors consisted of 5 BSc nurses with experience in ART clinics, who were working outside of the study, and 1 MSc supervisor.
Data Quality Assurance
A comprehensive two-day training session was conducted. The quality of the data was meticulously ensured throughout the data collection process. To maintain high standards, 5% of the cards from each data collection site were randomly selected by the principal investigator for a thorough examination. During data collection, the data collectors rigorously assessed the completeness, consistency, and accuracy of the data to identify any discrepancies or errors. Additionally, the investigators conducted further evaluations of the submitted data before entering it into the software.
Data Processing and Analysis
The data collected through ODK-Kobo Collect was exported into SPSS version 25 for further analysis. Various cleaning methods within the software were employed to ensure data accuracy and consistency. Checks and cleaning processes were carried out to rectify any inaccuracies or inconsistencies before proceeding with the final analysis. The normality of the data was assessed using graphical and statistical methods such as the Shapiro-Wilk test.
19
Bivariate and multivariate analyses were conducted to identify factors associated with the acceptance of index case testing. The significance level was set at
Ethical Considerations
Ethical clearance for this study was obtained from the Research and Ethical Committee (REC) of the Department of Public Health, Pharma College, Hawasa campus. Additionally, permission was sought and obtained from the respective health institutions before accessing and reviewing the registration books. Identifying information such as names and telephone numbers were not extracted from the checklist. Instead, individual unique ART numbers, specifically arranged for this study, were utilized. Access to the documents of ART patients included as study participants was restricted solely to the investigator and authorized data collectors to ensure confidentiality. Following data extraction, the records were promptly returned to the archive and securely placed on their designated shelves.
Result
Socio-Demographic Characteristics of the Study Participants
In this study, a total of 396 ART patient cards were reviewed. The mean age of the index cases who participated in the study was 35.89 years, with a standard deviation of 28.289 years. The majority of the study participants fell within the age group of 25-34 years, constituting 49.0% of the total. In terms of residence, 246 (62.1%) of the participants were from rural areas, while 150 (37.9%) were from urban areas. Regarding educational status, the largest proportion of participants (40.0%) had received primary education (Table 2).
Sociodemographic Characteristics of the Study Participants for Index Case HIV Testing among Adult ART Patient from Selected Public ART Facilities in Gedeo Zone, Southern Ethiopia (n = 396), 2023.

HIV/AIDS-Related Characteristics of the Study Participants
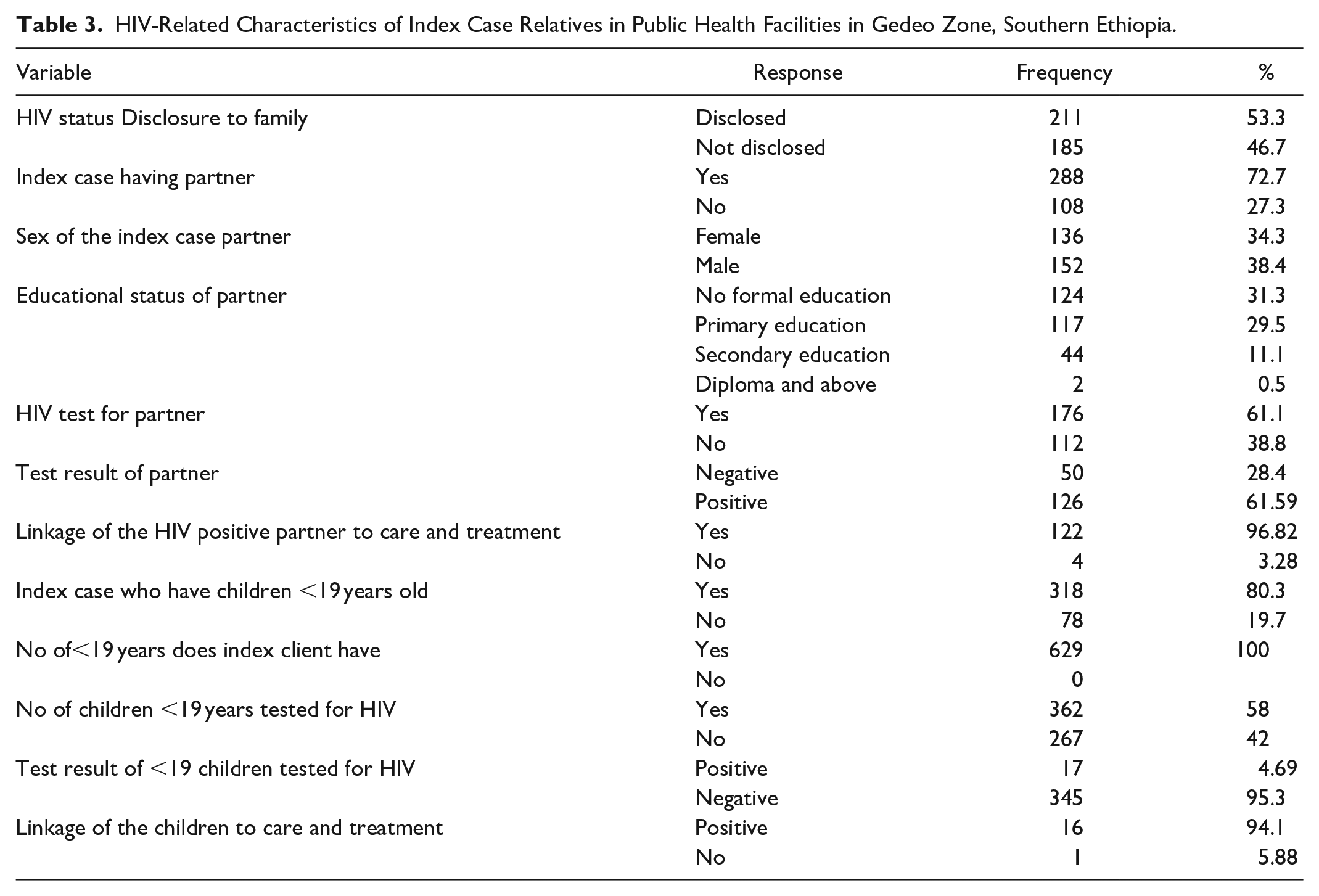
The acceptance rate of index case testing among ART patients was 240, 60.6% (95% CI: 56%-65%). In terms of the WHO clinical staging of the disease, the majority of index cases were classified under stage 1, comprising 292 (73.7%) individuals. Additionally, 56 (14.1%) were categorized as stage 2, 44 (11.1%) as stage 3, and 4 (1.0%) as stage 4 (Figure 2). Regarding adherence status, 156 (39.4%) index cases demonstrated good adherence, while 54 (13.6%) exhibited poor adherence (Figure 3). It is noteworthy that all ART patients were offered index case HIV testing services. Among the index cases with partners, which accounted for 288 (72.7%), a majority of 176 (61.1%) partners underwent HIV testing, and 126 (62%) of them tested positive for HIV. Importantly, the majority of HIV-positive partners were successfully linked to care and treatment services, totaling 122 individuals (96.82%) (Table 3).

The WHO clinical staging of the disease of index cases in Gedeo Zone public ART facilities.
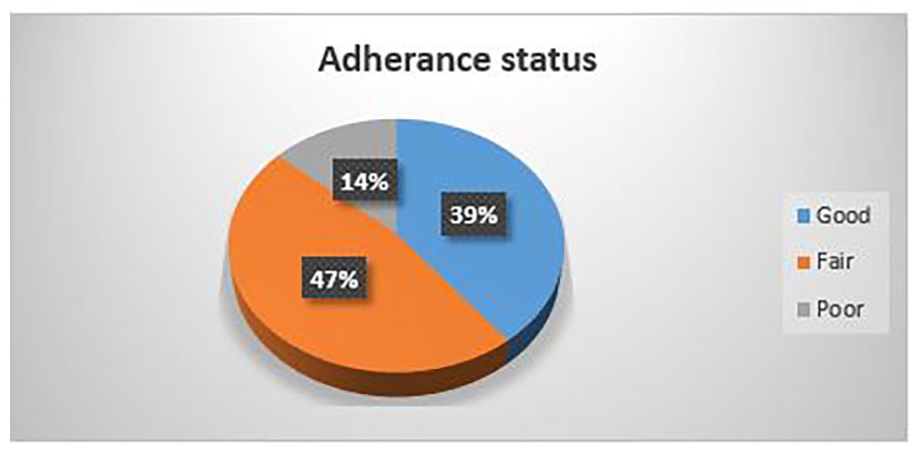
The adherence status of the index cases on ART in Gedeo zone public ART facilities.
HIV-Related Characteristics of Index Case Relatives in Public Health Facilities in Gedeo Zone, Southern Ethiopia.
Factors Associated With Acceptance of the Index Case Testing
In bivariate logistic regression, 5 variables, including residence 1.6 (95% CI: 1.087, 2.54), duration on AR 10.98 (95% CI: 6.824, 17.69), HIV disclosure 146.4 (95% CI: 60.05, 357.02), good adherence to ART 166.34 (95% CI: 51.93, 532.76), and educational status on index case partner 6.9 (95% CI: 3.53, 13.53), have an association with acceptance of the index case testing.
Those above-listed variables and other variables with a
The Factors Associated With Acceptance of HIV Index Case Testing Among ART Patient from Selected Public ART Facilities in Gedeo Zone, Southern Ethiopia (n = 396), 2023.

OR = odds ratio; CI = confidence interval.
Discussion
The present study revealed an acceptance rate of index case testing at 60.6%, which is lower compared to similar studies conducted in West Ethiopia (85.2%) 15 and refuge camps in southern Ethiopia 73%. 17 Additionally, it falls below acceptance rates reported in other studies conducted in Ethiopia 73%, 20 Tanzania 96.6%, 21 and Zimbabwe 95%. 22 Several factors may contribute to this discrepancy, including differences in study design, methodology, data collection tools, and study populations. Additionally, variations in healthcare infrastructure, access to services, and community awareness about HIV testing may influence acceptance rates across different settings. Conversely, the acceptance rate observed in this study was higher than that reported in another study conducted in Ethiopia (53%). 23 This difference could be attributed to variations in the study population and the study design utilized.
The odds of accepting index case testing among patients on ART for more than 12 months were found to be 2.3 times higher (AOR = 2.3; 95% CI: 1.86, 6.12) compared to those on ART for less than 12 months. This finding aligns with studies conducted in West Ethiopia, Jimma, Ethiopia, 24 and Gojam, Ethiopia. 25 It is plausible that as the duration of ART treatment increases, index-case patients become more aware of the risks to their family members. Additionally, prolonged ART treatment may reduce their fear and experience of discrimination, thus increasing their willingness to undergo index case testing.
Similarly, the acceptance of index case testing among patients who disclosed their HIV status was found to be significantly higher (AOR = 36.19, 95% CI: 7.26, 180.44) compared to patients who did not disclose their HIV status. This finding is consistent with a study conducted in West Ethiopia. 20 The act of disclosing one’s HIV status may empower patients to advocate for HIV testing among their family members, thereby increasing acceptance rates for index case testing.
Likewise, index cases with good ART adherence status were found to have a significantly higher chance (AOR = 14.52; 95% CI: 2.10, 100.2) of accepting index case testing compared to patients with poor adherence to ART. This may be attributed to the fact that patients with good adherence understand the benefits of ART care, which in turn motivates them to advocate for HIV testing among their contacts.
This study aimed to assess the acceptance rate of index case testing and identify associated factors among patients receiving ART treatment. The findings revealed that the acceptance rate of index case testing in the study area was lower than the global average. Additionally, the study identified factors associated with the acceptance of index case testing, which could serve as valuable evidence for enhancing acceptance rates. By shedding light on the challenges and determinants of index case testing acceptance among ART patients, this study contributes to the understanding of HIV prevention strategies in the local context. The identified factors can inform targeted interventions and programs aimed at improving acceptance rates and ultimately reducing HIV transmission within communities.
Strength of the Study
Comprehensive coverage of all ART facilities in the Gedeo zone ensures a representative sample, enhancing the study’s external validity.
Triangulation of incomplete data from various sources such as ICT registration books and SMART care clients' databases improves the reliability and completeness of the dataset, enhancing the study's robustness.
Weakness of the Study
The inability to include important variables due to the nature of the data source limits the depth of the analysis and may lead to potential biases or confounding factors not being accounted for.
Dependence on secondary data sources for triangulation may introduce errors or inconsistencies, impacting the accuracy of the findings.
Conclusion
The study reveals a concerning low overall acceptance rate of index case testing, highlighting the need for targeted interventions to improve HIV testing uptake in the Gedeo zone.
Factors such as duration of ART, HIV status disclosure, and medication adherence emerge as significant determinants of acceptance, providing valuable insights for designing targeted interventions.
Recommendations for improving index case testing rates include health education, awareness campaigns, and efforts to enhance treatment adherence, underscoring the importance of multi-faceted approaches to HIV care.
Ensuring the sustainability of ICT services and maintaining quality in HIV index case testing are essential for long-term improvements in HIV testing and care outcomes in the region.
Footnotes
Acknowledgements
We would like to thank the Gedeo Zone Health Office and Pharma college of Health Science, Hawassa campus.
Abbreviations/Acronyms
ART: Antiretroviral Therapy; ARV: Anti-retroviral; EDHS: Ethiopian Demographic Health Survey; EPHI: Ethiopian Public Health Institution; HTS: HIV Testing Service; ICT: Index Case HIV Testing; PLHIV: People Living with HIV/AIDS; PNS: Partner Notification Service
Author Contributions
Availability of Data and Materials
The authors presented the necessary data in the manuscript. The dataset associated with the conclusions of this article is available from the authors upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics,Approval,and Consent to Participate
Ethical clearance for this research was obtained from the research and ethical committee (REC) of the Department of Public Health, Pharma College, Hawasa, campus. Ethical approval number 053/2023.
Informed Consent
Not applicable
Consent for Publication
Not applicable.
