Abstract
To reduce pharmacy-related medical expenses, it is necessary to reduce drug costs. One way to achieve this is by increasing the usage rate of generic drugs. The purpose of this study was to identify platelet aggregation inhibitors (PAIs) that contribute to high drug costs and are sold as brand-name drugs in order to increase the usage rate of generic drugs, and to analyze the factors that affect the usage rate of generic drug. We conducted a cross-sectional study based on the National Database of Health Insurance Claims and Specific Health Checkups of Japan Open Data Japan (NODJ) of the Ministry of Health, Labor and Welfare and datasets containing related medical information from official statistical surveys such as the Basic Survey on Wage Structure. Monthly personal income in each prefecture were negatively correlated with outpatient out-of-hospital and outpatient in-hospital prescriptions of the PAIs clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg), but not between monthly personal income and outpatient out-of-hospital prescription of ticlopidine (100 mg). For outpatient out-of-hospital prescriptions and outpatient in-hospital prescriptions, negative correlation was generally observed between the usage rate of generic drug and monthly personal income, except for ticlopidine (100 mg), which has the lowest price among the brand-name drugs. The usage rate of generic PAIs is negatively correlated with monthly personal income. Promoting the use of generic drugs among high-income earners might be necessary to further increase the usage rate of generic drug.
Keywords
Introduction
To reduce pharmacy-related medical expenses, it is necessary to reduce drug costs. One way to achieve this is by increasing the usage rate of generic drugs. In fiscal year 2019, the medical expenses in Japan were 43.6 trillion yen, 1 which indicates an increase of 2.1 trillion yen from the 41.5 trillion yen spent in fiscal year 2015. Medical expenses related to pharmacies were 7.7 trillion yen, 5.7 trillion yen of which were due to drug costs. 2 Promotion of generic drug use is recognized as an effective means of controlling rising prescription drug costs. 3 According to a systematic review and meta-analysis conducted in the United States of America (USA), patients with lower income were more likely to use generic drugs than those with higher income. 4 It has also been reported that the generic drug discount programs of the USA provide affordable prescription medication to low-income individuals. 5 Hitiris and Posnett 6 reported that gross domestic product is an important factor that affects medical expenses, because there is a correlation between medical expenses and a patient’s monthly income from an economic point of view. However, few studies have investigated the relationship between income and usage rates of generic drug or individual drugs.
At the end of March 2020, approximately 16 000 items 7 were listed in the official bulletin as medical drugs that can used for medical treatment covered by insurance at medical institutions in Japan. The Basic Policy on Economic and Fiscal Management and Reform 2017 8 stipulated that the usage rate of generic drugs in Japan should be 77.4% at the end of 2019 and 80% by September 2020. 9 However, although generic drug use in the USA reached 95% in 2020, it reached only 70% in Japan.9,10 Further expansion of generic drug use is required.
In the past few decades, there has been an increase in the incidence and prevalence of cardiovascular disease (CVD) including acute coronary syndrome (ACS), which has become a leading cause of mortality and morbidity worldwide.11 -15 The number of CVD-related deaths has increased by 12.5% during the past decade, accounting for approximately one-third of all deaths globally, mainly because of population growth and aging. 13 Patients with ACS are at an increased risk of new ischemic events,16,17 and ischemic heart disease and stroke are main contributors to the global CVD burden. 13 Therefore, we focused on platelet aggregation inhibitors (PAIs) such as clopidogrel, ticlopidine, and cilostazol, which are used to prevent the recurrence of cerebral infarction and stent thrombosis during coronary intervention.
In Japan, various large-scale medical information databases have been developed for clinical and epidemiological research purposes.18,19 Based on the data given in the National Database of Health Insurance Claims and Specific Health Checkups of Japan Open Data Japan (NODJ), Basic Survey on Wage Structure (BSWS),20,21 Statistics of Physicians, Dentists and Pharmacists (SPDP), 22 and e-Stat websites, 23 we decided to identify PAIs that contribute to high drug costs and analyze factors associated with generic drug usage.
Materials and Methods
National Database of Health Insurance Claims and Specific Health Checkups of Japan Open Data Japan
The National Database of Health Insurance Claims and Specific Health Checkups of Japan (NDB) consists of data relevant to the outpatients, inpatients, a combination of diagnostic procedures, prescriptions, dental care, and specific laboratory data. Since its inception in 2009, this database has accumulated monthly data on health insurance claims along with annual data on specific health checkups focusing on metabolic syndrome, thus making it one of the most exhaustive national healthcare databases worldwide. Its insurance-related data are also useful in developing government policies for national healthcare insurance systems and academic research. NDB reflects healthcare trends in Japan because the national healthcare system oversees most of the medical care implemented in the country. 24 Compared to the data obtained through sampling surveys, the NDB consists of a more comprehensive dataset of individuals who have received specific health checkups in Japan. Because of its national coverage, it is also suitable for understanding the healthcare conditions in each prefecture. 25
The recently released NODJ database provides a variety of NDB summaries that are freely available to the public. As the NODJ contains open data, analyses entail less effort, cost, and ethical consideration, and can be conducted quickly. We obtained data from the Ministry of Health, Labor and Welfare (MHLW) website for the fourth NODJ in 2017. 26 The fourth NODJ contains data items, such as medical and dental fee schedules, dental injuries, drug data, specified insurance medical materials, specified medical examination test items, and a specified medical examination questionnaire from April 2017 to March 2018. The medication usage records in the fourth NODJ were restricted to the top 100 medication codes for each therapeutic category. The NODJ contains information on prescription drug usage, including outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions for internal use, external use, and injection, based on the standard unit of drug price listings. We evaluated PAIs from the NODJ based on the therapeutic categories set by the Standard Commodity Classification Number of Japan. 27 These PAIs were stratified by 47 Japanese prefectures, therapeutic categories, and drug prices from the data tables 000711952.xlsx, 000711957.xlsx, and 000711961.xlsx. 26 We then calculated the usage rates of generic drugs stratified by outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions. PAIs were classified into brand-name and generic drugs, and the usage rate of generic drugs was calculated.
Basic Survey on Wage Structure
Since 1948, the MHLW has annually conducted the BSWS,20,21 based on the Statistics Law. The BSWS was conducted in 49 541 offices across the country in FY2017 to obtain a clear picture of the employee/wage structure in major industries, 21 which is defined as the wage distribution stratified according to the types of employment and work, occupation, sex, age, school career, length of service, and occupational career. BSWS data is accessible to the public, and the data from 2017 are available from the MHLW and e-Stat websites (https://www.e-stat.go.jp). 23 In this study, we considered the monthly personal income by prefecture.
Statistics of Physicians, Dentists, and Pharmacists
The SPDP database clarifies the distribution of physicians, dentists, and pharmacists by gender, age, type of work, place of work, name of department, etc., and stores basic data for health, labor, and welfare administration. 22 Hospital doctor rate (defined as the ratio of the number of doctors working in hospitals to the total number of doctors working in hospitals and clinics), number of doctors per 1000 people in each prefecture are provided in the SPDP. This database is open to the public and can be obtained from the MHLW and e-Stat websites. 23
Data Analysis
We conducted a cross-sectional study using datasets from the NODJ, BSWS, and SPDP. Using 3 tables (000711952.xlsx, 000711957.xlsx, and 000711961.xlsx), PAIs that were not indicated for Kawasaki disease but were indicated for ischemic disorders were extracted from the fourth NODJ. In addition, we extracted PAIs for which the usage rate of generic drugs could be calculated for all outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions (Figure 1).

Flowchart depicting the inclusion/exclusion criteria for each prescription in the outpatient out-of-hospital, outpatient in-hospital, and inpatient prescription categories.
A linear regression analysis was performed using the hospital doctor rate, the number of doctors per 1000 people, and monthly personal income as independent variables. Cases wherein P-values were <.05 were considered to show statistically significant differences.
Data analysis was performed using Excel Statistics 2012 (Society Information Service Co., Japan) and JMP version 13.0 (SAS Institute, Cary, NC, USA).
Ethical Considerations
Ethics approval was not sought because this was a cross-sectional study without any research subjects. All results were obtained from data openly available online from the website of the MHLW (https://www.mhlw.go.jp/index.html) or e-Stat.
Results
The PAIs extracted from the fourth NODJ were clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg). Plavix® (75 mg) was the most expensive brand-name PAI.
The usage rates of generic drugs as a result of outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions for clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) by prefecture are summarized in Table 1 and Figure 2. The regression coefficients showing the relationship between the usage rate of generic drug and the hospital doctor rate, the number of doctors per 1000 people, and monthly personal income for the same drugs are summarized in Table 2.
Usage rate ate of generic drugs usage in Japan’s 47 prefectures.

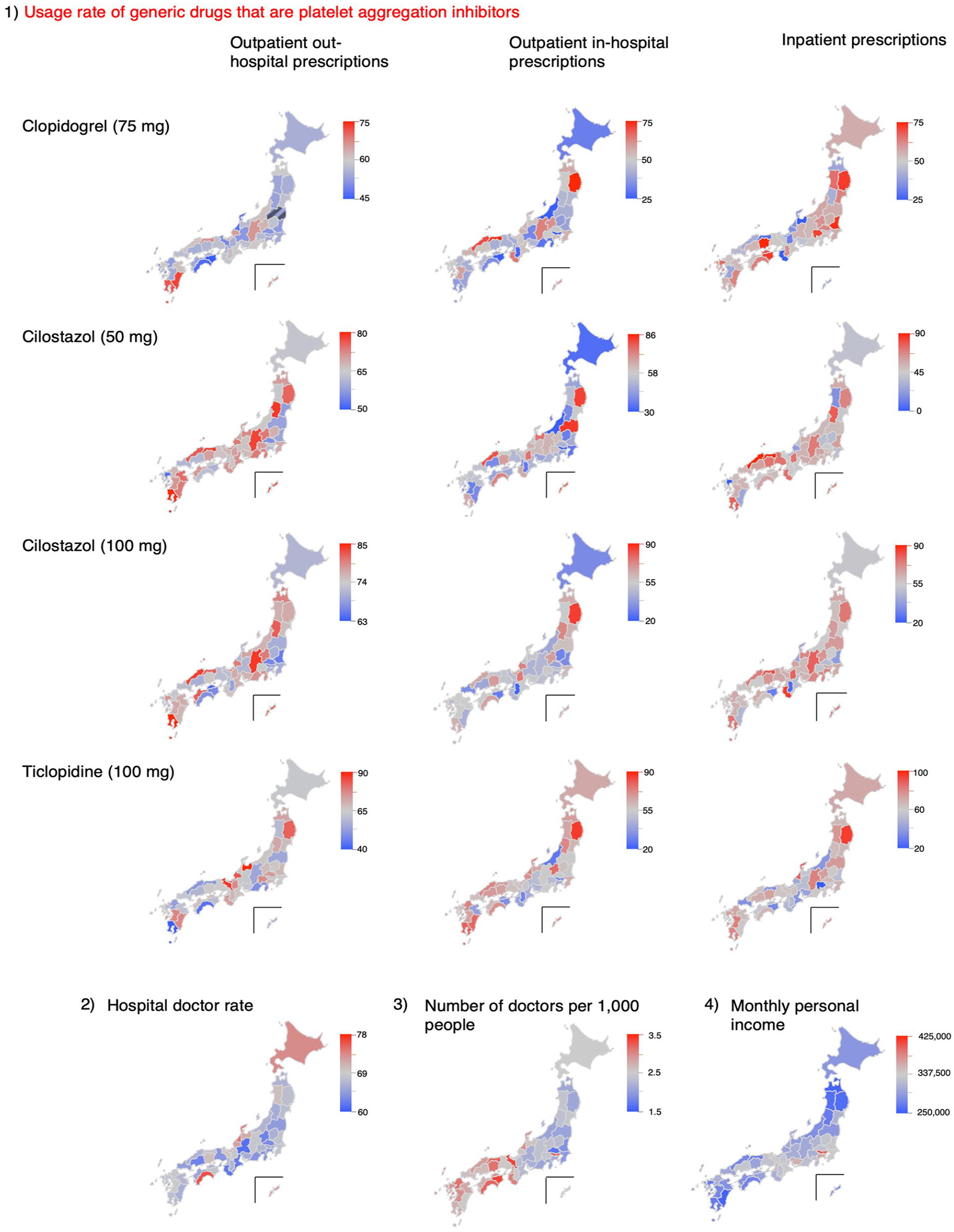
Extracted data categorized by the 47 prefectures of Japan: (1) usage rate of generic drugs that are platelet aggregation inhibitors, (2) hospital doctor rate, (3) number of doctors per 1000 people, and (4) monthly personal income.
Correlation between the usage rate of generic drugs and related factors.

P < .05.
The linear regression analyses of clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) revealed 8 combinations with P-values <.05 (Table 2). The correlation coefficient between the usage rate of generic drug of ticlopidine (100 mg) and monthly personal income of prefecture in outpatient in-hospital settings was −.5491 (P < .0001). The correlation coefficient between the usage rate of generic drug of cilostazol (100 mg) and monthly personal income of prefecture in outpatient out-hospital settings was −.4833 (P = .0006). The correlation coefficient between the usage rate of generic drug of cilostazol (100 mg) and monthly personal income of prefecture in outpatient in-hospital settings was −.3650 (P = .0117). The correlation coefficient between the usage rate of generic drug of ticlopidine (100 mg) and the hospital doctor rate in outpatient in-hospital setting was 0.3590 (P = .0132).
In outpatient out-of-hospital settings, the usage rate of generic drug of clopidogrel (75 mg), cilostazol (50 mg), and cilostazol (100 mg) were negatively correlated with monthly personal income. In outpatient in-hospital settings, the usage rate of generic drug for cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) were negatively correlated with monthly personal income. In outpatient in-hospital settings, the usage rate of generic drug for clopidogrel (75 mg) and monthly personal income were correlated negatively (P = .0525). For all drugs, there was no correlation between the usage rates of generic drug and monthly personal income in inpatient prescriptions.
The prefectures with a monthly personal income of 350 000 yen or more were Tokyo, Kanagawa, Aichi, and Osaka in descending order. In these 4 prefectures, the usage rates of generic drug in outpatient out-of-hospital settings and outpatient in-hospital settings for clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) were below the median of the other prefectures.
The drug prices of the brand-name and generic drugs for clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) are shown in Table S1. The drug prices for clopidogrel (75 mg) were 201.2 yen (brand-name drug), 100.6 yen (generic drug), 90.9 yen (generic drug), and 49.1 yen (generic drug). For cilostazol (50 mg), the drug prices were 80.8 yen (brand-name drug), 47.1 yen (generic drug), and 17.3 yen (generic drug). For cilostazol (100 mg), the drug prices were 144.2 yen (brand-name drug), 84.2 yen (generic drug), 47.5 yen (generic drug), and 33.1 yen (generic drug). For ticlopidine (100 mg), the drug prices were 50.7 yen (brand-name drug) and 5.8 yen (generic drug). The P-values of the linear regression analyses between the usage rate of generic drug and the monthly personal income for clopidogrel (75 mg), which had the highest drug price of all the brand-name drugs, were .0434 for outpatient out-of-hospital prescriptions, .0525 for outpatient in-hospital prescriptions, and .9861 for inpatient prescriptions. For the brand-name drug of cilostazol (50 mg), which had the third highest drug price, the P-values were .0255 for outpatient out-of-hospital prescriptions, .0143 for outpatient in-hospital prescriptions, and .7358 for inpatient prescriptions. For the brand-name drug of cilostazol (100 mg), which had the second highest drug price, the P-values were .0006 for outpatient out-of-hospital prescriptions, .0117 for outpatient in-hospital prescriptions, and .4789 for inpatient prescriptions. For the brand-name drug of ticlopidine (100 mg) with the lowest price, the P-values were .4504 for outpatient out-of-hospital prescriptions, .0001 for outpatient in-hospital prescriptions, and .0559 for inpatient prescriptions. For outpatient out-of-hospital prescriptions and outpatient in-hospital prescriptions, there was a correlation between the usage rate of generic drug and monthly personal income, except for ticlopidine (100 mg), which had the lowest drug price.
Discussion
In this study, we investigated the factors that affect the usage rate of generic drugs in outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions for PAIs, including Plavix® (clopidogrel: 75 mg, Sanofi), which is sold as a brand-name drug and contributes to high drug costs. We demonstrated the relationship between the usage rate of generic drugs and the hospital doctor rate, the number of doctors per 1000 people, and monthly personal income.
In outpatient in-hospital prescriptions, the correlation coefficients between the usage rate of generic drug and hospital doctor rate were significantly high for cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg). Clopidogrel (75 mg) did not exhibit significant correlation. It is assumed that there was little effect on generic drug prescriptions because generic drugs had not been on the market for very long when this correlation analysis was conducted. Hospital doctors are younger on average than clinical doctors. 28 Older doctors have a more negative perception of generic drug use than younger doctors do. 29 Therefore, it may be necessary to encourage elderly hospital doctors to use generic drugs.
In terms of monthly personal income, the usage rate of generic drug of clopidogrel (75 mg), cilostazol (50 mg), and cilostazol (100 mg), in outpatient out-of-hospital prescriptions, the usage rate of generic drug of cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) in outpatient in-hospital prescriptions were statistically significant.
In prefectures with monthly personal income of 350 000 yen or more, the usage rates of generic drug in outpatient out-of-hospital and outpatient in-hospital prescriptions for clopidogrel (75 mg), cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) were low. Of the 5 prefectures with the lowest monthly personal income, 3 prefectures were in the Tohoku region, with the lowest being Aomori, Iwate, and Akita prefectures. In these 3 prefectures, the usage rate of generic drug in outpatient out-of-hospital and outpatient in-hospital prescriptions for cilostazol (50 mg), cilostazol (100 mg), and ticlopidine (100 mg) were above the median in at least 2 prefectures.
In the Japanese healthcare system, additional insurance premiums are added according to the usage rate of generic drugs for outpatient out-of-hospital prescriptions, outpatient in-hospital prescriptions, and inpatient prescriptions. 30 However, the usage rate of generic drug differs between the 3 settings. The difference between outpatients and inpatients is thought to be because of the differences in the additional insurance system for the use of generic drugs between the 3 groups of patients in these settings. The self-pay rate is 10% for patients aged 75 years and above, 20% for those aged from 70 to 74 years, 30% for those aged from 6 to 69 years, and 20% for those aged under 5 years. 31 The correlation between the usage rate of generic drug and monthly personal income may differ depending on whether the prescriptions are categorized as outpatient out-of-hospital, outpatient in-hospital, or inpatient prescriptions.
The results of the present study show that the use of generics for drugs with high drug prices is negatively correlated with monthly personal income for both outpatient out-of-hospital and outpatient in-hospital prescriptions. Suzuki et al 32 reported that the proportion of negative correlation coefficients between the usage rate of generic drugs and monthly personal incomes for outpatient out-of-hospital and outpatient in-hospital prescriptions was over 70%, while that for inpatient prescriptions was 46.9%.
One possible reason why the usage rate of generic drug for inpatients is less affected by drug prices is that most inpatients are 70 years of age or older, and their self-pay are lower than those of outpatients. 33 In order to promote the use of generic drugs, it may be necessary to review the generic drug fee system for inpatient prescriptions. In outpatient out-hospital prescriptions and outpatient in-hospital prescriptions, a significant negative correlation was observed between the usage rate of generic drug and monthly personal income for most drugs (Table 2). The reason for this is unclear.
Regional differences in the use of generic drugs have been reported.34,35 Our results are consistent with previous studies showing that encouraging the use of generic antihypertensive drugs was ineffective in areas where the income levels were relatively high 34 ; Among seniors aged 75 years and older, high-income groups are more likely to choose brand-name drugs than general-income groups. 36
Federman et al 37 reported that older adults with low income or no prescription insurance were more likely to use generic cardiovascular medications than older adults with higher income and insurance. The reason for this is that people with lower incomes need to spend less and consequently need to reduce the cost of drugs for lifestyle diseases, which must be purchased on a regular basis. To aid such individuals, higher-priced brand-name drugs may be prescribed as generic drugs in pharmacies. To further increase the use of generic drugs, it is important to consider economic measures that promote the use of generic drugs among high-income earners.
In this study, the usage rate of generic drug for most of the PAIs investigated was negatively correlated with monthly personal income in outpatient prescriptions. Because this study targeted 4 types of PAIs, details about all PAIs are unavailable.
This study has several limitations. Because the usage of medications in the NODJ database is restricted to the top 100 medication codes in each treatment category, not all NDB medication codes were included. Therefore, the drugs studied are not an exhaustive list of all PAIs. These factors must be considered when interpreting the NODJ results. Generic drugs are used at a higher rate in large hospitals than in small medical institutions, such as clinics. 38 Physicians were more likely to select brand-name drugs to treat themselves, compared to non-physicians. 39 Generic drug use rates are influenced by patients, cost control, local government-specific policies, pharmaceutical company promotional activities, and educational initiatives. 40 However, our study did not consider the influence of these factors. In this study, we investigated correlations at the regional level. However, regional correlation studies cannot show an association between individual-level exposure and outcomes, nor can they elucidate the causal relationship and risk assessment of the usage rates of generic drug at the individual level. Overall, it is important to pay attention to these factors when the prefecture develops measures to increase the usage rate of generic drug.
Conclusion
The usage rate of generic PAIs is negatively correlated with monthly personal income in outpatient out-of hospital prescriptions and outpatient in-hospital prescriptions. It is important for prefectures with many PAI prescriptions to consider economic measures when increasing the usage rate of generic drugs. Our research observes an aspect of current situations of generic drug use and helps increase the proportion of generic drugs used in prefectures.
Supplemental Material
sj-xls-1-inq-10.1177_00469580231219094 – Supplemental material for Study of Factors to Increase the Usage Rate of Generic Drugs in Platelet Aggregation Inhibitors
Supplemental material, sj-xls-1-inq-10.1177_00469580231219094 for Study of Factors to Increase the Usage Rate of Generic Drugs in Platelet Aggregation Inhibitors by Takaaki Suzuki, Mari Iwata, Mika Maezawa, Kiyoka Matsumoto, Mizuki Tanaka, Riko Satake, Misaki Inoue, Yu Yoshida, Kazuhiro Iguchi and Mitsuhiro Nakamura in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
Not applicable.
Authors’ Contributions
TS and MN conceived of the study, participated in its design and coordination, and drafted the manuscript. TS and MI conceived of the study, participated in its design, helped the statistical analysis, and drafted the manuscript. KM, MT, YY, MI, RS performed the statistical analysis. MM and KI helped to interpret of data. All authors read and approved the final manuscript.
Consent for Publication
Not applicable.
Data Accessibility Statement
The dataset supporting the conclusions of this article is included within the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MI is an employee of Kifune Pharmacy. The other authors have no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant Numbers 17K08452, 20K10408, 21K06646, and 22K10446). No additional external funding was received for this study.
Ethics Approval and Consent to Participate
Ethics approval was not sought because this was an observational study without any research subjects. All results were obtained from data openly available online from the website of the PMDA (http://www.pmda.go.jp) and the e-Stat websites (![]() ).
).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
