Abstract
Chlorhexidine gluconate (CHG)-containing dressings are recommended to prevent central line associated bloodstream infections (CLABSIs) and other catheter-related infections. This study compared the effect of 2 CHG dressings on CLABSI, cost of care, and contact dermatitis. A retrospective analysis was conducted using the Premier Healthcare Database of hospitalized patients (n = 53 149) with central venous catheters (CVCs) and receiving either a transparent CHG gel dressing (n = 14 488) or an opaque CHG sponge dressing (n = 38 661) between January 2019 and September 2020. Two cohorts (n = 14 488 each), CHG-Gel and CHG-Sponge, were matched 1:1 using a propensity score method on 33 patient and facility characteristics. CLABSI and contact dermatitis rates, hospital length of stay (LOS), and hospitalization costs were compared using mixed-effect multiple regression. This approach effectively controlled for random clustering effects across hospitals and patients’ Diagnosis-Related Group (DRG) classifications. CHG gel dressings were associated with a 41% decrease in CLABSI rates (P = .0008) compared to CHG sponge dressings (0.35%vs 0.60%). A 0.4-day shorter LOS (9.53vs 9.90 days, P = .0001) and a cost saving of $3576 per hospital stay ($40 197 vs $43 774, P = .0179) was observed with CHG gel dressing use. There was no statistically significant difference in contact dermatitis rates (P = .7854) between the CHG-Gel and CHG-Sponge cohorts. The findings of this study suggest that the use of CHG gel dressings may be more effective in reducing the risk of CLABSIs and associated clinical costs compared to CHG sponge dressings in hospitalized patients. Moreover, there appears to be no significant discrepancy in contact dermatitis rates between CHG gel and CHG sponge dressings. Healthcare providers may consider using CHG gel dressings as a standard practice for patients with CVCs to reduce the risk of infections and improve patient outcomes.
The use of CHG dressings is recommended in published guidelines to reduce the risk of central line associated bloodstream infections.
There is limited literature available to compare different types of CHG dressings and our study is the first one comparing 2 dressings on clinical and economic outcomes.
Our findings suggest that the use of transparent CHG gel dressings significantly reduce CLABSIs and clinical costs when compared to opaque CHG sponge dressings for hospitalized patients; therefore, healthcare providers may consider using CHG gel dressings as a standard practice for patients with CVCs to reduce the risk of infections and improve patient outcomes.
Introduction
Central lines are commonly used for central venous access and for the administration of drugs or fluids in critically ill patients. Over 29 million central line days were reported in the US in 2021, with 11 million taking place in intensive care units (ICUs), 17 million in wards, and 1 million in neonatal intensive care units (NICUs). 1 Meanwhile, central line associated bloodstream infections (CLABSIs) cost the US healthcare system thousands of deaths and billions of dollars. In 2021, the Centers for Disease Control and Prevention (CDC) reported a total of 30 389 CLABSIs occurring in various inpatient locations. These infections result in an attributable cost of approximately $48 000 per episode, along with a 12% to 25% increase in mortality and extended hospital stays. 2 CLABSIs are linked to a 2.75-fold increased risk of in-hospital death 3 as well as an additional $32 000 in variable inpatient costs. 4
Chlorhexidine gluconate (CHG)-containing dressings to cover and protect catheter sites have been developed to reduce the risk of CLABSIs and other catheter-related infections (CRIs). These dressings maintain continuous antimicrobial activity on the skin at an insertion site, and therefore offer greater effectiveness than the transitory application of skin antiseptic agents prior to a catheter insertion or dressing change in standard care. A number of comparative clinical studies have reported favorable results in reducing CRIs with the use of CHG dressings compared to conventional site care for both hemodialysis patients and patients in the intensive care unit (ICU).5 -10 Multiple randomized controlled trials have reported similar findings, with a meta-analysis by Wei et al finding a risk ratio of 0.60 (95% CI, 0.42-0.85) for central venous catheter related bloodstream infections (CRBSIs) and a risk ratio of 0.46 (95% CI, 0.36-0.58) for catheter colonization. 11
Most studies have used either a transparent CHG gel-integrated film5 -7,10 or an opaque CHG sponge dressing to investigate the effectiveness of CHG dressings. 12 However, there is limited literature available to compare the two dressings directly on clinical and economic evidence. The CHG gel all-in-one dressing contains a transparent hydrophilic gel with 2% CHG integrated in a semipermeable transparent polyurethane dressing that absorbs fluid and allows skin inspection. The CHG sponge dressing consists of a CHG-impregnated sponge that needs to be secured in place and covered with an additional polyurethane dressing over the insertion site. Eyberg et al and Kohan et al found that the CHG gel dressing was preferred due to ease of application, removal, compliance, monitoring, and training compared to the CHG sponge dressing.13,14 Bashir et al also provided data to support the CHG gel and CHG sponge dressings reduced bacterial count on the skin similarly on days 1 and 4, but the CHG gel dressing maintained more robust suppression compared to the CHG sponge dressing on day 7. 15 A study by Maki et al compared the CHG gel and CHG sponge dressings in an in vitro time-kill study and in human volunteer in vivo studies, showing that both provided excellent long-term surface antimicrobial activity and cutaneous floral suppression and that the CHG gel dressing provided superior prevention of floral regrowth on prepped skin sites and progressive kill on unprepped sites. 16 Clinical studies have shown that both types of dressings are effective at reducing the rates of major CRI and CRBSI.6,12,17,18
As new evidence is being added to the existing literature, the medical guidelines for CLABSI prevention have evolved over time. In 2011, the CDC issued guidelines for the prevention of intravascular catheter-related infections and mentioned the use of CHG-sponge dressing if the rate of infection is not decreasing despite adherence to other strategies. 19 After a number of studies showing convergent results, the recommendation to incorporate CHG gel dressings in a prevention bundle as the standard care or as an addition has become more predominant in contemporary medical guidelines.20 -25
Only one publication comparing CHG gel dressing and CHG sponge dressing use in relation to CLABSI is available. This extensive 11-year-long real-world study demonstrated a sustained reduction of CLABSI as CHG sponge dressings were gradually replaced with CHG gel dressings. 26 Although this reduction in CLABSI rates was observed, statistical significance was not established. More real-world data are needed to shed light on the comparative effectiveness of CHG gel and CHG sponge dressings in terms of CLABSIs, as well as the associated clinical and economic implications. These findings will likely inform future development and utilization of CHG dressing products. This retrospective comparison using a nationwide, deidentified healthcare database with data from 217 US hospitals is the first to compare the 2 dressings on clinical and economic outcomes, including CLABSI, hospital length of stay (LOS), hospitalization costs, and contact dermatitis.
Materials and Methods
This study utilized data from the Premier Healthcare Database (PHD) of inpatients between January 1, 2019 and September 30, 2020, who had central venous catheters (CVCs). The PHD is a real-world database that has been collecting data since 2000 from a diverse population of over 231 million patients from >1041 healthcare systems. 27 The study utilized de-identified clinical data. Our institutional IRB determined that informed consent was waived and that this study did not require their oversight (IRB #23-003), in accordance with the Code of Federal Regulations, title 45 CFR 46 for this type of data. 28
Pre-identified CPT codes (eTable 1) were used to select hospitalized patients with CVCs (n = 53 149, 834 CLABSIs) from 217 hospitals. A total of 14 488 CVC inpatient cases that exclusively received a transparent film dressing with an integrated CHG gel pad (3M™ Tegaderm™ CHG Chlorhexidine Gluconate I.V. Securement Dressing, 3M, St. Paul, MN) were identified as the CHG-Gel cohort. A set of 38 661 CVC cases were assigned to the CHG-Sponge cohort which received an opaque CHG sponge dressing (BioPatch™, Ethicon, Johnson & Johnson Surgical Technologies, Somerville, NJ) as the only CHG dressing.
Patient and Facility Characteristics Between the CHG-Gel and CHG-Sponge Cohorts Before and After Propensity Score Matching.

COPD = chronic obstructive pulmonary disease; AIDS/HIV = acquired immunodeficiency syndrome/human immunodeficiency virus; ICU = intensive care unit; CCI = Charlson Comorbidity Index; RCC = ratio of costs-to-charges (to pay hospitals for services exempt from DRG payment); SD = standard deviation.
The CHG-Gel cohort was propensity score matched 1:1 to the CHG-Sponge patients on 33 patient and facility characteristics (Table 1), including patient demographics, comorbidities (eTable 2), and ICU admissions; facility size, type, and geographics. Patient comorbidities were assessed based on ICD-10 diagnosis codes presented at admission. These diagnosis code sets were primarily derived from the Charlson Comorbidity Index (CCI) algorithm. 29 The patient’s CCI score was calculated by examining the one-year medical history of ICD-10 codes leading up to admission. Information regarding facility attributes was extracted from healthcare provider-level data.
A Comparison of CLABSI Incidence, Hospital Length of Stay, and Hospitalization Costs Between CHG-Gel and CHG-Sponge Cohorts.

CI = confident Interval; CLABSI = central line associated bloodstream infection.
This matching process aimed to incorporate a comprehensive array of covariates associated with the endpoints, encompassing important risk factors for CLABSIs such as asthma, 30 liver disease,31,32 cancer,33 -35 peripheral vascular disease,36,37 rheumatic disease, 38 renal disease,31,35 blood disorders, 33 ICU admission, 39 and CCI score.31,40 -42 This approach was employed to enhance the precision of the estimate of endpoints. The process for selecting the final sample, comprising 28 976 individuals with 411 CLABSIs from 207 hospitals, was illustrated in a flow diagram (Figure 1).

Flow diagram for selection of the CHG-Gel and CHG-Sponge cohorts.
CLABSI, LOS, hospitalization cost, and contact dermatitis were selected as endpoints to compare the effectiveness of the 2 CHG dressings. CLABSI (T80.211A) 43 and contact dermatitis (L23-L25) were identified by scanning the post-admission ICD-10 diagnosis codes. CLABSI and contact dermatitis rates were measured as the percentage of hospitalizations where a diagnosis of CLABSI or contact dermatitis was recorded during a hospital day. LOS and costs were extracted from the PHD’s inpatient billing and utilization data representing the recorded hospital days and estimated cost of care per hospital stay.
The 2 matched cohorts were compared on CLABSI and contact dermatitis rates, LOS, and hospitalization costs, using logistic, Poisson, and linear mixed-effect regression, respectively. To account for potential clustering effects arising from patient case mix and hospital differences, the models included random effects for Diagnosis-Related Group (DRG) and hospital in the intercept. The DRG system hierarchically classifies hospital cases exhibiting analogous resource utilization patterns grounded in clinical coherence, accounting for factors like organ systems, surgical procedures, and critical care. All statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC).
Results
The cohort comparison before and after matching on the 33 characteristics is displayed in Table 1. The CHG-Gel cohort was composed of 52.2% male patients, 55.5% elderly patients (age ≥ 65), 70.7% white patients, and 86.2% of the cohort was managed in urban hospitals. These patients showed severe comorbidities with an average Charlson Comorbidity Index = 4.8 ± 3.6, with high percentages of patients having blood disorders (63.3%), diabetes (45.5%), renal disease (42.4%), and congestive heart failure (42.3%). Prior to implementing propensity score matching, a statistical disparity (P < .05) was evident between the 2 cohorts across 26 of the 33 assessed characteristics. After the propensity score matching, no differences were observed between the 2 cohorts across 30 characteristics (P > .05), and the remaining differences were all <4%. While variances persisted in provider location and facility cost type, these distinctions were accounted for within the regression models through the hospital clustering effect in the intercept. Moreover, potential remaining disparities among patients that could possibly impact the risk of infections, clinical utilization and associated expenses were also mitigated through the management of patient DRG clustering.
After comparing the 2 cohorts using mixed-effect multiple regression models, a 41% decrease in CLABSI rates (OR = 0.59, 95% CI: 0.43-0.80; P = .0008) was observed with an adjusted CLABSI rate of 0.35% for CHG-Gel compared to 0.60% for CHG-Sponge (Table 2, Figure 2). A 0.4-day shorter hospital stay was observed in the CHG-Gel cohort, with a mean length of stay of 9.53 days, compared to 9.90 days in the CHG-Sponge cohort (P = .0001) (Table 2, Figure 3). The hospitalization costs were decreased by $3576 per hospital stay in the CHG-Gel cohort (P = .0179), where the mean cost was $40 197 for CHG-Gel and $43 774 for the matched CHG-Sponge patients (Table 2, Figure 4). The 2 CHG dressing cohorts showed similar contact dermatitis rates (0.20%vs 0.18%, P = .7854) (Table 2, Figure 5).

Central line associated bloodstream infection incidence comparison.
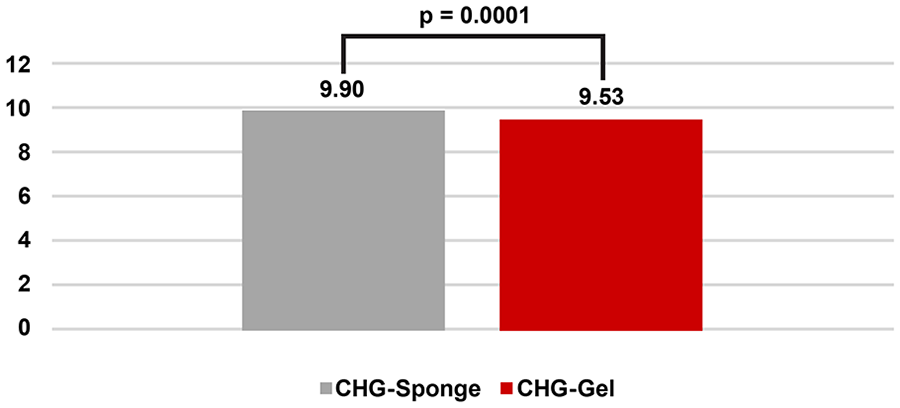
Hospital length of stay (days) comparison.

Hospitalization cost comparison.

Contact dermatitis incidence comparison.
Discussion
Patients with intravascular devices, such as CVCs, are at high risk of acquiring a bloodstream infection. One of the common causes of these infections is the migration of skin organisms from the insertion site into the cutaneous tract.44,45 Studies have shown that 5% to 25% of intravascular devices are colonized at removal; central venous catheter colonization rate is as high as 18%.46,47 As the United States Centers for Medicare and Medicaid Service ceased reimbursing for preventable healthcare-associated infections in 2008,48,49 health-care institutions have sought effective strategies to prevent CLABSIs. In 2011, the CDC recommended use of CHG sponge dressings for CLABSI prevention, though this recommendation was updated in 2017 to include CHG gel dressings.19,25 However, none of the contemporary studies have directly compared the rates of intravascular catheter-related infections or product-related adverse events as well as the economic impact between CHG gel and CHG sponge dressings. 25 The present study compared the effect of CHG gel and CHG sponge dressings on CLABSI, clinical utilization, cost of care and contact dermatitis in an inpatient setting using retrospective data from a nationwide, deidentified healthcare database. Results indicated that CHG gel dressing use had decreased CLABSI by 41%, reduced LOS by 0.4 day, and resulted in cost savings of $3576 per hospitalization compared to CHG sponge dressings.
Organisms that cause catheter related bloodstream infections originate mostly from the insertion site.26,44,45 The integrated aqueous CHG gel pad offers the advantage of CHG contact without requiring moisture, whereas the CHG sponge relies heavily on skin moisture to release CHG at the insertion site. The integrated design of the CHG gel dressing facilitates more accurate placement compared to the CHG sponge dressing. Eyberg et al and Kohan et al have both suggested that the utilization of CHG sponge dressings might not be intuitive.13,14 Despite adequate education and training, the risk of improper sponge application persists. Safdar et al and colleagues drew attention to the errors in CHG sponge placement and dressing disruption. Even after more than a decade of using the dressing in their health institution, instances of incorrect sponge placement were still observed. 50
The overall rate of CLABSIs exhibited a modest occurrence of 1.5% in our study population, which was in line with previous large, randomized trials where < 3% major-CRIs were expected.6,12 Despite this low infection rate, use of CHG gel dressing was associated with a 41% reduction in CLABSI. The observed decrease in CLABSI incidents associated with CHG gel dressings might stem from improper placement of CHG sponge dressings, if the CHG sponge dressings maintain antimicrobial activity as proficiently as CHG gel dressings when properly applied throughout the entire catheter dwell time. Such misapplication could potentially compromise dressing adherence, diminish skin contact, undermine the optimal release of CHG, and impede the suppression of skin flora regrowth. Our findings have highlighted the tangible efficacy of CHG gel versus CHG sponge dressings in a real-world inpatient context.
The COVID-19 pandemic has exacerbated challenges associated with CLABSIs, as hospitals confronted surging patient volumes, urgent care demands, and staffing and supply scarcities. Device utilization and CLABSI cases notably rose in 2020.51 -53 The pandemic strained central line management in hospitalized patients, resulting in decreased adherence to routine CLABSI prevention practices. 51 Remarkably, the first quarter of 2021 saw a substantial 45.3% CLABSI rate increase in 3394 US hospitals compared to the corresponding period in 2019. 54
Preceding the pandemic, the disruption of dressing integrity had already emerged as a substantial concern with up to 67% of catheter dressings required untimely replacement due to soiling or detachment, thereby concurrently elevating the risk of colonization associated with catheter removal. 55 Meanwhile, the CHG gel dressings have been found to have about 30% fewer dressing disruptions when compared to their CHG sponge counterparts. 18 During the pandemic, the inherent advantages attributed to CHG gel dressings in terms of ease of application, enhanced compliance, and superior dressing adhesion might have amplified the difference in CLABSI rates that we observed.
A cost saving of $3576 and a 0.4-day shorter LOS per hospitalization were observed with CHG gel dressing use compared to CHG sponge dressings, supporting the reduced incidence of CLABSI and related clinical expenses linked to the use of CHG gel dressings in hospitalized patients. The clinical and economic benefits associated with CHG gel dressings might extend beyond the scope of CLABSI mitigation, potentially encompassing a decreased risk for CRIs that were not specifically classified as CLABSI—a facet not examined within the present study. Additionally, the CHG sponge dressings conceal the insertion site whereas the CHG gel dressings allow for direct visual access to and ongoing monitoring of the insertion site. This capability is particularly significant as it likely played a crucial role in optimizing healthcare resources and thereby enhancing the quality of care during a challenging time period.
Contact dermatitis had overall lower incidence (≤0.2%) in the study population compared to previous published data (0.53%−1.1%).6,12 It is possible that contact dermatitis has been reduced considerably after the launch of improved breathable versions of the dressings. 17 Between 2008 and 2019, there were a series of improvements made to the CHG gel dressing used in this study, particularly in terms of better moisture management and catheter securement. CHG gel dressing and CHG sponge dressing were found to have similar rates of contact dermatitis after 2012.6,26 The comparison on contact dermatitis in this study echoed previous reports that the 2 dressings were similar in adverse reactions.17,26
The impact of human factors on the clinical and economic value of medical devices has gained momentum given the increasing emphasis on evidence-based practice. Our study highlighted the importance of weighing functionality and practicality in designing the optimal CLABSI prevention regimen. Such an approach holds the potential to optimize the advantages offered by current or prospective technologies, ensuring their maximal efficacy and benefits.
The present study relied on retrospective real-world data and is the first to compare the clinical and health economic impact of the 2 dressings. There are limitations to this study that could have influenced the results and interpretation. The study was constrained by the absence of data regarding CVC types, line days, and patient wards. The additional information could have further assisted in balancing the patient risk between cohorts. The lack of consistent microbiological criteria to define CLABSIs due to missing lab data may have led to an underestimation of CLABSI incidence. While the study incorporated hospital and DRG as random intercepts in the models to account for variances linked to service types and hospital-specific attributes, it is important to acknowledge that the cost structures across hospitals were not standardized. This absence of uniformity might have introduced additional uncertainty in estimating cost savings. Unfortunately, our findings could not be cross-validated due to the limited evidence available on the comparison of dressings. We were also unable to obtain data on dressing disruption and catheter colonization for the 2 types of CHG dressings, which are critical factors for future research to explore in-depth to confirm their correlation.
Conclusion
Hospitalized patients with intravascular devices are at high risk of acquiring CLABSIs. The present study provides real-world data demonstrating a population-based estimate of clinical and economic advantages of CHG gel dressing over CHG sponge dressing. The findings suggest that the use of transparent CHG gel dressings significantly reduce CLABSIs and clinical costs when compared to opaque CHG sponge dressings for hospitalized patients. Moreover, there appears to be no significant difference in contact dermatitis rates between CHG gel and CHG sponge dressings. Therefore, healthcare providers may consider using CHG gel dressings as a standard practice for patients with CVCs to reduce the risk of infections and improve patient outcomes.
Supplemental Material
sj-docx-1-inq-10.1177_00469580231214751 – Supplemental material for Comparative Effectiveness of 2 Chlorhexidine Gluconate-Containing Dressings in Reducing Central Line-Associated Bloodstream Infections, Hospital Stay, and Costs
Supplemental material, sj-docx-1-inq-10.1177_00469580231214751 for Comparative Effectiveness of 2 Chlorhexidine Gluconate-Containing Dressings in Reducing Central Line-Associated Bloodstream Infections, Hospital Stay, and Costs by Yuefeng Hou, Leah Griffin, Stéphanie F. Bernatchez, Joseph Hommes, Tarja Kärpänen and Maria Palka-Santini in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
The authors would like to thank Julie M. Robertson, PhD (3M) for providing editorial support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of 3M and contributed to writing this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 3M.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
