Abstract
The response by the biotech and pharmaceutical sectors to the COVID-19 pandemic has been historically unprecedented. Vaccines based in cutting-edge technologies, such as the mRNA platform, were invented, tested, and distributed to patients in less than a year. Yet politicians and activists argue that patents and other intellectual property (IP) have impeded the development and distribution of these vaccines. In explaining why this is profoundly mistaken, this essay first describes the medical and economic uncertainties inherent in the production of vaccines, especially those made in response to an emerging infectious disease like COVID-19. This makes clear the unprecedented achievement in the mass production and distribution of COVID-19 vaccines in less than 1 year after the start of the COVID-19 pandemic. It then describes the current manufacturing and licensing landscape, which was created on the basis of a market infrastructure built by reliable and effective patent rights. There is now a glut in the global supply of vaccine doses—and billions more doses are still being produced. The essay concludes by identifying the non-IP causes impeding global vaccine distribution, such as lack of infrastructure in the developing world, as well as regulatory restrictions and trade barriers. Those concerned with global vaccine equity should focus on policies to resolve these real-world problems.
The issue of global vaccine equity during the COVID-19 pandemic is well known among public health specialists, legal scholars, and policymakers who have been discussing and debating the appropriate legal and regulatory responses to the COVID-19 pandemic with respect to patent and other IP protections on vaccines and other therapeutic treatments.
This essay presents data and sets forth a novel, evidence-based explanation for why a waiver of patents on COVID-19 medical treatments is unnecessary for global distribution of vaccines, and that a waiver does not in fact address the real causes for the lack of global vaccine equity.
This essay has real-world implications for current policy debates concerning the legal and regulatory responses to the COVID-19 pandemic, including explaining why the World Trade Organization (WTO) should reject any further extension of the patent waiver and why the U.S. government should not implement the recently adopted WTO patent waiver in U.S. domestic law.
Introduction
By any historical standard, the response by the biotech and pharmaceutical sectors to the COVID-19 pandemic was unprecedented. Vaccines based in cutting-edge technologies, such as the mRNA platform, were invented, tested, and distributed to patients in less than a year. The World Health Organization (WHO) officially declared that the novel coronavirus constituted a public health emergency of international concern in late January 2020, and the first COVID-19 vaccines were distributed to patients by December 2020.
Yet, there remain continuing concerns about equitable worldwide distribution of these vaccines. By September 2021, 55% of the populations in developed countries had been inoculated, but less than 1% in developing countries. 1 By June 2022, the number of individuals who have received at least one vaccine dose increased to over 80% of populations in developed countries and to approximately 62% and 18% of populations in lower-middle-income countries and low-income countries, respectively. 2
The lower vaccination rates for developing countries raise valid policy concerns. Global vaccine access is especially important when combating a highly communicable infectious disease like COVID-19. If permitted to fester uncontrolled in any location in the world, however remote, this virulent pathogen continues to threaten disruption to the entire world.
The differences in vaccination rates between developed and developing countries has prompted a policy debate and international action concerning the causes of this disparity. Some politicians and activists blame patents, prompting ultimately the World Trade Organization (WTO) to adopt a waiver of patent rights on COVID-19 vaccines under international treaties. This is profoundly mistaken. Before the WTO patent waiver was adopted, a spokesperson for the European Union stated that “there is no evidence that IP rights in any way hamper access to COVID-19-related medicines and technologies.” 3 Yet, billboards appeared last year in Washington, D.C., and in major cities around the world proclaiming “Patents Kill” in promoting global vaccine distribution.
The WTO patent waiver is based on a profound misunderstanding about the role of patents in facilitating the invention and mass production of COVID-19 vaccines. In fact, there is now a global glut in the supply of vaccine doses. The blockades to global distribution of vaccines are found elsewhere, such as in the lack of infrastructure for distributing temperature-sensitive vaccines in the developing world and in regulatory and trade barriers.
Given the unique nature of vaccines, as opposed to standard therapeutic treatments, the response of the biotech and pharmaceutical sectors to the COVID-19 pandemic has been striking, if not downright impressive. This essay first describes the unprecedented WTO waiver on patents on COVID-19 vaccines It then details the medical and economic uncertainties inherent in the production of vaccines, especially those made in response to an emerging infectious disease like COVID-19. This makes clear the equally unprecedented achievement in the mass production and distribution of COVID-19 vaccines in less than 1 year after the start of the COVID-19 pandemic. It then describes the current manufacturing and licensing landscape for COVID-19 vaccines, which was built on a market infrastructure created by reliable and effective patent rights. The essay concludes by identifying the non-IP causes impeding global vaccine distribution, such as lack of infrastructure in the developing world. Those concerned with global vaccine equity should focus on policies to resolve these real-world problems.
The WTO Patent Waiver on COVID-19 Vaccines
India and South Africa first proposed in October 2020 that the WTO adopt a waiver under international treaties of IP rights in all COVID-19 medical treatments.
Notably, India and South Africa’s proposal was made only 8 months after the World Health Organization (WHO) declared COVID-19 to be a global pandemic—before any vaccines were fully tested for efficacy and approved by regulatory authorities for delivery to patients. With a large generic drug industry, India is known as the “pharmacy of the world,” supplying more than one-fifth of the total production of generic drugs worldwide. 4 Thus, it was unsurprising that India proposed a waiver of IP protections for all therapeutics and vaccines for COVID-19, as it has argued for similar waivers of IP rights in the past.
In a surprise announcement in May 2021, the Biden Administration declared its support for a patent waiver on COVID-19 vaccines, and negotiations began in earnest at the WTO. On June 16, 2022, the WTO approved a revised version of the original waiver covering only patent rights on vaccines. The WHO will consider in December 2022 whether to extend this waiver to patents on diagnostics and therapeutic treatments for COVID-19.
In prior global health emergencies, such as the AIDS epidemic, countries negotiated under existing international treaties special processes for compulsory licensing of patent rights to therapeutic treatments. 5 These past agreements at the WTO were confusingly referred to as “waivers,” but they implemented or amended processes and legal powers already provided for in existing international treaties. 5 The waiver for patents on COVID-19 vaccines is truly unprecedented.
Unique Market Challenges With Emerging Infectious Diseases and Vaccines
To understand the significant and unprecedented response by the biopharmaceutical industry to the COVID-19 pandemic, it is first necessary to understand the context in which biopharmaceutical companies and healthcare professionals respond to emerging infectious diseases. By their nature, emerging infectious diseases pose unique challenges that are unlike other human diseases for which biopharmaceutical innovators work to invent, produce, and deliver medical countermeasures. These challenges are important because they have resulted in a more limited commercial infrastructure in the development of vaccines as compared to therapeutic treatments for conditions like cancer, hypertension, or HIV.
Unlike with emerging infectious diseases, endemic diseases like HIV, MRSA, or hepatitis C have a known prevalence and incidence rate that make it possible for companies to plan manufacturing and forecast revenue to fund operational expenses, including funding ongoing research and development of new therapeutic treatments. By contrast, an emerging infectious disease has an uncertain magnitude and unclear duration, especially in the first stages of the outbreak. In the initial stages of the prior outbreaks of both SARS and MERS, for example, public health authorities believed there was a serious potential that either infectious disease could become a global pandemic, but it did not occur.
Even when an emerging infectious disease does result in a pandemic, this uncertainty can continue for several years after the initial outbreak. COVID-19 has exemplified many of these uncertainties, such as in the rapidity of the evolution of mutations and variants, and the resulting uncertainties about virulence and vaccine efficacy against these new variants.
It is a truism in economics that uncertainty undermines the incentives to make long-term investments in research and development, as well as reduces incentives to create capital-intensive manufacturing facilities and distribution infrastructure. This is especially true given the opportunity costs of creating therapeutic treatments for known medical conditions and their countermeasures. For example, when Moderna developed the COVID-19 vaccine in January 2020, the company considered this a very risky decision due to the lack of information about the virulence or trajectory of COVID-19, the lack of existing funds for the complete development and testing of the vaccine, and concerns about its ability to raise funds in this fundamentally uncertain medical and commercial environment. These were not merely concerns for startups like Moderna. 6 Pfizer reported similar concerns about the risks. 6
Heightening these concerns about uncertainties in response to emerging infectious diseases are basic biomedical and market differences in the production and delivery of vaccines as compared to standard therapeutic treatments. Medications for chronic, non-communicable diseases, such as hyperlipidemia or hypertension, are often taken daily by a substantial proportion of the population. In contrast, vaccines may result in only a single lifetime dose or possibly a single dose or several doses taken annually. As a result, despite similar costs in the research and development of vaccines as in standard therapeutic treatments, vaccines result in less revenue for biopharmaceutical companies. In 2007, vaccines accounted for just 2.2% of biopharmaceutical revenues. 7
As a result of these biomedical and market uncertainties, shortages in vaccine supplies are commonplace, which dramatically evidence the difficulties in the infectious disease market. There have been serious shortages in the supply of classic and well-known vaccine staples, such as influenza vaccine in 2004 and tetanus-diphtheria vaccine in 2017, despite there being a relatively stable and large market for these vaccines. Myriad other shortages have occurred in the U.S., ranging from Hib to yellow fever to pneumococcal conjugate vaccines. Since 2000, there have been at least 9 shortages of childhood vaccines, another relatively stable and large market. 7
These were the economic conditions and existing market structure for vaccines when SARS-CoV2, the virus that causes COVID-19, emerged out of China in late 2019 and early 2020. The SARS-CoV2 is a coronavirus, which is the same viral family as the virus that causes the common cold. The viruses that caused SARS and MERS, which emerged in humans in 2003 and 2012, respectively, are also in the coronavirus family, and these 2 viruses have much higher mortality rates compared to COVID-19. Yet, in the 2 decades since the emergence of SARS, there was no commercial development of any human coronavirus vaccines, coronavirus antivirals, or coronavirus monoclonal antibodies. Everyone knows the classic cliché, “there is no cure for the common cold,” and this aphorism seemed to apply to the entire coronavirus family, including even to MERS and SARS. This was the biomedical and commercial reality at the outbreak of the COVID-19 global pandemic in early 2020.
The Creation and Development of Vaccines in Response to COVID-19
The invention, development, testing, and rollout of vaccines in response to the COVID-19 pandemic was the fastest in global history. The delivery of vaccine doses to patients in the developing world has lagged behind the developed world, but this was not caused by patents or other IP rights. There is no evidence that patents have blocked the research, development, or distribution of any vaccines for the treatment of COVID-19. The evidence is to the contrary: patents prompted the investment of billions of dollars over several decades in research and development efforts, as well as in creating a manufacturing and commercial infrastructure in the biopharmaceutical sector, that made possible the COVID-19 vaccines.
These investments were made possible given the reliable and effective patents rights secured in biotech innovations in the U.S. since the early 1980s. 8 The result was the biotech revolution, driven by an explosion in biotech startups from Genentech in the early 1980s to Moderna today. The U.S. accounts for approximately 5% of the world’s total population and roughly 25% of global economic output, but over 50% of all new biotech innovations are created in the U.S. 9
This technological and commercial foundation was key to the biopharmaceutical sector’s response to COVID-19. There was some work by government scientists on the basic research, but it was patent-based venture capital investments and licensing that were the foundation of new biotech companies, like BioNTech and Moderna, that developed the mRNA platform to create the first COVID-19 vaccines in early 2020. Two decades earlier, many in the scientific community believed mRNA-based vaccines were an impossible pipedream. 6 After Chinese researchers published the genome of SARS-CoV2 on January 11, 2020, Moderna created its vaccine using its mRNA technology in 2 days. 10 BioNTech was even faster: researchers created its COVID-19 vaccine in several hours. 11
Prior to the COVID-19 pandemic, the fastest a vaccine had ever been invented and developed was 4 years, 12 and the average timeline for vaccine development was between 5 and 10 years. 13 Instead of years, it took only a few weeks for Moderna and BioNTech to deliver their vaccines to the U.S. Food & Drug Administration (FDA) in early February 2020 to start the regulatory approval process 1 month before the WHO officially declared COVID-19 to be a global pandemic. A year later, the U.S. government identified that, in addition to Moderna, Pfizer, Johnson & Johnson, and other vaccines, several more vaccines developed by Sanofi, GSK, and others were in various phases of the FDA’s testing process. 14
Patents functioned in more ways than merely serving as the mechanism for the venture capital investments that funded the research and development of the technologies that led to the mRNA-based vaccines. As property rights, patents have been the basis for the commercial development of the biopharmaceutical sector itself. Just as uncertainty undermines incentives to invest, reliable and effective property rights are the subject matter of contracts and other commercial agreements that drive growth in innovation economies.15,16
Thus, patents have been the basis of innumerable commercial agreements for decades in the biopharmaceutical sector. These economic activities created efficiency-maximizing supply chains, licensing of products and services to manufacture and distribute new therapeutic treatments, and ultimately formed a knowledge infrastructure in the growing biopharmaceutical sector. This preexisting commercial foundation served as the basis for the many cross-industry and cross-sector agreements that made possible the unprecedented response to the COVID-19 pandemic. 17
By May 2020, less than half a year after the emergence of a previously unknown novel coronavirus eventually classified by the WHO as SARS-CoV2, the biopharmaceutical sector was researching and developing 430 unique active compounds to treat the COVID-19 illness. 18 This number has almost doubled in 2 years. As of June 2022, there are now 854 unique active compounds under development, including 240 vaccines, 261 antivirals, and 353 therapeutic treatments. Almost half of these compounds under development (415) are by companies based in the U.S. (Figure 1). 19
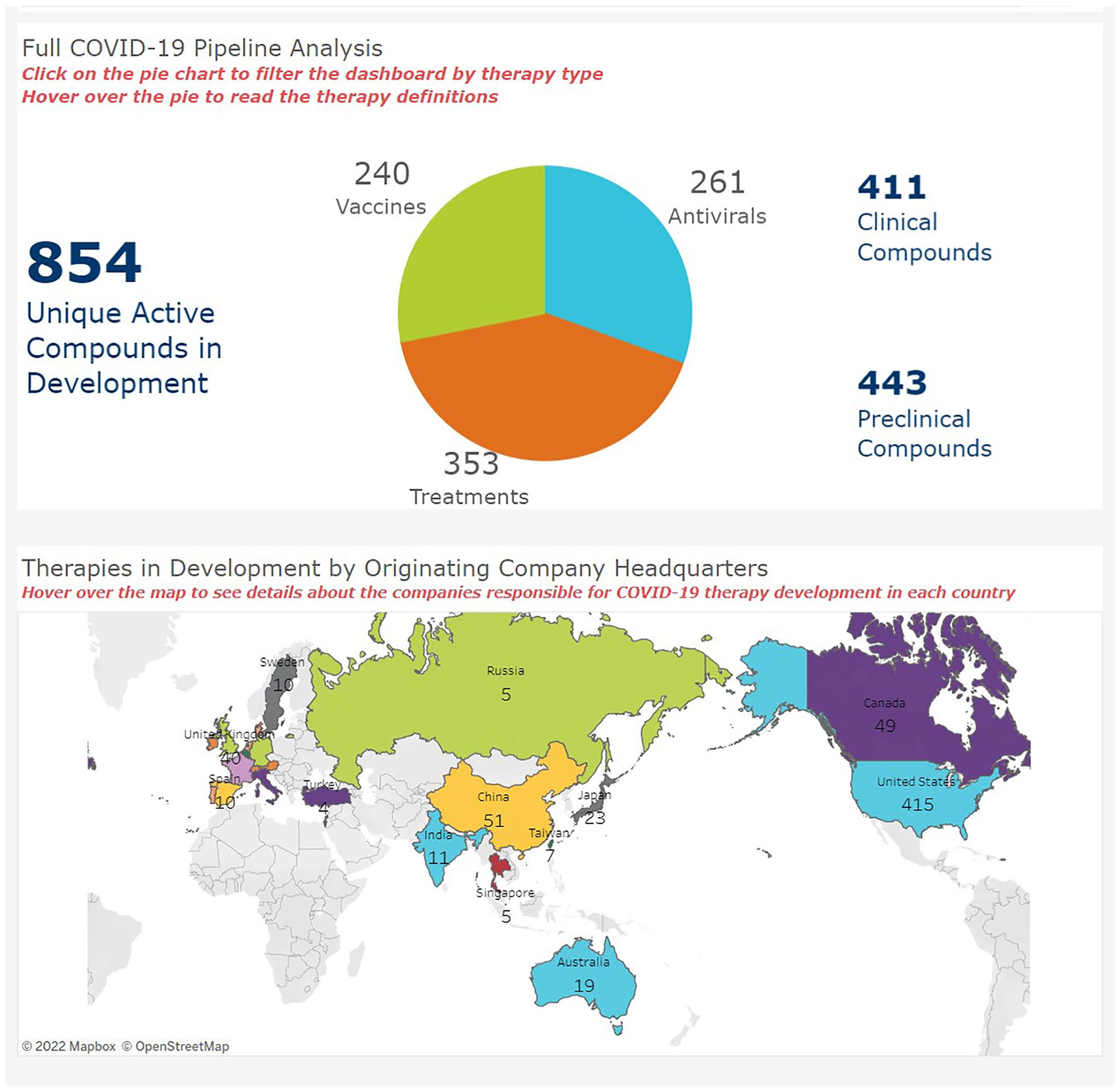
BIO COVID-19 Therapeutic Development Tracker (as of June 25, 2022). 19
The patents (and other IP rights) that have served as the basis for creating the complex global commercial and information-sharing infrastructure in the modern biopharmaceutical sector did more than incentivize research and development in vaccines. They also served as the basis for many commercial agreements to expand manufacturing capacity of vaccines (Figure 2).

The network of patent license agreements for only the mRNA vaccines as of May 2021. 20
One of the more well-known examples includes the license agreement BioNTech and Pfizer, which efficiently combined BioNTech’s innovation with Pfizer’s existing capital capabilities to quickly scale final development and manufacturing of vaccine doses. Other lesser-known licensing agreements entered into in 2020—the first year of the global pandemic—include AstraZeneca licensing the Serum Institute of India to manufacture its vaccine, Johnson & Johnson licensing Merck to manufacture its vaccine, and BioNTech-Pfizer licensing Novartis and Sanofi to manufacture its mRNA vaccine doses, among others. In total, there have been approximately 379 production and manufacturing agreements entered into between private and public entities for COVID-19 vaccines with over 50 of these agreements in the developing world.
The Production and Delivery of Vaccine Doses in Response to COVID-19
Governments around the world, including in the U.S., assumed sole responsibility for purchasing and delivering all vaccine doses in response to the COVID-19 pandemic. In 2020, governments in the developed world entered into advance-purchase agreements to secure supply of vaccine doses. These advance purchase agreements provided early-stage funding for biopharmaceutical companies to develop or scaleup their manufacturing facilities, especially for the unique manufacturing of the new mRNA-based vaccines. In the U.S., Operation Warp Speed was adopted in addition to advance-purchase agreements by the federal government to further subsidize late-stage FDA testing of the vaccines, as well as the large-scale manufacture of vaccine doses. Although some biopharmaceutical companies declined these subsidies for late-stage development and manufacture of vaccines—notably, Pfizer/BioNTech was not a participant in Operation Warp Speed—these additional government subsidies expanded production capabilities even more. 21 Many biopharmaceutical companies also shifted their existing manufacturing facilities used to make other vaccines and therapeutic treatments to the production of COVID-19 vaccine doses. COVID-19 vaccine production, at least for the extremely difficult and complex production of the mRNA vaccines, is at 100% capacity. 22
As a result, approximately 12 billion vaccine doses were produced worldwide in 2021—almost double the global population. 23 For 2022, total vaccine production is expected to top 24 billion doses. 24 There is now a worldwide glut of vaccine doses. In April 2022, the Serum Institute of India halted production of COVID-19 vaccines as a result of this supply glut. 25 Some governments are now declining the delivery of new vaccine doses. In November 2021, South Africa suspended all delivery of vaccine doses from Pfizer and Johnson & Johnson due to a supply glut and lack of demand for its existing stockpile of vaccines. 26
The delivery to patients of these billions of vaccine doses has been as equally unprecedented as the speed in which they were created and developed. In less than 2 years, more than 14 billion vaccine doses have been given to patients worldwide with 89% of adults worldwide receiving at least one vaccine dose. 2
Disparities in Global Vaccine Deliveries are Not Caused by Patents
Admittedly, the delivery of vaccine doses has not been as widespread in developing countries as it has been in developed countries; as of June 2022, approximately 4.8 billion doses were delivered to patients in lower-income and lower-middle-income countries out of the 14 billion total doses delivered worldwide. 2 But this has nothing to do with patents or any other IP rights. The causes are myriad, ranging from regulatory and trade barriers, lack of distribution infrastructure in the developing world, to vaccine hesitancy in some populations.
To prevent a repeat of the delays in delivery of vaccines to developing countries that occurred during the 2009 H1N1 influenza pandemic, the Coalition for Epidemic Preparedness Innovation (CEPI), the WHO, and several other partners developed the COVAX program. This program was designed to pool funds from participating countries to secure advance purchases of vaccine doses. The COVAX program faltered, but not due to patents or other intellectual property rights. Rather, COVAX failed due to classic organizational and legal problems that occur in coordinating multi-national efforts. This included lack of funding for the program. Governments in developed countries also engaged in “vaccine nationalism” by prioritizing the advance purchase of vaccine doses for their own populations. 21
For example, both the Trump and Biden Administrations invoked the U.S. Defense Production Act in response to the COVID-19 pandemic. This resulted in a presumptive legal mandate prohibiting exports of any materials necessary to address or respond to COVID-19, including prohibiting the export of personal protection equipment such as masks or the export of vaccine doses unused by U.S. patients. 27 Millions of doses of the COVID-19 vaccine produced by AstraZeneca that were purchased in advance by the U.S. government went unused and sat wasting away with looming expiration dates in warehouses for well over a year. 28 The reason was that the FDA did not approve the AstraZeneca vaccine for use in the U.S. due to the vaccine not meeting the FDA’s standards for efficacy. The AstraZeneca vaccine was approved for use in tens of other countries, including many in the developing world, but the trade barriers erected by the U.S. government prevented these countries from receiving these vital vaccine doses. Similar legal and regulatory restrictions were enacted in the EU and even in India, which prohibited the export of vaccine doses produced by the Serum Institute, all of which prevented quick and efficient global delivery of vaccine doses to the developing world.
But even if these regulatory restrictions and trade barriers had not been adopted and vaccine doses were quickly delivered to developing countries, this does not mean that they would have resulted in “shots in arms.” There remain pressing problems with lack of infrastructure in developing countries to facilitate the delivery to patients of the vaccine doses that are imported to these countries. Vaccines require storage at sub-zero temperatures, for example, which have to be maintained at the warehouses, in the delivery trucks, and in the medical offices or other facilities in which vaccine doses reside until they are delivered to a patient. Such storage requirements presented serious obstacles to the efficient and quick delivery of vaccine doses to small, rural communities in deserts or in tropical climates in developing countries.
Conclusion
Real-world solutions to disparities in global vaccine distribution should address investments in the creation and maintenance of infrastructure and vaccine distribution capacities in developing countries, removal of trade barriers to the export and import of vaccine doses produced in the developed world, and educational efforts on the efficacy and value of vaccines. The waiver of patents on COVID-19 vaccines, as well as any potential future patent waiver on diagnostics and therapeutic treatments, is unnecessary and ineffective in addressing these blockades to global vaccine equity. This waiver is also destructive of the incentives and commercial system that made possible the historically unprecedented response of the COVID-19 vaccines.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics and Consent Statement
The essay did not require an ethics board approval because it does not report a study using animal or human subjects.
