Abstract
We developed and tested the effectiveness of an algorithm to prevent medical device–related pressure injuries in intensive care unit patients. It was developed in four stages: literature review and analysis of medical records; preliminary algorithm development; validation of the preliminary algorithm by experts in two rounds; and practical feasibility verification of the revised algorithm by 109 intensive care unit nurses. To verify the algorithm’s effectiveness, we compared the incidence of medical device–related pressure injuries between 324 patients without algorithm application (control group) and 312 patients with algorithm application (experimental group). The outcomes were skin inspection of the medical device attachment, pressure injury evaluation, and implementation of pressure injury-preventive nursing care, based on the medical device type. The incidence rates were 1.46 per 100 devices (control group) and 1.19 per 100 devices (experimental group). Since there was no homogeneity in the previous score of the Braden scale in the experimental and control groups, the results regarding the incidence of pressure damage after applying the algorithm should be interpreted with care. Applying this algorithm was a safe intervention that helped prevent medical device–related pressure injuries in this population.
• Studies have investigated the current medical device–related pressure injuries status or medical device–related pressure injuries risk factors in Korea, but no studies have reported medical device–related pressure injuries prevention algorithms. • The medical device–related pressure injuries prevention algorithm may be conducive to providing safer care for intensive care unit patients by reducing the incidence of medical device–related pressure injuries. • We believe that the use of algorithm will improve the quality of nursing care.
Introduction
Medical device–related pressure injury (MDRPI) is a localized injury to the skin and tissues caused by continuous pressure inflicted by medical devices used for diagnostic or treatment purposes. MDRPIs are indicators for pressure injury management and have increasingly gained attention.1,2
A meta-analysis of 29 studies on MDRPI in inpatients showed that the estimated pooled incidence and prevalence of MDRPIs were 12% and 10%, respectively. 3 A systematic review of 13 studies reporting the incidence and prevalence of MDRPI in patients admitted to an intensive care unit (ICU) demonstrated that the incidence ranged from .9% to 41.2%, and the prevalence ranged from 1.4% to 121%, which are relatively high. 4 In a 2-year study of MDRPI prevalence among hospitalized patients, the incidence of pressure injury was 2.4 times higher when medical devices were attached than when the devices were not attached. 5 The prevention and management of MDRPIs are crucial for patient prognosis. 1
Arterial/venous catheters; gastrointestinal tubes; and antithrombotic, monitoring, assistive, oxygen-supplying devices, and all drainage tubes such as foley catheter and percutaneous catheter drainage can cause MDRPIs. 2 Risk factors for MDRPI include the use of medical devices made of hard materials, prolonged use of medical devices at the same site, simultaneous use of multiple medical devices, and inappropriate size and fixation of medical devices.5,6 The risk of MDRPI increases by 5.79-fold with endotracheal tube usage. 7 Devices that secure endotracheal tubes are usually attached to skin with little fat tissue, such as lips, cheeks, and mouth, thus leading to high risk of pressure injury. 8
Compared to patients admitted to general wards, ICU patients have many risk factors for pressure injury such as low consciousness and mobility level; malnutrition; use of pressor agents, sedatives, or muscle relaxants; and edema. 9 Furthermore, several medical devices such as mechanical ventilators, extracorporeal membrane oxygenation system, and equipment for renal replacement therapy and therapeutic hypothermia5,9 are simultaneously used in ICU patients, which increase their MDRPI risk. The MDRPI risk was 5.79, 5.79, and 5.54 times higher among semi-comatose and comatose patients and sedated patients, respectively, 7 and the risk was highest among patients receiving mechanical ventilation for more than 20 days. 6
Careful evaluation of an injury is crucial for MDRPI prevention. Examining all areas of the skin and mucosal surface potentially injured by any medical device rather than investigating only areas affected by invasive medical devices is important. 10 The affected area of the skin must be examined at least twice daily. 2 To prevent pressure injury, medical devices of appropriate size should be used and fixed properly to avoid uneven pressure application on the skin, and prophylactic dressings should be applied. 2 Regularly repositioning and lifting the device and positioning the patient in a way that minimizes pressure on the skin can reduce medical device–related pressure on the skin, while keeping the skin clean and dry can prevent MDRPI.2,11,12
The key factor associated with MDRPI prevention is the accurate assessment of the degree of skin injury caused by medical devices and appropriate management. Early evaluation is effective; however, ICU nurses occasionally fail to detect MDRPI because of low awareness or knowledge regarding MDRPI, excessive workload, and varying proficiency levels.13,14 Furthermore, when lifting medical devices to examine the skin underneath, nurses often inappropriately assess the skin because of concerns regarding medical device displacement. Moreover, MDRPI management may be inappropriate because nurses must obtain the physician’s consent or cooperation before altering the size or position of a medical device. 15 Therefore, to ensure early assessment and effective management of MDRPIs, an algorithm needs to be developed that lists reasonable, sequential activities to be followed in clinical practice. 16
A multidisciplinary and international consensus on MDRPI, held in London, United Kingdom in 2020, made evidence-based considerations of the etiology, assessment, prevention, and management of MDRPI. 17 Cooper et al. 18 suggested the preferrable materials to be used for manufacturing medical devices, size, skin assessment cycles, and dressing methods for the prevention and management of MDRPI. These findings indicate the importance of MDRPI prevention and management, but the immediate applicability of these considerations under various situations in a hospital setting is limited.
A review of studies on MDRPI management algorithms showed that MDRPI-related recommendations are outlined in the international guidelines for clinical practice developed by the National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel, and Pan Pacific Pressure Injury Alliance. 2 In their algorithm, Black and Kalowes 19 state that the medical device used should be identified, but they did not provide guidance on how to manage the device.
Purpose
In Korea, evidence-based clinical practice guidelines for the treatment of pressure injuries 12 include recommendations for the management of medical device–related injury but lack detailed MDRPI guidelines. The primary objective of this study was to develop an algorithm aimed at preventing MDRPI in ICU patients. The secondary objective was to test its effectiveness by applying the developed algorithm in an ICU setting.
Methods
We developed an MDRPI prevention algorithm for ICU patients by using a modified four-stage clinical practice guideline development strategy.
20
We changed the four-step (periodic algorithm update) to testing to verify the effectiveness of the developed algorithm (Figure 1). Development and testing of a medical device–related pressure injury prevention algorithm for patients admitted to intensive care units using a modified four-stage clinical practice guideline.
Stage 1: Review of the Evidence
We conducted literature reviews and medical record analysis to identify the evidence for the development of preliminary algorithms. We reviewed the MDRPI-related literature published till August 2019 in Korean and international journals on medicine, nursing, and health care. To identify relevant literature, we conducted a search in PubMed, Cumulative Index to Nursing and Allied Health Literature, National Guideline Clearinghouse, Research Information Sharing Service, Korea Citation Index, and Korean Medical Guideline Information Center using different combinations of the following terms: “pressure ulcer,” “pressure injury,” “pressure ulcer algorithm,” “pressure ulcer algorithm,” “pressure injury algorithm,” “pressure injury algorithm,” “medical device-related pressure ulcer,” “medical device-related pressure injury,” “equipment and supply,” “medical device,” “medical and device,” “intensive care units,” “ICU,” “nursing algorithm,” “nursing algorithm,” “critically ill patients.” The search was limited to papers that included participants aged ≥18 years and were written in Korean and English. The PubMed search terms used in the present study are presented in Figure 2. Keywords used in the search strategy.
The total number of papers retrieved by entering the search terms into each database was 241. Abstracts from these articles were reviewed. The complete paper was reviewed if it addressed MDRPI. After full-text reviews, 10 articles or guidelines were selected (Figure 3, Supplementary Appendix S1). Flowchart of the study selection process. CINAHL, Cumulative Index to Nursing and Allied Health Literature; NGC, National Guideline Clearinghouse; RISS, Research Information Sharing Service; KCI, Korea Citation Index; KoMGI, Korean Medical Guideline Information Center.
To determine the overall incidence of MDRPI and incidence by type of medical device, the medical records of 751 adult patients (age, ≥18 years) were analyzed in a 2-week period (June 1-14, 2019). Based on the analysis results of the medical records of 751 patients in the surgical cardiovascular ICU, surgical gastrointestinal ICU, neurosurgical ICU, medical ICU, and emergency ICU, 25 patients had a total of 37 MDRPIs. Pressure injuries were induced most often by an intermittent pneumatic compressor (IPC; n = 12, 32.4%), followed by endotracheal tubes (n = 8, 21.6%), oximeters (n = 4, 10.8%), high-flow nasal cannulas (n = 4, 10.8%), physical restraints (n = 3, 8.1%), nasogastric tubes (n = 3, 8.1%), tracheotomy tubes (n = 1, 2.7%), nasal cannulas (n = 1, 2.7%), and transcutaneous catheter drainage tubes (n = 1, 2.7%).
Stage 2: Constructing Preliminary Algorithm
The contents of the preliminary algorithm were constructed using the results of the literature review and the medical records analysis, which identified the medical device types to be included in the algorithm.
To construct the preliminary algorithm, four articles reporting common sites of injury and preventive measures of MDRPI19,21-23 and six guidelines containing information regarding MDRPI on websites posted on a clinical practice guideline development facility were referenced.2,10,12,24-26 Information on the methods used for MDRPI management based on the medical device type was obtained from four relevant articles.19,21-23 Among the 10 selected literature reports, if a study did not present the level of evidence and strength of recommendations,10,24,26 our research team conducted an objective evaluation of the study design and quality. To provide consistent evidence level, the level was evaluated using the criteria of the National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel, and Pan Pacific Pressure Injury Alliance, 2 which are most often reflected in the content of this algorithm. Each evidence-based element of the algorithm is shown in Supplementary Appendix (S2). The evidence leveling system described in each reference is provided in Supplementary Appendix (S3).
Six types of medical devices that cause MDRPI were identified and included in the algorithm: restraints; oximeters; endotracheal, nasogastric, and tracheotomy tubes; and IPCs or antithrombotic stockings. A detailed algorithm for each medical device type was developed. First, nurses were required to check the medical device being used and the site of attachment, keep the skin dry and clean, and provide corresponding preventive care for pressure injuries for each medical device type. If the patient had a pressure injury, then nurses were required to provide care, based on the injury stage, and to record the type of medical device involved and stage and size of the pressure injury.
Stage 3: Validation of the Αlgorithm (expert validation and practical feasibility)
The validation of the algorithm was confirmed through the two expert validations and one practicality feasibility process.
Expert Validation
In the first round, two items had an item-level content validity index of .80 or lower: “oximeter is repositioned at least three times a day” and “check if the size of the nasogastric tube is appropriate.” The item related to the oximeter was modified to change its position at least once per shift, and the item related to the nasogastric tube size was deleted. Experts recommended restructuring the medical device list, based on the body area where the device was placed and the order of devices that required more frequent evaluation. The expert opinion indicated the need for additional space to list the medical device types because devices such as nasal cannulas or high-flow nasal cannulas often cause pressure injury.
Thus, the algorithm was revised after the first round of expert validity testing. First, nurses were required to check which listed medical devices were being applied to each patient, based on the frequency of use, and then to evaluate the attachment area for signs of MDRPI and provide preventive care, as specified for the six medical device types. The final step was to ensure that the skin was dry and clean. The algorithm included instructions for methods of fixation of medical devices, based on the device type. After fixing the medical device onto a new area, dressing of the pressure injury varied, based on the stage of severity, and the results of skin assessment were recorded. If no pressure injury existed, then the nurses evaluated the skin at pre-established time intervals or changed the device position, based on the pressure injury-prevention strategies for each medical device. The nurses additionally assessed the need for prophylactic dressing and recorded the skin assessment results.
Practical Feasibility
The practical feasibility of the preliminary algorithm, modified based on the results of expert validity testing, was verified among the nurses. Verification was performed with 109 nurses (work experience >3 months) working in the surgical cardiovascular, surgical gastrointestinal, neurosurgical, medical, and emergency ICUs. Each nurse applied the algorithm to all their patients during the 1-month period and completed the practical feasibility verification questionnaire on the last day of the algorithm application.
Before the practical feasibility verification, we visited each of the surgical cardiovascular, surgical gastrointestinal, neurosurgical, medical, and emergency ICUs at least three times to train the nurses on how to use the algorithm. Relevant training materials were also provided. On average, three nurses were trained per session, and each nurse received one 20-minute session. The researcher visited the nurses’ stations immediately before their shift changed, before work initiation, and after work completion. Multiple copies of the algorithm were printed for the nurses. Two boxes, A and B, were on a table at the entry of the nurses’ station in each ICU. Nurses were instructed to take one copy of algorithm per patient from Box A at the beginning of their shift and to leave the copies of the algorithm in Box B after their shift. Algorithm implementation required approximately 5 minutes, and the copies of algorithm were retrieved on the last day of algorithm application.
The clinical feasibility questionnaire devised by Noh and Lee, 28 which comprised 13 items rated on a four-point scale, was modified, and used with the authors’ permission. In the original tool, the usefulness, convenience, accuracy, implementation, speed of work, intellectual satisfaction, and algorithm evaluation were identified with 23 items. We selected and used a total of 13 out of 16 items that assessed the usefulness, convenience, accuracy, implementation, speed of work, and intellectual satisfaction of the algorithm, except those with similar contents. The reply “not very much” was scored 1 while “very much so” was scored 4.
Results of the Verification of the Feasibility of Nurses’ Practice (N = 109).

SD, standard deviation; MDRPI, medical device–related pressure injury.
The results of practical feasibility for nurses showed that all items had more than 3 points, except for item 1, and we confirmed the testing algorithm.
Stage 4: Testing the Effectiveness of the Algorithm
To test the algorithm on patients, we examined whether the MDRPI incidence reduced after the algorithm was applied. The patients were divided into two groups: the control group, which included patients for whom the algorithm was not applied, and the experimental group, which included patients for whom the algorithm was applied. The MDRPI rates in the algorithm application were compared. In total, 636 patients were admitted to the surgical cardiovascular ICU, surgical gastrointestinal ICU, neurosurgical ICU, medical ICU, and emergency ICU at X Hospital. Of these, 324 patients were discharged from intensive care without the application of the algorithm (control) and 312 patients received application the algorithm (experimental group). The control group was surveyed January 1-31, 2020 (before), and the experimental group was surveyed February 4, 2020, to March 6, 2020 (after). We completed the effectiveness assessment questionnaire by referring to the patients' electronic medical records, which allowed us to review what the nurse in-charge wrote about the pressure injury in the medical record and to record it in the investigation paper. We excluded patients with pressure injuries at ICU admission and we included only pressure injury cases that were newly discovered during the study period. MDRPIs were specified in the medical records to ensure consistent application of the same patient’s algorithm in each nursing shift.
The structured effectiveness assessment questionnaire included items related to the patients’ general and MDRPI characteristics. General characteristics included sex, age, and medical department, and MDRPI-related characteristics such as pre-Braden Scale scores, post-pressure injury incidence by medical device type, stage of pressure injury, and area of pressure injury. The Braden Scale for pressure injuries comprises six subscales: (1) sensory, (2) moisture, (3) activity level, (4) mobility, (5) nutritional status, and (6) friction and shear. 29 The score range is from 6 to 23, and the lower the score, the higher is the risk of pressure injury. The stage of pressure injury is presented as the frequency, and the incidence of pressure injury by medical device type is presented as the frequency and percentage (number of medical devices that induced pressure injury among the total medical devices used).
The effectiveness of the algorithm was tested, and we confirmed the final algorithm (Figure 4). The final algorithm consists of checking the type of medical device being used and the attachment of the medical device, checking whether the pressure injury is present or not, and implementing pressure injury prevention care for each type of medical device. If a pressure injury occurs, the skin of the area where the medical device is to be newly attached should be assessed and preventive dressing applied in advance. Since then, the method of re-stabilizing each type of medical device has been specifically presented, such as changing its location or fixing it not too hard, and then re-stabilizing it. After fixing the medical device in the new area, the dressing was applied to the pressure injury stage of NPUAP to manage the pressure injury in the existing area and the results of the skin condition were recorded. If pressure injury did not occur, it was required to assess the skin or change the location of the medical device every set time according to the pressure injury prevention method for each type of medical device. In addition to the types of medical devices presented, medical devices being used are described under other medical devices to ensure that the skin assessment process is not omitted. The next step is to identify the need for preventive dressing. Preventive dressing may be applied if sensation is impaired, humidity is present underneath the medical device, perfusion is poor, tissue durability is altered, nutrition is poor, or if there is local or whole-body edema. Because the medical device itself causes pressure injuries, preventive dressing may be applied depending on the anatomical location or purpose or shape of the medical device. After the dressing is applied and the results of the skin condition are recorded, the skin is always kept dry and clean, and the algorithm can be used repeatedly. The final medical device–related pressure-injury-prevention algorithm.
Results
Characteristics of the Participants
No significant differences existed in general characteristics such as sex, age, and medical department, but significant differences did exist in MDRPI-related characteristics (eg, Braden Scale score) between the experimental and control groups (P<.001); the patient groups were homogenous with regard to general characteristics but not MDRPI-related characteristics (Supplementary Appendix S4).
Incidence and Stage of Pressure Injury
Comparison of the Incidence Site, Stage, and Frequency of Pressure Injury by Type of Medical Device between the Experimental Group and the Control Group (N = 636).
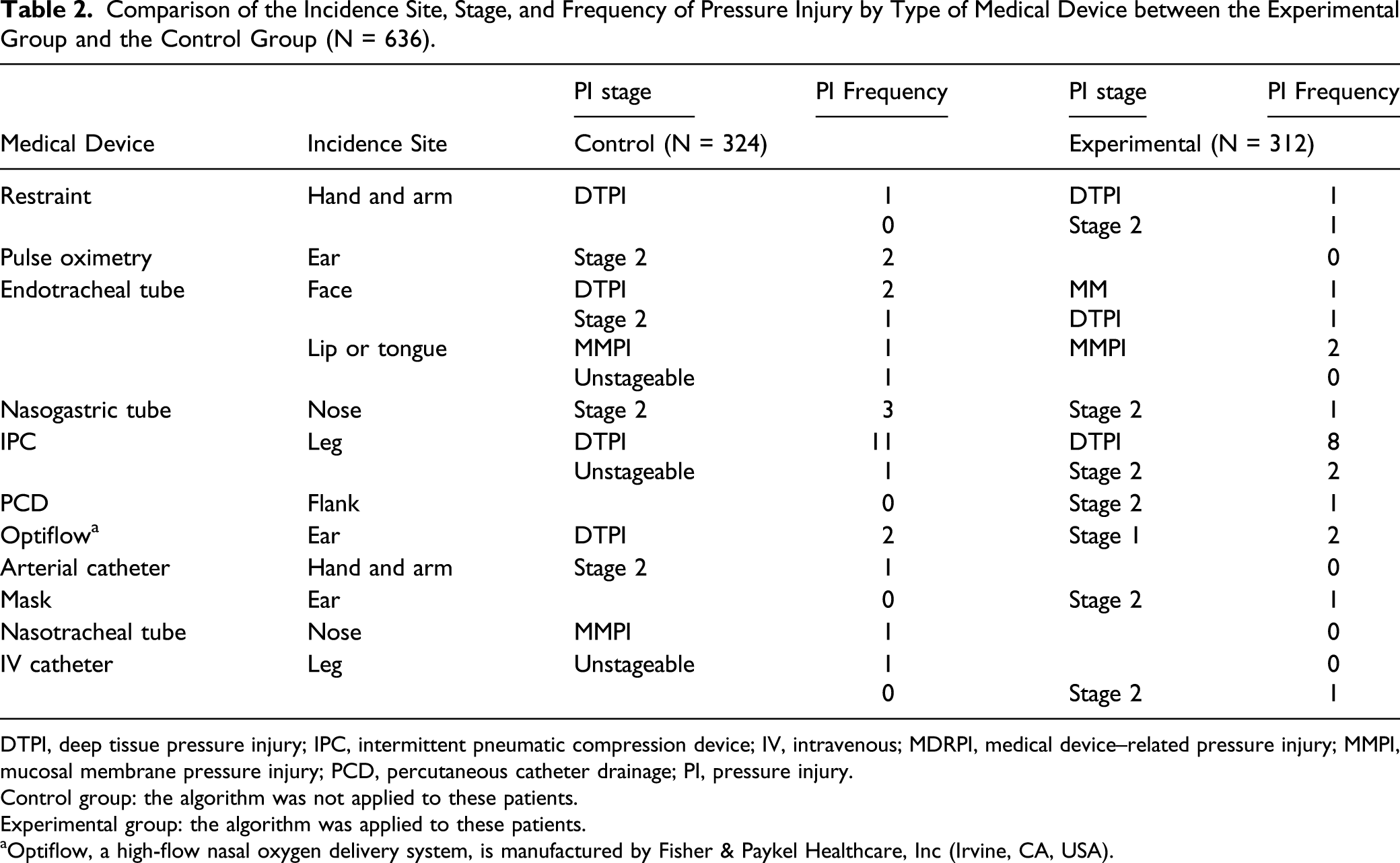
DTPI, deep tissue pressure injury; IPC, intermittent pneumatic compression device; IV, intravenous; MDRPI, medical device–related pressure injury; MMPI, mucosal membrane pressure injury; PCD, percutaneous catheter drainage; PI, pressure injury.
Control group: the algorithm was not applied to these patients.
Experimental group: the algorithm was applied to these patients.
aOptiflow, a high-flow nasal oxygen delivery system, is manufactured by Fisher & Paykel Healthcare, Inc (Irvine, CA, USA).
Incidence of Pressure Injuries
Incidence of Pressure Injury between the Experimental Group and the Control Group (N = 3759).

CVC, central venous catheter; IPC, intermittent pneumatic compression device; IV, intravenous; MDRPI, medical device–related pressure injury; PCD, percutaneous catheter drainage
Control group: the algorithm was not applied to these patients.
Experimental group: the algorithm was applied to these patients.
aOptiflow, a high-flow nasal oxygen delivery system, is manufactured by Fisher & Paykel Healthcare, Inc (Irvine, CA, USA).
Comparison of the MDRPI Incidence
The incident rate of MDRPI was 1.46 per 100 devices in the control group and 1.19 per 100 devices in the experimental group.
Discussion
In this study, we developed an MDRPI prevention algorithm through a four-stage process. A preliminary algorithm was constructed, based on literature review and analysis of medical records, and the algorithm was subjected to expert review for content validity. The validated algorithm was verified for practical feasibility by nurses. The final algorithm was applied to the patients to verify its effectiveness.
The algorithm items were selected by analyzing the guidelines for pressure injury management. The National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel, and Pan Pacific Pressure Injury Alliance guidelines for pressure injuries 2 were primarily used as a reference because they include recommendations for selecting and fixing medical devices, evaluating the skin and medical devices, preventing MDRPI, and applying prophylactic dressings. We attempted to increase the validity of this algorithm by clearly tabulating the evidence for each item of this algorithm. Based on the literature reports used for the development of the algorithm content, if the report presented evidence level and recommended strength, it was specified in the evidence table. However, if the literature report did not present the evidence level and recommended strength, only the results of the research team’s discussion were reported. Integrating the evidence level and recommended strength by applying different criteria obtained from various studies was limited to some extent. Therefore, if the research team determined the level, then the ratings in the most referenced literature were used. A meaningful fact is that existing international evidence-based pressure injury guidelines have been developed as local clinical practice algorithms applicable to current clinical sites, and their application has been proven as appropriate and feasible for nurses and patients.
The time intervals for skin evaluations differ, based on the medical device type, and conducting evaluations at accurate time intervals is important. The experts involved in the current study recommended that the timing of skin evaluations should be explicitly specified such as on ICU admission or during shift changes. Therefore, we specified the timepoints for nurse inspection of the skin, based on medical device type. The experts involved in this study discussed the possibility that nurses would omit skin evaluations, given the wide range and number of medical devices used for ICU patients and the number of measures necessary to address them. We accordingly provided blank spaces for nurses to record the type of “other” medical devices that were not included in the prespecified types of medical devices.
The overall algorithm content, which included maintaining dry and clean skin areas by skin inspection at least twice a day, applying prophylactic dressings, changing the location of the medical device, and using medical devices of appropriate sizes, was similar to the National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel, and Pan Pacific Pressure Injury Alliance guidelines for pressure injuries. 2 However, we included information on proper methods for medical device fixation in ICU patients and specific time points for skin evaluations to ensure consistent nursing interventions.
The practical feasibility of the algorithm was verified among the ICU nurses. The results showed high scores for items stating that using the algorithm will enable early MDRPI detection, decrease omissions of nursing interventions for pressure injury prevention and management, and aid in pressure injury-prevention care (all; score, >3.37). This result may be attributable to the fact that the algorithm presents detailed methods of pressure injury prevention by medical device type, ensuring that nursing interventions will not be omitted, and that the algorithm was designed as a brief record form. However, the item on reducing nurse workload was ranked the lowest (score, 2.75), presumably because presenting a more detailed algorithm for pressure injury care, compared to the existing care, may have increased nurse workload. As the long-term use of this algorithm becomes embedded within the practice of nursing interventions for the prevention and management of pressure injuries, the workload of these nurses is expected to decrease.
We compared the MDRPI incidence without and with algorithm implementation on patients to verify the effectiveness of the algorithm. The incidence rate without algorithm application was 1.46 per 100 devices; it decreased to 1.19 per 100 devices with algorithm application. We could not compare our results with those of other Korean researchers, owing to the lack of Korean studies on MDRPI prevention; however, Black and Kalowes(19) reported a similar reduction (.3% to .0%) in the incidence of MDRPI by using a pressure injury-prevention model. In this study, the specific rates of reduction were 3.9% to 3.1% for endotracheal tube-induced pressure injuries, 2.6% to .9% for nasogastric tube-induced pressure injuries, and .6% to .0% for oximeter-induced pressure injuries. These results are attributable to the fact that the developed algorithm provided specific instructions for the cycle of location change such as changing the position of an endotracheal tube at least once daily, and it provided detailed methods of care such as instructing nurses to ensure that the nasogastric tube does not meet the nasal cavity, which promoted pressure injury-prevention care and consequently reduced the incidence of pressure injuries. However, since there was no homogeneity in the pre-Braden scale scores of the experimental and control groups, the results concerning the incidence of pressure damage after applying the algorithm should be interpreted with care.
Contrastingly, the incidence of pressure injuries caused by restraints, transcutaneous catheter drainage tubes, and facial masks slightly increased after algorithm implementation. The algorithm has instructions requiring nurses to remove restraints at least once every 2 hours to examine the skin and to avoid placing restraints too tightly. However, the incidence of pressure injuries did not decrease, probably because these devices were placed tightly on ICU patients on account that many ICU patients had delirium and tried to remove medical devices. 30 Transcutaneous catheter drainage tubes are generally inserted in a patient’s flank, but ICU patients are often placed in the lateral position to prevent pressure injuries, which then causes the body to impose pressure on the medical device and thus increases the risk of pressure injuries. With facial masks, stage 2 pressure injuries developed on the ears, which are more vulnerable to pressure injuries because of thin tissues, although applying prophylactic dressings seemingly cushioned the ears and reduced the stage of pressure injuries.
In this study, pressure injuries were most commonly induced by IPCs and no marked changes occurred between patients for whom the algorithm was not implemented and patients for whom the algorithm was implemented. The Hospital Nurses Association 21 recommends removing the IPC every day to assess for any problems on the skin and to prevent MDRPI on the legs. The present algorithm has instructions for nurses to remove the compressors once a day to evaluate the skin, but this action was not particularly effective in preventing pressure injuries, and the time of day when the compressors were removed for skin evaluation was unknown. In the future, studies should examine the optimal number of skin inspections and duration (min) of compressor removal to prevent pressure injuries.
The deep tissue pressure injury rate was highest among the pressure injury levels investigated without and with the algorithm applied. Many patients were hospitalized long-term, and IPCs were continuously worn to prevent deep vein thrombosis. A possibility is that the area where the restraint and IPC are worn such as the wrist, ankle, and leg lower extremities could not be detected until the pressure injury deepened because the sites were not prominent areas as are the face, neck, and fingers. Regular use of this algorithm for skin assessment would help reduce the incidence of deep tissue pressure injury.
This study has some limitations. First, most studies related to MDRPI collected study data from 3 months to >2 years; however, we could use the algorithm only for 1 month, which hinders sufficient examination of the algorithm’s effectiveness. This aspect should be confirmed in further investigation/evaluation. Second, verification of practical feasibility for nurses and assessment of the effectiveness of algorithms for patients were conducted simultaneously. We applied an algorithm with proven expert validity to the patient; hence, we determined that no harm to the patient would occur. Third, the algorithm has instructions for nurses to check and record the site and stage of MDRPIs, however, the possibility that records were omitted or that pressure injuries were undetected because of the high workload is difficult to completely rule out. Fourth, the pre-Braden Scale scores of the experimental and control groups did not have homogeneity. Results regarding the incidence of pressure injury after applying the algorithm should be interpreted with caution. Further modified studies need to be conducted to ensure a balanced group. Fifth, we failed to investigate other characteristics that could affect the outcome, such as the duration of the patient’s stay in the ICU and the number of devices per patient. In the future, repeated studies will need to be conducted to investigate all of these characteristics to perform homogeneity verification and to analyze the effectiveness of the algorithm.
Conclusion
We developed a MDRPI prevention algorithm with reference to the four-step clinical practice guideline development guidelines. 19 This algorithm is an effective and safe intervention that can contribute to providing a higher quality of nursing care. Subsequent studies should be conducted on the application and assessment of the effects of this MDRPI prevention algorithm on patients admitted to wards other than an ICU, and the long-term effects of this algorithm on MDRPI prevention need to be examined.
Supplemental Material
sj-pdf-1-inq-10.1177_00469580211050219 – Supplemental Material for Development and Testing of an Algorithm to Prevent Medical Device–Related Pressure Injuries
Supplemental Material, sj-pdf-1-inq-10.1177_00469580211050219 for Development and Testing of an Algorithm to Prevent Medical Device–Related Pressure Injuries by Yeong-Mi Seong, Hyejin Lee and Ji Min Seo in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the institutional review board of XXX (IRB No. XXXXXXX), which waived the need to obtain informed consent from patients undergoing the practical feasibility verification. Written consent was obtained from nurses who performed the verification.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
