Abstract
Split cornea transplantation can reduce the shortage of donor corneas. Therefore, this study aimed to evaluate the effect of split graft storage time on the outcomes of split corneal transplantation through Descemet membrane endothelial keratoplasty (DMEK) and deep anterior lamellar keratoplasty (DALK) surgeries. Split corneal transplantation was performed in 80 eyes using 41 donor corneas. The mean before and after splitting storage times and total storage times were recorded. Donor corneal buttons and split grafts were stored in short-term solution at 4°C. In both surgeries (DMEK and DALK), donor corneas were divided into groups depending on their storage times. Mean postoperative 12th month best corrected visual acuity (BCVA), endothelial cell density (ECD), endothelial cell loss (ECL), central corneal thickness (CCT), refractive spherical equivalent (RSE), refractive astigmatism, and complication rates were compared among the groups. Correlation between storage times and 1-year BCVA, ECL, and complication rates were assessed. Clinical outcomes of the groups 1 year after the surgeries were also compared. DALK and DMEK were performed in 41 and 39 eyes, respectively. Storage times were not correlated with 1-year DMEK outcomes and only weakly correlated with post-DALK ECD, ECL, and RSE values. Except for CCT in those that underwent DALK, the outcomes of DMEK and DALK surgeries with stored and non-stored split grafts were not significantly different (P = .02). The storage times of donor corneas and split grafts do not have any impact on outcomes.
Keywords
• What do we already know about this topic?
Previous studies that were performed with organ cultures show that endothelial Descemet grafts can be safely stored for 1 week in split corneal transplantation. • How does your research contribute to the field?
Our study was performed with short-term corneal solution contributes to the field that endothelial Descemet grafts can be safely stored for 1 week in split corneal transplantation and post-splitting storage and total storage times did not have any significant impact on the clinical outcomes of split corneal transplantation. • What are your research’s implications toward theory, practice, or policy?
We conclude that split corneal transplantation can reduce donor cornea shortages through DMEK and DALK procedures with 2 split grafts derived from 1 cornea.
Background
Corneal disease is the third leading cause of blindness. Majority of patients with corneal disease can be treated with corneal transplantation.
1
There are reportedly 185 000 corneal transplantations performed annually. However, there are millions of patients worldwide who are waitlisted for transplantation.
2
The disparity between the low number of transplants and the high number of waitlisted patients results in long waiting periods. There are 2 procedures performed in corneal transplantation: the deep anterior lamellar keratoplasty (DALK) and the Descemet membrane endothelial keratoplasty (DMEK). DALK is the preferred procedure for the treatment of stromal corneal diseases, while Descemet membrane endothelial keratoplasty (DMEK) is the treatment of choice for endothelial corneal diseases.3,4 These 2 procedures enable split corneal transplantation, wherein 1 donor cornea is divided into 2 partial grafts, which can then be used by 2 recipients.5–7 In split corneal transplantation, the anterior part of the donor button is used for DALK in 1 recipient, whereas the donor’s endothelial Descemet membrane layer is used for DMEK in another recipient (Figure 1). Previous studies reported that split corneal transplantation reduced the need for donor corneas and had several technical advantages over penetrating keratoplasty.5–8 Despite numerous studies on the advantages of split corneal transplantation, the ideal storage time of the donor corneas and/or split grafts and the potential effects of storage times on the outcomes of the aforementioned procedures remain unclear. In this study, we investigated the relationship between the storage times of split corneal grafts stored in short-term corneal solution and associated clinical outcomes. Principle of split cornea transplantation for 2 recipients. A single donor cornea can be split into 2 parts. The anterior cornea graft (ACG) is transplanted using deep anterior lamellar keratoplasty (DALK) in corneal diseases affecting solely the anterior part and not the endothelium and Descemet membrane layer (eg, in keratoconus). The endothelial Descemet graft (EDG) is grafted using Descemet membrane endothelial keratoplasty (DMEK) in corneal endothelial diseases such as pseudophakic bullous keratopathy (PBK) (modified from Heindl et al, 2011; surgery photographs provided by Kandemir and associates).
Materials and Methods
Study Design, Setting, and Population
This study was conducted in accordance with the 1964 Declaration of Helsinki and approved by the Research Ethics Committee of University of Healthy Science Istanbul Dr. Lutfi Kırdar Kartal City Hospital (protocol number: 2019/59/150/15). All patients provided written consent for the inclusion of their information in our database and its potential use for later analyses.
Sampling
Data of patients who underwent DALK or DMEK procedures at the Department of Ophthalmology of the Istanbul Dr. Lutfi Kırdar Kartal Training and Research Hospital between January 2016 and January 2018 were retrospectively reviewed. Pregnant patients and patients with a postoperative follow-up period of less than 1 year were not included in the study. Patients who underwent DMEK procedures were diagnosed with either pseudophakic bullous keratopathy (PBK), Fuchs’ endothelial dystrophy (FED), or iridocorneal endothelial syndrome with a best corrected visual acuity (BCVA) of ≤20/200 without any anterior corneal scarring. Patients who underwent DALK had either advanced keratoconus or corneal dystrophy with a BCVA of ≤20/200. The donors’ ages ranged from 45 to 70 years old. In this age range, the endothelial Descemet graft (EDG) is healthier and can be prepared more easily. Donor tissue was accepted with a postmortem time of up to 24 hours and a donor endothelial cell density (ECD) of >2000 cells/mm2. When short-term medium (Eusol-C, Corneal Chamber, Alchimia, Ponte San Nicolo, Italy) at 4°C was used, the preservation time of the donor corneas was increased to up to 21 days, as described previously. 9 Depending on the availability of the patients, either the DALK procedure was performed first, followed by the EDG storage for subsequent DMEK procedure. An alternative was the performance of the DMEK procedure first, followed by the storage of the anterior corneal graft (ACG) in Eusol-C for the subsequent DALK procedure.
Data Collection and Outcomes Assessment
The ECD of the donor corneas was measured using a specular microscope (Konan Eye Bank KeratoAnalyzer EKA-04, Hyogo, Japan) provided by the eye bank. Pre- and post-splitting storage and total storage times of the donor corneas were noted separately for both the DMEK and the DALK procedure. The entire sample was divided into 4 subgroups based on storage times: 0–2, 2–4, 4–6, and >6 days. The subgroups were compared in terms of mean postoperative 12th month BCVA, ECD, endothelial cell loss (ECL), central corneal thickness (CCT), refractive spheric equivalent (RSE), refractive astigmatism (RA), and complication rates. Furthermore, effects of immediate transplantation or storage of the split grafts on post-surgical outcomes were also assessed for both types of surgery. DALK, DMEK, and split donor graft preparations were all performed by an experienced surgeon (B.K.) using techniques described previously (Figure 1).10,11 The storage times of the split corneal grafts stored for subsequent transplantation surgeries were recorded. Since the storage times for the grafts prepared at the time of surgery were all shorter than an hour, they were ignored. For the treatment of clinically significant coexisting cataract and FED, DMEK was combined with phacoemulsification and posterior chamber lens implantation. Before postmortem excision, serological and microbiological tests were conducted. Only seronegative donor corneas were used. The splitting procedure was performed in the operation room. The ACG or EDG, which was the remaining donor lenticule, was stored in storage solution for the subsequent surgery. In all surgeries, a 2 mL sample was taken from the storage solution before and after splitting. Samples were examined for the presence of aerobic, anaerobic, and fungal microorganisms. Thioglycolate broth (THIO) was used for the isolation and culture of organisms from the corneal samples. THIO was then incubated at 35°C for 24 hours and inoculated on the plate medium consisting of 5% sheep blood agar, chocolate agar, and MacConkey agar for aerobic organisms and Brucella agar for anaerobic organisms.
All plates were incubated at 35°C in 5% to 7% CO2 for 72 hours. The plates were examined daily for growth of microorganisms. If growth was detected, antimicrobial susceptibility tests (VİTEK 2 Compact Systems, BioMerieux, Craponne, France) were performed. After the surgery, all eyes were treated with .5% moxifloxacin hydrochloride (Vigamox, Alcon Pharma GmbH, Freiburg, Germany) and .1% dexamethasone (Maxidex, Alcon Pharma GmbH) 5 times daily. The topical antibiotic was discontinued after the completion of a 10-day treatment regimen. Dexamethasone, the topical steroid treatment, was replaced with .5% loteprednol etabonate (Lotemax, Bausch + Lomb, Bridgewater, NJ, US), which was applied 4 times daily for 3 months after surgery. Topical steroid therapy was continuously administered for at least 2 years after the surgery and was gradually reduced to at least once a day according to the patient’s clinical outcomes. Standardized eye examinations were carried out both preoperatively and 1 day, 1 week, and 1, 3, 6, and 12 months after surgery. These examinations included the Snellen BCVA assessment using a Snellen chart converted to logMAR units, slit lamp examination, tonometry, fundoscopy, subjective refractometry, corneal topography (Sirius Scheimpflug-Placido topographer, Costruzione Strumenti Oftalmici, Florence, Italy), corneal pachymetry (Optikon Pacline, Optikon Corp., Rome, Italy) for the CCT measurement, and slit lamp-adapted optical coherence tomography (Optos PLC., Dunfermline, Scotland, United Kingdom) of the anterior segment for the detection of endothelial graft detachment. ECD was measured using a specular microscope (Topcon Corporation, Tokyo, Japan) during each follow-up assessment. Intraoperative and postoperative complications were recorded. For the DMEK procedures, the rates of rebubbling, in which air re-injection was performed for partial graft detachment, were recorded. All patients were followed up for at least 1 year. The main outcomes were visual and refractive, ECD, ECL, CCT, and postoperative complication rates.
Data Analysis
Statistical analyses were performed using the SPSS software, version 21.0 (IBM Corp. Armonk, NY, US). Normality of data distribution before the group comparisons was assessed using the Shapiro–Wilk test. For the comparisons, the Kruskal–Wallis test, one-way ANOVA, chi-square test, Fisher’s exact test, independent-samples t-test, Wilcoxon’s test, and paired-samples t-test were used. The correlation analysis was conducted using Spearman’s correlation coefficient. Statistical significance was set at P < .05.
Findings
Recipient and donor characteristics
The study included 80 patients, 45 of whom were women and the remaining 35 were men A total of 41 patients underwent DALK or DMEK procedures; hence, 41 donor corneas were used altogether. The mean age of the patients was 52.62 ± 16.30 years (range, 14–94 years). A total of 82 grafts from 41 donors (100%) were successfully prepared. There were 39 eyes that underwent DMEK procedures. The remaining 2 of the 41 EDGs were not viable. In total, 26 EDGs were transplanted after preparation during DMEK surgery. The remaining 13 EDGs that were prepared during DALK surgery and stored in storage solution following splitting were all used for DMEK surgeries. All 41 DALK procedures were successful; thus, a switch to penetrating surgery was not necessary. The ACGs of the 37 eyes were used after preparation during DALK surgeries, while 4 ACGs were used for DALK surgeries after storage in the solution. Overall, 41 DALK and 39 DMEK surgeries were performed using 41 donor corneas for 80 eyes. Since 2 EDGs could not be used for DMEK surgeries, one of which was due to a relatively long storage time of 16 days and the other due to a size mismatch with the recipient eye that had microcornea, 39 donor corneas were saved by our approach. Consequently, corneal tissue demand decreased by 48% when split donor corneas from 41 donors for 80 eyes were used.
The mean age of the donors was 59.72 ± 6.81 years (range, 40 to 70 years). The mean total pre- and post-splitting storage times for the donor corneas were 94.2 ± 59.8 (range, 24–322 hours) and 8.64 ± 23.2 (range, 0–168 hours), respectively. The mean total storage time was 105.65 ± 73.18 hours (range, 24–408 hours). For the donor corneas used for DMEK, the mean pre- and post-splitting storage times were 99.9 ± 65 (range, 24 to 322 hours; maximum, 13 days; n = 39) and 45.76 ± 40.00 hours (range, 19 to 168 hours; maximum, 7 days; n = 13), respectively. The mean total storage time for all donor corneas used for DMEK was 115.23 ± 85.04 hours (range, 24 to 408 hours; maximum, 17 days). For the donor corneas used for DALK, the mean pre- and post-splitting storage times were 94.2 ± 59.8 (range, 24 to 322 hours; n = 41) and 24 (n = 4) hours, respectively. The mean total storage time for all donor corneas used for DALK was 96.53 ± 59.43 hours (range, 24 to 264 hours). When samples were grouped according to the pre-splitting storage time for the donor cornea in Eusol-C, there were no significant differences between groups and different initial donor ECD values (P = .588).
Relationship Between Clinical Findings and Storage Times
Demographic characteristics and clinical findings in the DMEK and DALK groups according to the storage time.

Note. DALK, deep anterior lamellar keratoplasty; DMEK, Descemet membrane endothelial keratoplasty; SD, standard deviation, ECD, endothelial cell density, BCVA, best corrected visual acuity, logMAR, logarithm of the minimum angle of resolution, CCT, central corneal thickness, RA, refractive astigmatism, RSE, refractive spherical equivalent, D, diopters.
* Kruskal–Wallis and chi-square tests were performed.
** P<.05.
Correlations between clinical findings and storage times in the DMEK and DALK groups.

Note. DALK, deep anterior lamellar keratoplasty; DMEK, Descemet membrane endothelial keratoplasty; ECD, endothelial cell density; BCVA, best corrected visual acuity; logMAR, logarithm of the minimum angle of resolution; CCT, central corneal thickness; RA, refractive astigmatism; SE, spherical equivalent; D, diopters.
* Spearman’s correlation analysis was performed.
Clinical findings in the DMEK and DALK groups according to the use of stored or non-stored grafts.

Note. DALK, deep anterior lamellar keratoplasty; DMEK, Descemet membrane endothelial keratoplasty; SD, standard deviation; ECD, endothelial cell density; BCVA, best corrected visual acuity; logMAR, logarithm of the minimum angle of resolution; CCT, central corneal thickness; RA, refractive astigmatism; SE, spherical equivalent; D, diopters.
* Independent-samples t-test and Fisher’s exact test were performed.
** P<.05.
Recipients Outcomes
The mean BCVA was 1.37 ± .37 logMAR (.7–2.0 logMAR) before the DMEK procedure and had increased to .40 ± .41 logMAR (.0–1.7 logMAR) at the postoperative .2 logMAR. The mean ECD of the donor endothelium was 2777 ± 374 cells/mm2 (2000–3881 cell/mm2) before the DMEK surgery and decreased to 1151.05 ± 460.64 cells/mm2 (378–2147 cell/mm2) 12 months after surgery. The mean ECL was 57.46 ± 17.23 12 months after the DMEK procedure. The mean CCT of the recipient corneas was 770 ± 103µ (510–1031µ) before surgery and 542 ± 92µ (435–818µ) at the 12-month follow-up. The mean RSE was −.6 ± 1.5D (−3.25 to +2.75) preoperatively and .7 ± 1.0D (1.6–3.5D) 12 months after surgery. The mean RA was −.8 ± 1.0D (−3.75 to .50) preoperatively and .66 ± 1.61D (P = .00, P = .698) 12 months after surgery (Figure 2). Complications were noted during follow-up in 9 (16%) patients (n = 39) who underwent DMEK surgery. Primary graft failure developed in 7 eyes, which necessitated re-DMEK surgery. Secondary glaucoma developed in 2 eyes and was controlled with medical treatment. Within the first postoperative month, partial graft detachment occurred in 6 (15%) eyes; thus, rebubbling of the anterior chamber was required (once in 3 cases, twice in 2, and thrice in 1). There were 3 patients who underwent split corneal transplantation immediately after the donor cornea splitting procedure, while the remaining 3 underwent split corneal transplantation after corneal storage in short-term solution after splitting. The mean BCVA was 1.67 ± .31 logMAR (.7–2.0 logMAR) before the DALK procedure and .42 ± .30 logMAR (.1–1.3 logMAR) 12 months after the procedure. The mean ECD of the recipient eyes was 1973.80 ± 279.52 cells/mm2 (range, 1199–2457 cells/mm2) preoperatively and 1724.26 ± 301.82 cells/mm2 (759–2120 cells/mm2) 12 months after the surgery. Total ECL was 12.43 ± 7.35 until up to 1 year after DALK. The mean CCT was 453 ± 130µ (324 ±702µ) preoperatively and 516 ± 37µ 12 months after the surgery. The mean RSE was −9.88 ±2.99D (−14.5 to −3.00D) preoperatively and −2.62 ± 3.66D (−7.25 to +6.0D) 12 months after the surgery. The mean RA value decreased from −7.86 ± 3.54D to −2.41 ± 3.78D 12 months after the surgery (Figure 3). Fuchs’ endothelial dystrophy (A-B) and iridocorneal endothelial syndrome (C-D) before and 1 year after Descemet membrane endothelial keratoplasty (DMEK) with (A-B) and without (C-D) endothelial Descemet graft (EDG) storage. Keratoconus before and 1 year after deep anterior lamellar keratoplasty (DALK) with (A-B) and without (C-D) anterior corneal graft (ACG) storage.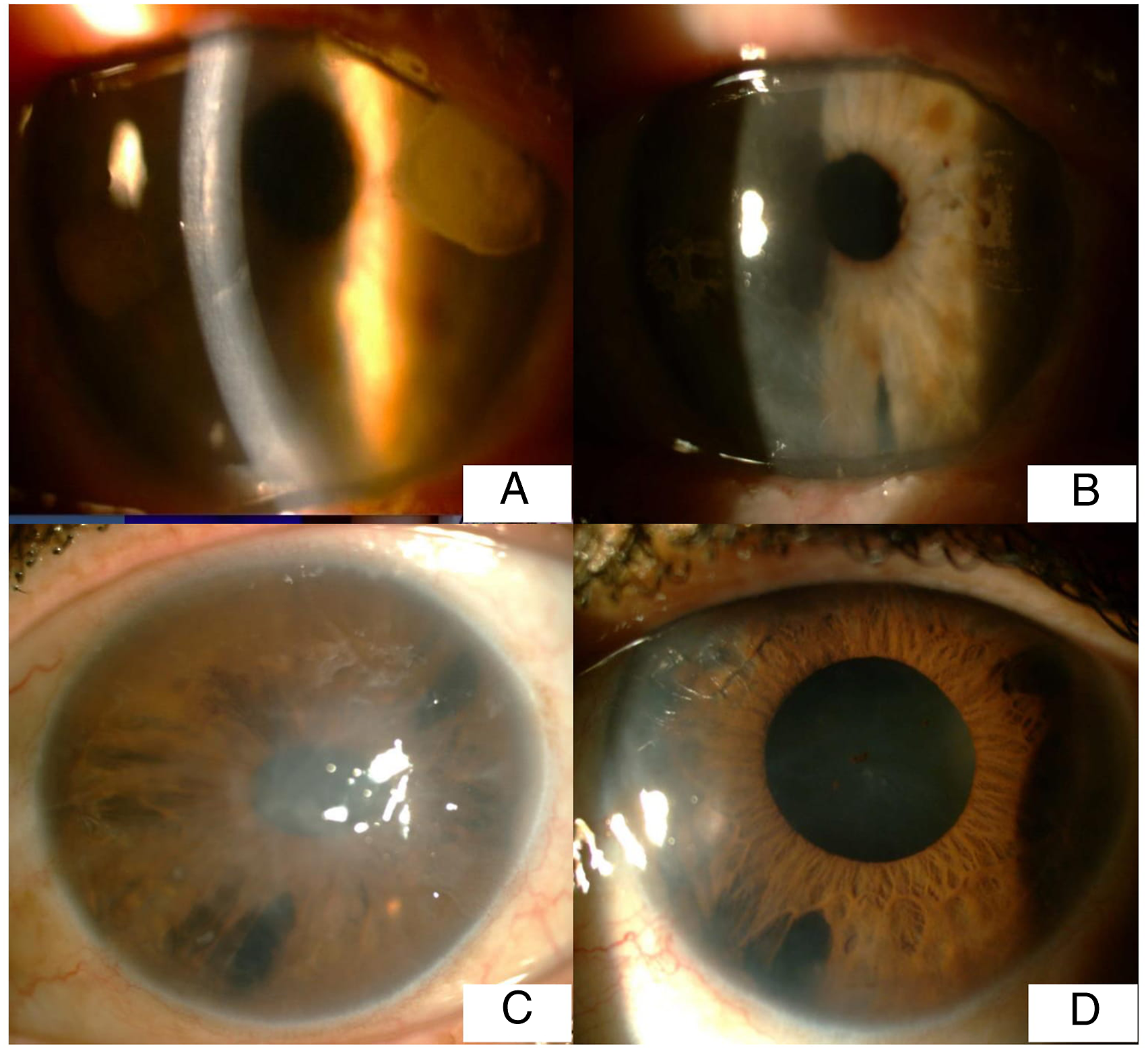
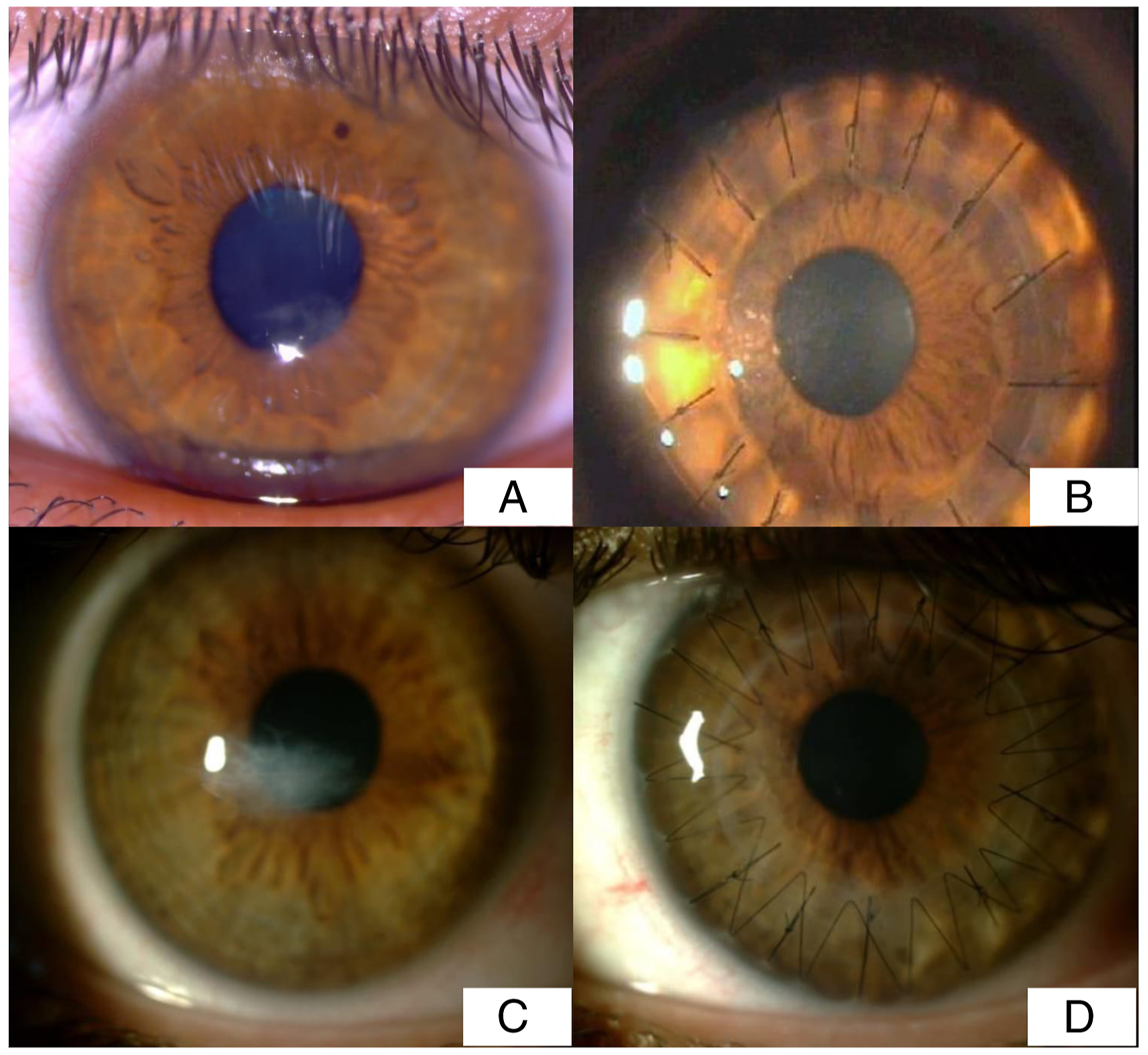
Complications were detected during follow-up in 3 patients (7.3%) (n = 41) who underwent DALK surgery. Delayed re-epithelization that was diagnosed in 1 eye improved with medical treatment. Acute graft rejection secondary to herpetic keratitis was diagnosed in 1 eye, while bacterial keratitis developed in another. Both complications were later treated with penetrating keratoplasty. We did not encounter any other postoperative complications in our cohort of patients.
Discussion
Split donor organs and tissues have been used in transplantation surgeries for years. 12 It was reported that split liver transplantation reduced the need for donor livers by approximately 40%.13,14 In line with this finding, donor cornea shortages might also be reduced by approximately 45% and 47% when split grafts in corneal transplantation is applied in practice.5–8 Here, 82 grafts prepared from 41 donor corneas were used in 80 patients. As a result, the demand for donor corneal tissue thus decreased by 48%.
In our study, the pre-splitting maximum storage time of the donor corneas in Eusol-C solution was 322 hours (13 days). Yuksel et al 9 reported that the viability of donor corneas can be preserved for up to 8 days in Eusol-C solution. Recently, several studies reported the storage of EDG in organ culture medium or short-term corneal storage solution at 4°C and the effects of these media and storage times on cornea transplantation outcomes.6,8,15 In these studies, the maximum split donor storage time was 7 days, while the total storage time was 115.16 ± 17 days. In a study involving 100 patients, Heindl et al stored EDGs in Dulbecco’s Modified Eagle Medium organ medium at 34°C either after a DALK procedure for utilization within 72 hours for DMEK or after the first DMEK procedure for utilization in ACG for a DALK procedure within 72 hours. 8 The authors emphasized that this approach was feasible and that EDGs can be stored in an organ culture medium for 3 days after splitting. 8 Our correlation analysis on storage times and surgical outcomes revealed that the storage time of the split donor EDGs and total storage time in short-term solution were not correlated with the postoperative 12th month surgical outcomes of the DMEK procedure. Heindl et al 16 also conducted a correlation analysis between post-splitting storage times of EDGs and surgical outcomes but did not detect a significant correlation. However, the authors of another study reported that long storage times had negative effects on ECDs when donor corneas were stored in organ culture medium for approximately 13.5 days or longer. Further, this study found that ECL became more prominent 6 months after a DMEK procedure. 15 Feng et al used grafts in the DMEK surgery either immediately after splitting or after storage in short-term corneal storage solution (Optisol-GS, Bausch + Lomb Surgical, Irvine, CA, US) for 1 to 2 days and compared the rebubbling and primary graft failure rates and ECLs between the groups. 17 The authors did not find any significant difference between the stored and non-stored graft group in terms of the aforementioned parameters. 17 Here, our visual acuity values after DMEK procedures with the EDGs stored for up to 7 days after splitting or with non-stored EDGs were similar to those reported by earlier studies. However, these were lower than the ones proposed by other studies.6,7,18 The difference may be attributed to the exclusion of patients with pre-existing ocular comorbidities or other complicated cases. Our DMEK series using split donor tissue stored up to 7 days led to a mean 1-year ECL of 57.46 ± 17.16%, which was similar to the rates reported for stand-alone and triple DMEK surgeries. A mean 1-year ECL rate of 34% was reported by Dirisamer et al, 19 who used EDGs stored for 5 to 10 days in an organ culture medium until the time of transplantation and 1 other study by Guerra et al, 20 who reported a 36 ± 20% rate. This study used non-stored grafts for DMEK procedures.19,20 Based on these results, we attributed our relatively higher ECL rate to early surgical inexperience and inclusion of complicated cases.19,20 The mean CCT, refractive values, and complication and rebubbling rates of the eyes undergoing DMEK procedures in our study were similar to those reported in other DMEK studies, including those that used non-stored grafts.21–23
In the present study, there was a statistically significant difference in the ECL values of the DALK procedure. However, in our opinion, this difference was not clinically important because there were studies that showed that the donor endothelial cells were the least decisive criterion in assessing the quality of an ACG.8,24 Indeed, Borderie et al 25 showed that a lower donor ECD (<2000 cells/mm2) was more favorable for anterior lamellar keratoplasty (ALK) surgery and that storage of ACGs in the storage solution did not affect outcomes. 25 We found only a weak correlation between storage times and postoperative 12th month values of ECD, ECL, and RSE values for the DALK procedure. These findings imply that ACGs can be safely stored in short-term storage solution. Heindl et al 16 indicated that storing the anterior lamella after splitting did not have an impact on surgical outcomes such as CCT. Furthermore, the grafts can be stored in an organ culture medium for up to 1 week. 16 In our study, ACGs that were prepared during the DALK surgery were subsequently transplanted to the recipient eye in 37 of 41 patients. In 4 eyes, the ACG was transplanted after its storage on Eusol-C for 24 hours. When preoperative and postoperative clinical outcomes were compared for the eyes that received stored and non-stored ACGs after splitting, only CCT was found to be higher in the stored group; however, graft survival and visual outcomes were not affected. Our final visual acuity results for the DALK surgery resemble those reported by other authors who performed DALK surgeries using non-stored tissues.26–28 Similarly, our mean ECL, CCT values, and complication rates for the DALK procedure were close to those reported in the DALK series performed with non-stored anterior grafts.19,24 Other DALK series that used anterior grafts stored in organ culture medium also reported similar rates.6,8
One of the reservations regarding the usage of split grafts was potential infections. In order to prevent contamination and subsequent complications, 2 mL of each sample was taken from the storage solution before and after splitting. Since THIO was an excellent medium for anaerobe recovery and unsuitable for aerobic microorganisms such as yeasts, THIO was used for the isolation of microorganisms and graft cultures in this study. 29 All microbiological tests showed negative results in terms of bacterial or fungal pathogens.
These results indicated that split cornea transplantation was a microbiologically safe procedure, provided that precautions were taken. Schaub et al 30 also confirmed that split corneal transplantation was microbiologically safe, regardless of the cultivation method, frequency and time points of performed splits, and corneal transplantation technique. Our study has several limitations, which should be taken into consideration when evaluating our findings. This was a retrospective study and therefore may have been affected by inherent weaknesses due to its design. Other limitations include the inclusion of patients with ocular comorbidities and the absence of a randomization procedure for the storage times.
Conclusions
Our results showed that EDGs can be stored in Eusol-C at 4°C for a relatively long time after splitting for DMEK surgery. We also found that this approach did not affect outcomes. To the best of our knowledge, this is the first study that shows the feasibility of this approach in split corneal transplantation. This research builds on the findings of previous studies that were performed with organ cultures and shows that EDGs can be safely stored for short-term preservation solution for 1 week. We conclude that split corneal transplantation can reduce donor cornea shortages through DMEK and DALK procedures with 2 split grafts derived from 1 cornea. Moreover, pre- and post-splitting storage and total storage times did not have any significant impact on the clinical outcomes of split corneal transplantation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
