Abstract
The burden of complications associated with peripheral intravenous use is underevaluated, in part, due to the broad use, inconsistent coding, and lack of mandatory reporting of these devices. This study aimed to analyze the clinical and economic impact of peripheral intravenous–related complications on hospitalized patients. This analysis of Premier Perspective® Database US hospital discharge records included admissions occurring between July 1, 2013 and June 30, 2015 for pneumonia, chronic obstructive pulmonary disease, myocardial infarction, congestive heart failure, chronic kidney disease, diabetes with complications, and major trauma (hip, spinal, cranial fractures). Admissions were assumed to include a peripheral intravenous. Admissions involving surgery, dialysis, or central venous lines were excluded. Multivariable analyses compared inpatient length of stay, cost, admission to intensive care unit, and discharge status of patients with versus without peripheral intravenous–related complications (bloodstream infection, cellulitis, thrombophlebitis, other infection, or extravasation). Models were conducted separately for congestive heart failure, chronic obstructive pulmonary disease, diabetes with complications, and overall (all 7 diagnoses) and adjusted for demographics, comorbidities, and hospital characteristics. We identified 588 375 qualifying admissions: mean (SD), age 66.1 (20.6) years; 52.4% female; and 95.2% urgent/emergent admissions. Overall, 1.76% of patients (n = 10 354) had peripheral intravenous–related complications. In adjusted analyses between patients with versus without peripheral intravenous complications, the mean (95% confidence interval) inpatient length of stay was 5.9 (5.8-6.0) days versus 3.9 (3.9-3.9) days; mean hospitalization cost was $10 895 ($10 738-$11 052) versus $7009 ($6988-$7031). Patients with complications were less likely to be discharged home versus those without (62.4% [58.6%-66.1%] vs 77.6% [74.6%-80.5%]) and were more likely to have died (3.6% [2.9%-4.2%] vs 0.7% [0.6%-0.9%]). Models restricted to single admitting diagnosis were consistent with overall results. Patients with peripheral intravenous–related complications have longer length of stay, higher costs, and greater risk of death than patients without such complications; this is true across diagnosis groups of interest. Future research should focus on reducing these complications to improve clinical and economic outcomes.
Keywords
Nearly 200 million peripheral intravenous (PIV) catheters are used annually in the United States, but PIV-associated complications such as bloodstream infection are currently underevaluated, which may be due, in part, to the challenge of identifying PIV-associated complications in real-world data because of the broad use, inconsistent coding, and voluntary reporting of PIV devices.
Contributes to limited research regarding evidence on PIV-associated complications and subsequent outcomes, which is warranted to improve safety procedures for using PIV catheters in hospital settings.
Patients with PIV-associated complications have longer hospital stays, higher inpatient costs, and greater risk of death than patients without such complications; by implementing policies focused on quality improvement and making procedures for using PIV catheters more safe, PIV-associated complications may be reduced and clinical and economic outcomes may be improved
Introduction
Intravenous catheters of all types are ubiquitous in health care settings in the United States. Over 5 million central venous catheters, 1 or central lines, and nearly 200 million peripheral intravenous (PIV) catheters2-4 are used annually in the United States. The widespread use of intravascular devices has raised concerns about the risk of infection and other adverse outcomes occurring from their use.2,5,6
It is well established through prior research that central lines can increase the risk of complications, such as bloodstream infections (BSI), and lead to intensive health care resource utilization. 7 Central-line–associated bloodstream infections (CLABSI) occur in about 3% to 5% of catheterizations.5,8 According to a recent Healthcare-associated Infections (HAI) Progress Report, an estimated 21 000 CLABSI occurred in the United States in 2014. 9 The Centers for Disease Control and Prevention (CDC) reported an adjusted inpatient hospital cost of nearly $30 000 per patient and a total annual cost of $0.67 to $2.68 billion due to infections associated solely with central-line use. 10 Catheter-related bloodstream infections (CRBSI), which include infections from any catheter type (eg, central lines and PIVs), are also associated with increased hospital length of stay (LOS) and health care costs.11-13
Despite the common use of PIVs, research on the complications and related burden associated with PIV catheter use is limited. Existing studies have focused mainly on rates of specific complications, such as BSIs or particular causative organisms, on small populations,14-18 or on the management of catheter-related infections.17,19,20 In addition, prior work may underestimate rates of PIV-associated complications due to potential underreporting of PIV infections.3,15,17,18 Furthermore, research on the costs associated with IV catheter-related complications has focused on central lines alone or combined with PIV, but not on PIV alone. 12
Additional evidence of PIV-associated complications and subsequent outcomes is needed to inform policies that could improve procedures for using PIV catheters in hospital settings as well as for improving patient safety.3,15,17,18 Therefore, the aim of this study was to quantify the rates of selected PIV-associated complications and the associated health care utilization and costs among hospitalized patients.
Methods
Design and Data Source
A retrospective cross-sectional analysis of hospital discharge records in the United States was conducted to examine the prevalence of PIV-associated complications and associated clinical and cost outcomes. The Premier Healthcare Database (PHD; formerly known as the Premier Perspective® Database at the time of this study), a large comprehensive electronic health care database, was used. The PHD covers over 108 million inpatient visits and over 765 million outpatient visits and contains data from over 208 million unique patients. Data include complete clinical coding, hospital cost, patient billing data (eg, medications and devices, laboratory tests, diagnostic and therapeutic services), type of admission, discharge status, and patient demographics from more than 970 hospitals (representing the 4 geographic regions) throughout the United States. 21 All discharge data were deidentified and compliant with the Health Insurance Portability and Accountability Act (HIPAA) of 1996. Institutional review board approval was not required.
Patient Selection
Hospital admissions occurring between July 1, 2013 and June 30, 2015 were identified, and this included patients who had 1 of 7 admission diagnoses listed as the primary diagnosis (via International Classification of Diseases, Ninth Revision, Clinical Modification [ICD-9-CM] code) in at least 1 inpatient admission claim: pneumonia, chronic obstructive pulmonary disease (COPD), myocardial infarction (MI), congestive heart failure (CHF), chronic kidney disease (CKD), diabetes with complications (DM), and major trauma (hip, spinal, cranial fractures) (see Appendix Table A1 for full list of codes). These admission diagnoses were selected because it was determined, based on expert clinical input, that patients admitted with such conditions would likely not have a PIV-associated complication, defined below, prior to admission.
Admissions that included dialysis, surgery, and central-line use (see Appendix Table A2 for codes) were excluded because such services could potentially cause complications independent of PIV. Also excluded were readmissions for the same primary diagnosis. Finally, it was assumed all patients in the sample received a PIV upon admission, in line with standard hospital admitting procedures. This approach was used to avoid underreporting PIV use in our sample, as dedicated procedure codes for PIV are often not used in the hospital setting.
Within each admission diagnosis and the overall group (7 admission diagnoses combined), patients were stratified according to whether they experienced a selected PIV-associated complication during the admission (yes/no). PIV-associated complication was defined as the presence of an ICD-9-CM code in the admission record for at least one of the following complications: BSI, cellulitis, phlebitis, infection not elsewhere classified, and extravasation (Table 1).
Peripheral Intravenous–Associated Complications Code List.

Note. ICD-9-CM = International Classification of Diseases, Ninth Revision, Clinical Modification.
Study Measures
The rate of PIV-associated complication in the sample was calculated by the proportion of patients with each and any selected complication. The following key outcomes were compared according to PIV-associated complication status: hospital LOS in days, cost of hospitalization (USD), admission to the intensive care unit (ICU), and discharge status. Admission to the ICU was measured as the proportion being admitted (yes/no), and discharge status was measured as the proportion of patients either being sent home (reference), being sent to another facility, or having died prior to discharge.
Other variables included patient demographics (age, gender, and race), payment source (Medicare vs other), the Chronic Condition Indicator (CCI), 22 and admitting hospital characteristics, which were reported. The CCI categorizes 5-digit ICD-9-CM codes as either chronic or not chronic, which was used to calculate the number for chronic conditions for each patient. 22 Admitting hospital characteristics included admission type (urgent/emergent or elective), hospital geographic region, bed size (over 500 beds vs 500 beds or less), and whether the admission took place in an urban location (yes/no) or teaching hospital (yes/no).
Statistical Analyses
Descriptive statistics (eg, mean, standard deviation, and frequency) were reported for the patient, payer, and admitting hospital characteristics and number and frequency were for PIV-associated complication rates among each diagnosis and the overall group. Descriptive characteristics were also compared by PIV-complication status (with versus without) to assess potential confounders.
Multivariate analyses were conducted to compare the key outcomes of LOS, cost, admission to ICU, and discharge status for patients with versus without a PIV-associated complication. All models were adjusted for patient demographics (ie, age, gender, and race), number of comorbidities, and admitting hospital characteristics (ie, region, teaching hospital, and location). Analysis of covariance (ANCOVA) was conducted to compare LOS and the cost of index hospitalization between the study groups. Logistic regression and multinomial logistic regression were used to examine the outcomes of admission to ICU and discharge status, respectively. All models were conducted separately for the CHF, COPD, DM, and overall cohorts. In the model for the overall cohort, we also included 2-way interactions between diagnosis and the individual variables above. The 3 diagnosis groups were chosen based on clinical considerations, such as likeliness to have a PIV without a central line (CHF), potential vulnerability with a PIV infection (COPD), and the inclusion of younger patients (DM), and were analyzed separately to account for potential between-group differences that might obscure the relationship between PIV complication status and the outcomes.
Sensitivity analyses were conducted to examine the impact of a small percentage (1.5%) of patients who were observed in multiple diagnosis groups. For patients with multiple hospitalizations due to different diagnoses, the first hospitalization in time was chosen and those hospitalizations that followed were dropped. All data transformations and statistical analyses were performed using SAS® version 9.4 (SAS Institute, Cary, NC).
Results
Patient, Payer, and Hospital Characteristics
A total of 588 375 qualifying admissions were identified, ranging from 15 637 trauma patients to 187 904 patients admitted with pneumonia (Figure 1). In the combined group (including all 7 admission diagnosis cohorts), the mean age was 66.1 years (range: 50.5-75.0 years). Overall, 52.4% of patients were female (range: 47.8%-56.0%), and 71.2% of patients were white (range: 52.8%-78.8%). The main payer type among admissions was Medicare (66.2% overall; range: 38.2%-77.7%). Hospital admissions were mainly nonelective (95.2%; range: 94.7%-96.1%) and were distributed across all geographic regions. Overall, 39.5% (range: 35.7%-50.9%) of admissions occurred in teaching hospitals, and the majority (83.5%; range: 80.9%-89.3%) in urban settings. Nearly one quarter (24.3%; range: 20.6%-35.7%) of admissions took place in large hospitals with over 500 beds (Table 2).

Patient identification.
Patient Demographics, Payer Source, and Admitting Hospital Characteristics.

Note. CHF = congestive heart failure; CKD = chronic kidney disease without dialysis; COPD = chronic obstructive pulmonary disease; DM = diabetes with complications; MI = myocardial infarction; Trauma = hip, spinal, or cranial fracture.
PIV-Associated Complication Rates
Overall, 1.76% of patients (n = 10 354) had at least one of the selected complications (Table 3), and rates varied by admission diagnosis cohort: pneumonia (2.67%), MI (1.98%), diabetes (1.95%), CHF (1.25%), trauma (1.03%), CKD (1.00%), and COPD (0.98%). BSI was the most common complication (1.45%, or 82.2% of all complications) and ranged from 0.67% (CKD) to 2.46% (pneumonia). Rates of cellulitis, phlebitis, other infections, and extravasation were also evident, but at lower rates than BSI, and varied according to admission diagnosis (Table 3; Figure 2).
Selected Complication a Rates Associated With Peripheral IV.

Note. IV = intravenous; CHF = congestive heart failure; CKD = chronic kidney disease without dialysis; COPD = chronic obstructive pulmonary disease; DM = diabetes with complications; MI = myocardial infarction; Trauma = hip, spinal, or cranial fracture.
Complications include bloodstream infection, upper extremity cellulitis and abscess, upper extremity superficial phlebitis and thrombophlebitis, PIV-associated infections not elsewhere classified (ie, infection or inflammatory reaction following infusion, injection, transfusion, vaccination, vascular device, implant, or graft), or possible extravasation.
Patients might have more than 1 complication.
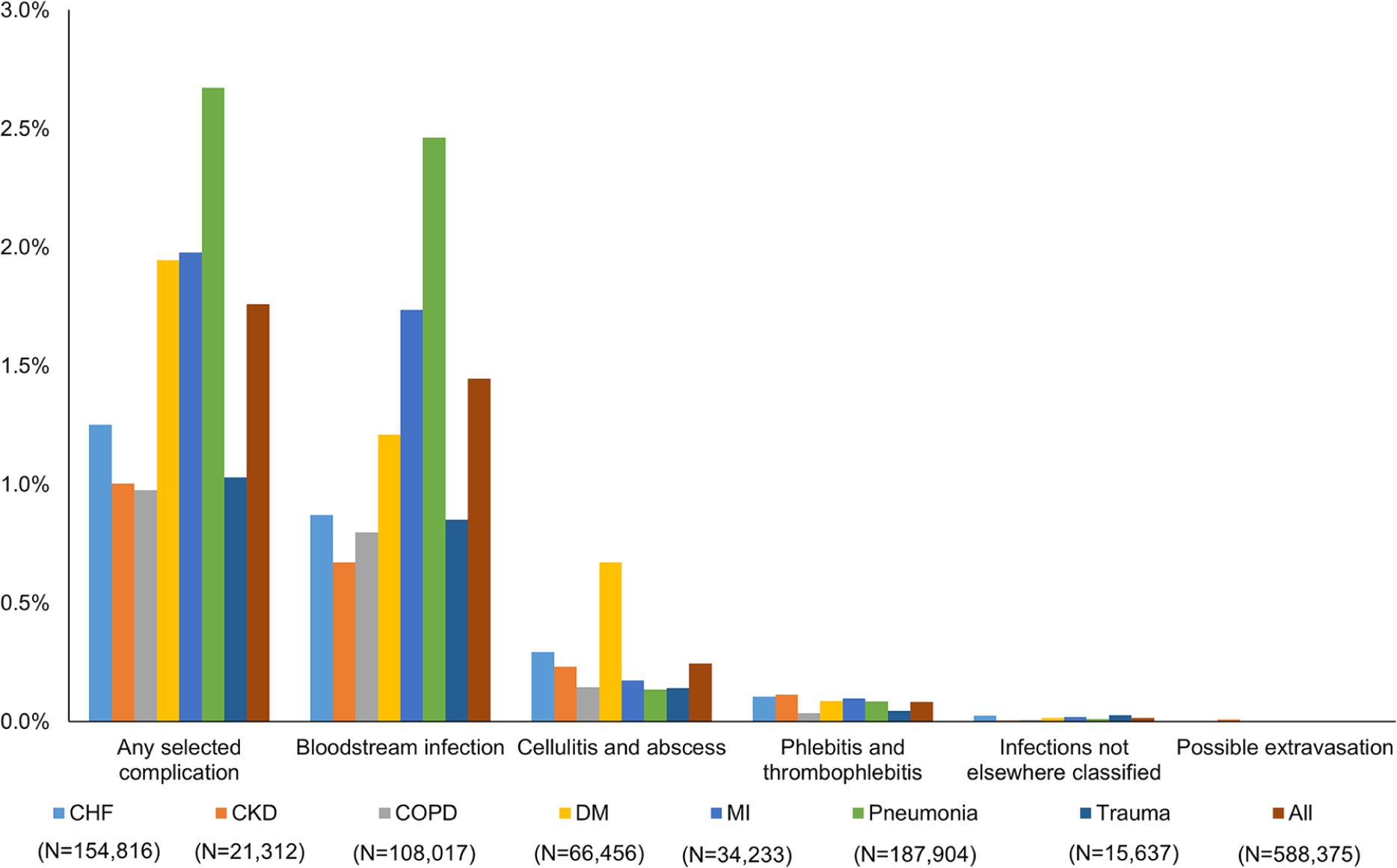
Selected complication rates associated with PIV.
Patient, Payer, and Hospital Characteristics by PIV Complication Status
Small differences in several characteristics were observed according to PIV complication status, including age, gender, race, payment source, and hospital region and location (Table 4). The patient group with a PIV-associated complication was slightly older (67.7 vs 66.1 years; P < .001), had fewer females (50.5% vs 52.4%; P < .001), and more white patients (73.5% vs 71.1%; P < .001) compared with the group without a complication. The patient group with a PIV-associated complication had a higher proportion who used Medicare as payment source (67.6% vs 66.1%; P = .009) and a lower proportion who went to urban hospitals (82.5% vs 83.5%; P = .006) versus patients without a complication. Differences in hospital region were slight albeit statistically significant (P < .001). Admission type, teaching hospital, and number of beds were similar by PIV-associated complication status. Within the diagnosis cohorts, similar patterns in characteristics according to complication status were found.
Patient Demographics, Payer Source, and Admitting Hospital Characteristics, by Peripheral Intravenous Complication Status.

Adjusted Utilization and Costs
In the main cohort, the adjusted mean LOS for patients with a PIV complication was 5.9 days compared with 3.9 days for those without a complication (Table 5; P < .001). Adjusted costs were higher among patients with a complication compared with those without (mean $10 895 [95% confidence interval [CI], $10 738-$11 052] vs $7009 [$6988-$7031]; P < .001). Patients with complications were more likely to be admitted to the ICU (20.4% [19.6%-21.3%] vs 11.0% [11.0%-11.1%]; P < .001). They were also less likely to be discharged home than those without complications (62.4% [58.6%-66.1%] vs 77.6% [74.6%-80.5%]; P < .001) and were more likely to have died (3.6% [1.9%-4.2%] vs 0.7% [0.6%-0.9%]; P < .001). The above differences in adjusted LOS, costs, admission to ICU, and discharge status between comparison groups were directionally similar for the 3 diagnosis groups (Table 5), yet were most pronounced in the CHF group: LOS (7.5 [7.2-7.7] days vs 4.2 [4.2-4.2] days), costs ($13 132 [$12 852-$13 411] vs $7394 [$7363-$7426]), admissions to the ICU (22.5% [20.7%-24.4%] vs 10.1% [10.0%-10.3%]), and discharge status of death (8.9% [7.6%-10.1% ] vs 1.8% [1.7%-1.8%]) (P < .001 for all).
Adjusted a Means and Rates for Length of Stay, Cost, ICU Admission, and Discharge Status Among CHF, COPD, DM, and Overall Groups.

Note. ICU = intensive care unit; CHF = congestive heart failure; COPD = chronic obstructive pulmonary disease; DM = diabetes with complications; CI = confidence interval; PIV = peripheral intravenous.
Adjusted by age, gender, race, number of chronic conditions, hospital region, teaching versus nonteaching hospital, and hospital location.
CHF cohort includes 154 816 patients.
COPD cohort includes 108 017 patients.
DM cohort includes 66 456 patients.
All diagnosis groups (N = 588 375).
Sensitivity analyses that restricted patients to a single admitting diagnosis (ie, allowing only unique patients in the overall cohort) showed results that were consistent with main findings.
Discussion
This retrospective analysis of hospital discharge records revealed evidence of PIV-associated complications and associated burden. It was found that nearly 2% of patients in the sample experienced a PIV-associated complication and that most of these complications were in the form of a BSI. Patients with pneumonia, diabetes, or MI had the highest rate of PIV complications (~2%) and BSI (>1%) among any of the admission diagnosis cohorts. In addition, it was found that admitted patients with a PIV-associated complication spent on average 2 additional days and required over $3000 more dollars while hospitalized compared with those without a complication. Patients with a PIV-associated complication were also twice as likely to be admitted to the ICU and nearly 3 times as likely to die while hospitalized than patients without a complication.
These results suggest that PIV use contributes to medical complications at a significant rate in US hospitals. Furthermore, such complications are associated with higher spending, longer and more intensive care, and higher mortality, all of which create burden for the health system and the patient.
The PIV-associated complication rates found in this study are plausible, as they are consistent with previously reported estimates.2,23 In particular, a robust 2006 systematic review of 200 prospective studies that examined and clinically verified intravascular device–related BSIs reported pooled rates of 0.1% to 3.7% (infections per 100 devices; 0.5-9.0 infections per 1000 intravascular device days) depending on the type of PIV used (plastic, steel, venous cutdown). 2 Several other studies have reported lower rates of PIV-associated complications but have focused on specific pathogens15,16,18,24 and on other countries,15-17,25 and used different study designs and units15,16,18 making results incomparable with those of our study. Moreover, reported rates may be lower than ours due underevaluation of such complications despite increasing use of PIV.15-18,25,26
Studies linking PIV-associated complications to outcomes were limited for comparison and appeared to focus on the effect of specific causative pathogens 15 rather than comparing outcomes according to the presence (vs absence) of a PIV catheter. Alternatively, other research examined predictors of PIV catheter infection, such as the clinical setting or placement location where a PIV catheter was used. 18
In addition to the main results, this study found that PIV-associated complications were linked to worse outcomes among the 3 admission diagnosis groups. Within each subgroup of CHF, COPD, or diabetes, patients with PIV-associated complications experienced longer hospital stays, higher costs, and higher mortality compared with those without complications, adjusting for key characteristics. Interestingly, the magnitude of these differences was considerably greater for patients with CHF compared with overall and other diagnosis groups. For CHF patients with a complication, length of hospital stay and admission to ICU were about double and risk of death about 5 times that of patients without complications. This finding may reflect a modified effect of PIV-associated complications among CHF patients, potentially due to an increased vulnerability in that patient population; however, our study design precludes confirmation of this.
The findings in this study underscore the need to reduce PIV-associated complications through a multistage approach as illustrated with the case of central line catheters. The identification of central-line catheter infections as public health problem, the development and implementation of quality measures and best practices for the insertion of central lines, and the creation of federal and state prevention programs have all led to a dramatic reduction in infections rates20,27-30 and conferred other clinical and economic benefits. Efforts such as these resulted in the reduction of 25 000 CLABSI in 2009, reflecting a 58% reduction from 2001 and corresponding to potentially 6000 lives saved and $414 million avoided in excess health care costs (ie, $1.8 billion cumulative costs) according to the CDC.27,30 Another study reported that CDC-sponsored prevention efforts saved $5.7 to $31.5 billion in direct medical costs. 10 Through similar recognition and effort, including programs such as the Hospital-Acquired Condition Reduction Program (HACRP) under the Affordable Care Act, which creates monetary incentives for hospitals to reduce hospital-acquired conditions, similar health and economic returns may be gained for PIV catheters.31-33
This study has several limitations. First, it was assumed that all patients received a PIV upon admission, which could not be verified in the data due to underuse of PIV-associated procedure codes in hospital discharge records; however, we believe this assumption was reasonable based on expert clinical input that the placement of a PIV catheter in every patient upon admission is standard practice. 34 Second, infections due specifically to PIV catheters may have been underreported in the data as there is no mandatory reporting for these devices, as required for central lines. To address this, we attributed the selected complications to PIV by ruling out other potential causes, both prior to and following admission. Nevertheless, the rate of PIV-associated complications in the sample may have been overestimated because we did not exclude other potential sources of the complications, such as sepsis related to ventilator-associated pneumonia. Third, although our findings appear consistent with previous estimates, 2 the approach we used to identify PIV-related complications was not validated pointing an area for future research. Finally, ANCOVA regression was used to estimate mean LOS and costs; as these variables often lack normal distribution, the use of ANCOVA allowed for interpretable results. However, we performed a sensitivity analysis on costs using median regression and found the results to be of a different magnitude albeit directionally similar (see Supplementary Materials) compared with ANCOVA with increased costs associated with a PIV-related complication in all groups (CHF: mean difference $5738, median difference $3712; COPD: $3538, $2008; DM: $2943, $2031; overall group: $3886, $2430).
Conclusion
This analysis of hospital records uncovered rates of PIV-associated complications that are meaningful and consistent with prior research. In addition, patients with PIV-associated complications have longer hospital stays, higher inpatient costs, and greater risk of death than patients without such complications. This relationship is present for multiple diagnosis groups. Future research should focus on expanding the current evidence base such that PIV-focused quality measures and policy interventions can be developed and implemented to reduce PIV-associated economic and disease burden.
Supplemental Material
Supplemental_table – Supplemental material for Increased Clinical and Economic Burden Associated With Peripheral Intravenous Catheter–Related Complications: Analysis of a US Hospital Discharge Database
Supplemental material, Supplemental_table for Increased Clinical and Economic Burden Associated With Peripheral Intravenous Catheter–Related Complications: Analysis of a US Hospital Discharge Database by Sangtaeck Lim, Gaurav Gangoli, Erica Adams, Robert Hyde, Michael S. Broder, Eunice Chang, Sheila R. Reddy, Marian H. Tarbox, Tanya Bentley, Liza Ovington and Walt Danker in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Appendix
Potential Non-PIV Causes of Complication Code List.

| Exclusion criteria | ICD-9-CM code |
|---|---|
| Dialysis | 38.95 Venous catheterization for renal dialysis
38
39.27 Arteriovenostomy for renal dialysis a 39.42 Revision of arteriovenous shunt for renal dialysis a 39.43 Removal of arteriovenous shunt for renal dialysis a 39.95 Hemodialysis a 50.92 Extracorporeal hepatic assistance (liver dialysis) 458.21 Hypotension of hemodialysis a 996.56 Mechanical complication due to peritoneal dialysis catheter a 996.68 Infection and inflammatory reaction due to peritoneal dialysis catheter 996.73 Complications due to renal dialysis device, implant, and graft E879.1 Kidney dialysis as the cause of abnormal reaction of patient, or of later complication, without mention of misadventure at time of procedure E870.2 Accidental cut, puncture, perforation or hemorrhage during kidney dialysis or other perfusion E871.2 Foreign object left in body during kidney dialysis or other perfusion E872.2 Failure of sterile precautions during kidney dialysis or other perfusion E874.2 Mechanical failure of instrument or apparatus during kidney dialysis or other perfusion V45.11 Renal dialysis status (renal, hemodialysis, peritoneal) V45.12 Noncompliance with renal dialysis V56.xx Encounter for dialysis and dialysis catheter care |
| Surgery, graft, implant or non-PIV device | Procedural codes: 00-86 (includes operations, obstetrical procedures, and procedures not elsewhere classified) 996.60 Infection and inflammatory reaction due to unspecified device, implant, and graft 996.61 Infections and inflammatory reaction due to cardiac device, implant, and graft 996.63 Infections and inflammatory reaction due to nervous system device, implant, and graft 996.64 Infections and inflammatory reaction due to indwelling urinary catheter 996.65 Infections and inflammatory reaction due to other genitourinary device, implant, and graft 996.66 Infections and inflammatory reaction due to internal joint prosthesis 996.67 Infections and inflammatory reaction due to other internal orthopedic device, implant, and graft 996.69 Infections and inflammatory reaction due to other internal prosthetic device, implant, and graft 998.5x Infected postoperative seroma and other postoperative infections 39 998.83 Nonhealing surgical wound b |
| Central line use | 38.97 CVC placement with guidance 89.62 Central venous pressure monitoring 999.31 Other/unspecified infection due to CVC 999.32 Blood stream infection due to a CVC 999.33 Local infection due to a CVC |
Note. PIV = peripheral intravenous; ICD-9-CM = International Classification of Diseases, Ninth Revision, Clinical Modification; CVC = central venous catheter.
See Taneja et al 38
See Suaya et al 39
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sangtaeck Lim, Gaurav Gangoli, Erica Adams, Robert Hyde, Liza Ovington, and Walt Danker III are employees of Ethicon, Inc. Tanya Bentley is a former employee, and Michael S. Broder, Eunice Chang, Sheila R. Reddy, and Marian Tarbox are employees of Partnership for Health Analytic Research, LLC, a health services research company hired by Ethicon to conduct this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Ethicon, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
