Abstract
Hemophilia is an inherited bleeding disorder that mainly affects males. Globally, there are about 400 000 people with hemophilia and only 25% of them receive adequate treatment. There is insufficient epidemiological data on hemophilia in Iraq; so, this study was conducted to determine the trend of the prevalence and incidence and to identify certain clinical and epidemiological characteristics of patients with hemophilia in Baghdad, Iraq, 2016. This cross-sectional study was conducted in the 4 hemophilia centers in Baghdad. The data were compiled by reviewing all hemophilia patients’ records and the centers’ registry books between 2007 and 2016. Corresponding population data of Baghdad were obtained from the Ministry of Health. The total number of currently registered patients who are residents of Baghdad was 654. The prevalence of hemophilia increased from 7.2/100 000 males in 2007 to 15.9/100 000 males in 2016. Similarly, the incidence of hemophilia had increased from 8.4/100 000 livebirths in 2007 to 16.3/100 000 livebirths in 2016. Severe hemophilia represented 63.4% of all types. The prevalence of hepatitis C virus (HCV), hepatitis B virus (HBV), and HIV infections were 22.9%, 0.9%, and 0.2%, respectively. Clotting factor inhibitors were positive in 11.6% and target joints were found among 45.1% of patients. About 27% were on prophylactic therapy. Only one death was recorded in 2016. The prevalence and incidence of hemophilia in Baghdad was doubled in 10-year period. The prevalence of different complications was almost close to the rates in the neighboring countries.
What do we already know about this topic?
Available information on the prevalence of hemophilia in Iraq is based on the estimates of the World Federation of Hemophilia (WFH). Few Iraqi studies were published and tackled mainly the clinical aspects of the disease.
How does your research contribute to the field?
This study provides more accurate measure of the incidence, prevalence, and the trend of the disease and using more accurate denominators. Also, we provide a full description of the types and the complications of hemophilia in Baghdad.
What are your research’s implications toward theory, practice, or policy?
The study provides more accurate estimates of the incidence and prevalence of the disease in the country instead of relying on external estimates. Also, the study demonstrates the increasing trend of both the incidence and the prevalence over the last decade.
The study provided a valid database to the Ministry of Health and it is already used to build upon and expended to all Iraqi provinces. It was also useful to elaborate the defects in registration. As example we discovered that 9% of the registered cases were duplicated.
The result of the study was useful in assessment of the needed amount of the treatment materials and other resources that are now accurately calculated for the whole country.
Introduction
Hereditary bleeding disorders (HBDs) are a group of multiple diseases that include inherited abnormalities of primary and secondary hemostasis. 1 HBDs result from the deficiency or functional abnormality of one of the plasma proteins involved in providing normal coagulation.2,3
Hemophilia and von Willebrand disease (VWD) are the most common HBDs. Hemophilia is classified into hemophilia A (Factor VIII Deficiency), hemophilia B (Factor IX Deficiency), and hemophilia C (Factor XI Deficiency). 2 Hemophilia A (also called Classic Hemophilia) is the second most common type of all HBDs after VWD, and the most common type of hemophilia in the world. 2 It is 5 times more common than hemophilia B (also called Christmas disease), while Hemophilia C is the rarest form.2,4,5 Hemophilia A and B are the only HBDs that are inherited in a sex-linked model, and the affected gene is located on the long arm of the X-chromosome. 4 Today, the treatment for hemophilia is very effective, and without treatment, most children with severe hemophilia will die young. 6
Globally, there are about 400 000 people with hemophilia and only 25% of them receive adequate treatment. 6 After introducing clotting factors, the life expectancy of patients with hemophilia has improved. In developed countries, life expectancy of patients with hemophilia is more than that in developing countries, but it is less than that of other people without the disease by 10 to 15 years. 7
Hemophilia A has been recognized in all areas of the world and in all ethnic groups. Estimates of its incidence approximate 1 in 5000 male livebirths, or 1 in 10 000 livebirths. Hemophilia B is less common with an incidence of 1 in 30 000 male livebirths.1,2 However, hemophilia C affects about 1 in every 100 000 populations.2,5 India ranked first in number of registered patients with hemophilia (A, B, and C) in 2016, with a total number of 18 383 patients with a prevalence of 1.4/100 000 populations, followed by the United States and China. 8
According to the annual report of World Federation of Hemophilia (WFH) in 2016, the estimated total number of patients with all types of hemophilia in Iraq in 2016 was 1346, making a prevalence of 3.7/100 000. 8
There is scarce data on the epidemiology of HBDs in Iraq. Therefore, this study aimed to estimate the incidence and prevalence of hemophilia in Baghdad, Iraq, determine the trend over the period of 2007 to 2016, and identify the main subtypes and complications.
Methods
Study Design and Study Population
This descriptive study was conducted on all registered patients with hemophilia in the 4 HBD centers in Baghdad and who resided in this city through 2016. These centers were Child Welfare Teaching Hospital, the National Center of Hematology/Al Yarmuk Teaching Hospital, the Central Pediatrics Teaching Hospital, and Al-Imamain Al-Kadhimiyain Medical City.
Data Collection Form
A structured data collection form had been used to abstract the following data:
Demographic characteristics: sex, age, residence, marital status, education, and occupation;
Clinical profile of the patients: type of hemophilia, blood group, date of diagnosis, severity of the disease, and type of treatment;
Surgical history: synovectomy, circumcision, dental intervention, and other types of surgical operations (if present);
Family history, parental consanguinity (relatives or not), and number of patients with hemophilia in the family;
Complications: arthropathy, viral infections (hepatitis B virus [HBV], hepatitis C virus [HCV], and HIV), presence of inhibitors, and other complications were also included;
Patient outcomes: maintained on treatment, death, or defaulter.
The data collection form was piloted in Child Welfare Teaching Hospital–HBDs center.
Data Sources
Data were collected by reviewing all patient records at these centers. Trend over the last 10 years was calculated using the data obtained from the registry books, regarding total number of patients with hemophilia and the newly registered patients every year. Data of Baghdad population and livebirths were obtained from Iraqi Ministry of Health (MOH)—Directorate of Planning and Resource Development—Health and Vital Statistics Department.
Operational Definition
Patient with hemophilia was defined as any person diagnosed by a physician as having bleeding tendency and confirmed with lab evidence of low activity level (below 40% of normal level) of any of the clotting factors (VIII, IX, or XI). 8 Patient with hemophilia A is any patient with hemophilia with clotting factor VIII (Factor 8) deficiency since birth.
Patient with hemophilia B is any patient with hemophilia with clotting factor IX (Factor 9) deficiency since birth. Patient with hemophilia C is any patient with hemophilia with clotting factor XI (Factor 11) deficiency since birth. Patient with acquired hemophilia is any person diagnosed by a physician as hemophilia that did not develop since birth. 6
Mild hemophilia: any person (male or female) with >5% to 40% of the normal level of clotting factor VIII or factor IX in the bloodstream. 4
Moderate hemophilia: any person (male or female) with 1% to 5% of the normal level of clotting factor VIII or factor IX in the bloodstream. 4
Severe hemophilia: any person (male or female) with less than 1% of the normal level of clotting factor VIII or factor IX in the bloodstream. 4
Target joint: it is that joint which had recurrent bleeding, as diagnosed by the physician and denoted in the patient’s record. It is usually defined as a joint in which at least 3 or 4 bleeds occurred within 3- to 6-month period. 2
Ethical and Official Approval
Ethical and official approvals were granted from the committee of health research/Iraq Ministry of Health and the administration of the 4 centers. All data were kept confidential and used exclusively for the sake of the research.
Statistical Analysis
Microsoft Excel and Epi info 7 programs were used for data entry and analysis. Categorical data were compared using chi-square test. A P value <.05 was considered statistically significant.
Results
Patients’ Characteristics
Of all registered cases in Baghdad centers, 654 patients were living in Baghdad in 2016. Male patients represented 97.1% of all patients with hemophilia. The mean (±SD) age of all patients with hemophilia was 19.6 (±15.0) years. The mean age was 18.5 (±14.0) years for patients with hemophilia A, 21.4 (±15.0) years for patients with hemophilia B, and 34.1 (±24.6) years for patients with other types of hemophilia. About 40.5% aged 19 to 44 years and 28.4% aged 5 to 13 years. Of the total number of patients with hemophilia aged ≥18 years (n = 315), 14.6% attended university, 59.6% were married, and 17.7% were ever-employed (current employees or retired). Of the 329 patients aged ≥6 years, 7.6% did not attend school. The proportion of patients with relative parents was 50.2%. About three quarters (72.7%) of the families of patients with hemophilia had only one patient.
The mean (±SD) age at first bleeding was 6.2 (±31.4) months and the mean (±SD) age at diagnosis was 4.4 (±24.2) months. More than one third of patients with hemophilia (35.2%) was with O Rh(D) Positive blood group, followed by B Rh(D) Positive (22.9%) and the lowest blood group was AB Rh(D) Negative (0.3%).
Prevalence of Hemophilia
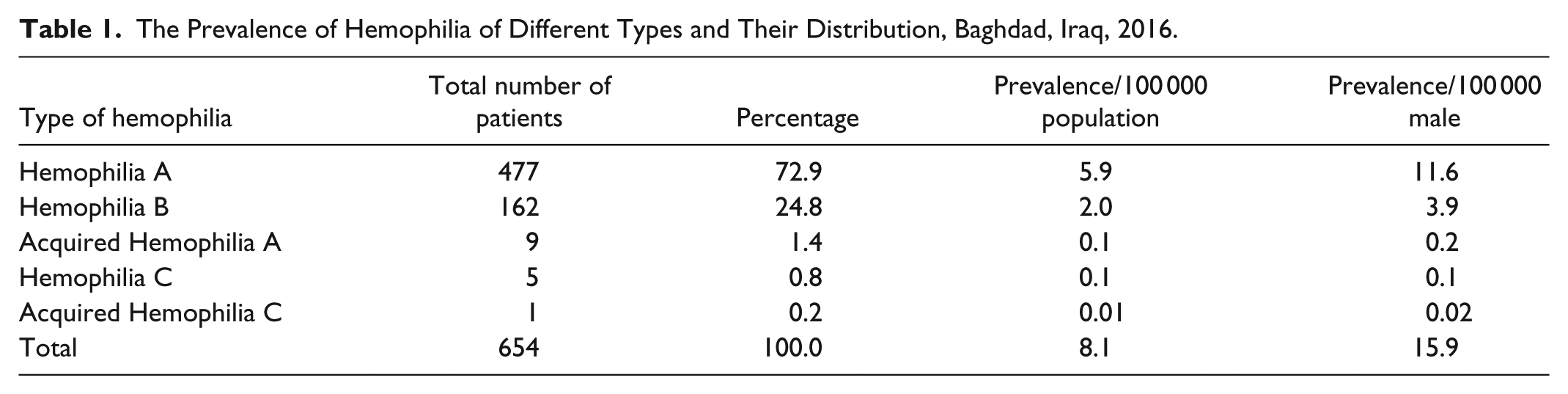
The prevalence of hemophilia was 8.1/100 000 population (15.9/100 000 males). Hemophilia A represented 72.9% of all types of registered hemophilia with a prevalence of 5.9/100 000 population and 11.6/100 000 males. The prevalence and frequency distribution of each type of hemophilia is shown in Table 1. The majority of patients with hemophilia were males (97.1%) with a prevalence of 15.5/100 000 male (11.6/100 000 male for hemophilia A, 3.7/100 000 male for hemophilia B, and 0.2/100 000 male for other types). Patients with hemophilia A and hemophilia B were classified according to the severity of the disease (Table 2). The highest percentage was the severe type (63.6%) followed by moderate type (20.1%) and mild type (16.3%). Other types of hemophilia have different types of classification and the number of patients of each type was so small.
The Prevalence of Hemophilia of Different Types and Their Distribution, Baghdad, Iraq, 2016.
Frequency Distribution of Hemophilia A and B Patients by Severity of the Disease and the Type of Hemophilia, Baghdad, Iraq, 2016.

Trend of Hemophilia
The prevalence of hemophilia A had increased from 2.8/100 000 population (5.6/100 000 male population) in 2007 to 5.8/100 000 population (11.5/ 100 000 male population) in 2016. The incidence of hemophilia A increased from 6.9/100 000 livebirths in 2007 to 11.6/100 000 livebirths in 2016. Meanwhile, the prevalence of hemophilia B increased from 0.8/100 000 populations in 2007 to 1.9/100 000 in 2016, and the incidence increased from 1.6/100 000 livebirths in 2007 to 4.7/100 000 livebirths in 2016. The incidence of hemophilia B showed marked increase in 2010 (7.3/100 000 livebirths) and another spike in 2014 (5.9/100 000 livebirths). In general, the prevalence and incidence of both hemophilia A and hemophilia B had increased between 2007 and 2016. The percent annual growth rate of prevalence of hemophilia from 2007 to 2016 was 11.4%; 10.7% for hemophilia A and 13.8% for hemophilia B. Figure 1 shows the trends in the prevalence rates per 100 000 populations and incidence rates per 100 000 livebirths of both hemophilia A and hemophilia B together. Table 3 shows the change in the prevalence and incidence rates of hemophilia A and hemophilia B over time, separately.

Trend of prevalence and incidence of hemophilia A and B in Baghdad, Iraq, 2007-2016.
Trend of Prevalence and Incidence of Hemophilia A in Baghdad, Iraq, 2007-2016.

Common Transfusion-Transmitted Viral Infections Among Hemophilia Patients
Regarding common transfusion-transmitted viral infections among patients with hemophilia, HCV was recorded in 22.9% of all patients with hemophilia (25.4% of patients with hemophilia A and 17.9% of patients with Hemophilia B), HBV was recorded in 0.9% of patients, and only one case of HIV was recorded in a patient with hemophilia B.
Inhibitors Against the Clotting Factors
Inhibitors against the clotting factors were present in 11.6% of patients (14.3% in patients with hemophilia A, 0.6% in patients with hemophilia B, and 77.8% in patients with other types).
Target Joints
Target joints were recorded among 45.1% of patients with hemophilia. Less than half (43.7%) of patients with hemophilia with target joints had only one target joint, while those with 2 target joints represented 32.9% and those with more than 2 target joints represented 23.4%. Among all joints, knee joints represented about half (48.6%) from all types of target joints, followed by elbow joints, and the least affected joint was the wrist joint.
Treatment of Hemophilia
Only about one quarter (26.9%) of patients with hemophilia was on prophylactic therapy. Other patients were either on demand therapy (43.6%) or both (on demand and prophylactic therapy) (24.9%), with 4.6% of data were missing. Treatment of hemophilia included different types of medications and blood products, in addition to factor concentrates. About half (48.6%) of the treatment products used for patients with hemophilia was factor concentrate, then tranexamic acid (27.8%), while desmopressin was the least product used (0.2%). Around 31% of patients with hemophilia received only one treatment product, and 36.2% received 2 products.
Surgical Interventions and Bleeding History
More than one third (35.3%) of patients with hemophilia had undergone surgical intervention in some point of their lives. The first ranking type of surgical intervention was circumcision with 43.3% from all interventions (15.3% from all patients), followed by dental extraction with 37.3% (13.1% from all patients). Patients with hemophilia experience bleeding from different parts of their bodies and the common bleeding sites among patients with hemophilia were joints (25.9% of all bleeding sites), followed by skin (and subcutaneous tissues) in 20.9% of all bleeding sites. The lowest percentage was for bleeding within the eyeball (0.2% of all bleeding sites).
The most common mechanism of bleeding among patients with hemophilia was trauma (38.5% of all mechanisms) and 13.6% of bleeding was after surgical operations. Spontaneous bleeding accounted for 29.8% of all mechanisms of bleeding.
Deaths Among Patients With Hemophilia
In 2016, there was only one death among patients with hemophilia, and the deceased was a 12-year-old child with a history of severe hemophilia A. The cause of his death was intracranial hemorrhage after exposure to trauma. During the period 2007-2016, there were 7 deaths among patients with hemophilia, 1 due to complications of a tumor in the left thigh, 1 after explosion, 2 due to internal bleeding after trauma, and 3 due to intracranial hemorrhage after trauma.
Discussion
Globally, there are about 400 000 people with hemophilia and only 25% of them receive adequate treatment. Without treatment, most children with severe hemophilia will die young, 2 and if treated properly, patients can live normal healthy lives. 6
Prevalence
This study revealed that the prevalence of all types of hemophilia was 8.1/100 000 population (15.9/100 000 male) which was higher than the global prevalence of hemophilia in 2016 which was 2.8/100 000 populations 8 and higher than the range of hemophilia prevalence among upper middle countries (4.8–13.2/100 000 male population). 9
The estimated prevalence of hemophilia in some neighboring and regional countries per 100 000 populations was Iran 7.1, Turkey 7, Egypt 6, Jordan 4.4, Syria 3.5, and Saudi Arabia 1.4.8,10
The highest estimated prevalence for hemophilia A, B, and C, in the world, was in the United Kingdom with 16.7/100 000 populations. 8
This variation in the reported prevalence among different countries could be due to inaccurate reporting practices between countries or due to actual differences in the prevalence resulted from varying availability of the treatment and consequent death of affected patients. 9 A study observed that the majority of people with hemophilia in the world have not been identified because of lack of diagnostic capability, no access to care, no economic means, and little to no available factor replacement therapy. 11 Without treatment, those with severe hemophilia often die in childhood or early adult life, thereby resulting in a decreased prevalence relative to the number of cases born.11,12 In addition, the reported hemophilia prevalence in lower income countries is often considerably less than that in higher income countries and less than expected from average international incidence. 9
Trend of Hemophilia
In this study, within 10-year period, the prevalence and incidence of hemophilia had increased. Between 2006 and 2016, there was a slight increase in the prevalence of hemophilia in some countries; in the United Kingdom from 10.4 to 10.7/100 000 populations, in the United States from 5.2 to 5.4/100 000 populations, in China from 0.2 to 0.8/100 000 populations, and in Brazil from 3.7 to 5.7/100 000 populations.8,13
The increasing prevalence over time could be due to many factors such as increased survival, improvement of diagnostic procedures and treatments, or due to improved reporting procedures, improvement in the access of care for hemophilia, or due to the effect of migration of patients to areas with better care. 9
The increasing incidence, in almost all countries, is because that hemophilia is an inherited disease, and there is no known way to prevent it, till now. However, genetic counseling for parents is available. 9 Individuals from high-risk populations, or those with family histories of hemophilia, may meet with genetic counselors to determine whether they carry a genetic mutation linked to hemophilia. Carriers can be determined through detailed family histories or by genetic testing. 14 Known hemophilia carriers may undergo genetic counseling before conceiving a child. Genetic counselors can explain the options and the associated risks of various tests, such as amniocentesis, chorionic villus sampling, or preimplantation diagnosis. 15 Some countries use prenatal diagnosis that followed by the option of medical termination of pregnancy. 16
Age Groups
The highest proportion of patients with hemophilia aged 19 to 44 years (40.5%) and only 6% aged ≥45 years. A previous study in Anbar governorate, Iraq, 2013, revealed that 65% of patients with hemophilia aged ≤15 years and only 11.7% aged ≥26 years. 17
According to WFH, 2016, the highest proportion of patients with hemophilia in Iraq was among those aged 5 to 13 years (38%), followed by age group 0 to 4 years (23%) and patients ≥45 years represented 2%. 8 There is considerable variation in the age distribution of patients with hemophilia among countries. In developed countries, patients with hemophilia live longer than those in developing countries. 7
Severity of the Disease
In this study, around two thirds of patients with hemophilia had severe type followed by the moderate type and the mild. The percentage of severe hemophilia A among upper middle income countries in 2016 was 19.3%, moderate hemophilia A was 10.1%, mild hemophilia A was 52.6%, and the rest of the percentage (18%) was unknown. 8 However, the percentages for hemophilia B were 16.9% severe, 8.1% moderate, 61.2% mild, and 13.8% unknown. 8
Many patients with mild hemophilia were not registered in many hemophilia centers, because the disease may go undiagnosed and be discovered only because of excessive hemorrhage postoperatively or following trauma. 4 In addition, many countries with lower income do not provide resources (both personnel and treatment products) for treating hemophilia, so many cases with severe hemophilia often die in childhood or early adult life. 9
Common Transfusion-Transmitted Viral Infections Among Patients With Hemophilia
The prevalence of HCV in this study (22.9%) was consistent with the prevalence of HCV among patients with hemophilia in Iraq, according to WFH report in 2016, which was 25.1%.
The prevalence of HCV among patients with hemophilia in 46 countries was 8.9%. In some neighboring countries, the prevalence of HCV was as follows: 22.6% in Saudi Arabia, 13.2% in Jordan, and 5.7% in Iran. Meanwhile, the prevalence of HCV was 21.7% in the United States and 10.9% in the United Kingdom. 8
The prevalence of HBV in this study was less than that reported in a previous study in the Medical City Complex in Baghdad in 2006 which was 3%. 18 Almost similar prevalence of HBV was revealed by a study conducted in Iran which was 1.1% 19 and another study in Pakistan which was 1.7%. 20 Our result was less than that of a study in Poland (7.8%). 21
Only one case of HIV was recorded in a patient with hemophilia B in Baghdad, which represented 0.2% from all patients with hemophilia. However, no cases of HIV were reported in 2016 in Iraq. 8 The prevalence of HIV among patients with hemophilia in 46 countries was 2.2%. 8 In some neighboring countries, the prevalence of HIV was as follows: 8.0% in Saudi Arabia, 0.6% in Jordan, and 0.5% in Iran. Meanwhile, the prevalence of HIV was 6.7% in the United States and 4.3% in the United Kingdom. 8
This variation in the prevalence of common transfusion-transmitted viral infections among patients with hemophilia depends on multiple factors, such as the usage of recombinant factors, type of blood component used, safety precautions, scientific activities in transfusion, strict donor selection, the prevalence of these infections in that country, and other causes.14,22
Inhibitors Against the Clotting Factors
The percentage of inhibitors among patients with hemophilia reported in this study (11.6%) was higher than the WFH, 2016 report for Iraq. 8 The prevalence of inhibitors among patients with hemophilia A in 82 countries was 2.3%, and in some neighboring countries, the prevalence was as follows: 14.0% in Saudi Arabia, 6.7% in Jordan, 5.0% in Iran, and 3.2% in Turkey, whereas in other countries, the prevalence was as follows: 23.7% in Morocco, 5% in Brazil, 1% in Egypt, and 3.9% in the United Kingdom. 8
The prevalence of inhibitors varies from country to country, and this variation is related to several factors which can be patient related (genetic, ethnicity, or immune system), treatment related (type of product, exposure to factor VIII/IX in terms of the age at the first treatment, treatment duration, and intensity), or diagnostics related (type and sensitivity of test detecting the inhibitor, frequency of inhibitor testing). 23
Target Joints
The percentage and type of the target joints in patients with hemophilia in Baghdad were consistent with the results of a previous study in the Medical City Complex in Baghdad, 2008, 24 and with a study in India, 2013. 25
Type of Treatment
About one quarter of patients with hemophilia was on prophylactic therapy, which is less than the proportion reported for Iraq in WFH, 2016 (45%). 8 The reported percentage in some countries was as follows: 82.5% in the United Kingdom, 81% in Japan, 74% in the United States, 60% in Qatar, 54% in Brazil, 25% in Morocco, 10% in Iran, and 1% in Egypt. 8
We can conclude that the prevalence of hemophilia of all types is higher than the global prevalence but within the range of hemophilia prevalence among upper middle countries. The prevalence and incidence had increased by 2-fold over the last 10 years. The prevalence of HBV and HCV among patients with hemophilia was almost similar to the rates reported in the neighboring countries, but HIV prevalence was less. The prevalence of other complications like target joints and inhibitors was also close to the rates in neighboring countries.
This study had a number of limitations, such as missing information and duplicate reporting resulted from lack of standard reporting system of HBDs in the hemophilia centers. Also, these centers received patients from other governorates in Iraq despite the presence of hemophilia centers in these governorates.
Footnotes
Acknowledgements
Authors would like to acknowledge the Eastern Mediterranean Public Health Network (EMPHNET) for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
