Abstract
Biocidal fabrics can reduce the transmission of pathogens caused by contaminated personal protective equipment (PPE). N-halamines are very effective and fast-acting biocides against bacteria and viruses. To explore the relevance of N-halamine compounds for use in PPE and operational clothing and equipment (OCE), this study investigates the impact of an N-halamine-based finish on the functional and aesthetic properties of fabrics used for medical gowns and military uniforms, and examines the effect of conditions simulating the PPE and OCE practical use on the N-halamine-based finish. It was observed that the presence of a water-repellent finish on the fabrics reduced the chlorine loading for the fabric made of hydrophilic fibers, whereas no effect was observed for the polyester fabric. No major effect of the finish application was measured on the fabric strength. In terms of the color, the gown fabric was strongly affected by the finish application and subsequent chlorination, whereas the effect on the military fabric was more limited. The treated fabrics remained within the requirements for Class 1 in terms of flammability. The results showed no impact of low chlorination temperature and different water quality levels on the chlorination efficiency. On the other hand, laundering, repeated abrasion, and exposure to UV radiation and perspiration simulating use conditions reduced the chlorine content in the fabric. These results provide some insight into the strengths and remaining challenges of N-halamine fabric finishes when considering practical applications for protective clothing.
Keywords
With more than 768 million confirmed cases and 6.9 million deaths worldwide, 1 the COVID-19 pandemic has been a devastating reminder of the difficulty in controlling the propagation of viruses. Although personal protective equipment (PPE) used by medical personnel can protect them from being exposed, their contaminated PPE may act as a vector for the coronavirus and lead to the contamination of other patients, other medical personnel, and personal objects/family members. For instance, researchers have shown that viable SARS-CoV-2 coronavirus, which causes the COVID-19 disease, is still detected on plastic surfaces 72 h after application. 2 Yet, masks, respirators, protective clothing, and other PPE are generally fabricated using petroleum-based polymer fibers.
Self-decontaminating PPE made from fabrics treated with a biocide finish could prevent this issue. For instance, N-halamines are a very efficient and fast-acting biocide thanks to their nitrogen–halogen bond, which acts as an oxidizing agent for the virus or other pathogens. 3 They are safe to humans and their great advantage lies in their ability to be recharged by treatment with sodium hypochlorite (bleach) once the halogen has been consumed by the oxidation/decontamination process. In addition, the development of resistance to pathogens has not been observed, possibly because of non-specific oxidation by the N-halamine functional groups. N-halamine compounds can easily be covalently bonded to the surface of textiles, which provides resistance to laundering. Their effectiveness has been demonstrated with viruses in addition to bacteria. 4
Various N-halamine compounds have been synthesized since their efficiency at purifying water was first revealed in 1931. 5 Three categories of N-halamine compounds can be identified, amine, amide, or imide, depending on the type of halogenated compound. They can be cyclic or acyclic, with cyclic N-halamines displaying the best stability. Hydantoin-containing N-halamines are the most frequently encountered cyclic N-halamines, as they are easily synthesized and provide excellent antimicrobial performance. They also display good laundering and better UV resistance. They can be used for textiles, water treatment, and paints, for instance. In the case of textiles, most of the work evaluating the performance of N-halamine finishes has been done on cotton fabrics. Recently, researchers developed an N-halamine finish based on 1-chloro-3-benzophenone-5,5-dimethylhydantoin (Cl-BPDMH) and successfully used it to functionalize a polyester fabric. 6
Recent research on N-halamines has looked at improving the resistance of these compounds to light, which has generally limited their application to undergarments and the inner surfaces of textile products. 5 A strategy explored by researchers involves combining the N-halamine-containing molecules with UV absorbers such as certain metal oxide nanoparticles. For instance, the UV stability of the N-halamine compound poly[5,5-dimethyl-3-(3′-triethoxysilylpropyl)hydantoin] (PSPH) that was grafted on cotton was strongly improved with the addition of titanium dioxide nanoparticles to the finish. 7 With the added titanium dioxide nanoparticles, it took 48 h of UV irradiation to decrease to the chlorine loading that was reached after 4 h of UV irradiation without nanoparticles. This improved UV resistance was attributed to an increase in the activation barrier of the scission reaction responsible for the loss of the biocidal active group. An improved UV stability of an N-halamine coating on cotton was also obtained using ZnO nanoparticles. 8 Alternatively, researchers have also directly functionalized photoactive nanoparticles with the N-halamine compounds.9,10
Another area of recent progress regarding the use of N-halamine compounds for biocidal function relates to the improvement of their efficiency and ability to target specific microorganisms. For instance, a quaternary ammonium salt was used to add a cationic center in the vicinity of two N-halamine bonds while forming a silicone antibacterial polymer. 11 This allowed the attraction of anionic bacteria to the N-halogen sites, thus overcoming the hydrophobicity of the N-halamine finish. Another strategy to improve the biocidal efficiency of N-halamine compounds takes advantage of the properties of matter at the nanoscale. A 6-phenyl-1,3,5-trialkane-2,4-dione N-halamine compound was grafted onto polyethyleneimine-modified graphene oxide. 12 The N-halamine-grafted graphene oxide nanosheets were immobilized onto cotton webs with polyurethane nanofibers and simultaneously spun into yarns. High active surface area and excellent immobilization of the N-halamine compound led to a 99.99% inhibition rate of Escherichia coli and Staphylococcus aureus bacteria after 50 washes. Moreover, selective antibacterial action against S. aureus was provided with nanoparticles formed by cross-linking methacrylamide with N,N-methylene bis(acrylamide) and further converting the amide bonds with sodium hypochlorite. 13 Reactive oxygen species were only generated by exposure to organic media and not upon suspension in water. In addition, the active nanoparticles displayed a unique, bacteria-encircling process with the S. aureus bacteria.
Recent research has also focused on preparing multifunctional fabric finishes incorporating N-halamine compounds. For example, combined biocidal efficacy and flame retardancy was provided to a cotton fabric using an ammonium salt of tris(methylphosphonate)aminomethane (TAMPU). 14 The heat release upon exposure to flame was reduced due to the char-forming ability of TAMPU, while the fabric exhibited biocidal activity within 5 min of exposure to S. aureus and E. coli bacteria. Researchers have also combined the N-halamine bacteria-killing function with a bacteria-release ability. 15 They achieved it by treating a cotton fabric with poly(N-isopropyl-acrylamide) (PNIPAM). After chlorination, the fabric displayed biocidal activity. The bacteria-release function was observed with the PNIPAM-treated fabric when brought to low temperature.
Thanks to the progress achieved, N-halamine-enabled textile products have started reaching the market. To foster further advances in the application of N-halamine compounds to PPE and operational clothing and equipment (OCE), this study investigated the impact of an N-halamine-based finish on the functional and aesthetic properties of fabrics currently used for medical gowns and military uniforms. It also examined the effect of the presence of a water-repellent finish on the fabric and of simulated use conditions such as abrasion, laundering cycles, and exposure to perspiration and UV light on the fabric chlorine content. In addition, the study assessed the sensitivity of the self-decontaminating function to variations in the chlorination application conditions that would be associated with the severe environments and austere conditions encountered, for instance, during military missions.
To the authors’ knowledge, this is the first published account of attempts to apply an N-halamine-based finish on fabrics of various fiber contents that are used for medical gowns and military uniforms. It is also the first time the effects of the water quality for the sodium hypochlorite recharge, presence of a water-repellent finish, application of abrasion cycles, and exposure to perspiration on the efficiency of the N-halamine-based fabric finish are reported.
Materials and methods
Materials
Fabrics
Three fabrics were used for this study. A summary of their physical properties is presented in Table 1. The first fabric, F1, is a plain weave material used for medical gowns. It is woven from filament polyester yarns with the insertion of an anti-static carbon fiber after every 30 warp yarns (see Figure 1(a)). The use of anti-static fiber is often observed in medical gown fabrics made with hydrophobic materials in low-humidity climates to reduce charging issues. Two versions of the fabric were included in the study. Medical gown fabric F1a had no finish, whereas F1b had a water-repellent finish. The second fabric, F2, is a military uniform fabric with a wrinkle-resistant finish. It is a ripstop weave made from blended, 2-ply yarns of cotton and nylon (see Figure 1(b)). The third fabric, labeled F5, is also a military uniform fabric. It is a plain weave made from 2-ply yarns that combine filament nylon in one ply with cotton in the second ply (see Figure 1(c)). Two versions of fabric F5 were included in the study. Military uniform fabric F5a had no finish, whereas F5b had a water-repellent finish. Images of the fabrics were taken by a stereomicroscope with an integrated camera Leica (Model EZ4W, Leica microsystems (Schweiz) AG, Heerbrugg, Switzerland).
Fabric physical properties
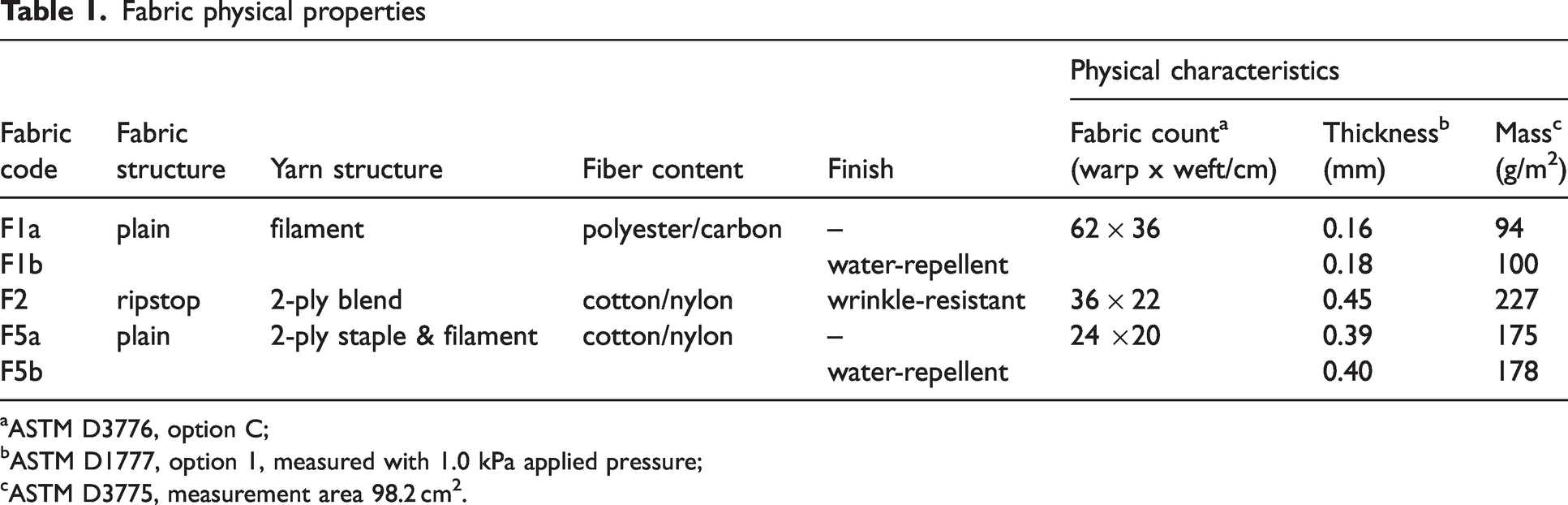
ASTM D3776, option C;
ASTM D1777, option 1, measured with 1.0 kPa applied pressure;
ASTM D3775, measurement area 98.2 cm2.

Surface light stereomicroscope images of the fabrics: (a) medical gown fabric F1b, (b) military uniform fabric F2, and (c) military uniform fabric F5a. (scale bar 0.25 mm).
Chemicals
The chemicals used for the synthesis of the N-halamine PSPH are 5,5-dimethylhydantoin (purity ≥ 97%), potassium hydroxide (purity ≥ 85%) and 3-chloropropyltriethoxysilane (purity ≥ 95%) (Sigma-Aldrich, USA), N, N-dimethylformamide (DMF) (purity ≥ 99.8%) and 100% ethanol (purity: 99.9%) (Fisher Scientific, USA), and hydrochloric acid (HCl) (36.5–37%, Caledon Laboratories, USA). Ethanol (Commercial Alcohols, Ontario, Canada) was also used to prepare the finish solution using the PSPH polymer.
A 5% sodium hypochlorite solution (Sigma-Aldrich, Missouri, USA) and 0.1 M sulfuric acid (Caledon Laboratories Ltd., Alberta, Canada) were used for the chlorination process. The titration process used potassium iodide (KI) (99.6%, Fisher, Ontario, Canada), 1 M acetic acid pH 4 buffer (Fisher, Ontario, Canada), 0.001 N sodium thiosulfate (Sigma-Aldrich, Missouri, USA), and 1% starch indicator (Fisher, Ontario, Canada).
For the antibacterial testing, S. aureus (SA) (ATCC 25923) and E. coli (EC) (ATCC 25922) bacteria were used. Supplies for the test include agar plates (Tryptic Soy Agar, Dalynn Biological Inc., Canada), 0.02 N sodium thiosulfate (99%, Sigma-Aldrich, St. Louis, MO, USA), phosphate buffer saline solution (pH 7.4, no calcium, no magnesium, Fisher Scientific, USA), and Triton × 100 (Fisher Scientific, USA).
The acid perspiration solution was prepared using sodium chloride (Fisher Scientific, Ontario, Canada), lactic acid (Fisher Scientific, Ontario, Canada), sodium phosphate (Fisher Scientific, Ontario, Canada), and l-histidine monohydrochloride (JT Baker, Fisher Scientific, Ontario, Canada).
Sample preparation
PSPH was selected for the study as it has been reported to display a good resistance to washing when applied to cotton. 7 The synthesis protocol is described in Worley et al. 16 In summary, a solution of 0.5 M of 5, 5-dimethylhydantoin and 0.5 M of potassium hydroxide in ethanol was refluxed until the solution became clear. The solid 5, 5-dimethylhydantoin potassium salt was obtained upon drying in a vacuum oven at 60°C for 3 days. Anhydrous DMF was added to the dry salt and the mixture was heated at 60°C until a clear solution was formed. 3-chloropropyltriethoxysilane (0.5 M) was added drop-wise while stirring at ambient temperature. Then the mixture was heated to 95°C for 4 h and further cooled down. The precursor monomer 3-triethoxysilyl propyl-5, 5-dimethyl hydantoin was obtained after filtration of the potassium chloride produced in the reaction and removal of the DMF solvent by vacuum distillation. Ethanol and water were added to the precursor monomer and the pH was adjusted to 3.5 by adding HCl. The mixture was refluxed with stirring for 5 h and then vacuum-distilled to reduce the volume of the solution. The remaining solution was poured into a beaker and then left in a vacuum oven at 60°C for 3 h, then 100°C for 3 h, and finally at 150°C for 3 h. When cooled down, it yielded PSPH in the form of a glossy solid. Figure 2 shows the spectrum by proton nuclear magnetic resonance spectroscopy (1H NMR, Agilent i400, Agilent Technologies, Santa Clara, California) of the PSPH produced using dimethyl sulfoxide-D6 as a solvent. The peaks characteristic of PSPH can be observed at 0.52 ppm (2H), 1.26 ppm (6H), 1.53 ppm (2H), 1.74 ppm (1H), 3.30 ppm (3H), and 8.14 ppm (1H). 16 The extra peaks at 3.30 ppm (4H) and 3.56 ppm (1H) are possibly attributed to the oligomer or unreacted chemicals.
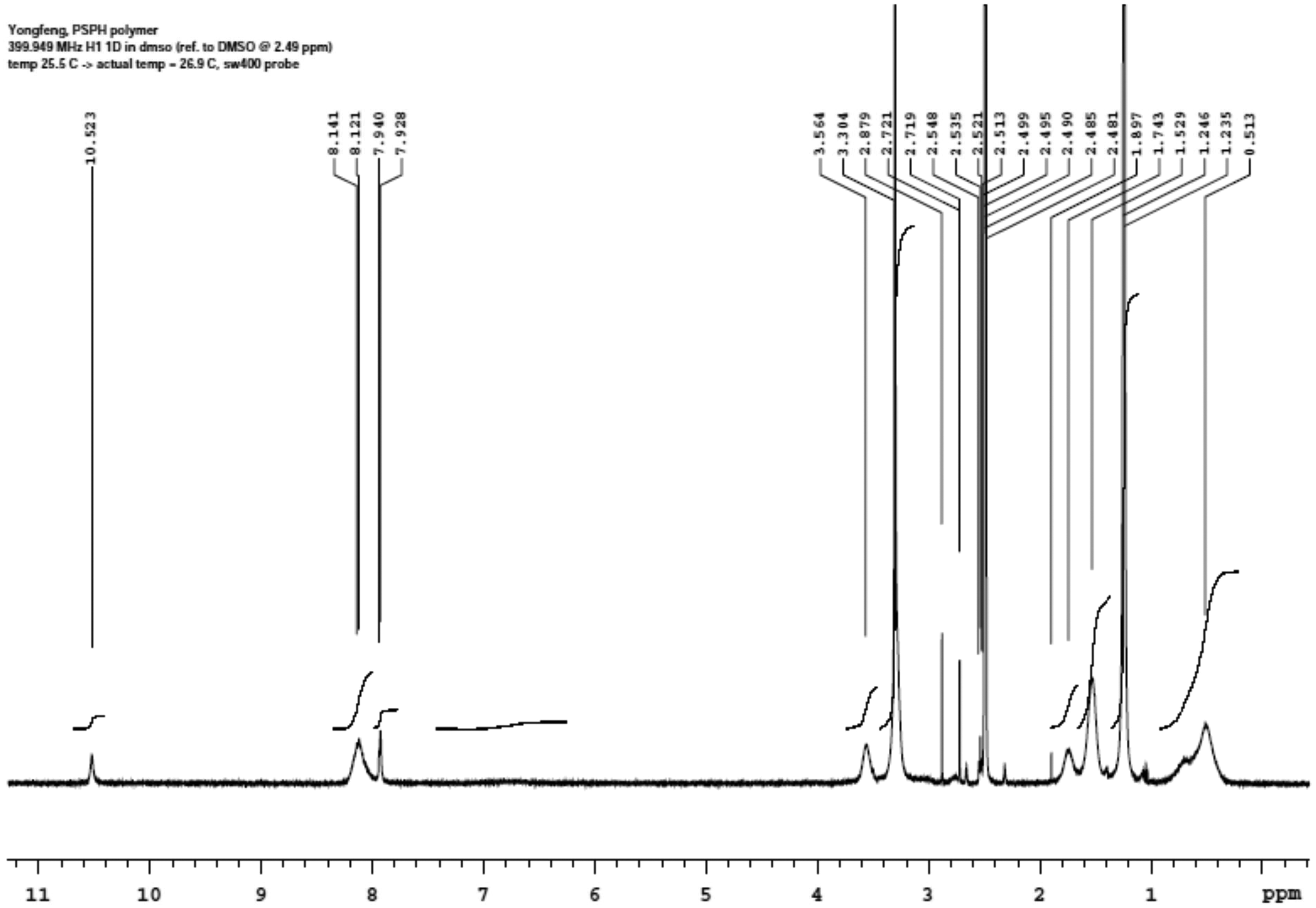
1H NMR of PSPH.
The protocol for the PSPH coating and chlorination of the sample was adapted from Liu et al. 7 Fabrics were cut into 80 × 200 mm pieces before polymer application. A 3–5% PSPH solution was prepared in 100 mL of 100% ethanol and stored in a glass jar. The PSPH solution was stirred using a magnetic stir plate until the polymer was completely dissolved. Once the PSPH solution was prepared, cut fabrics were placed in a 240 × 240 × 20 mm plastic tray. The PSPH solution was poured overtop the cut fabrics and was left to soak for 15 min. A lid was placed on top of the tray to avoid evaporation losses. After soaking for 15 min, fabrics were removed with tweezers and placed in an oven set to 95°C for 1 h. After removal from the oven, fabrics were placed in a glass beaker containing 75°C deionized (DI) water for 5 min. Following this, fabrics were placed in an oven set to 50°C for 1 h. After this procedure, the coated fabrics were stored in a dry location away from light until chlorination.
For the chlorination step, a 5% sodium hypochlorite solution was diluted tenfold with DI water to create a 100 mL mixture using a glass jar. The pH of the dilute sodium hypochlorite solution was adjusted by adding a 0.1 M sulfuric acid solution drop-wise until a pH of 7 was achieved, indicated by adding a drop of the solution to pH paper. Once the chlorination solution was ready, 80 × 200 mm specimens of fabrics that had been coated with PSPH were placed in a 24 × 24 × 2 cm plastic tray. All 100 mL of dilute hypochlorite solution was poured onto the fabric cuttings inside the plastic container. The chlorination lasted 1 h. Every 5 min, tongs were used to flip each piece of fabric during the chlorination period. After chlorination, fabric cuttings were rinsed three times with DI water then placed in an oven set to 50°C for 1 h to remove excess free chlorine. The specimens were wrapped in aluminum foil and stored in a dry environment until testing or exposure to selected conditions occurred.
Performance characterization
Antibacterial test
The antibacterial efficiency of the finished and chlorinated fabrics F1b and F5a was assessed according to the standard test method AATCC 100 with some modifications. 17 Unsterilized stacks of 5 cm diameter fabric specimens were placed in 175 mL jars. The stacks included five layers for fabric F1b and three layers for fabric F5A so that the weight of each stack equals 1.0 ± 0.1 g. A 24 h culture broth with each bacteria strain (SA and EC) was diluted (1:20) in DI water amended with 0.05% Triton X 100. 1.0 mL of the diluted broth was inoculated onto the control and treated specimens, which were incubated at 35 ± 2°C for 24 h. These control specimens were produced by subjecting the fabric pieces to the same protocol as the treated fabric except that the PSPH polymer was omitted during the finish application and no sodium hypochlorite was used for the chlorination treatment. Measurements on the control samples were also performed at “time 0” of bacterial contact.
Then, 5 mL of the sodium thiosulfate solution was added to the jars followed by 95 mL of phosphate saline buffer solution. The jars were shaken on a vortex for 1 min, followed by serial dilutions. Then, 20 µL of the solutions were pipetted onto the nutrient agar plates. The agar plates were incubated at 35 ± 2°C for 24 h prior to counting of colony-forming units (CFU). Triplicates were produced for the control and treated specimens.
Titration
The efficiency of the chlorination treatment and the impact of the simulated use conditions on the fabric finish were quantified by determining the chlorine content on the fabrics via iodometric/thiosulfate titration. The protocol was adapted from Liu et al. 7 Fabrics were cut to produce 0.2 g pieces. The 0.2 g fabric cutting was placed in an Erlenmeyer flask containing 20 mL of DI water; 0.5 g of KI was then added to the Erlenmeyer flask, and 1 mL of 1 M acetic acid pH 4 buffer was dispensed into the Erlenmeyer flask as well. The solution was stirred on a magnetic stir plate for 10 min at room temperature. After 10 min, the solution was ready to be titrated using 0.001 N sodium thiosulfate as the titrant. 1% starch indicator was added to the solution part way through to enhance the visualization of the end point. Titrations were performed in triplicate.
To determine the percentage of chlorine present on the fabric sample, the following equation was used:
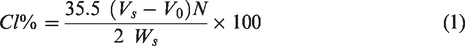
Strength
The breaking strength of fabric F1b and F5a before and after the PSPH finish application and chlorination treatment was measured in accordance with ASTM D5035. 18 Five specimens for each fabric were prepared in the warp direction. A specimen size of 35 mm in width (raveled to 25 mm) by 150 mm in length was used. All specimens were conditioned at 20°C and 65% relative humidity for at least 24 h prior to testing. They were tested at the same standard conditions. Testing was performed at a constant rate of extension of 300 mm/min, using an Instron tensile testing apparatus (Model 5565, Instron, Norwood, MA, USA). The maximum force at break was recorded in Newtons (N) for each specimen and the average breaking strength calculated for each fabric and treatment condition.
Color
The original fabric color and the color change resulting from the PSPH finish application and chlorination treatment on fabric F1b and F5a were recorded with a spectrophotometer (Model LabScan XE, Hunter Lab, Reston, VA, USA) following AATCC EP 1. 19 The test was performed at room temperature. Five specimens of fabric F1b and 10 specimens of fabric F5a were stacked with a white background and their colors measured under daylight settings, 10° standard observer, and D65 illuminant. The measurements were performed using a 44.5 mm diameter target mask.
Color differences were calculated using the CIELAB color space: 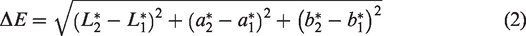
Flammability
The flammability of fabrics F1b and F5a before and after the PSPH finish application and chlorination treatment was evaluated in accordance with 16 CFR Part 1610. 21 Five specimens of fabric F1b and F5a were prepared in the warp direction for each condition. When tested by the prescribed method of this standard, these fabrics are expected to exhibit normal flammability characteristics and fall into Class 1. The fabric specimens were conditioned prior to testing by oven drying for 30 ± 2 min at 105 ± 3°C, and cooling in a desiccator with anhydrous calcium sulfate desiccant for at least 15 min. The ignition gas was butane, and the flame application time was 1 s. The time for the specimens that ignited during the flammability test to burn to the stop cord at a distance of 127 mm was recorded.
Application of simulated service conditions
Exposure to light
Five specimens of finished and chlorinated fabrics of F1b and F5a were exposed for 12.2 h to artificial light in a xenon arc Weatherometer (Model Ci35W, Atlas, Chicago, IL, USA), following test method ISO 105-B02. 22 The test conditions included an irradiance of 1.1 W/m2 at 420 nm, borosilicate (Type S) inner and soda lime outer lamp filters, black panel temperature of 65 ± 3°C, and relative humidity of 30 ± 5%. After exposure, the specimens were stored in aluminum foil until titration.
In addition, three treated fabric samples of both F1b and F5a were left outside (Sherwood Park, Alberta, Canada) for 48 h during June 25–27, 2021; the specimens received approximately 34 h of sunlight over this 48 h period. After the 48 h exposure period, the specimens were stored in aluminum foil until titration.
Exposure to perspiration
To determine the effect of exposure to perspiration on the chlorine content in the finished fabric, three specimens of the treated fabrics F1b and F5a were exposed to a test procedure used in the assessment of colorfastness to perspiration, AATCC TM15-2021e. 23 The acidic perspiration solution, with a pH 4.3, consisted of 10.00 ± 0.01 g of sodium chloride, 1.00 ± 0.01 g of lactic acid, 1 ± 0.01 g of sodium phosphate, and 0.250 ± 0.001 g of l-histidine monohydrochloride, in 1 L of distilled water. Each specimen was soaked in the perspiration solution for 30 min, then drip-dried until the saturation weight was about 2.25 times the dry weight. The specimens were then squeezed between plexiglass plates of the perspiration tester and placed in an oven for 4 h at 37 ± 2°C. After 4 h in the oven, the specimens were removed from the tester and allowed to air dry for 8 h. While drying, the specimens were loosely covered with aluminum foil to protect them from light and dust. After the 8 h drying period, the specimens were stored in aluminum foil until titration.
Laundering
Five specimens of the treated fabrics F1b and F5a were exposed to an accelerated test procedure for colorfastness to laundering, ISO 105-C06, 24 using a Launderometer (Atlas, Chicago, IL, USA) test apparatus and small stainless steel canisters for washing individual specimens. The wash solutions for all tests contained 0.4% w/v of 1993 AATCC Standard Reference Detergent without optical brightener. For fabric F1b, test number C1M was followed, using 50 mL of detergent solution with 50 steel balls, a 60°C wash temperature, and washing time of 45 min. For fabric F5a, test number B1M was followed, using 150 mL of detergent solution with 50 steel balls, a 50°C wash temperature, and washing time of 45 min. Following the wash cycles, the specimens were rinsed in distilled water. Excess water was removed by blotting on cotton towels and then the specimens were allowed to air dry for 8 h. While drying, the specimens were loosely covered with aluminum foil to protect them from light and dust. After the 8 h drying period, the specimens were stored in aluminum foil until titration.
Abrasion
Four specimens of the treated fabrics, F1b and F5a, were exposed to the abrasion test procedure, ASTM D4966, 25 using a Martindale pilling and abrasion tester (Model M235, SDL Atlas, Rock Hill, SC, USA). Each specimen was exposed to 1000 cycles of abrasion against plain weave crossbred worsted wool abradant fabric: this condition with a low number of abrasion cycles was selected so that it would not damage the fabric itself but would reveal if the finish is easily rubbed off from the fabric surface. At the end of the abrasion treatment, the specimens were stored in aluminum foil until titration. For the calculation of the residual chlorine content after abrasion, a correction was made for the edge of the specimen, which was held in the clamp and thus was not subjected to abrasion. In addition to evaluating the effect of the abrasion on the residual chlorine content, the specimens were also weighed before and after exposure to abrasion to assess the change in mass resulting from the abrasion.
Statistical analysis
T-tests or ANOVA were performed when relevant to determine the statistical significance of the data. A p-value lower than 0.05 was considered to be indicative of significant differences between results for two groups of specimens.
Results and discussion
Antibacterial efficiency
The antibacterial efficiency of fabric F1b and F5a treated with the PSPH finish and further chlorinated was assessed by comparison with control samples. These fabrics were selected for this part of the study as they are the versions of fabric F1 and F5 that are commercially available and effectively used for medical gowns and military uniforms, respectively. The results are listed in Table 2.
Results of the antibacterial test on fabric F1b and F5a
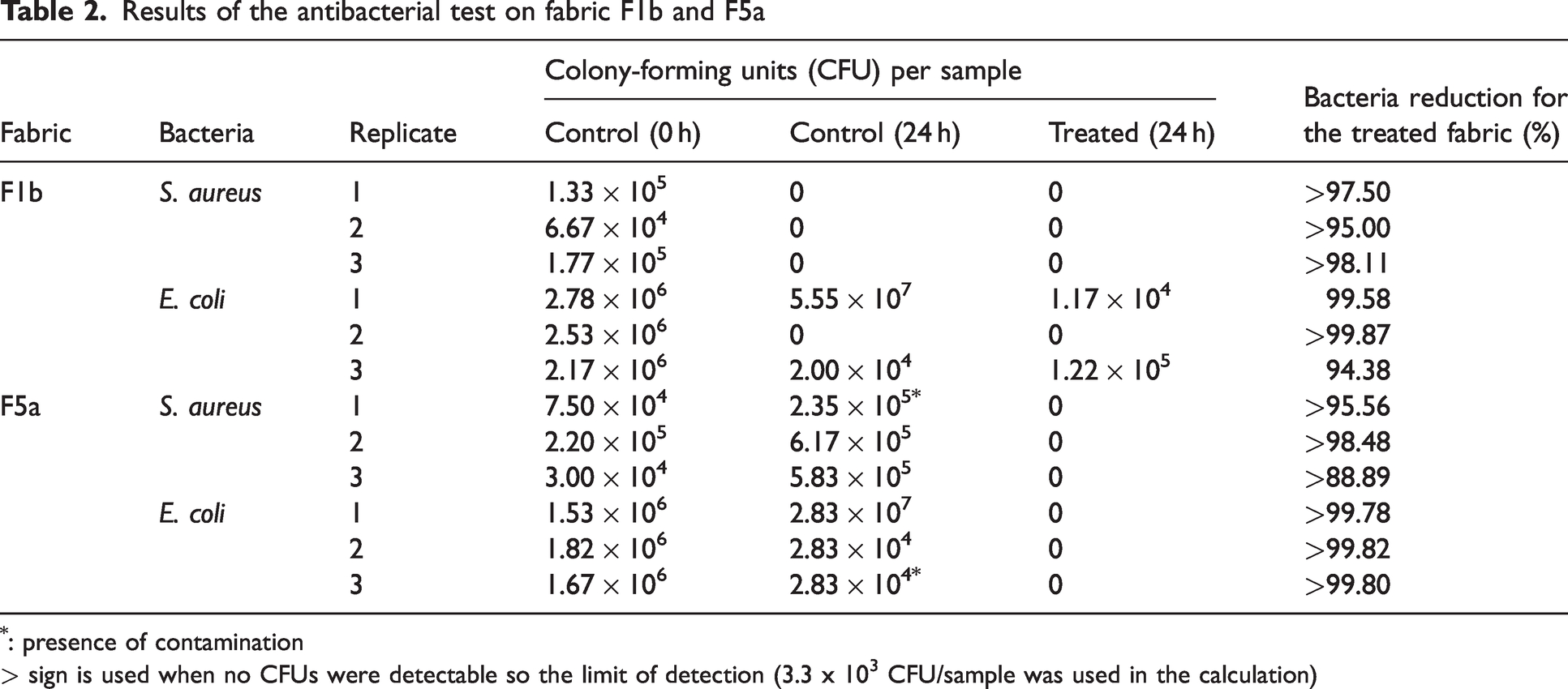
: presence of contamination
> sign is used when no CFUs were detectable so the limit of detection (3.3 x 103 CFU/sample was used in the calculation)
The results with fabric F5a clearly show the antibacterial activity of the finish: none of the treated samples showed any bacterial growth, both for SA and EC, whereas consistent bacterial growth was recorded on all the control replicates (Figure 3). In the case of fabric F1b, no bacterial growth was observed either on the control and treated samples with SA. In the case of EC, some control and treated replicates showed the presence of bacteria whereas others did not. This may have been caused by the presence of the water-repellent finish on fabric F1b, which prevented the proper inoculation of the specimens. For SA, it is also possible that the water-repellent finish itself contributed to preventing the growth of the bacteria. 26
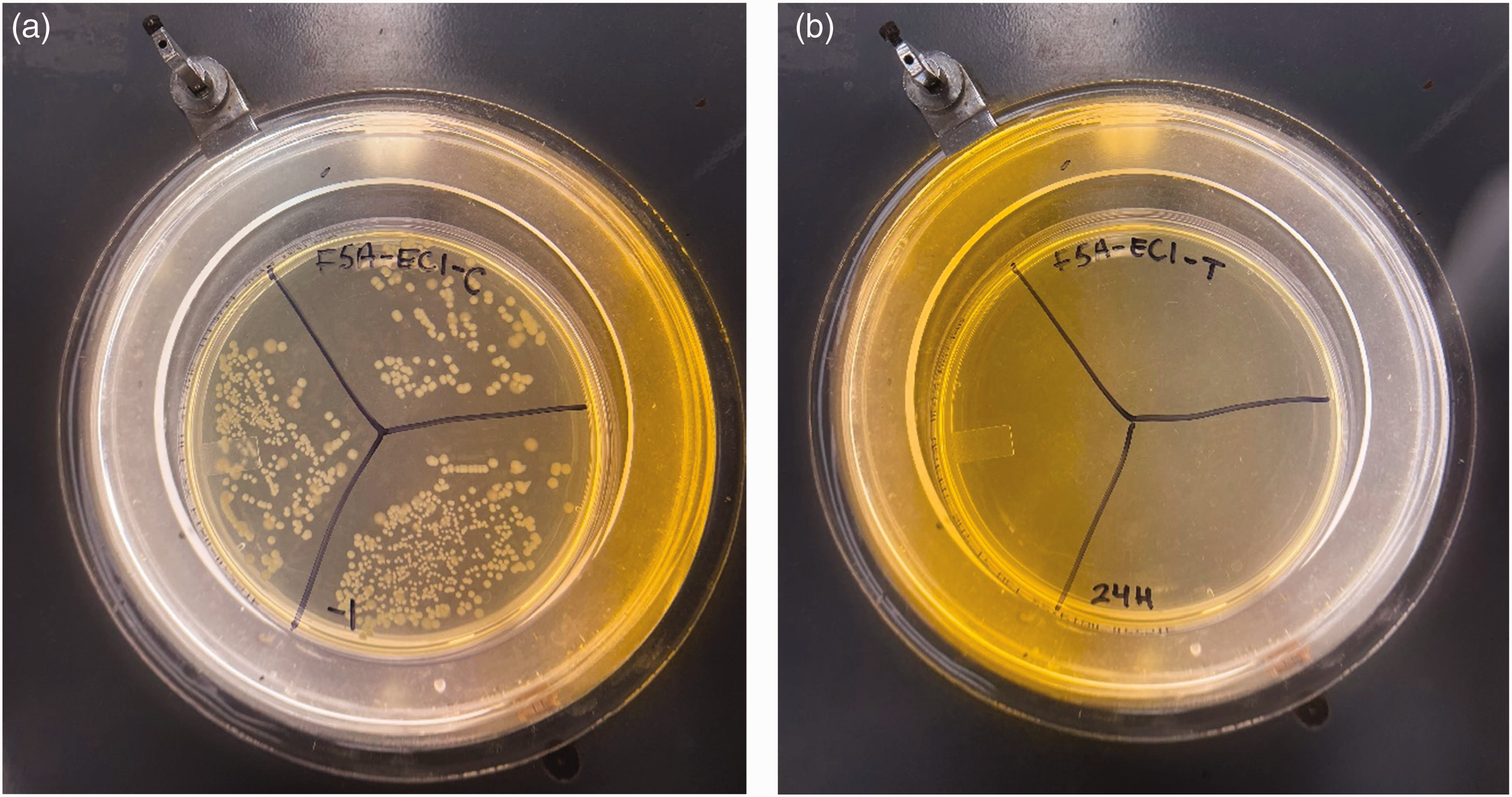
Example of results of antibacterial tests obtained with fabric F5a with E. coli bacteria: (a) control (no PSPH polymer used during the finish application and no sodium hypochlorite used for the chlorination treatment) and (b) with the PSPH finish and further chlorination.
Using the value of the detection limit of 3.3 × 103 CFU per sample when no growth was detected after 24 h of incubation, the reduction in bacteria was calculated using the 0 h values for control samples as a reference (according to Equation 2 in AATCC TM100) 17 . For fabric F5a, EC reduction was higher than 99.78% with the chlorinated PSPH finish, whereas it was higher than 88.89% for SA. However, in the case of fabric F1b, bacterial reductions of 95% or higher were observed. However, it is not clear if the effect can be attributed to the presence of the water-repellent finish and/or the antibacterial finish on the surface of the fabric.
Effect of the chlorination conditions on the chlorine content
The effect of the chlorination conditions on the chlorine loading in the finished fabric was tested with fabric F1a and F2. The two fabrics correspond to two types of fiber contents: polyester for fabric F1a and cotton/nylon blend for fabric F2. The conditions studied are the chlorination temperature, water quality, duration, and fabric to chlorine solution volume ratio.
For the study of the effect of the chlorination temperature on the chlorine loading in the finished fabric, the protocol described in the “Sample preparation” section was used with two different temperatures for the chlorination solution. In addition to the usual room temperature condition (21°C), specimens were also chlorinated at 4°C. This temperature was selected to represent the cool conditions experienced by deployed soldiers, and people on hiking, hunting, or fishing trips in temperate and continental climate zones. The results of the titration of the specimens chlorinated at the two different temperatures are shown in Figure 4. The temperature did not appear to influence the amount of chlorine in these fabrics (p-values of 0.872 and 0.899 for F1a and F2, respectively). A large standard deviation was observed for the results at 21°C for fabric F2 compared with the other conditions in terms of fabric and chlorination temperature. No explanation was found for it other than the variability of the finish application process.
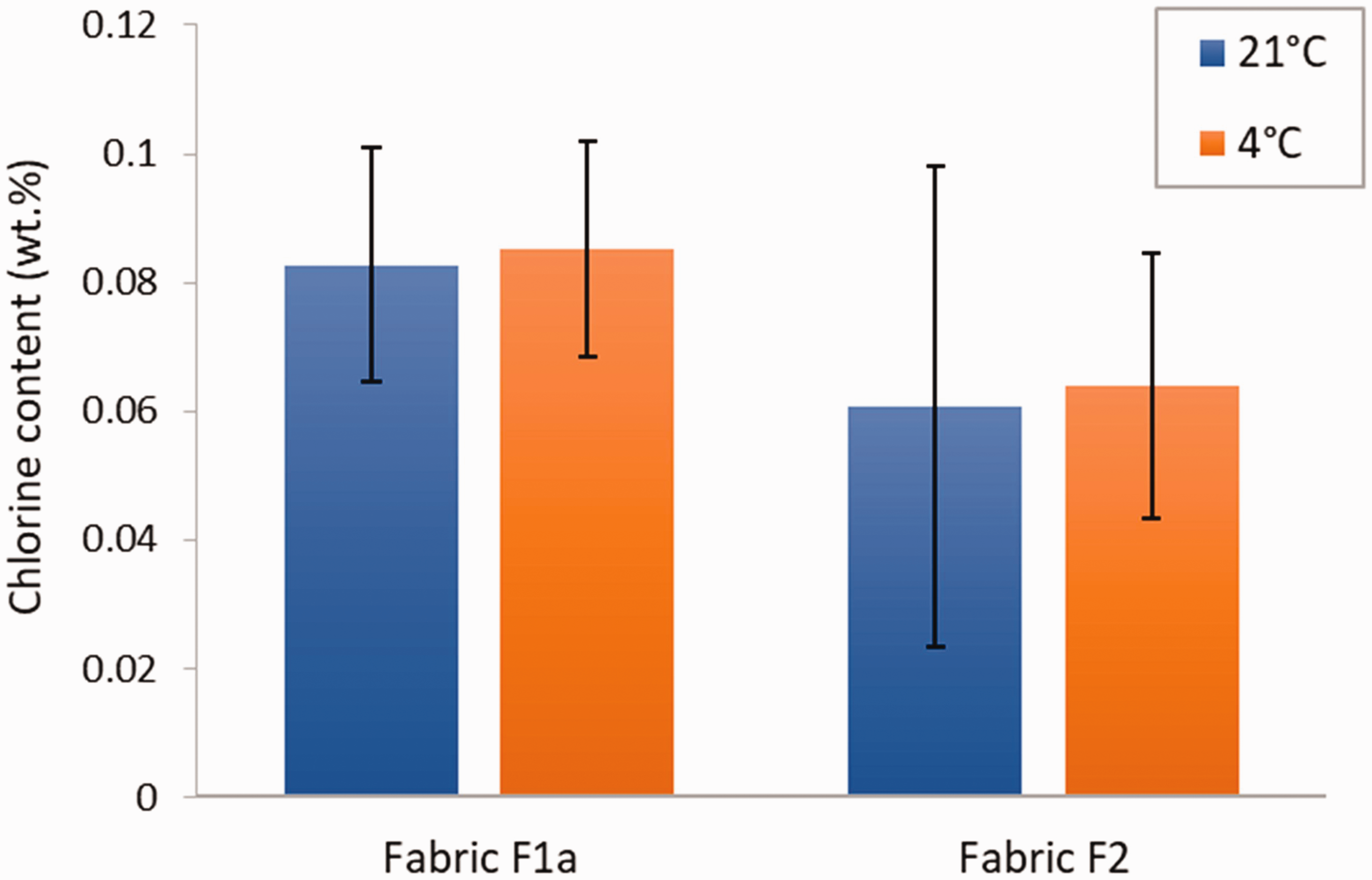
Chlorine content in coated fabric specimens chlorinated at 21 and 4°C. Error bars represent the standard deviation (N = 3).
Studies in the literature have shown conflicting results regarding the effect of the chlorination temperature. Some researchers have reported a large increase in the chlorine content and antibacterial activity of an N-halamine polyurethane polymer 27 and silica nanoparticles coated with polystyrene and functionalized with N-halamines 28 when the chlorination temperature was increased from 0°C to room temperature. Both studies reported that the chlorine content and antibacterial activity then decreased with further increase in the chlorination temperature.27,28 On the other hand, other researchers observed an increase in chlorine content with N-halamine functionalized polymethacrylamide nanoparticles when the chlorination temperature was increased from 30 to 60°C. 29 However, none of these studies involved N-halamine-functionalized fabrics or PSPH. In the case of our results, the absence of a significant effect of the chlorination temperature on the finished fabric chlorine content shows the robustness of the biocidal finish when chlorination is performed at low temperature.
The study of the effect of the water quality used for the chlorination on the chlorine content of the finished fabric was conducted using DI water, tap water (municipal water system, Edmonton, Alberta, Canada), and lake water collected in May 2021 from Hastings Lake (Alberta, Canada). The use of lake water simulated the conditions that deployed soldiers and people on hiking, hunting, or fishing trips may face in remote locations. The results in terms of chlorine content are shown in Figure 5. For fabric F1a, a slightly lower chlorine loading was recorded for tap water. In the case of fabric F2, the chlorine content with DI water was about 30% lower compared with the case where chlorination was conducted using tap and lake water. No reasons were identified to explain this lower chlorine loading with DI water. However, the results indicate that chlorine contents with tap and lake water, which are more relevant to practical use in the field compared with DI water, would not produce lower chlorine contents in the fabric. For both types of fabrics, a one-way ANOVA indicated that the difference in the chlorine content between the three types of water was not significant (p-value of 0.718 and 0.514 for fabric F1a and F2, respectively). The results show the robustness of the biocidal finish to variations in the chlorination water quality. To our knowledge, this is the first time that the effect of chlorination water quality on the chlorination efficiency of fabrics with an N-halamine finish has been assessed.
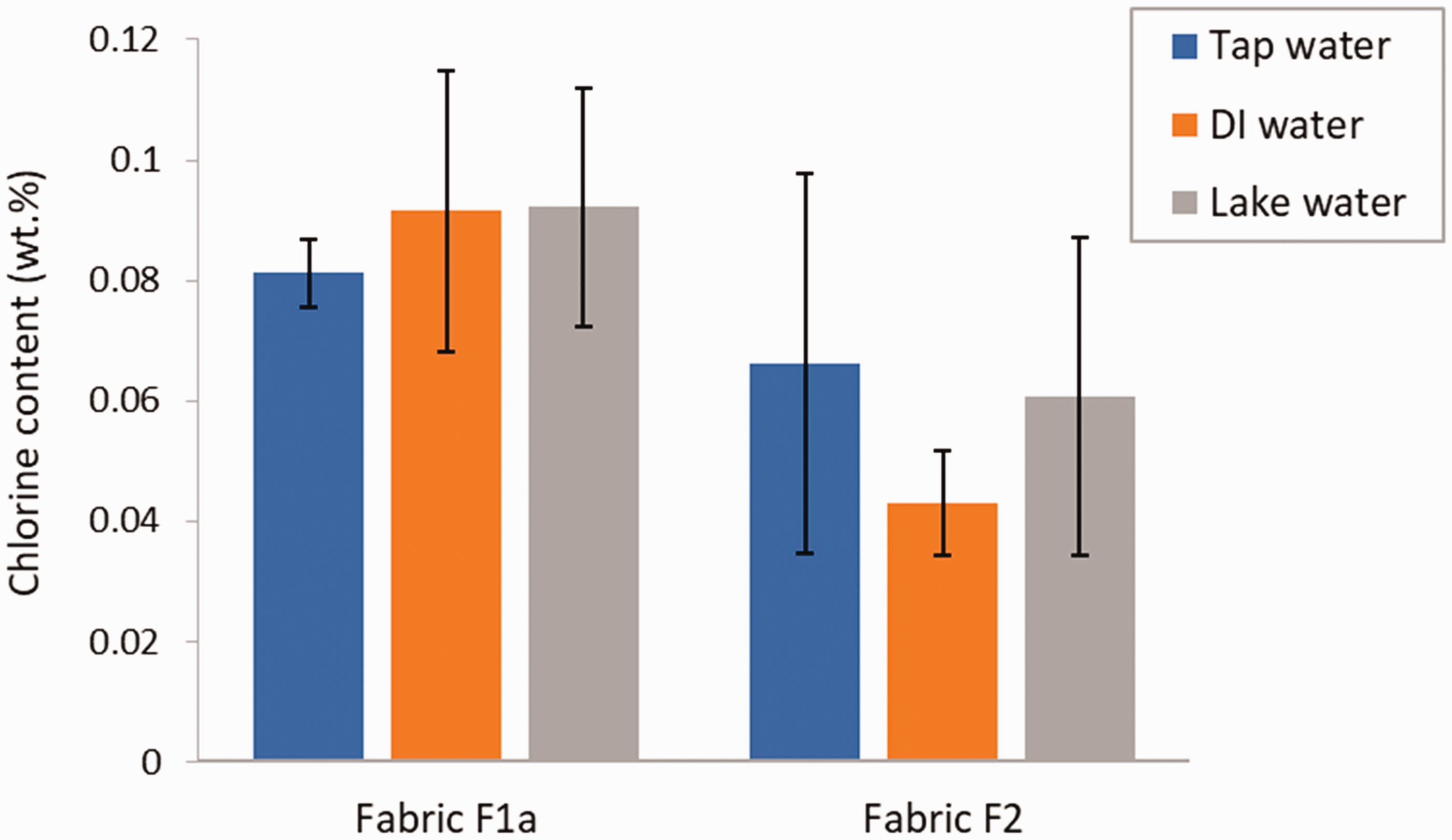
Chlorine content in coated fabric specimens chlorinated with DI water, tap water, and lake water. Error bars represent the standard deviation (N = 3).
To study the effect of the chlorination time, fabric specimens were chlorinated for 15, 30, and 60 min. Figure 6 shows that the chlorination time appears to influence the amount of chlorine loaded onto both fabric types, with the highest chlorine content being measured for the 60 min chlorination treatment. However, due to the large variability recorded in some instances, the differences measured were not statistically significant for fabric F1a (ANOVA, p = 0.217). They were close to significance in the case of fabric F2 (ANOVA, p = 0.051). This increase in chlorine content with an increase in chlorination time is in agreement with what has been reported in the literature for N-halamine-functionalized polymethacrylamide nanoparticles. 29
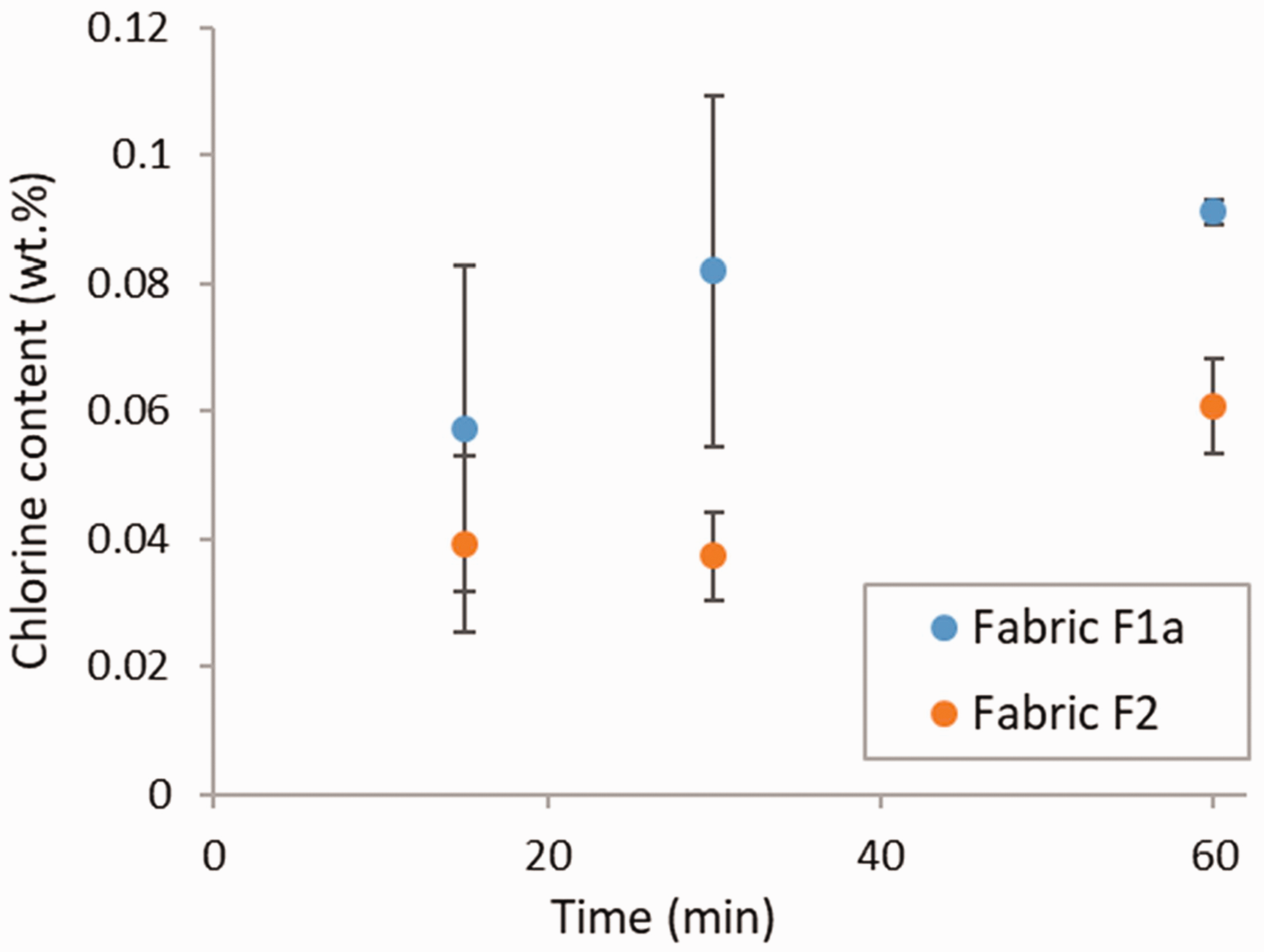
Chlorine content in coated fabric specimens as a function of the chlorination time. Error bars represent the standard deviation (N = 3).
The effect of the fabric to chlorination solution volume ratio on the treated fabric chlorine content was studied by using two sizes of specimens, 40 × 100 mm and 80 × 200 mm, while keeping the volume of the chlorination solution in the tray constant. The objective of this test was to determine the maximum fabric load that could be effectively recharged by a given amount of chlorination solution. The results in Figure 7 show a decrease in the specimen chlorine content when the fabric to chlorination solution volume ratio was increased by a factor of 4. However, because of the high variability in the results for some of the tests, the differences measured were not statistically significant (p-values of 0.103 and 0.0835 for fabric F1a and F2, respectively).
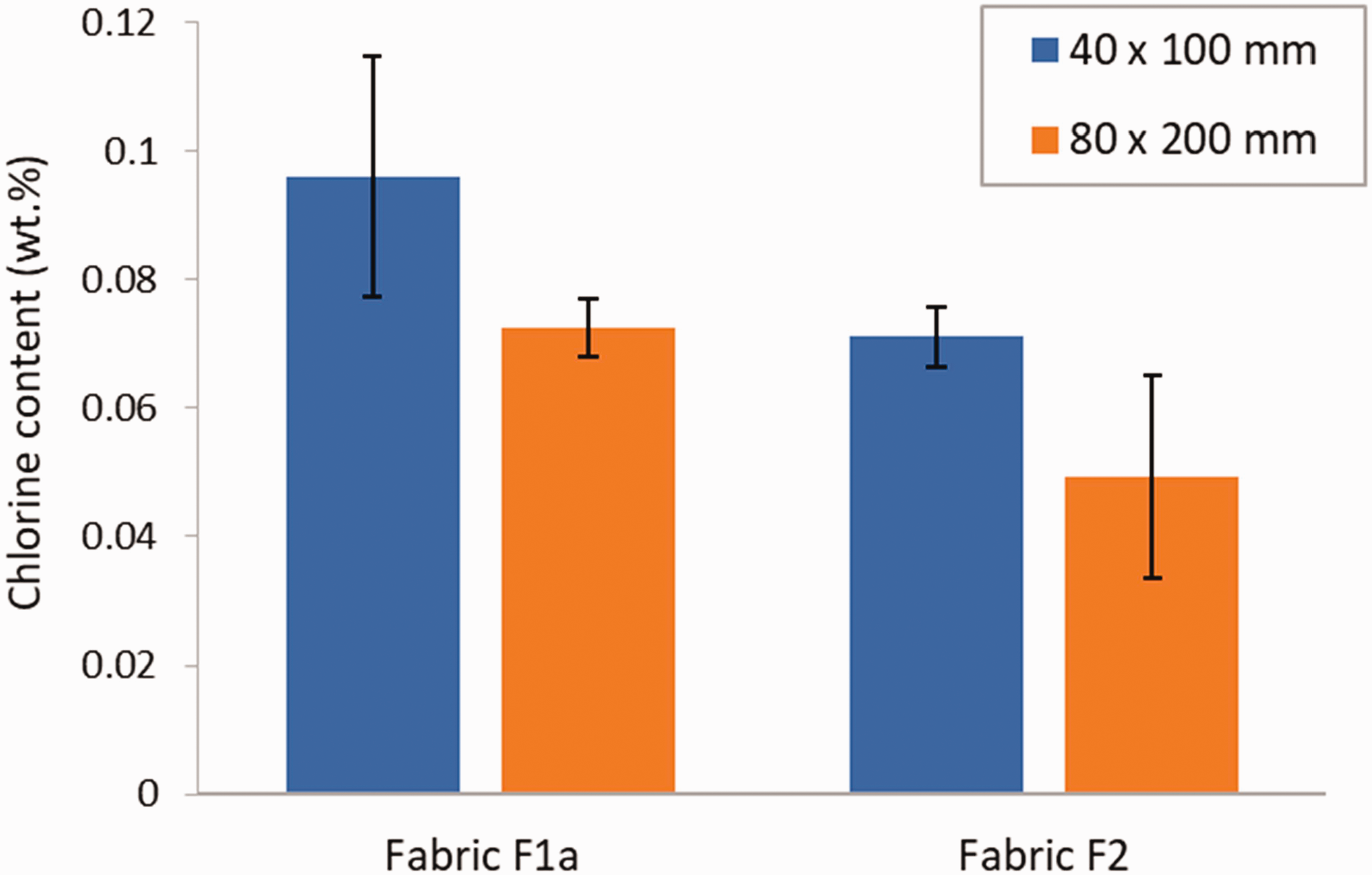
Chlorine content in coated fabric specimens chlorinated using 40 × 100 mm and 80 × 200 mm specimen sizes. Error bars represent the standard deviation (N = 3).
Effect of the water-repellent finish on the chlorine content
The effect of the presence of a water-repellent finish on the fabric on its ability to be successfully coated with PSPH and further chlorinated was studied with variations of fabric F1 and F5 without a water repellent (F1a and F5a) and with a water repellent (F1b and F5b). No information was available on the nature of the water repellents used for the two fabrics. As a note, the version of fabric F1 commercially available and used for medical gowns has a water-repellent finish, whereas fabric F5 is used without a water-repellent finish to manufacture military uniforms.
Figure 8 shows the results in terms of chlorine content for the fabrics with and without a water repellent. In the case of fabric F1, the presence of a water-repellent finish on its surface did not affect the chlorine content (p = 0.4802). This can be attributed to the fact that fabric F1 is made of polyester, which is a hydrophobic fiber. In both instances, the chlorination solution had difficulties wetting the fabric specimens. On the other hand, the presence of a water repellent at the surface of fabric F5, which was made of nylon/cotton combination yarns, strongly affected the chlorine content in the treated fabric (p = 0.000004462). Indeed, cotton fibers are highly hydrophilic; they were easily wetted by the aqueous chlorine solution when the fabric had no water-repellent finish. The presence of the water repellent on the surface of fabric F5b strongly reduced the ability of the fabric specimens to be wetted by the chlorination solution and thus the amount of chlorine uptake. This also explains why the chlorine content in the version of fabric F5 without a water-repellent finish was higher than the values observed for fabric F1, both in its versions with and without water repellent.
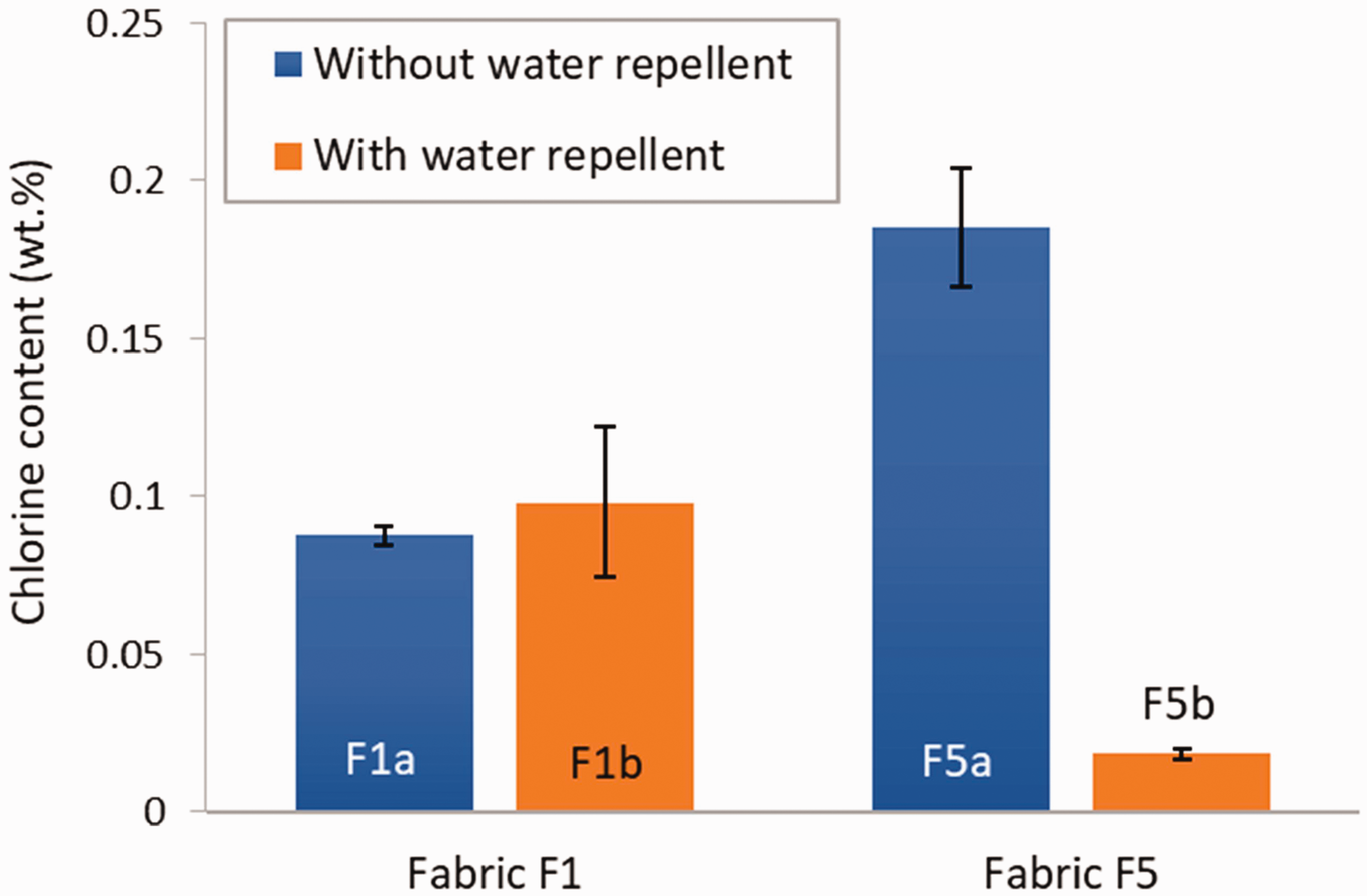
Chlorine content in fabric F1 and F5 with and without a water repellent. Error bars represent the standard deviation (N = 3).
Effect of the PSPH finish and chlorination on the fabric performance
The effect of the PSPH finish application and chlorination treatment on the fabric performance was assessed in terms of tensile strength, color, and flame resistance with fabric F1b and F5a. These fabrics were selected for this part of the study as they are the versions of fabric F1 and F5 that are commercially available and effectively used for medical gowns and military uniforms, respectively.
Figure 9 shows the breaking strength data for fabric F1b and F5a untreated (labeled as control) and after the PSPH finish application and chlorination treatment. Fabric F1b showed only 0.9% loss of strength after the PSPH finish application and chlorination treatment. The difference with the mean breaking strength of the control specimens was not statistically significant (p = 0.26). Fabric F5a experienced a 4.4% decrease in breaking strength after the PSPH finish application and chlorination treatment. In this case, the difference in breaking strength was statistically significant (p = 0.005). By comparison, Li et al. reported a 30% decrease in tensile strength for a cotton fabric treated with a hydantoin diol finish with and without addition of TiO2 nanoparticles. 30 The authors attributed this decrease in strength of their samples to the high curing temperatures (170–180°C) and acidic conditions (pH 4–5) used, which would hydrolyze bonds in the cellulose molecule. A 14% decrease in tensile strength of a cotton fabric treated with PSPH and further chlorinated was also reported in the warp direction by Ma et al., whereas no difference was measured in the weft direction. 8 They cured their specimens at 130°C for 3 min. Even though it was associated with a longer curing time, the lower curing temperature of 95°C used in our study, which is based on the protocol by Liu et al. 7 , can possibly explain the lower negative impact of the PSPH finish application and chlorination treatment on the fabric strength observed here.
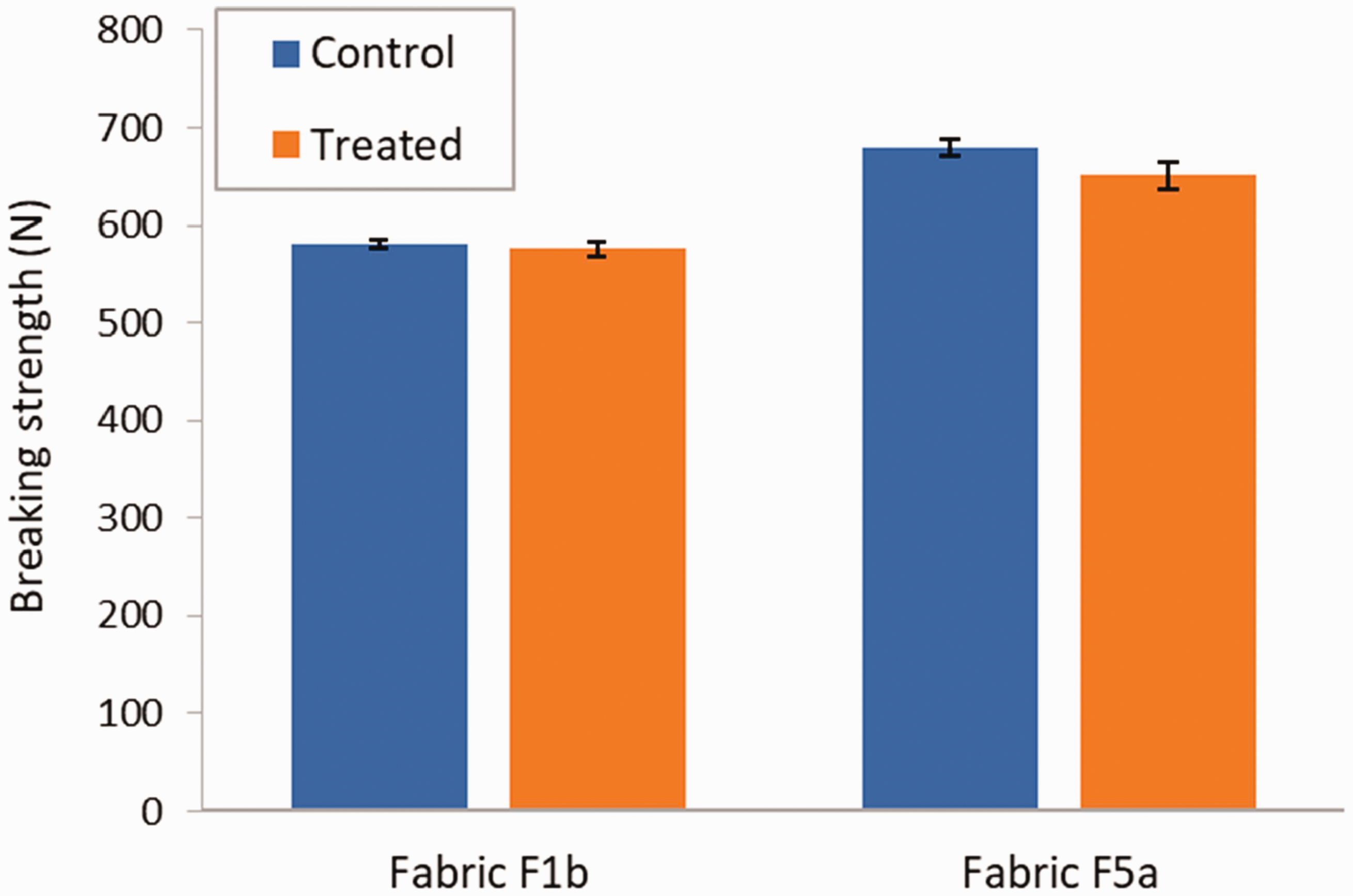
Mean breaking strength of fabric F1b and F5a in the warp direction before and after PSPH finish application and chlorination treatment. Error bars represent the standard deviation (N = 5).
Fabric color is often an important aspect for PPE and OCE: in the case of military clothing, it is subject to very specific requirements. 31 Figure 10 shows examples of pictures of the fabric specimens before and after the PSPH finish application and chlorination. The color of fabric F1b has become lighter. On the other hand, no obvious changes in the color of the different colored zones of fabric F5a are visible.

Pictures of specimens of fabric F1b and F5a before and after PSPH finish application and subsequent chlorination.
Table 3 gives the values of the color change of fabric F1b and F5a after the PSPH finish application and chlorination. For fabric F1b, the values of ΔL*, Δa*, and Δb* indicate a lighter and less yellow shade. In the case of fabric F5a, the positive value for Δa* points toward a more reddish shade. It may be noted that, although fabric F5a has a military camouflage pattern including dark and light green, black and brown, the large size of the target mask (44.5 mm diameter) allowed us to capture a representative area of the pattern.
Results of the measurement of the fabric color change after PSPH finish application and chlorination for fabric F1b and F5a

The value of ΔE after the finish application and chlorination was quite high for fabric F1b, which aligned with the large color change observed visually. On the other hand, the change in color produced in fabric F5a was more limited, with a value of ΔE below 3, which corresponds to the limit of acceptable color changes based on Canadian military standards. 31
The results of the flammability tests performed on specimens of fabric F1b and F5a that were untreated (labeled as control) and treated with PSPH finish and chlorination are listed in Table 4. The value reported is the average time for the specimens that ignited during the flammability test to burn to the stop cord. For the untreated fabric F1b, one out of the five specimens ignited and burned slowly, giving an average time of 15.3 s to burn to the stop cord. The remaining four specimens did not ignite during the test. After treatment, three out of the five specimens of fabric F1b ignited and burned slowly with an average time to burn to the stop cord of 13.5 s. The presence of the PSPH finish may have limited the ability of fabric F1b to shrink away from the flame, which is the typical behavior observed when polyester fabrics are exposed to a flame. 32 This may explain the larger number of specimens of fabric F1b that ignited for the treated condition. However, both average burn time values were greater than 3.5 s, i.e. within the requirement for Class 1 – normal flammability according to 16 CFR Part 1610. 21 For fabric F5a, none of the untreated and treated specimens ignited during the test. The fabric with and without the treatment was thus assigned to Class 1 – normal flammability.
Result of the flammability test for control and treated (with PSPH finish and chlorination) specimens of fabric F1b and F5a
Effect of the simulated use conditions on the chlorine content
This section describes the results obtained in terms of the resistance of the fabric finish to simulated use conditions: exposure to light, perspiration, repeated laundering, and abrasion. The effect was assessed through the measurement of the residual chlorine content in the finished and chlorinated fabric specimens after application of the simulated use conditions. The tests were conducted on fabrics F1b and F5a. These fabrics were selected for this part of the study as they are the versions of fabric F1 and F5 that are commercially available and effectively used for medical gowns and military uniforms, respectively.
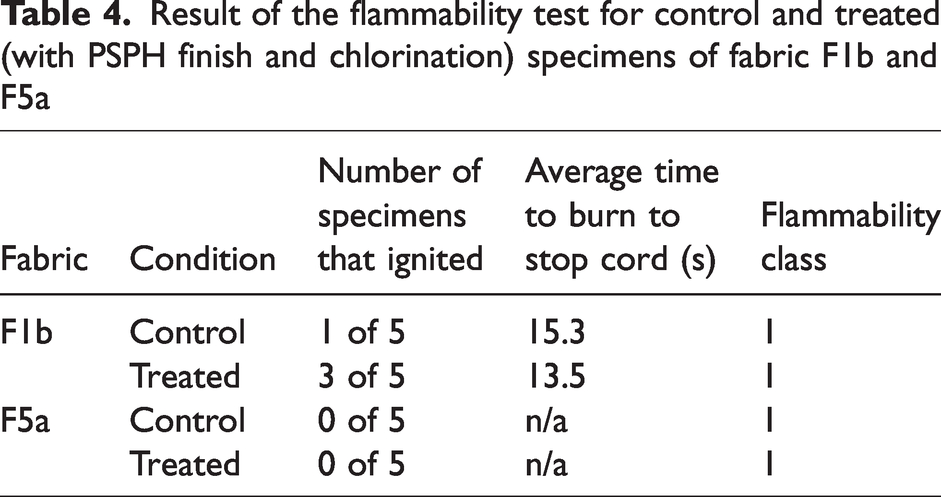
The results of the measurement of the effect of UV radiation from simulated sunlight exposure using a xenon arc weatherometer on the chlorine content are shown in Figure 11. The control specimens were not exposed to the UV radiation. Exposure to UV radiation significantly reduced the chlorine content in both fabrics: p = 0.001278 for fabric F1b and p = 3.128 ×10−7 for fabric F5a. The reduction in chlorine content was 46% in the case of fabric F1b and 71% for fabric F5a. These results are in line with what has been reported in the literature regarding the sensitivity of N-halamine compounds, including PSPH, to UV radiation. For instance, reductions in chlorine content from 0.26 to 0.01% and from 0.32 to 0.06% were reported respectively for PSPH 7 and PSPH-polyhexadecyltrimethoxysilane 8 coated on a cotton fabric after exposure for 12 h under fluorescent UVA at an irradiance of 0.89 W/m2 (measured at 340 nm) and a temperature of 60°C. The difference in the effect of UV radiation between fabric F1b and F5a may possibly be attributed to the effect of the fabric color (yellow for fabric F1b and a camouflage pattern with dark and light green, black and brown for fabric F5a) on their UV absorption 33 . Although fabric F5a started with a higher chlorine content compared with fabric F1b, their residual content was similar after the 12 h of exposure to UV radiation, with a residual chlorine content in the range of 0.05 wt.%.
Chlorine content in treated fabric F1b and F5a for the control specimens and after exposure to UV radiation using a xenon arc weatherometer. Error bars represent the standard deviation (N = 5).
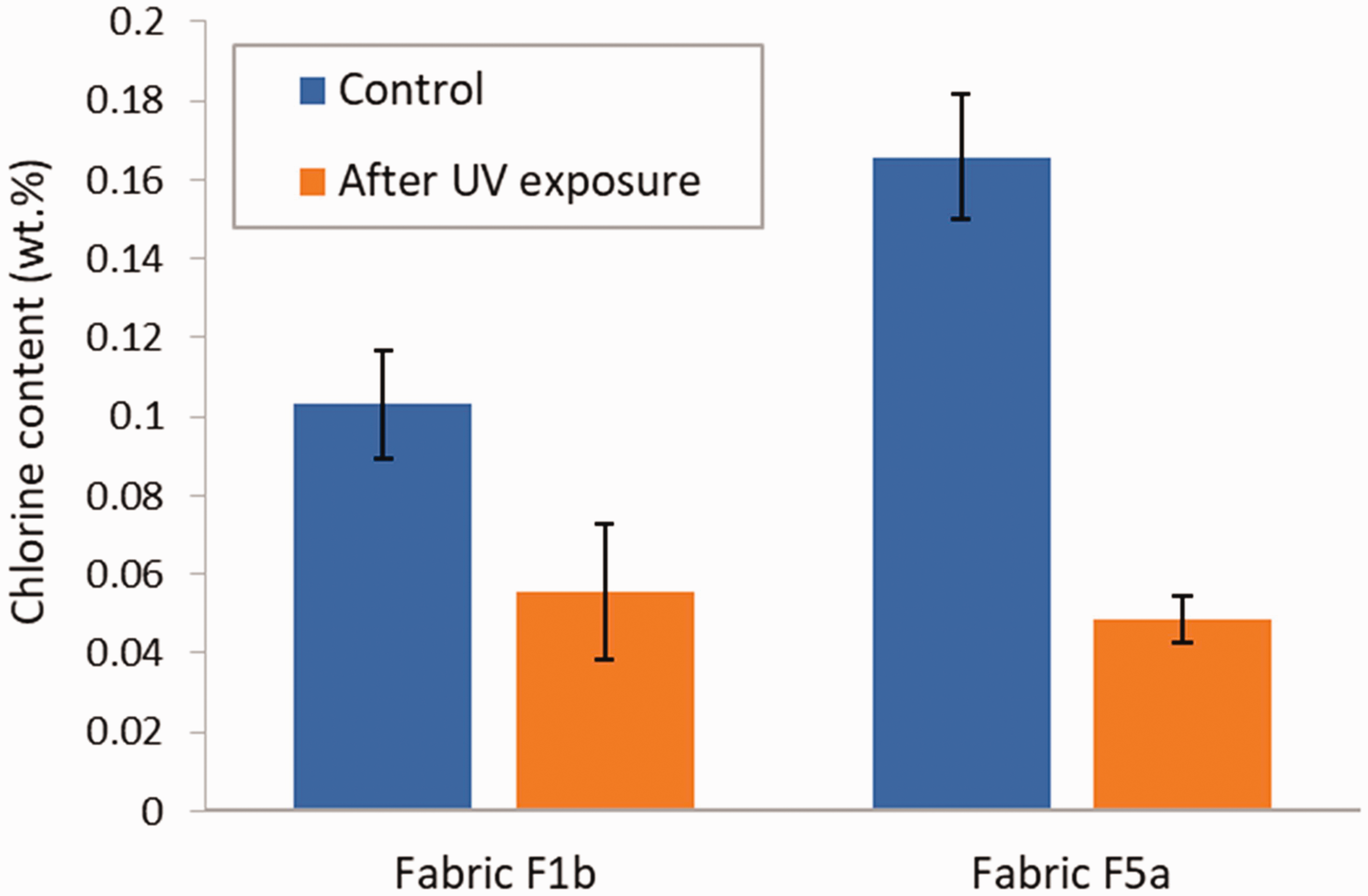
Specimens of treated fabric F1b and F5a were also exposed to direct sunlight in natural conditions. As shown in Figure 12, the total percent chlorine largely decreased after sun exposure when compared with the unexposed control condition, by 61% for fabric F1b and by 49% for fabric F5b. In both instances, the differences in chlorine content in the fabric as a result of exposure to natural sunlight were significant, with p-values of 0.0002513 and 0.0001678 for fabric F1b and F5a, respectively. The reduction in the finish chlorine content observed with exposure to natural sunlight was higher in the case of fabric F1b and lower in the case of fabric F5b by comparison with the xenon arc weatherometer (Figure 11). This difference in the effect produced by natural sunlight and the xenon arc weatherometer may possibly be attributed to the interplay between the effect of the fabric color on their UV absorption 33 and the differences in UV spectrum between the xenon arc lamp with a borosilicate-soda lime filter, which is aimed at stimulating sunlight through a window, and direct natural sunlight. 34
Chlorine content in treated fabric F1b and F5a for control specimens and after exposure to direct sunlight. Error bars represent the standard deviation (N = 3).
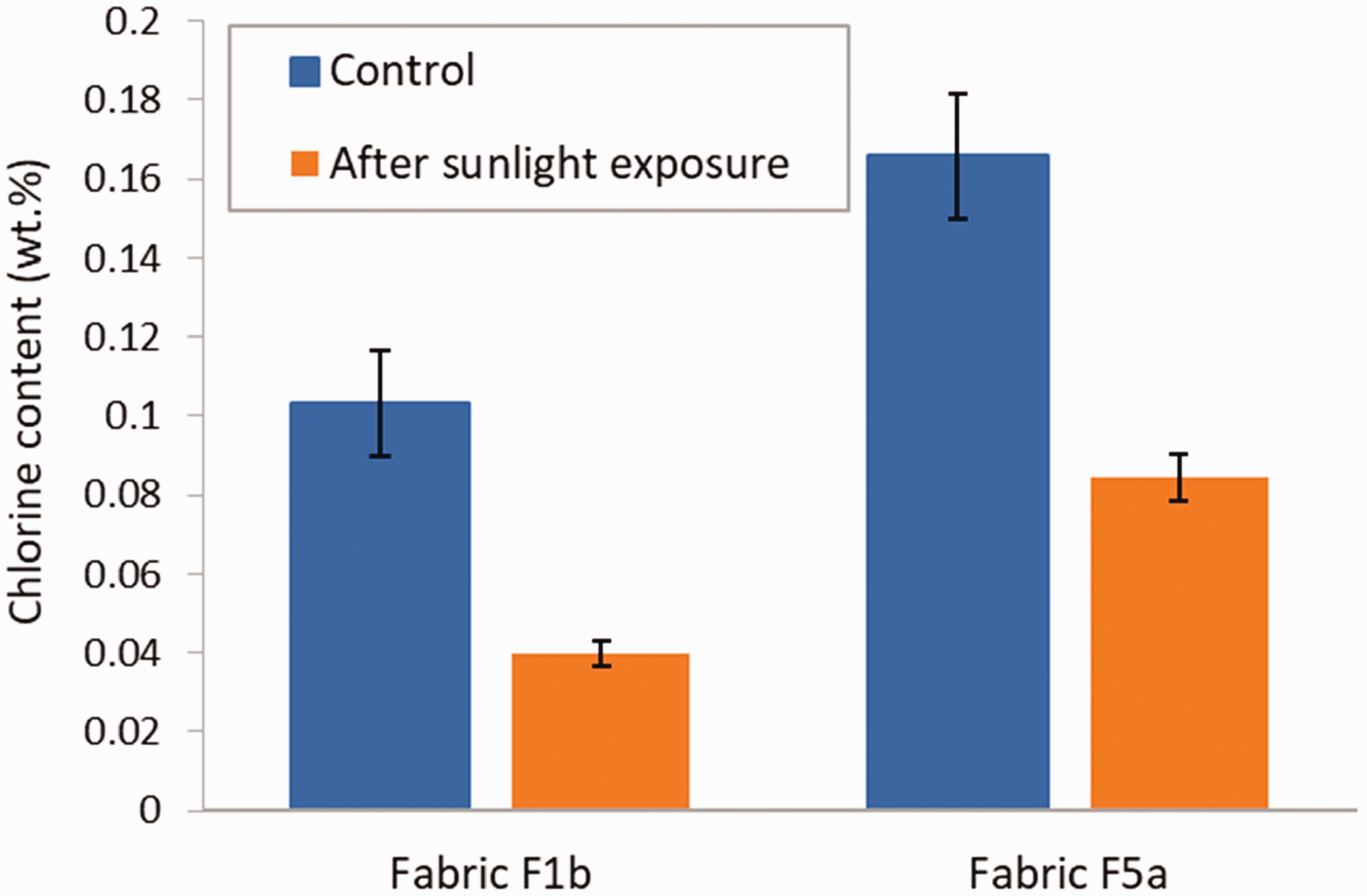
The results of the effect of exposure to acidic perspiration on the residual chlorine content of the treated fabrics are shown in Figure 13. Control specimens were not exposed to the perspiration solution. In the case of fabric F1b, an increase of 36% of the chlorine content was observed after exposure to perspiration, whereas the chlorine content in fabric F5a decreased by 54%. In both instances, the difference with the control was significant (p-values of 0.01679 and 0.0001684 for F1b and F5a, respectively). Results from the literature show that N-halamine compounds are more stable in low pH solutions compared with high pH. 5 However, they may still be affected by acids. The difference in behavior between fabric F1b and F5a is possibly due to the presence of the water-repellent treatment on fabric F1b, which limited the wetting of the fabric by the perspiration solution and its penetration into the fabric. The reason for the increase in the chlorine content in fabric F1b after exposure to the acidic perspiration might be related to the presence of sodium chloride in the synthetic perspiration solution.
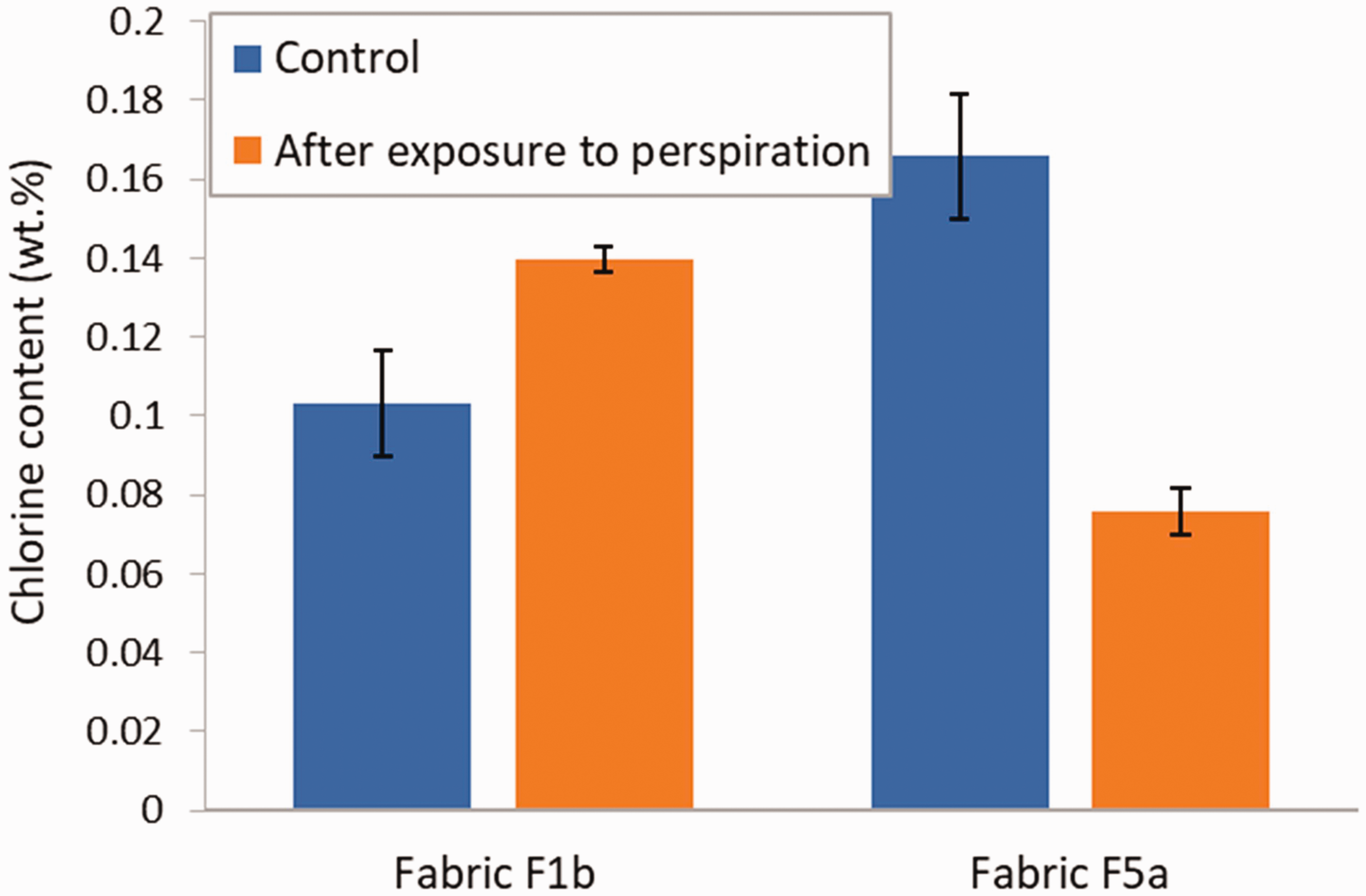
Chlorine content in fabric F1b and F5a for control specimens and after exposure to perspiration. Error bars represent the standard deviation (N = 3).
Figure 14 shows the effect of accelerated laundering on the chlorine content of the treated samples of fabric F1b and F5a. Control specimens were not subjected to the laundering treatment. In both instances, a significant decrease was measured after the 45 min accelerated laundering treatment, which has been determined to represent the equivalent of five domestic washings when considering colorfastness. 24 The decrease was 47% for fabric F1b (p = 0.0001333) and 63% for fabric F5a (p = 0.000003553). By comparison, Worley et al. reported a reduction of 37.5% in the chlorine content of a cotton fabric coated with PSPH after five cycles of accelerated laundering using the same washing conditions as those used for fabric F5a. 16
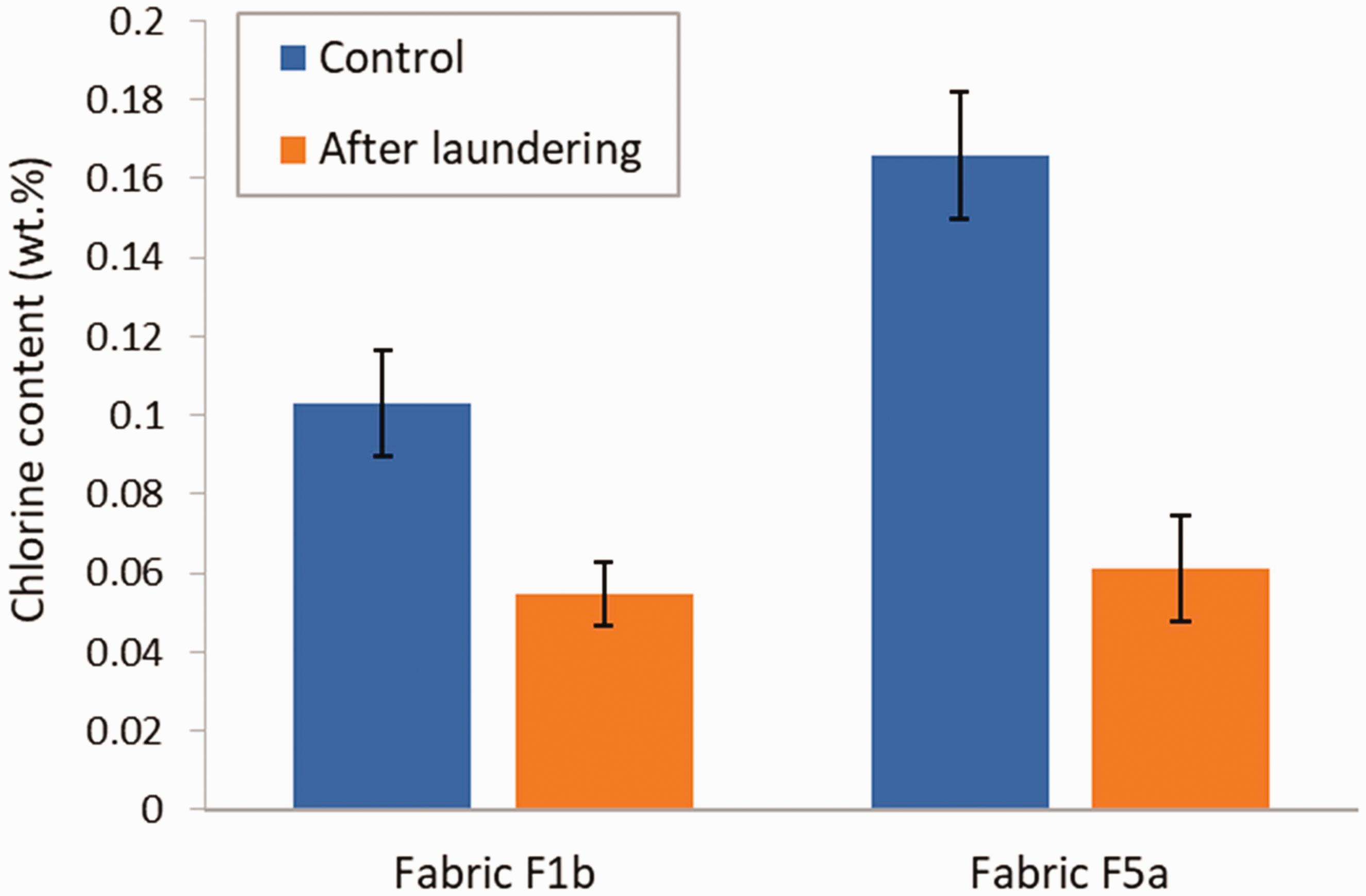
Chlorine content in fabric F1b and F5a for control specimens (not washed) and after accelerated laundering. Error bars represent the standard deviation (N = 5).
The last simulated use condition tested was abrasion, which can occur when PPE/OCE is worn or during laundering, for instance. The effect of 1000 abrasion cycles on the chlorine content of treated fabric F1b and F5a is shown in Figure 15. Control specimens were not subjected to the abrasion cycles. No significant difference in the chlorine content was observed for fabric F1b (p = 0.4849). On the other hand, a 68% decrease in the chlorine content was measured for fabric F5a, with a p-value of 0.000005201. These results can be explained when considering the mass of the specimens after abrasion. In the case of fabric F1b, there was no measurable change in the mass of the specimens after the 1000 abrasion cycles, whereas the specimens of fabric F5a lost between 1 and 1.5% in weight after abrasion.
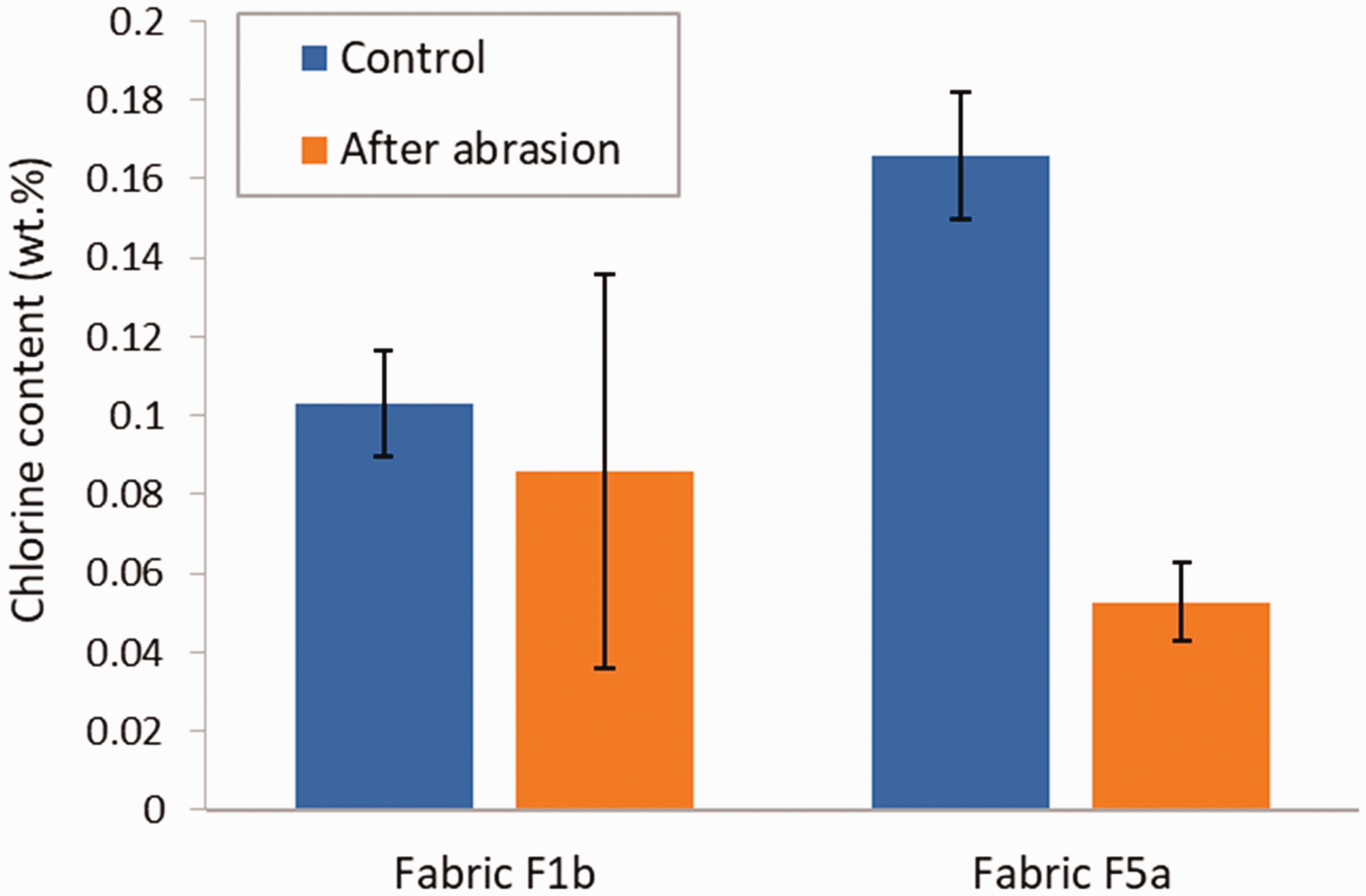
Chlorine content in treated fabric F1b and F5a for control specimens and after abrasion. Error bars represent the standard deviation (N = 4).
Conclusion
This study investigated for the first time the impact of an N-halamine-based finish on the functional and aesthetic properties of fabrics of various fiber contents currently used for medical gowns and military uniforms. It also assessed the effect of the presence of a water-repellent finish on the fabric on the chlorine content as well as the sensitivity of the self-decontaminating function to variations in the chlorination application conditions that would be associated with the severe environments encountered, for instance, during military missions. Finally, we examined the effect of simulated use conditions on the fabric chlorine content.
The antibacterial activity of the finish was confirmed with both gram-positive and gram-negative bacteria. The presence of a water-repellent finish on the fabrics reduced the chlorine loading for the fabric made of hydrophilic fibers, whereas no effect was measured for the polyester fabric. The presence of the water-repellent finish on the medical gown fabric may also have led to the antibacterial activity observed in the untreated specimens for the gram-positive bacteria. In terms of the effect of chlorination conditions, no significant effect of the temperature (between 4 and 21°C) and water quality was measured. On the other hand, the chlorine content in the fabric increased with the chlorination time and decreased with the fabric/chlorine solution volume ratio. The finish application of the fabric and further chlorination had low to no effect on the breaking strength, depending on the fabric. The effect of the treatment on the fabric color was strong for the yellow-colored medical gown fabric. It was more limited for the military fabric. The treated fabrics remained within the requirements for Class 1 in terms of flammability. The study of the effect of simulated use conditions on the chlorine content in the fabrics generally showed a reduction after exposure to UV/sunlight, perspiration, laundering, and abrasion. In general, the chlorine loading in the polyester medical gown fabric better resisted the simulated use conditions compared with the cotton/nylon military fabric.
These results provide some insights into the strengths and remaining challenges of N-halamine fabric finishes when considering practical applications for protective clothing, in particular for use in severe environments and austere conditions. Solutions will have to be found to limit the loss in color of the fabrics when subjected to the chlorine recharge treatment. It will also be important to assess the effect of the finish on other aspects of the fabrics’ performance, for instance the anti-static properties of the medical gown fabric and infrared reflectance of the military fabric. Once fully developed and optimized for application for PPE and OCE, N-halamine textile finishes can provide a solution with a lower environmental impact compared with other antibacterial strategies because of the absence of residual antibacterial activity when the textile item is disposed of.
Footnotes
Acknowledgments
The authors would like to thank Dr. Rachel McQueen, Ankita Shroff, Jennifer Beaudette, René Arredondo, Anita Amir Labonno, Dylan Long, and Benita Malaba for their contribution to the experimental work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by the Innovation for Defence Excellence and Security (IDEaS) Program of the Department of National Defence of Canada (# CovCA-0017).
