Abstract
Body odorants typically transfer to clothing fabrics by way of liquid sweat, yet investigations of odor retention in textiles often neglect this route of exposure in their test procedures. This paper describes a novel method for transferring selected odorous volatile organic compounds to six types of textile fibers in yarn bundle form by an aqueous sweat solution. Headspace volatile organic compounds varying by chemical class (ketones, aldehydes, carboxylic acids) were monitored at discrete time intervals (30 min, 3 h, 24 h) using proton transfer reaction mass spectrometry. Lower intensities of ketones and aldehydes were detected in the headspace above cellulosic fibers (cotton, mercerized cotton, viscose) than above wool, nylon, and polyester fibers at 30 min. A rapid decrease in ketones occurred for all fibers, but lower intensities of ketones were released after 3 h for cellulosic and wool fibers. Nylon fibers typically released the highest amounts of ketones and aldehydes at 30 min, but by 24 h higher intensities of these compounds were released from polyester. Carboxylic acids exhibited minimal differences in intensities between 30 min and 3 h, with few differences evident among fiber types. Understanding the preferential sorption of odorants when clothing is exposed to volatile organic compounds in aqueous solutions such as sweat is enhanced from the results of this investigation.
Keywords
Textile fibers in clothing and interior products act as a reservoir for the sorption of many organic volatile and semi-volatile organic compounds in the indoor environment.1,2 Furthermore, due to the close contact of clothing next to the body, sweat, sebum and bacteria can transfer to the textile fibers and yarns. The sorption and subsequent release of odorous volatile organic compounds (VOCs) present in human sweat within apparel fibers/yarns can cause an unpleasant experience for end users. 3 The problem can be exacerbated if odorants, or the source of odors, are not completely removed during laundering.4 –6 Understanding how odors are generated, retained and released from textiles is vital to addressing this important consumer issue. Attending to the problem of odor in textiles has broader implications for the longevity of clothing, 7 and facilitating textile reuse, which is essential for a circular economy. 8
Different types of textile fibers have different odor profiles and intensities following next-to-skin wear.9 –11 Protein fibers, such as wool, typically have lower odor intensity, followed by cellulosic fibers, with synthetics such as polyester and nylon often perceived as more odorous.10,12,13 Munk et al. 9 found polyester to have more odorants released following contamination with sweat, leading to a complex odor profile when compared with cotton. Differences in odor that appear among generic fiber types have been linked to the growth of selective microorganisms,14,15 and inherent differences in fiber chemistry and the resulting chemical interactions with odorous VOCs.4,16,17 Measurable differences occur even among fibers with the same molecular chemistry due to differences in the fiber fine structure and physical morphology, which provide access to more potential reactive sites in the internal fiber structure. This has been observed with cellulosic fibers, such as between cotton and viscose, 2 and mercerized and nonmercerized cotton fabrics. 18 Fabric and yarn structural parameters influencing mass, thickness and surface area can also impact odor sorption and release, although they play a less significant role than fiber type.10,19 Despite the increase in odor research over the past 20 years,6,14,20 there is still much to learn about the complex interactions of odorants and textiles.
Clothing fabrics absorb sweat released from the human body. 21 Sweat is mainly made up of water and sodium chloride (NaCl), with many other micronutrients and chemical constituents also present. 22 During periods of physical activity or increased ambient temperatures, the human body sweats as part of thermoregulatory processes to enable cooling through evaporation. 23 The density of sweat glands and sweat rates vary across the body,24,25 with some individuals being able to sweat over 2 L.h−1 during high-intensity exercise. 24 Considerable quantities of liquid can thus be transported to adjacent clothing. Liquid sweat can also be produced during less strenuous physical activity due to increased ambient temperatures or during periods of mental stress.26,27 Although sterile sweat is odorless, it can become odorous due to bacterial action. 28 Therefore, odorous VOCs can be transported through sweat and be sorbed by/to textile fibers during wear. Hence, examination of the transfer of odorants to textiles through liquid sweat is pertinent.
Many studies have examined the effect of single odorants applied directly to textiles in a solvent,4,9 transfer and development of odor through human wear trials,10,12,13 and adsorption of volatiles in gaseous form.16,17,29 Human wear trials are the most representative method for evaluating odor in apparel fabrics, but are often complicated by the requirement for lengthy trials, acquiring suitable participants, and variability of the odor source. 5 Although odorants will transfer to textiles in gaseous form, the major route of odorant transfer will be through bulk liquid. 30 Hence, during wear the majority of odorants, but also the precursors to odor (e.g. long-chain fatty acids) will transfer to textiles through liquid sweat. Therefore, examining the transfer of odorants to textile fibers/yarns/fabrics through aqueous solutions is a critical step toward addressing a gap in textile odor research.
After odorous VOCs are ab/adsorbed into textile substrates, odorants are released from fibers at different rates.16,17 The extent to which odorants release from fibers has implications for odor detection by the human nose, because odorous VOCs will be smelled by the wearer and potentially other people. The rate and duration at which odorous VOCs are released from different generic fibers may have implications for understanding how effectively clothing could be ‘aired’. Furthermore, the chemistry of the odorous volatiles, as well as their precursors, can vary depending on their physicochemical properties, their polarity and solubility in water. 4 Polarity affects how easily compounds can be removed by laundering, 4 and how well they are absorbed by textiles. 18
Proton transfer reaction mass spectrometry (PTR-MS) is a tool that can detect multiple compounds in the headspace of a sample in real time. Unlike traditional electron ionization, PTR-MS utilizes chemical ionization with the primary ion being the hydronium ion (H3O+). Chemical ionization is useful when analyzing a mixture of gases, such as VOCs emitted from the axillary region. 20 PTR-MS has been used successfully to assess the sorption and desorption of odorous VOCs from textiles varying in fiber content.16,17,29 It offers an instrumental means that better represents how odorous VOCs can be detected by the human nose, as the VOCs are present in the headspace above the textiles, rather than requiring complex extraction and clean-up procedures. 5
Therefore, the aim of this study was to develop a method for transferring selected odorous VOCs to textile fibers in yarn form by an aqueous solution (simulated sweat). The release of odorous VOCs which varied in polarity and water solubility were measured at selected time periods to monitor the changes in release from fibers/yarns with time using PTR-MS.
Materials and methods
Preparation of experimental yarns/fibers
Experimental fabrics were purchased from Testfabrics Inc. (West Pittston, PA). All fabrics were woven from spun yarns with 100% fiber content (wool [ISO 105-F01], 31 viscose [ISO 105-F02], 32 nylon 6.6 [ISO 105-F03], 33 polyester [ISO 105-F04], 34 cotton [style 400] and mercerized cotton [style 400M]). Before experimental work, fabrics were laundered six times continuously according to ISO 6330-2012. 35 The nonphosphate reference detergent A was included in the first two washes only. Fabrics were line dried, cut into 100 mm × 100 mm swatches, yarns separated from the fabric structure and loosely grouped together in bundles. The study focused on fiber types and their behavior in relation to VOC absorption and release, and although yarn parameters such as size and twist level may also influence sorption behavior, the yarn bundles enabled easy handling than further disaggregating to fiber components. This approach aligns with that of previous work.17,29 The yarn bundles were cleaned in dichloromethane prior to beginning experiments to remove surface impurities and wool oils. Yarn bundles were conditioned at 20 ± 2°C, 65 ± 4% relative humidity 36 for a minimum of 24 h, then weighed to 1.00 g of their dry weight based on moisture regain values.
Preparation of odorous sweat solution
Nine odorous VOCs were selected to represent a homologous series of compounds within three chemical classes known to be present in body and laundry malodors: three carboxylic acids, two aldehydes and four ketones. 37 Butanoic acid, 2-hexenal, 2-nonenal and 2-heptanone were sourced from McCormack Chemicals (Ireland); isovaleric acid, 2-butanone and 2-hexanone were sourced from Sigma (Germany); and hexanoic acid and diacetyl (2,3-butanedione) were sourced from Merck (USA). Selected properties of each VOC are listed in Table 1. Four stock solutions were prepared to 50,000 ppm of each VOC in propylene glycol (PG) (McCormack Chemicals). A stock solution for each chemical class was prepared, with diacetyl prepared separately from the other four ketones. The stocks were stored in a refrigerator at 4°C and sonicated for 5 min in a warm water bath (35°C) prior to preparation of the odorant-sweat solution.
Properties of volatile organic compounds and target m/z
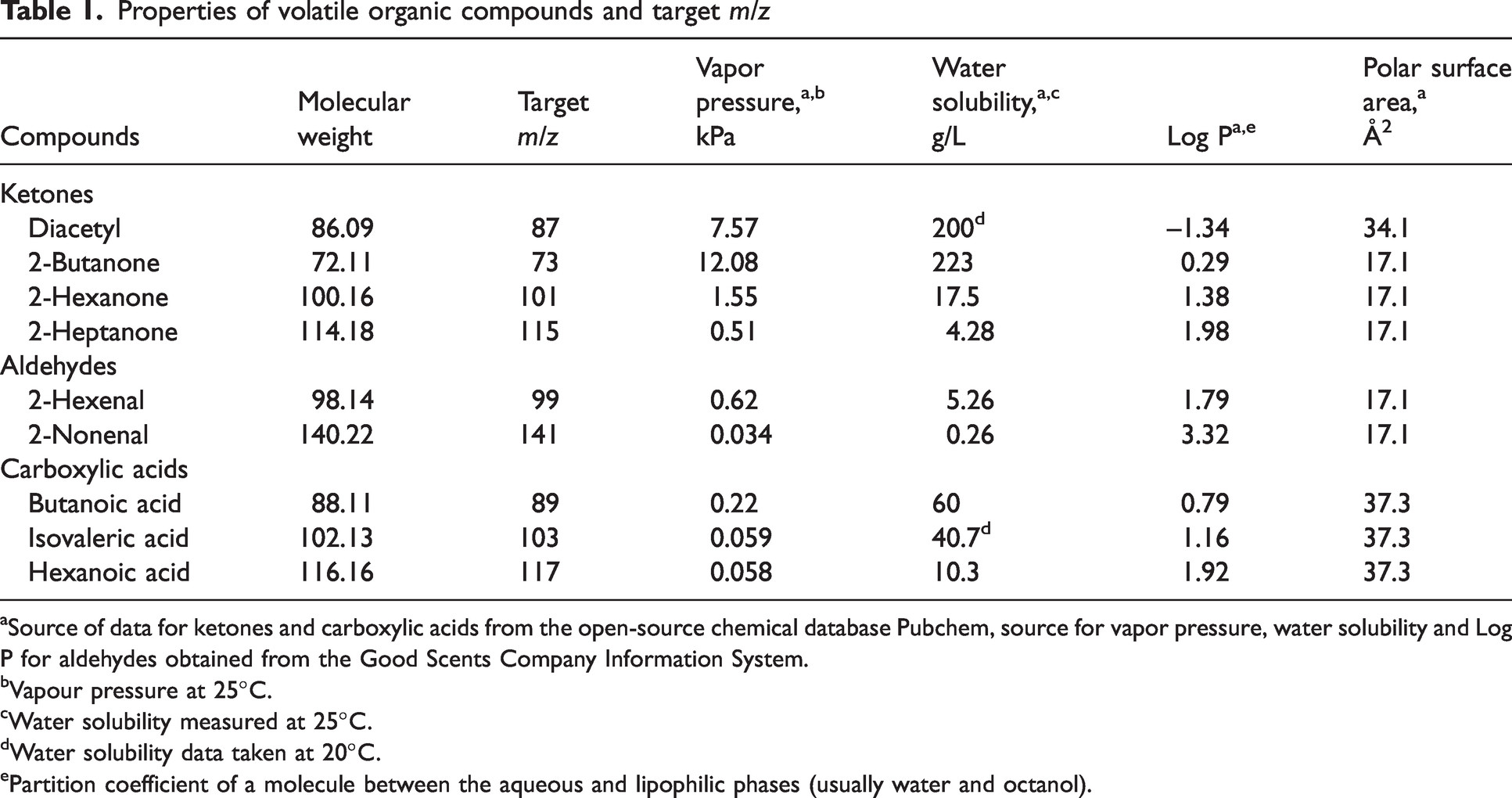
Source of data for ketones and carboxylic acids from the open-source chemical database Pubchem, source for vapor pressure, water solubility and Log P for aldehydes obtained from the Good Scents Company Information System.
Vapour pressure at 25°C.
Water solubility measured at 25°C.
Water solubility data taken at 20°C.
Partition coefficient of a molecule between the aqueous and lipophilic phases (usually water and octanol).
A sweat solution was prepared according to the AATCC test method 15 38 to a pH of 4.3 ± 0.2. [1.0% (w/v) of sodium chloride (NaCl) (BDH Chemicals); 0.025% L-histidine monohydrochloride monohydrate (C6H9O2N3•HCl•H2O) (BDH Chemicals); 0.25% disodium hydrogen orthophosphate (Na2HPO4•12H2O) (Merck), 0.1% lactic acid (85%) (Sigma)]. The volume was made up with MilliQ water.
From each stock solution, 200 µL was taken and added to a 250 mL volumetric flask. The sweat solution was then added to make up the volume, creating a 40 ppm odorant/sweat solution. As a control, a solution was prepared by adding 800 µL of PG to a volume of 250 mL with sweat solution. The test and control solutions were shaken by hand until thoroughly mixed.
Experimental procedure
A procedure for applying odorous VOCs to yarn bundles was adapted from the method used by Obendorf et al. 39 for applying aroma chemicals on cotton fabrics in aqueous solutions. In 100 mL glass bottles, 40 mL of odorant/sweat solution was added. Yarn bundles were placed into the sweat solution and the cap was secured. The bottles were shaken by hand for 2 min and allowed to stand for a further 30 min. This length of time reflects a contact time of intense sweating during vigorous exercise. Following the standing period, the bottles were shaken for 30 s, and the liquid and yarn bundles were poured into a Buchner funnel lined with filter paper (11 µm pore size). The bulk of the liquid was rapidly removed due to the suction of the Buchner system. Yarn bundles remained in the funnel for 2 min during which time they were loosely separated using tweezers. The yarns were moved to a glass dish, weighed to determine the liquid retained, and tied with a polyester thread. The tied yarn bundle was hung in the center of a 500 mL glass Schott Duran bottle (Figure 1). The polyester thread was taped to the outside of the bottle to hold the yarn bundle in place. The bottle was sealed with a modified PTFE cap that had two access points for PTFE tubing (1.59 mm internal diameter) to connect with the PTR-MS set-up. The tubes were capped at the end to avoid loss of volatiles. The yarns were left to stand for 30 min to allow the headspace to build up in the bottle. Following PTR-MS analysis, the yarn bundles remained in the glass bottle and were placed in a fume hood. They were allowed to stand for two additional periods of time without a cap to allow the yarns to dry and VOCs to be released into the fume hood to simulate ‘airing’. PTR-MS analysis was conducted again at 3 h and 24 h following the removal of yarn bundles from the sweat solution. Before each 3 h and 24 h test period, the bottles were sealed with the PTFE modified cap 30 min prior to the end of the period to allow headspace to equilibrate prior to PTR-MS analysis.
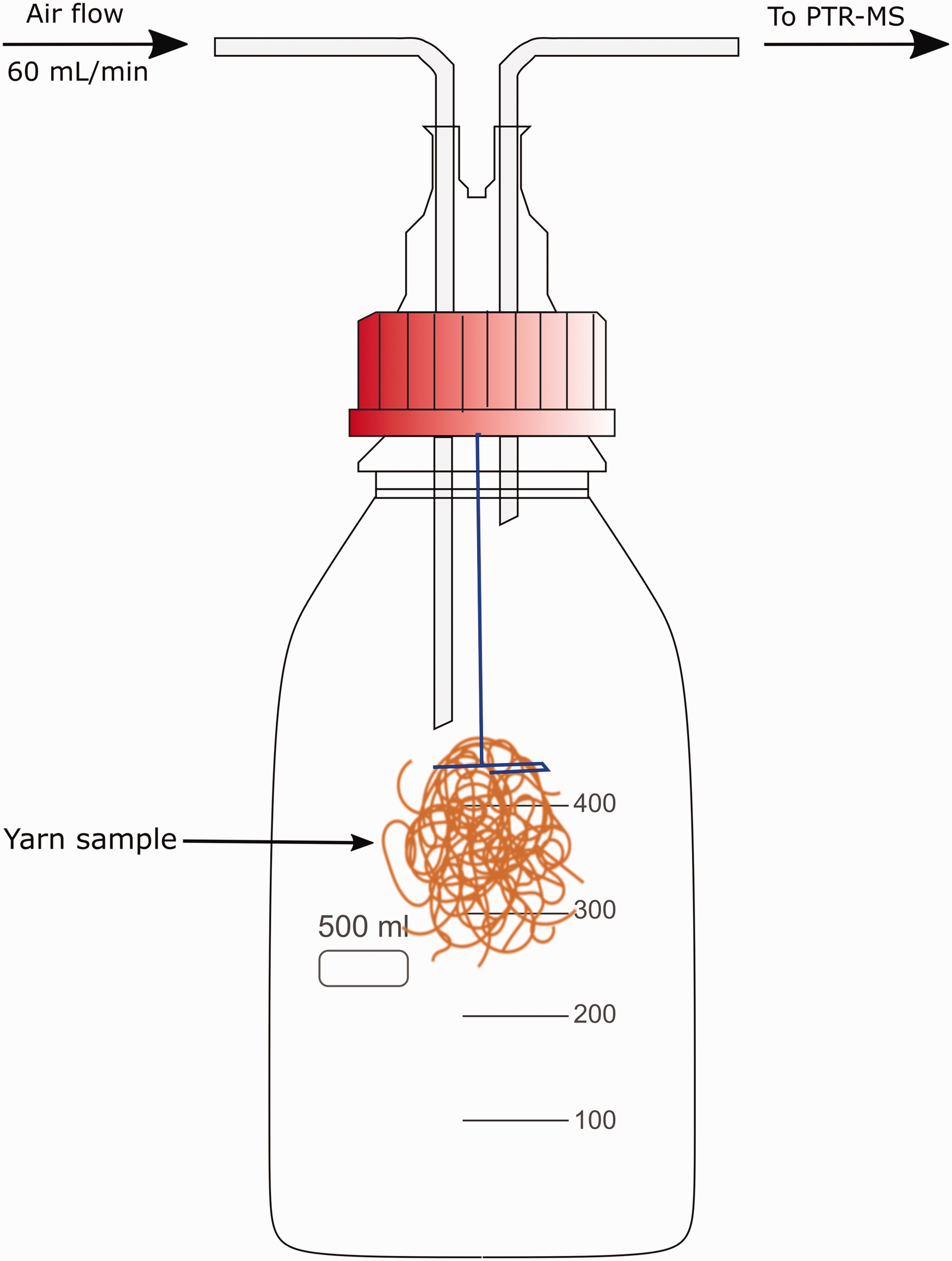
Experimental set-up of yarn sample.
PTR-MS measurements
The PTR-MS (Ionicon Analytik GmbH, Innsbruck, Austria) operating conditions were as follows: inlet temperature at 90°C, drift tube pressure at 2.23 mbar, chamber temperature at 70°C, drift tube voltage at 600 (±0.4 V), field density 136–138 Td, and a flow of 60 mL/min through a heated capillary (90°C). Mass spectrometric data were collected in scan mode covering the range of m/z 20 to 180 amu with a dwell time of 200 ms per mass. Seven cycles of measurements were taken, with the first two cycles excluded from analysis, and the mean of the last five cycles was calculated. Before each control and test sample measurement, cycles of background air were monitored to confirm background masses were low. Instrument performance was monitored through the masses of m/z 21.0 (H3O+), m/z 30.0 (NO), m/z 32.0 (O2) and m/z 37.0 (water cluster).
Statistical analysis
The measured signal intensities were normalized by correcting for variations in the primary ion (H3O+) and water cluster (H2O.H3O+), using the isotopologue at m/z 21 and m/z 37, respectively.
Statistical analysis was carried out on each compound (m/z) separately. Repeated measures analysis of variance (ANOVA) was conducted with time as the repeated measure on log10 transformed normalized counts per second (ncps) data (sphericity assumed). Fiber type was a between-subjects factor. In addition, for each compound and time, a series of one-way ANOVAs was conducted to determine whether the fiber type differed within each time period. When significant differences were found, Tukey’s honestly significant difference (HSD) tests and Games–Howell tests were carried out to establish significant differences between means at the P < 0.05 level. All statistical analyses were completed using IBM SPSS, version 26.0 (IBM, USA).
Results
Estimate of retained water in yarn bundles by fiber type
The mean weight of yarn bundles for each fiber type after sweat solution treatment before being placed in the bottle for PTR-MS measurements are presented in Table 2. The amount of retained liquid approximately relates to the hygroscopic nature of the fiber type, with wool and viscose retaining more liquid and therefore being heavier than nylon and polyester.
Mean weight (g) of yarn bundles following immersion in aqueous solution
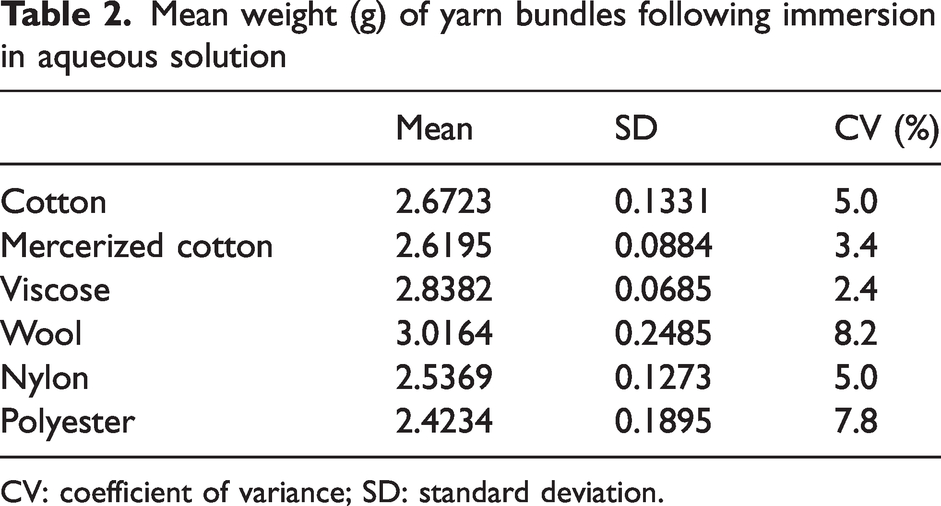
CV: coefficient of variance; SD: standard deviation.
Figure 2 shows the ratio of m/z 37/21 according to the time period for each of the fiber types. This figure represents humidity changes and is a measure of the free water. As absolute humidity decreases the ion intensity for the ratio m/z 37/21 will decrease. 40 Within the first 3 h, little difference in the absolute humidity for all the fiber types was evident between 30 min and 3 h, as the yarn bundles were still releasing moisture as they dried. By 24 h, the ion intensity was considerably lower, indicating that the yarn bundles had dried.
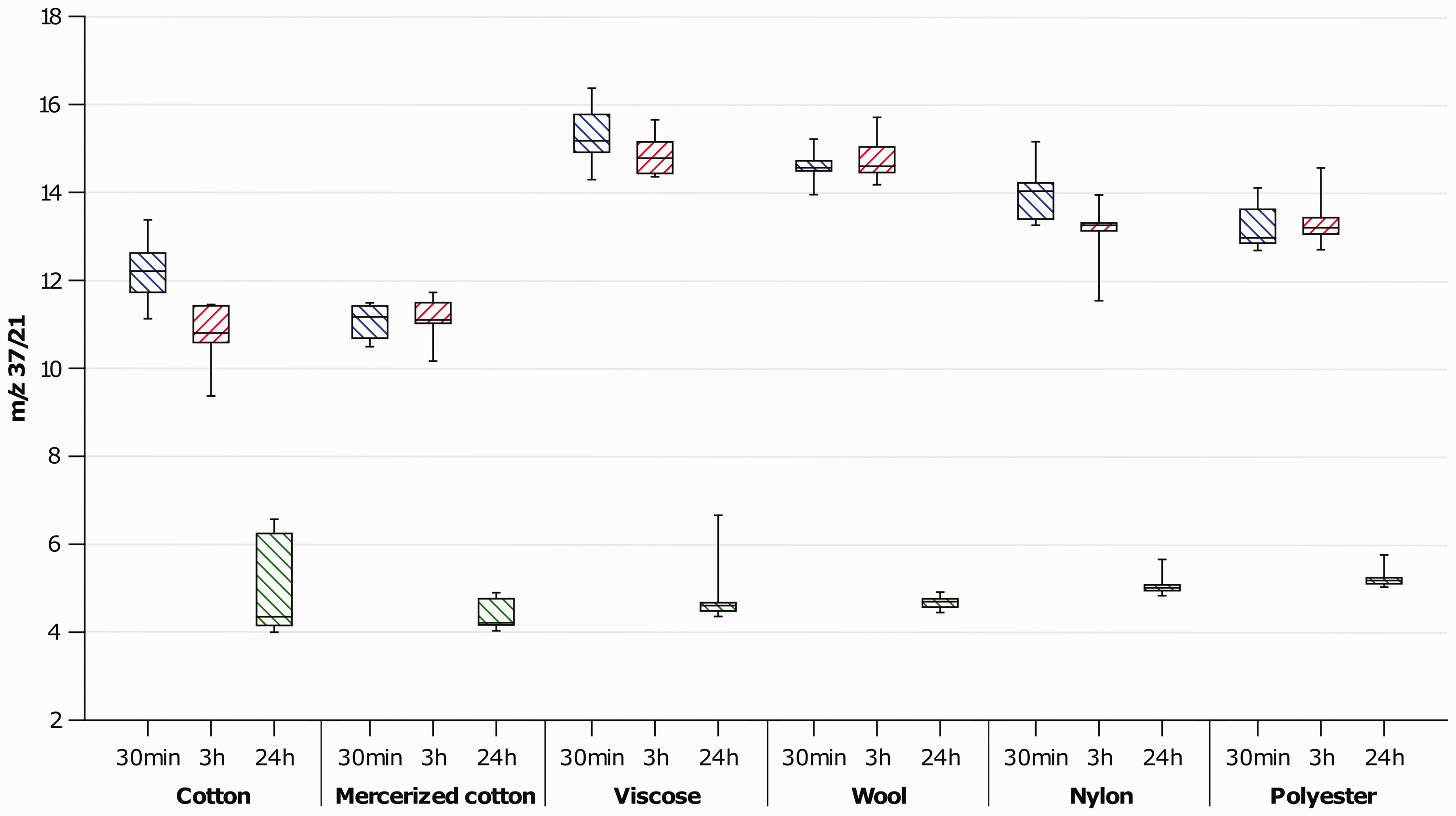
Ion intensities for m/z 37/21 representing the change in humidity as yarn bundles dry.
Changes of intensity of odorous VOCs with time
Results for each odorous VOC are presented by their respective mass to charge (m/z) ratio by fiber type at each time period (30 min, 3 h, 24 h) (presented in Figure 3 and Table 3).
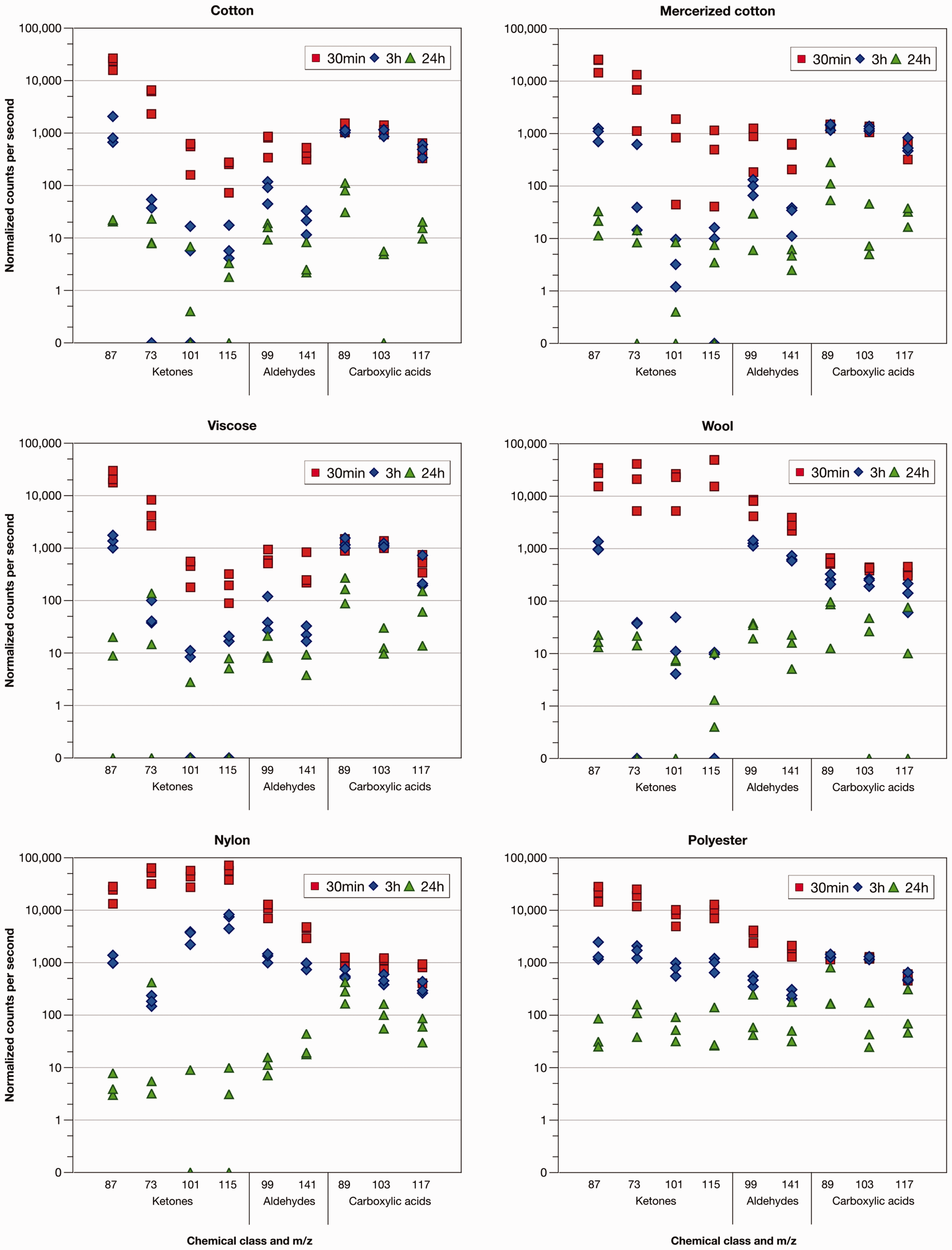
Normalized counts per second (ncps) of odorous volatile organic compounds (VOCs) released from fibers following contamination in odorous sweat solution.
Mean normalized counts per second (ncps) of odorous VOCs released from fibers following contamination in odorous sweat solution
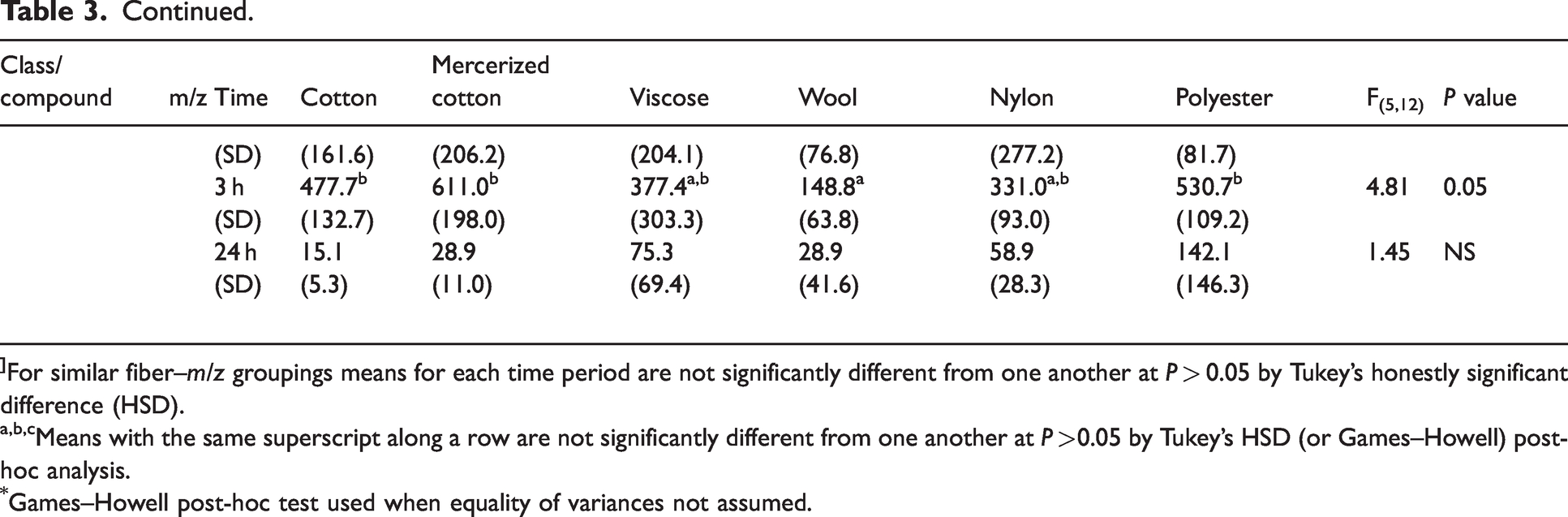
]For similar fiber–m/z groupings means for each time period are not significantly different from one another at P > 0.05 by Tukey’s honestly significant difference (HSD).
a,b,cMeans with the same superscript along a row are not significantly different from one another at P >0.05 by Tukey’s HSD (or Games–Howell) post-hoc analysis.
Games–Howell post-hoc test used when equality of variances not assumed.
As time increased, the VOCs dissipated from the fiber/yarn bundles, as shown by the decreasing intensity signals (ncps). Time significantly affected the amounts of VOCs detected in the headspace above treated fibers/yarns, as determined by a series of one-way ANOVAs on each like fiber × VOC group (given by Table 3). For the ketones and aldehydes, a significant decrease between 30 min and 3 h in the intensity of VOCs was detected for all fiber types. However, between 3 h and 24 h, differences were insignificant for the cellulosic or wool fibers/yarns for 2-butanone, 2-hexanone (except viscose) and 2-heptanone. A similar trend was found with mercerized cotton for 2-hexenal and viscose for 2-nonenal, in which between 3 h and 24 h time periods, no significant differences were evident.
A different effect with time was apparent for the carboxylic acids. Differences in intensities of carboxylic acids between the 30 min and 3 h periods following exposure to the sweat solution were not significant for any of the fiber types. However, between 3 h and 24 h the quantity of carboxylic acids decreased significantly for all fiber types (except for 2-hexanoic acid (m/z 117) on the wool, the nylon, and the polyester).
Fiber differences by chemical class
Ketones
Ketones were represented by four compounds (diacetyl [m/z 87], 2-butanone [m/z 73], 2-hexanone [m/z 101], 2-heptanone [m/z 115]). The release of diacetyl from the different fibers/yarns showed a different behavior than the other three ketones, but in particular, from the two largest ketones. No significant differences among fibers/yarns were found at 30 min (F5,12 = 0.10) and 3 h (F5,12 = 0.68) following exposure to the odorous sweat solution in the amount of diacetyl released, whereas, for the other ketones, significant differences among the fiber types were apparent at both these shorter periods (given by Table 3). For 2-butanone, the only significant difference among the fiber/yarn types was the greater release from nylon compared with the three cellulosic fibers (i.e. cotton, mercerized cotton, viscose), and although the mean values were higher for both wool and polyester than for the cellulosic fibers, these differences were not statistically significant.
Nylon fibers/yarns emitted the highest amounts of ketone VOCs at 30 min, differing significantly from the three cellulosic fibers. The amount of these compounds emitted from the polyester and wool fibers was lower than nylon but not significantly at 30 min. Three hours after exposure, far lower quantities of ketones were detected from wool, indicating a relatively rapid release rate from wool. In fact, in wool after the 3 h period, three ketones (2-butanone, 2-hexanone, 2-heptanone) did not differ significantly from those emitted from the cellulosic fibers. Instead, wool significantly differed from those detected in the headspace of nylon and polyester fibers (except nylon for 2-butanone). This indicates that although wool has a high sorption capacity for ketones in an aqueous solution, it desorbs them relatively rapidly compared with the rate of desorption from other fiber types.
At 24 h following exposure to the VOCs in the sweat solution, polyester exhibited the highest mean values for all ketones, although differences were not necessarily significant overall (i.e. 2-butanone: F5,12 = 1.97, NS) or from some other fiber types (e.g. for 2-hexanone polyester did not differ from wool at the 5% level of significance). No significant differences were observed among all other fibers. As nylon and wool consistently had higher abundances (ncps) at 30 min (e.g. for 2-heptanone: 37,807 ± 19,445 [wool]; 55,609 ± 17,059 [nylon] cf. 9983 ± 3024 [polyester]) with lower mean values at 24 h, this indicates that both nylon and wool were more effective at releasing ketones than was polyester.
Aldehydes
Two aldehydes, 2-hexenal (m/z 99) and 2-nonenal (m/z 141), used in this study exhibited release patterns from textile fibers similar to the ketones. At 30 min, differences among fiber types were found for 2-hexenal (F5,12 = 17.12, P < 0.001) and 2-nonenal (F5,12 = 16.84, P < 0.001), with post-hoc analysis indicating that quantities released in the headspace above the three cellulosic fiber types were lower than amounts detected above polyester, wool, and nylon at the 5% level of significance. Although higher mean values were detected from nylon than from either wool or polyester (given by Table 3), differences were not statistically significant at 30 min (e.g. for 2-hexenal: 6946 ± 2464 [wool]; 10,251 ± 2991 [nylon]; 3408 ± 915 [polyester]). Similar trends to results at 30 min were also noted at 3 h (i.e. 2-hexenal: F5,12 = 35.14, P < 0.001; 2-nonenal: F5,12 = 63.72, P < 0.001). However, at 3 h, the amount of 2-nonenal released from nylon was significantly greater than that released from polyester (i.e. 250 ± 52 [polyester]; 894 ± 138 [nylon]), whereas for 2-hexenal the wool, polyester and nylon did not differ significantly from one another. At 24 h, higher quantities of aldehydes were detected in the headspace of polyester fibers, although this did not differ significantly from wool and mercerized cotton (2-hexenal), or from either wool and nylon (2-nonenal).
Carboxylic acids
The three carboxylic acids in this study were butanoic acid (m/z 89), isovaleric acid (m/z 103) and 2-hexanoic acid (m/z 117). The trends in carboxylic acids detected at 30 min, 3 h and 24 h following contamination in the sweat solution by fiber type varied from the ketones and aldehydes. At 30 min, differences among fiber types were found for butanoic acid (F5,12 = 9.86, P < 0.001) and isovaleric acid (F5,12 = 19.42, P < 0.001), but post-hoc analysis revealed that the amounts emitted from cellulosic fibers did not differ significantly from those emitted from both nylon and polyester fibers. Carboxylic acids released from wool fibers/yarns at 30 min were significantly lower than all other fiber types. No statistically significant differences among fiber types for hexanoic acid were found at 30 min (F5,12 = 1.08). Similar trends were apparent at 3 h following exposure. Differences among fiber type were found for all carboxylic acids (butanoic acid: F5,12 = 44.77, P < 0.001; isovaleric acid: F5,12 = 62.04, P < 0.001; hexanoic acid: F5,12 = 4.81, P < 0.05), with wool fibers emitting the lowest amounts into the headspace. These results for wool were significantly different from all other fiber types. The three cellulosic fibers did not differ from polyester at the 5% level of significance. However, significantly lower amounts of butanoic and isovaleric acids were emitted from nylon compared with polyester and cellulosic fibers at 3 h.
Discussion
Several parameters can influence mechanisms of sorption by various adsorbing materials: (a) characteristics of the adsorbent itself; (b) characteristics of the adsorbate; and (c) media and conditions in which sorption takes place. For example, the sorbent (i.e. fiber) surface area, chemical composition, morphology and surface energy; the sorbant (i.e. VOCs) chemistry and initial concentration within a solute; and the pH, temperature, contact time, pressure, and competing compounds within the solute all play a role. 41 In the current study, the media and conditions were held constant; therefore, differences were associated with characteristics of the fibers and the VOCs evaluated. As noted, a general decrease in VOCs intensity occurred over time, and different behaviors were apparent depending on fiber content and VOC chemistry.
Initial amounts of ketones and aldehyde VOCs released from the six textile fibers could be generally categorized into two groupings (except for diacetyl). The first grouping comprised the three cellulosic fibers (cotton, mercerized cotton, viscose) with lower quantities of VOCs detected in the headspace. The second grouping comprised the two synthetic fibers (nylon, polyester) and wool. These three fiber types tended to release higher amounts of ketones and aldehydes, with nylon releasing more 2-heptanone. This suggests that cellulosic fibers sorbed less of the ketones and aldehydes from the sweat solution at the onset, accounting for the lesser amounts released in the headspace at 30 min. Conversely, comparatively more ketones and aldehydes VOCs were sorbed from the sweat solution by polyester, nylon, and wool fibers. This explanation aligns with the findings of Vaezafshar, 42 who found cotton and viscose retained lower quantities of nonenal, undecanone and undecanal than polyester and nylon fibers. Vaezafshar 42 used a similar method to that in the current study for immersing fabrics in aqueous sweat/odorous VOCs solution; however, the author extracted VOCs directly from contaminated fabric swatches immediately after removal from the sweat solution, thereby providing a direct measure of the quantity of compounds sorbed and initially retained by the fibers. An important interpretation of the current findings, supported by the work of Vaezafshar, 42 relates to the lower quantity of less polar VOCs detected in the headspace above cellulosic fibers, and the higher quantity in the headspace above polyester and nylon fibers. A difference between cellulosic fibers and synthetic fibers is related to the strong propensity of cellulose to absorb liquid water, and the preferential sorption of water over larger, less polar odorous compounds. Competitive sorption of VOCs and water on the different fibers can occur, further attributing to differences observed in the multi-compound solution. 41
Diacetyl did not exhibit the same trend as the other ketone VOCs, as there were no differences in the amount of diacetyl in the headspace among the fiber types. Compared with the other ketones, diacetyl has a high polar surface area of 34.1 Å 2 . This greater polarity of the compound leads to a greater affinity for forming hydrogen bonds with the functional hydroxyl groups in the cellulosic fibers. The higher detection of diacetyl in the headspace above the cellulosic fibers may relate to greater amounts initially sorbed, resulting in less of a difference in headspace analysis, as was observed for carboxylic acids.
The lack of a difference in intensity for the carboxylic acids between the 30 min and 3 h period for any of the fibers may be explained by the moisture content of the yarn bundles. Dissociation of the acids would have occurred within the aqueous solution, thus accounting for lower signal intensities at 30 min. By 3 h, although some drying would have occurred and some VOCs would have dissipated, the yarn bundles were still wet as evident in Figure 2.
Fiber characteristics influencing VOC sorption and release
That the three cellulosic fibers would behave in a similar way based on their chemistry was not unexpected. However, some differences in the sorption of VOCs were expected based on their inherent moisture sorption behavior that is affected by their physical morphology and fine structure. Cotton has a complex morphological structure composed of a lumen, primary cell wall, and secondary cell wall. Cellulose is arranged in microfibrils that reverse direction in the secondary cell wall, resulting in the fiber collapsing into a crenulated morphology. The process of mercerization swells the cotton fiber, thereby reducing the crenulation, creating a smoother surface and rounder cross-sectional shape. 43 Due to these changes in the physical morphology, mercerized cotton is more absorbent because of increased pore size and greater accessibility to hydroxyl groups for hydrogen bonding. 44 Viscose, a regenerated cellulosic fiber, has a much simpler microstructure with shorter cellulose chains, and is considerably less crystalline (more amorphous) than cotton. As a result, more hydroxyl groups are accessible to water and other polar compounds within the internal structure of the viscose fiber. These differences in the fine structure result in viscose being more absorbent than cotton, with 11% moisture regain in standard conditions (cf. 8.5% for mercerized cotton and 7–8% for natural-dyed cotton). 45 Such differences in fiber morphology were associated with differences between mercerized cotton and untreated cotton in work by Liu et al., 18 in which a slightly higher absorption of aroma compounds transferred by an aqueous solution in mercerized cotton was observed. In a study examining the sorption and release of selected VOCs in a gaseous environment, viscose absorbed greater amounts of polar VOCs such as hexanoic acid and phenol than cotton, 16 but not for any of the other VOCs evaluated in their study. Our findings, however, showed no significant differences in the amount of VOCs detected in the headspace above the cellulosic fibers, and are in agreement with Vaezafshar. 42 In that study, cotton (nonmercerized) and viscose were compared, and no significant differences were found in the sorption of six VOCs (i.e. isovaleric acid, octanoic acid, 2-heptanone, 6-undecanone, nonenal and undecanal).
Both wool and nylon fibers initially released the highest intensities of ketones and aldehydes, indicating the high sorption among the six fiber types. However, they also released VOCs at a much higher rate because ketone and aldehyde intensities at 24 h were comparable with those of the cellulosic fibers. Both wool and nylon carpets have been found to have higher sorption capacities to toluene and α-pinene, with wool having a higher equilibrium constant during the desorption phase. 46 The wool keratin protein molecule is made up of a complex sequence of amino acids that enables chemisorption with an array of molecules due to the many reactive sites, such as the peptide bonds and polar and ionic functional groups on the amino acid side chains. The high surface area associated with many pores of the keratin fibers enables physisorption. 47 Similar findings have been reported elsewhere, in which wool fibers and powders made by pulverizing waste wool had van der Waals forces dominant in the physisorption and Lewis acid-base interactions for chemisorption.48,49
Moisture also enhances sorption compared with sorption in dry environments. 50 The method of complete submersion of fibers/yarns within the sweat solution would have also played a role in facilitating sorption but also a high initial release. The yarns were initially completely saturated, and on removal from the bulk liquid, unbound VOCs would quickly begin to dissipate, as evidenced by the highest initial counts at 30 min, and significantly lower intensities at 3 h. The release rate slows as the wool yarns dry because chemically bound VOCs remain within the fibers. 50 Accounts of an initial rapid release of formaldehyde leading to incomplete desorption with residue compounds remaining bound within the fiber structure have been noted elsewhere. 51
The high initial sorption of compounds in the aqueous environment by nylon can also be explained due to fiber chemistry. Nylon is a polyamide with many repeating amide linkages: amide groups separated by methylene bridges. The methylene chains within nylon enable nonpolar interactions to occur, while the hydrophilic amide groups allow for the sorption of water molecules into the fiber interior, 52 confirming that nylon fibers bind both nonpolar and polar compounds within/to its structure. As a synthetic fiber, nylon has been shown to be more ab/adsorbent than hydrophobic polyester fibers. Nylon fabrics exhibited a higher overall sorption capacity of isovaleric acid and 2-nonenal VOCs in a gaseous form than polyester. 12 In solution, far greater sorption of the surfactant dodecyldimethylphosphine oxide occurred in nylon fabrics compared with polyester, as well as higher sorption than cotton and viscose fabrics. 53 The rapid release of VOCs from nylon as the fiber dried compared with polyester, coupled with the binding of compounds at reactive sites on nylon, may lead to lower odor intensity following the use of nylon fabrics. 54 In fact, the higher quantities of aldehydes emitted from polyester 24 h following treatment when compared with the amount at 30 min, in contrast to higher values on nylon and wool, at 30 min and lower quantities at 24 h, suggests that nylon and wool release aldehydes initially more rapidly. This may also suggest that nylon and wool would be more effectively ‘aired’ than polyester. However, VOCs that are chemisorbed within the wool fibers may not be released to be detected by the sense of smell; to a lesser extent, this may also occur with nylon.
Unlike the other fibers examined in the current study, polyester (polyethylene terephthalate) is effectively inaccessible to moisture with a moisture regain of about 0.4% at 21°C and 65% relative humidity. 45 As water will not enter the fiber interior, adsorption of VOCs on the fiber surface will occur. Attraction between the nonpolar ketones and aldehydes occurs due to the lack of affinity that either the polyester fiber or the compounds have for water, leading to the adsorption of odorous VOCs to the polyester surface. Adsorption of odorous VOCs to polyester will occur primarily through van der Waals interactions, by which, on drying and removal from the sweat solution, desorption into the headspace above the fibers occurs. 55 Polyester does not have multiple functional groups, like the other fibers examined; therefore, none to minimal chemical binding may occur within the amorphous regions of the fiber internal structure under test conditions of the current study (i.e. room temperature). Based on the published literature, polyester fibers typically exhibit higher rates of release of VOCs than either nylon or wool fibers/yarns during a 6–8 h period.16,17,42 This may also be relative to the higher quantities of compounds, particularly nonpolar compounds, that are adsorbed by polyester initially. 42
Implications
Compared with other studies in which the sorption and release of odorous compounds in textiles have been examined, the current study offers insights that have been largely overlooked. Most investigations have examined the sorption of odorous VOCs in air. Sorption of VOCs through air is the basis of the international test standard ISO 17299, in which an overall odor reduction rate is calculated based on the amount of odorant detected in a container with a textile present relative to an empty container. 56 A similar approach for measuring the sorption of VOCs on textiles was carried out using PTR-MS as the instrument for detection.16,17,29 Other methods for introducing odorous VOCs to textiles have involved applying discrete quantities of single VOCs in a solvent4,9 or a sweat solution. 30 Because of the proximity of clothing in use to the human skin, liquid sweat passes through clothing fabrics, providing a major route for transporting odorants to apparel textiles. The relevance of odorant transfer in the presence of moisture, particularly liquid sweat, was noted by Hammer et al., 30 in which the binding of isovaleric acid was measured using an artificial skin model to transfer the VOC to textile substrates. In the current study, yarns were completely saturated in a synthetic sweat solution. Although this procedure does not reflect the complexity of fabric structural parameters, which can affect liquid moisture sorption, nor the compression and friction that occurs between the human skin and textiles, it does provide a new method for examining generic fiber differences when exposed to odorous VOCs in an aqueous liquid medium. As excessive sweating occurs during intense exercise, assessing the sorption of odorous VOCs to textile fibers through an aqueous liquid rather than through air is warranted. The novel method simulates the saturation of textile materials with aqueous sweat rather than unrealistic solvents or contact with only small discrete quantities of the same volume of liquid. When saturated, the more nonpolar VOCs had a greater affinity to hydrophobic fibers (i.e. nylon, polyester) or to those with multiple active functional groups (e.g. wool). For these fibers the attraction of the nonpolar compounds toward the fibers will be greater than their affinity to remain in the aqueous solution. Comparatively, the cellulose fibers with hydroxyl functional groups had a preferential attraction to absorb water, and excluded more of these hydrophobic compounds.
This work contributes to our understanding of odorant transfer and sorption within common fiber-based textiles by way of sweat. Clothing composed of cellulosic fibers may be less odorous than synthetic clothing following wear, because lower quantities of odorous nonpolar VOCs (and presumably nonpolar precursors to odor) are ad/absorbed at the onset. Furthermore, this study offers insights into the release of VOCs from different fiber-based textiles, showing how ‘airing’ may be more effective for minimizing odor emitted from both wool and nylon fabrics and less so for polyester. PTR-MS offers the advantage of real time monitoring of headspace at selected time periods, and has been used for measuring textile odor in previous research.17,20,29
Recommendations for future research
Notwithstanding the novel approach in this investigation, several limitations to the method may be addressed in further research. The temperature of the sweat solution was that of the room (approx. 20°C) and was not consistently monitored. However, sweat transfer from the body to apparel fabrics will typically occur at skin temperatures (32–35°C). Temperature is an important parameter influencing sorption. Sorption of compounds can increase with increasing temperature in aqueous solutions, so at a 10–15°C increase in temperature, higher amounts of compounds would have been absorbed in all fibers, including the cellulose fibers. 53 Contact time is another parameter that influences sorption. 57 Yarn bundles were exposed to the sweat solution for only 30 min. Equilibrium of sorption of other compounds, such as the surfactant dimethyldodecylphosphine oxide, can take over 8 h. 53 Therefore, it is expected that the quantity of VOCs ad/absorbed would increase with increasing duration of exposure up to equilibrium. Higher quantities of VOC sorption would likely occur for all fiber types if temperature and contact time were increased (and therefore, the trends would remain the same). Future work should examine contact under extended contact times that may simulate different body–clothing interactions (e.g. an 8-h wear day).
In addition to investigating the parameters of temperature and contact time, future research should also examine other parameters surrounding the sorption of sweat-derived odorants from sweat solutions. The pH of the sweat solution in the current study was 4.3. However, the axillary region has a higher pH ranging from 5.2 to 6.6, depending on the part of the axillae, or other endogenous and exogenous factors.58,59 Thus, in future research, the sorption of odorous VOCs at varying levels of pH could be conducted. Conducting the experiments under conditions in which the kinetics of odorant adsorption and desorption are examined and modeled is also recommended in future work. 57
The focus of the current study was to develop a method for applying odorous VOCs that varied in polarity and solubility in a sweat solution to textiles to determine differences among fibers in sorption and desorption processes in a laboratory setting using real time headspace monitoring. However, human sweat comprises a complex mixture of many VOCs that vary even more widely than those odorants selected here.
Conclusions
This study utilized real time PTR-MS to examine the release of six odorous VOCs from cellulosic, wool and synthetic fibers/yarns exposed to the VOCs in an aqueous solution. Shortly following treatment with the odorous sweat solution, cellulosic fibers released lesser amounts of nonpolar ketone and aldehyde VOCs into the headspace compared with wool, nylon, and polyester fibers. This finding suggests that cellulosic fibers ad/absorb lower quantities of nonpolar compounds initially when transferred through an aqueous sweat solution than textiles composed of wool, nylon, and polyester fibers. Conversely, textiles consisting of polyester and nylon fibers released higher quantities of nonpolar VOCs. This research enhances our understanding of odorant transfer and absorption in various textiles through sweat. Clothing made from cellulosic fibers may tend to be less odorous than synthetic garments when worn, because they adsorb lower quantities of odorous nonpolar VOCs (and presumably odor-related precursors) initially.
Footnotes
Acknowledgements
The author(s) would like to thank Ms Michelle Leus for her assistance in using the PTR-MS and Dr Pat Silcock for his assistance with developing the PTR-MS protocols.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the support of the Natural Sciences and Engineering Research Council of Canada (NSERC).
