Abstract
Introduction
Clothing made from tight material worn by operating room staff adds to the effect achieved by ventilation in the effort to keep a low level of bacteria in the operating room air. The material used for clean air suits should be tested according to the standard EN 13795-2.
Aim
The aim of the study was to investigate whether there was a difference in protective capacity between three clean air suits made from materials fulfilling the requirements of EN 13795-2:2019 and designed as described in Annex E of the standard.
Materials and methods
Eight people (five men and three women) performed standardized movements in a dispersal chamber with a fixed supply air flow. Each person performed two test cycles with each clean air suit. Counts of colony-forming units (CFU) in the air were measured during testing and source strength was calculated for each test cycle.
Results
The mean values of source strength for the three clean air suits were 1.4 CFU/s, 0.8 CFU/s and 0.7 CFU/s. The difference between the most and the least protective garment was statistically significant (p < 0.05).
Conclusions
Clean air suits made from material fulfilling the requirements for ‘high performance’ in the standard EN 13795-2:2019 might show a significant difference in protective capacity when comparing source strength. Tests for measuring of source strength in a dispersal chamber can be performed as suggested in Annex E of the standard, i.e. ‘The test person is male, 20–50 years old, with no visible skin disorder’.
It is well known since the comprehensive study of orthopedic prothesis surgery performed by Lidwell et al. in the late 1970s that tight clothing worn by operating room staff adds to the effect achieved by ventilation in the effort to keep low levels of bacteria in operating room air. 1 The microbial quality of the air is expressed as colony-forming units (CFU) per cubic meter (m3).
Both before and after the above-mentioned studies by Lidwell et al., several studies have compared the performance of staff clothing made from different materials, mainly polyesters, microfiber and mixed materials regarding their impact on CFU levels in the operating room.2–7 This has led to the development of new materials for clothing and has also led to a stepwise progress of the requirements for clothing in the European standard where the demands are described together with the testing methods. A suit that is meant to be part of measures taken to protect the patient from contamination from operating staff is called a clean air suit and is, by definition, a medical device.
The latest version of the European standard EN 13795-2 from 2019 describes the performance requirements for clean air suits with specification of the two levels ‘standard performance’ and ‘high performance’. 8 The test procedure in the standard, however, only concerns characteristics of a material that can be used when creating the actual clean air suit. To investigate the protective capacity of the whole garment (such as coverall or blouse and trousers), testing must be performed in an operating room or in a dispersal chamber.
The aim of the present study was to investigate whether there was a difference in protective capacity between three modern clean air suits made from materials fulfilling the requirements for high performance in EN 13795-2:2019 and designed as described in Annex E of the standard. We chose to perform tests in a dispersal chamber to avoid unforeseen situations that might appear during surgical procedures in an operating room.
Materials and methods
Clothing systems
Three reusable clean air suits entitled A, B and C were evaluated in the dispersal chamber.
The characteristics of the fabrics of the clean air suits are given in Table 1.
Characteristics of the fabrics used for the three clean air suits
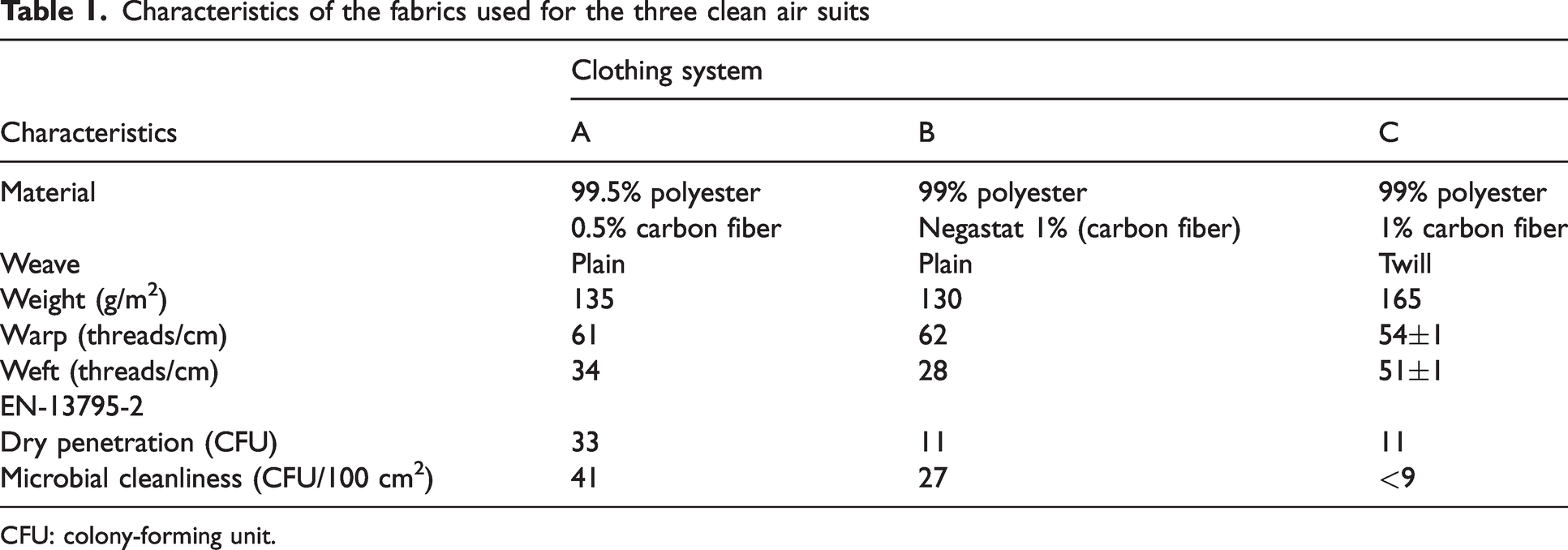
CFU: colony-forming unit.
Each clean air suit consisted of a textile hood with cuffs around the face and buttons below the chin, a blouse with cuffs at arms, neck and waist and a pair of trousers with cuffs at the wrists.
The three clean air suits are shown in Figure 1.

Clean air suits tested. From left to right: A, B and C.
All parts of the clean air suits were delivered new and washed twice (70°C for 10 minutes) and packaged in sealed bags. Washed socks were delivered in separate sealed bags. The laundering company uses the standards EN 14065 and ISO 15797 for control purposes.9,10
Test subjects
The test subjects were five men age between 20 and 51 years and three women aged between 20 and 50 years.
Performance of test
During the evaluation tests the hood was well tucked under the blouse and the blouse was tucked into the trousers. Additionally, subjects wore a disposable facemask, nonsterile gloves, washed but not new socks (65% cotton and 35% polyester) and clean, disinfected open plastic shoes (sandals). During the evaluation of clean air suit A, the face masks had loops behind the ears under the hood, whereas in the tests of the two other clean air suits the face masks had drawstrings tied outside the hood.
During the measurements, the test subjects performed standardized cycles of movements that included 3 minutes of arm movements (one every 5 seconds), 3 minutes of knee bends (one every 20 seconds) and 3 minutes of calm walking on the spot at a set speed (one step per second). Before each cycle of movements, the test subject stood still to avoid the influence of particle generation from the previous test cycle. The evaluated clothing systems each had the same eight test people (1–8) performing the standardized cycles of movements twice (a and b).
Dispersal chamber
A dispersal chamber is a qualified and validated chamber with a volume of approximately 2 m3, with tightly sealed walls and door and with a specified supply of HEPA-filtered air at positive pressure (≈5 Pa) and controlled outflow. The principal arrangement of the dispersal chamber is shown in Figure 2. Descriptions of the dispersal chamber located at Chalmers University of Technology, Gothenburg, Sweden, which was used in this study, are given in previous publications by Ljungqvist et al. and Lytsy et al.7,11
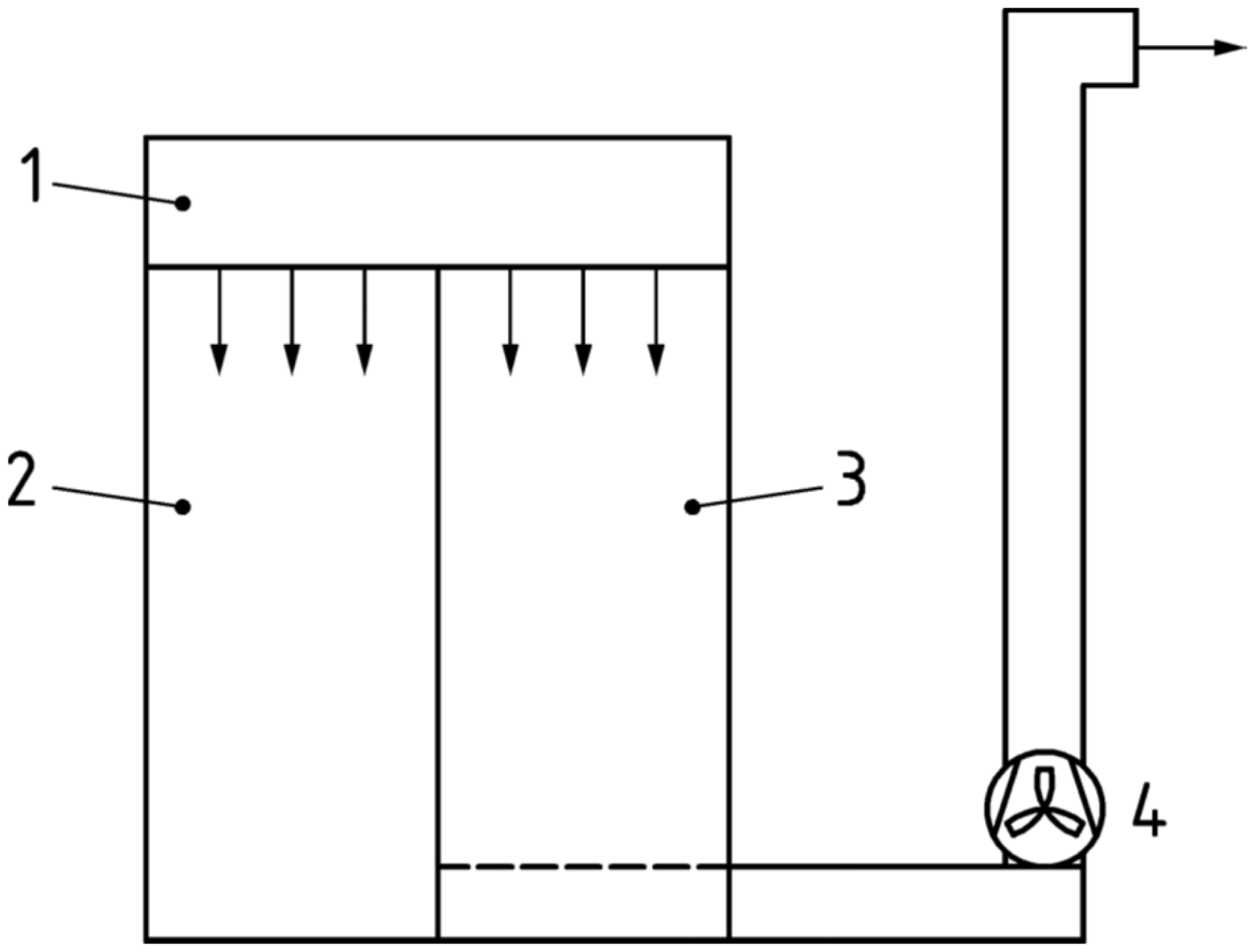
Principal arrangement of dispersal chamber with its changing area and test cabin (body box). 1. HEPA filter. 2. Changing area. 3. Test cabin (body box). 4. Exhaust fan.
In the exhaust air of the test cabin, viable particles were collected using a slit sampler (FH3®; d50 value of 1.6 µm). The instrument was operated according to the manufacturer’s instructions.
Microbial growth medium for all tests was standard Tryptic Soy Agar (TSA) in Petri dishes with a diameter of 9 cm, pre-sterilized and double packed. The TSA plates were incubated for at least 72 hours at 33°C followed by at least 48 hours at room temperature. After incubation, the number of CFU were counted, characterized and recorded as aerobic CFU per m3.
Source strength
The source strength is described as the number of viable airborne particulates per second emitted from one person.
By using the air volume flow in the dispersal chamber of 0.29 m3/s, in combination with the measured CFU concentrations, the source strength for each test cycle was calculated.
Source strength (outward particle flow):
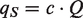
c = concentration; bacteria-carrying particles (CFU/m3)
Q = total air flow (m3/s)
Data analysis
A two-sided Mann–Whitney U-test was used to compare the values for source strength obtained with clean air suit A with the values obtained with the other two suits. Software provided from Statistics Kingdom (http://www.statskingdom.com) was used for analysis.
Results
The test results are shown in Tables 2 (men) and 3 (women), where the source strength values (CFU/s) are given for every test cycle and test subject.
Calculated source strength values (CFU/s) for every test cycle with five male test subjects
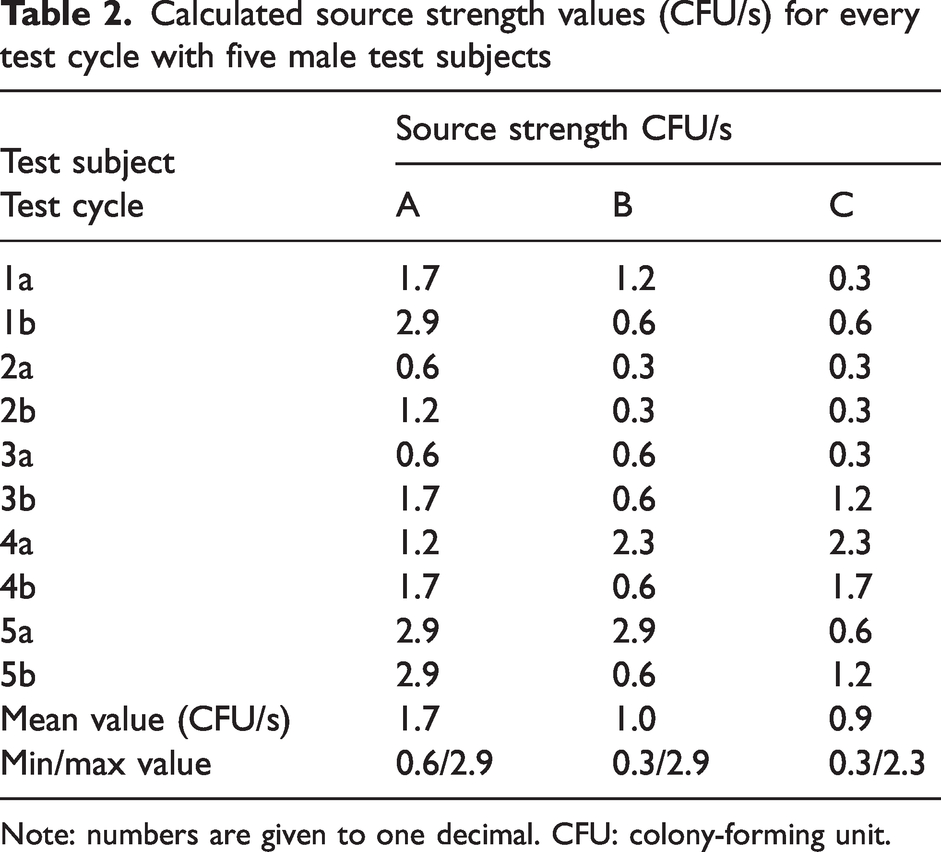
Note: numbers are given to one decimal. CFU: colony-forming unit.
Calculated source strength values (CFU/s) for every test cycle with three female test subjects
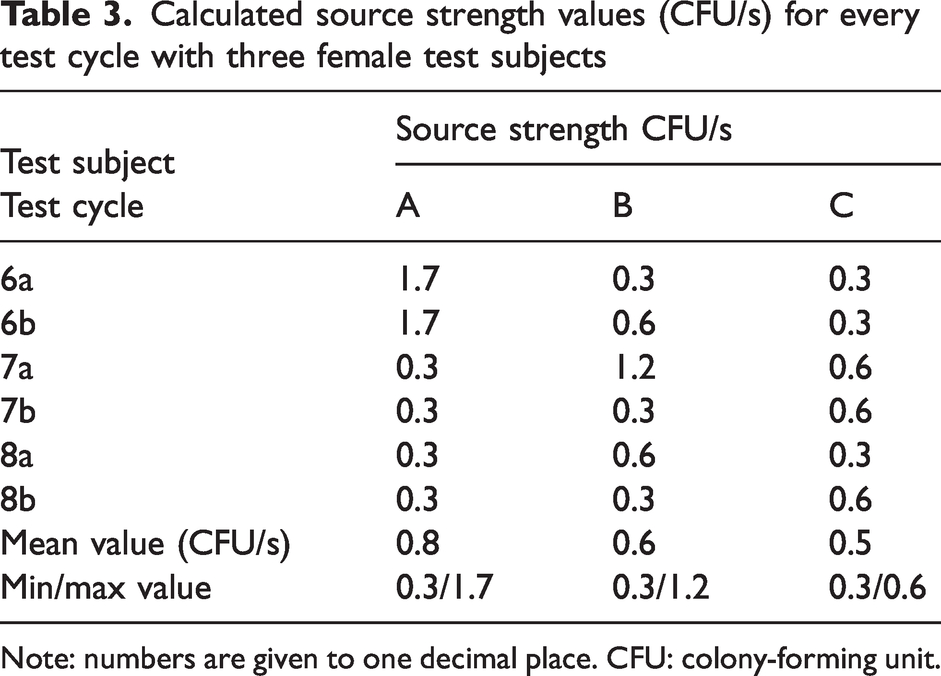
Note: numbers are given to one decimal place. CFU: colony-forming unit.
For men, the difference between the values for source strength obtained with the clean air suits A and B was statistically significant (p < 0.05) as well as the difference between clean air suits A and C (p < 0.05).
For women, there were no significant differences between values for source strengths, neither between clean air suits A and B nor clean air suits A and C.
Mean values of source strength for each clean air suit calculated from all test cycles from men and women presented in Tables 2 and 3 are:
A: qs = 1.38 ≈ 1.4;
B: qs = 0.83 ≈ 0.8;
C: qs = 0.72 ≈ 0.7.
For the whole group of men and women the difference between the values for source strength obtained with clean air suits A and C was statistically significant (p < 0.05), whereas there was no significant difference between clean air suits A and B.
Discussion
All three materials used for the clean air suits in this study passed the requirements for ‘high performance’ in the standard EN 13795-2:2019. 8 There was, however, a significant difference in protective capacity between suit A and the other two when comparing source strength. This is important information for a buyer of clean air suits such as a hospital or a county or region providing healthcare for the inhabitants.
The description of the test method for measuring source strength in Annex E of the standard EN 13795-2:2019 suggests that all test subjects should be men, which is based on several studies performed in the 1960s and 1970s comparing dispersal from men and women (where men were found to shed more skin scales than women).12,13 In this study we chose to use both men and women as test subjects as we know that more women than men are employed in the operating departments where clean air suits are used. The results, however, show that the average source strength is lower for women and that significant differences between the protective capacity of the clean air suits was found in the male but not the female group, thus justifying the choice of men as test subjects to simulate a ‘worst case’ scenario.
In this study, each test subject performed two test cycles with each of the three clean air suits. As seen from the results, there was a difference between source strength calculated for the same person from the two test cycles with a specific clean air suit. In 12 out of 24 pairs of test cycles the source strength was at least double in one of the tests compared with the other. This indicates that an intra-individual difference in dispersal of skin scales exists and is not always eliminated by the clean air suit. It shows the necessity to perform multiple test cycles with each test subject, as suggested in Annex E (E.3) of the standard EN 13795-2:2019.
The guiding technical specification, SIS-TS 39:2015, published by the Swedish Institute for Standards suggests that the requirement for a clean air suit should be achievement of a source strength of ≤1.5 CFU/s when the garment is used in an operating room during ongoing surgery with an activity level close to hip joint surgery. 14 About twice this value is achieved when the evaluation is performed in a dispersal chamber.15–18 This indicates that for the three tested clothing systems, the expected source strength mean values will be <1 CFU/s during ongoing hip joint surgery.
Defining a value for performance of the whole garment, not only the material, is a promising development, which would be helpful to differentiate between clean air suits offered by different manufacturers. It has, however, to be decided whether the suggested value of 1.5 CFU/s should refer to a clean air suit made from a material fulfilling the requirements for ‘standard performance’ or ‘high performance’ in the standard EN 13795-2:2019. This could be an area for further studies of clean air suits.
Conclusions
Clean air suits made from material fulfilling the requirements for ‘high performance’ in the standard EN 13795-2:2019 might show a difference in protective capacity when comparing source strength.
Tests for measuring of source strength in a dispersal chamber can be performed according to what is suggested in Annex E of the standard EN 13795-2:2019 (i.e. ‘The test person is male, 20 years to 50 years old, with no visible skin disorder’). Each test subject should perform multiple cycles with the same clean air suit when testing to detect intra-individual differences in shedding of bacteria-carrying particles.
All calculated values of source strength for each person should be presented together with figures for minimum, maximum and average source strength for the group of test subjects to give a complete basis for decisions when choosing a clean air suit.
The results from testing of clean air suits in a dispersal chamber should be included together with results from testing of material according to the standard EN 13795-2:2019 as a basis for decision when purchasing staff clothing for surgical departments.
Limitations
In this study we chose to use more test subjects than suggested in the standard EN 13795-2:2019 but on the other hand, each test subject performed fewer test cycles. The standard says ‘The source strength is an approximation based on 20 measurements in the dispersal chamber, with five male test persons, each performing a standardized exercise four times on separate days.’ Fewer test cycles per person might mask an even larger intra-individual difference in dispersal of skin scales than we detected.
Source strength values based on CFU concentration <4 CFU/m3 gave unsecure values. This means that source strength values <1.2 CFU/s in this study are indicative, which should be considered when comparing protective efficiency between the tested clean air suits. It should also be noted that source strength values <0.6 CFU/s are based on CFU concentrations near the detection level of the slit sampler.
Footnotes
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Textilia Tvätt and Textilservice AB.
