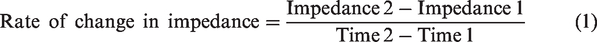Abstract
In this paper, we evaluated the impedance of textile electrodes for electroencephalography measurements. We compared the commercially available standard dry silver/silver chloride dry electrodes, copper-based, and silver-based textile electrodes by measuring impedance on the skin. First, on dry skin, the impedance range of the electrodes was 93–350 kΩ at 0 min; after 60 min, the range was 20–56 kΩ. The commercial electrode showed the lowest impedance of 20 kΩ, and the silver-based textile electrode showed the highest impedance of 56 kΩ in dry skin conditions after 60 min. When tap water was applied as an electrolyte, the impedance range was 13–15 kΩ at 0 min; after 60 min, the range was 17–22 kΩ for all three types of electrodes. In this case, the copper-based textile electrode showed the lowest impedance of 17 kΩ and the commercial electrode showed the highest impedance of 22 kΩ after 60 min. When saline water was applied as an electrolyte, at 0 min, the impedance range was 8–11 kΩ; after 60 min, the range became 11–12 kΩ for all three types of electrodes. Thus, all electrodes showed similar results. It was concluded that the copper-based textile electrodes were more suitable than the silver-based textile electrodes and comparable to commercial electrodes regarding impedance in all three skin conditions.
Mental health problems, work-related stress, and brain health disorders contribute to the emergence of many diseases and cause substantial financial losses. Identifying such issues early is the key to reducing their impact. Brain health and brain load could be most reliably assessed by directly measuring the brain’s electrical activity.
Extensive research is currently being conducted to integrate wearable technology into daily life to make human life safer, easier, and more accessible. Wearable solutions have spread rapidly in the healthcare and consumer markets because of their portability, lightweight quality, washability, and long-term monitoring capabilities, among other advantages. 1 Wearable exoskeletons for movement and rehabilitation, 2 nonstop sweat pH monitoring, 3 continuous blood glucose monitoring, 4 and continuous skin temperature monitoring and analysis 5 are a few examples of the versatile wearable health monitoring and rehabilitation sector.
The current wearable devices for measuring electrical activity in the brain (electroencephalography; EEG) are unsuitable for long-term measurements as they are still too uncomfortable and clumsy to use. There is a need to develop a measurement method that travels effortlessly and unobtrusively with the user and provides reliable information in all operating environments. Clothing is a natural place for these measurement technologies, which are also more emotionally and socially acceptable than current devices. When integrated into clothing, flexible textile-based electrodes enable comfortable, long-lasting, and unobtrusive measurements. Many brain functions can be especially observed from the frontal lobe EEG signal. 6 For example, cognitive functions such as motor control, 7 neuropsychiatric functions, attention deficit, 8 planning, working memory functions, 9 and language learning functions 10 are detectable brain functions in the frontal area.
The skin-electrode impedance is an important parameter affecting the quality of the EEG signal, and it is affected, for example, by electrode material and size, sweat, temperature, pressure, and skin preparation. 11 This study compared the skin impedance of copper and silver-based textile dry electrodes with commercial standard dry silver/silver chloride (Ag/AgCl) electrodes under three different skin conditions: dry skin measurement, with tap water applied as an electrolyte, and with saline water used as an electrolyte for long-term EEG signal acquisition.
Basics of EEG and current measurement devices
Monitoring the biosignal of the human brain’s electrical activity from the scalp’s surface (EEG) 12 is the safest, most straightforward, and most noninvasive way to acquire a neurophysiological response. The brain wave electrical signal was first recorded in 1924; the alpha brain wave was recorded as low-frequency brain wave fluctuations in 1929 by Hans Berger.13,14 For example, EEG is used to diagnose epilepsy, sleep disorders, and attention-deficit hyperactivity disorder. 15
There are five common types of brain biosignal frequency ranges in the medical sector, denoted by the Greek alphabet: delta (0.5–3 Hz), theta (4–7 Hz), alpha (8–13 Hz), beta (13–30 Hz), and gamma (30–100 Hz). 16 Postsynaptic potential is responsible for brain electroactivity. An electric dipole is created due to the current flow in the brain. Na+, K+, Ca++, and Cl− movement creates current flow. Only the sum of the electrical activity of the numerous neuron cells can be recorded as a brain signal. 17 An accepted electrode placement system is known as the 10–20 system for EEG measurement. 18 Odd numbering would be on the left side; thus, even numbering would be on the right. 18 There are five sections for electrode designation for brain wave detection: frontal, temporal, central, parietal, and occipital. 19 In the 10–20 system, the skull and head area are divided into 23 standard positional points. From these points, fronto-polar 1 (FP1) and fronto-polar 2 (FP2) are on the forehead (the frontal lobe) area for measuring the EEG signal, and A1 and A2 are the earlobe regions used for reference, driven ground leg, or the common-mode rejection ratio point of the measuring circuit. 18
The human body’s inner tissues and outer surface area are divided by different layers of the skin. The stratum corneum (SC) layer is the topmost layer of skin, with a thickness of 10–15 µm. 20 Between the dermis and the SC is the epidermis.20,21 The hypodermis underneath the dermis contains the blood vessels, whereas the dermis contains sweat glands.20,21
Electrodes are utilized as a transducer to collect EEG biosignals; skin impedance is a prominent issue for EEG measurements. A large contact impedance causes the EEG signal attenuation, making a system more susceptible to power line interferences and motion artifacts during measurement.22,23 So, while fabricating the new electrodes, considering the effect of the new electrode and the contact skin impedance to obtain a better EEG signal is essential. The skin impedance should be between 5 and 20 kΩ to generate an accurate EEG signal.12,13 Figure 1 represents the basic electrical model of a skin layer. 24
Basic skin electrical model. 24
Here, R0 is the skin resistance, C, the skin capacitance, and R, the inner resistance of the internal tissues. The effect of C and R0 varies with the skin preparation. For example, R0 will decrease when the skin is introduced to moisture. 24
Wet electrodes are commonly used for acquiring biosignals in medical sectors. However, in the case of long-term monitoring, wet paste in electrodes is accused of creating skin irritation or allergies, and the signal quality can be corrupted when the gel or wetness dries up.25–27 Semi-dry or dry electrodes are used to overcome these issues. Conductive yarn, 28 electro textile 29 and conductive ink, 30 or paste 31 have been used to fabricate flexible dry electrodes 32 for various health monitoring or biosignal detection purposes. EEG measurement during sleep 33 and EEG monitoring for epilepsy 34 are two examples of textile-based electrodes for health monitoring. However, until now, there has been no direct comparison of different textile materials and their effect on skin impedance.
Several commercial wearable EEG devices are on the market. Some of the gadgets are the ‘BrainBit’ a four-channel EEG device used to monitor brain signals, meditation, and sleep recording, 35 ‘MuseS’ a four-channel EEG device that records brain signals during sleep and meditation and provides raw EEG data, 36 ‘Imec’ an EEG device compatible with a computer or tablet for data visualization, 37 and ‘NEUROON’ a biorhythm monitoring device that measures EEG and electrooculogram. 38 However, all these devices are still too uncomfortable and clumsy for daily use. Thus, the goal is to create a comfortable and attractive textile-based EEG measurement system.
Electrode materials and fabrication
Commercial electrodes are made of Ag/AgCl. 39 The textile electrodes were fabricated from two different conductive textiles. ‘Stretch conductive fabric’ is a medical-grade plainly knitted fabric of 76% nylon and 24% elastane and finally coated with silver, which was from the Less EMF Inc., USA supplier. The unstretched surface resistivity of this conductive fabric is less than 1.00 Ohm/square, and the fabric thickness is 0.40 mm.40,41 The second material, ‘pure copper polyester taffeta’ fabric, is a plain-woven polyester-based with copper plated and has a surface resistivity of less than 0.05 Ohm/square from the supplier Less EMF Inc., USA.41,42 The fabric has a thickness of 0.08 mm and a copper content of 35% per 880 g/m2. 42
According to the study, the best projection of brain waves is demonstrated when the electrode diameter is 10 mm. 43 Therefore, our experiment used an electrode with a diameter of 10 mm.
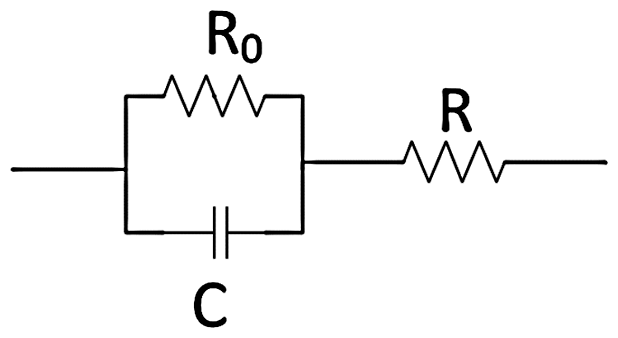
The fabricated textile electrodes were constructed according to Figure 2. First, the electro textile was cut into 10 mm diameter; a conductive wire was placed between the conductive textile and copper tape. Then the copper tape side was glued with ethylene propylene diene monomer (EPDM) foam of 2 mm thickness to make a measuring base. The dimension of the base foam was 3 cm × 1.5 cm. This EPDM substrate was attached with velcro by using glue, which helped to hold the electrode to the adjustable headband. Figure 3 shows the finalized fabricated textile electrodes and commercial electrodes.

Electro-textile electrode structure.
Copper textile-based electrode (left); silver textile-based electrode (middle); commercial electrode (right).
Measurement setup
The OpenBCI ganglion device is an open-source brain–computer Interface (BCI), a four-channel device that can communicate with the laptop through bluetooth low energy for data acquisition, was used for this study. An MCP3912 integrated circuit is the main IC for operation. Also, OpenBCI has a graphical user interface for signal representation and data recording. 44
Skin impedance was measured under three skin conditions: dry skin measurement, tap water applied as an electrolyte, and saline water used as an electrolyte. Saline water was made according to the manual from Florida Research Instruments, Inc. This saline water-based electrolyte was concocted with one tablespoon of sodium chloride and 200 ml of water. 45 A cotton pad was used to apply the electrolytes only to the exact points to avoid the bridge connection between adjacent electrodes.
Previous research also supports using tap water and saline water as electrolytes. For example, tap water increased the clean signal acquisition quality for a longer time for the electrode structure, made of a small pellet electrode with a rolled cotton layer. 46 Tap water and saline water-based electrolytes reduced the contact impedance and showed better performance in a BCI than the dry electrode.47–49 Finally, a saline water-based electrolyte decreased the contact impedance of polymer-based micro sponge electrodes more than tap water. 49
All the three types of electrodes including commercially available dry electrodes, silver textile-based electrodes, and copper textile-based electrodes were tested twice in this study with three participants each (one man and two women) for three skin conditions. All participants were healthy and fell into the 20–32 years age group. All the measurements were taken at stable normal office temperature and thus the temperature of the environment did not change. Throughout the hour-long measurement, the electrodes were attached to the band (and were thus held with a constant pressure) to maintain the electrode placement intact for all the participants.
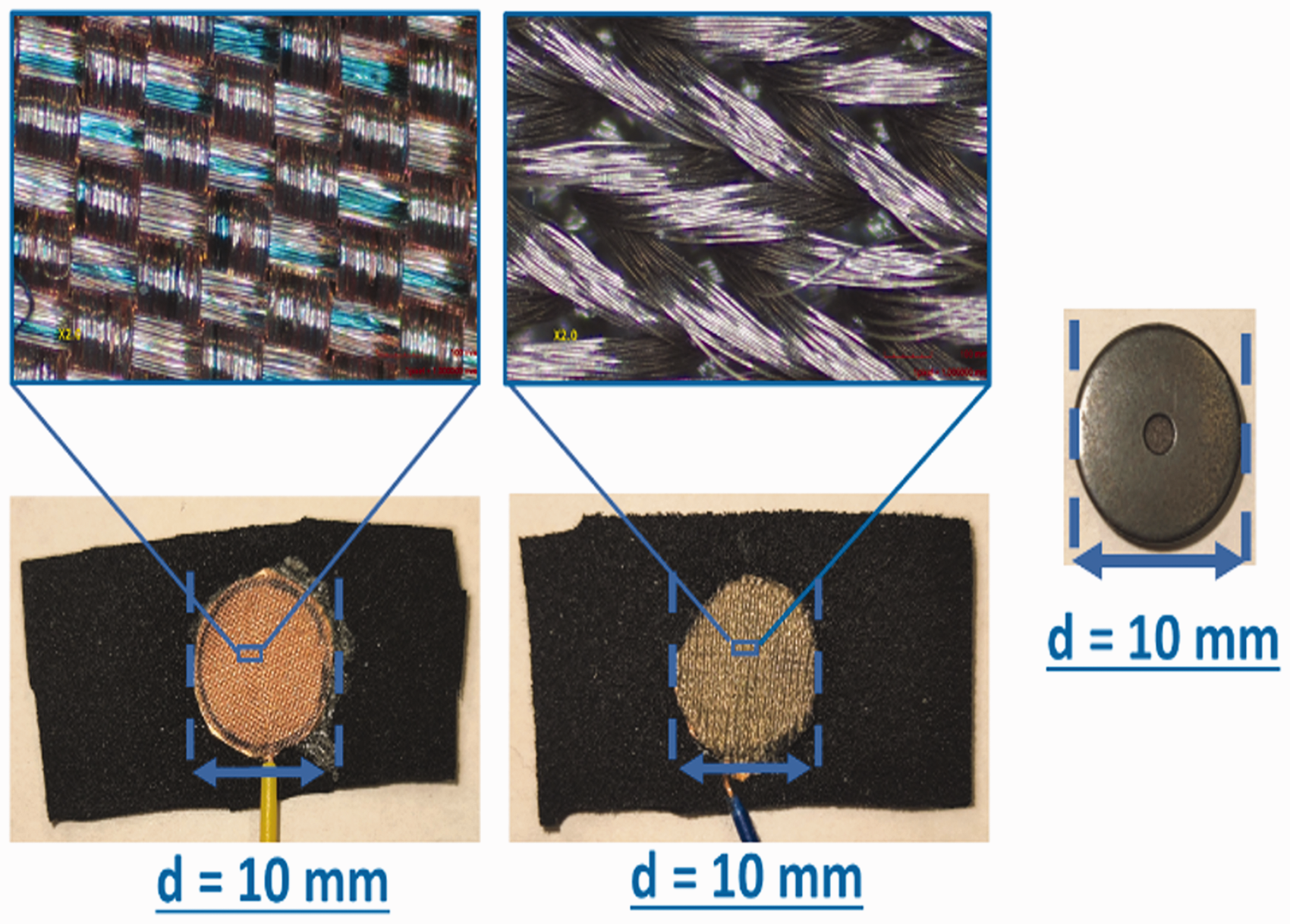
The electrodes were attached to the skin with a band throughout the measurement (see Figure 4); the impedance data were collected for 1 h. The measuring electrodes were placed at the FP1 and FP2 points according to the 10–20 EEG electrode placement system (see Figure 4). Starting from 0 min to 10 min, impedance was recorded every 2 min to observe the impedance changes closely. From 10 min onwards, impedance was measured and recorded five more times (after every 10 min) for up to 60 min.
Placement of electrodes according to 10–20 system for impedance measurement at points fronto-polar 1 (FP1) and fronto-polar 2 (FP2) for impedance measurement, A1 for driven ground leg pin of the circuit, and A2 for reference. 18
Results and discussion
The recorded impedance measurement was analyzed by using IBM SPSS software version 27. The impedance variation can be observed from the rate of change of impedance over time. The rate of change equation (1) of impedance can be given as follows:
Impedance 1 is the impedance value for the previous point of time, impedance 2 is the impedance value for the current point of time, time 1 is the previous point of time, and time 2 is the current point of time. For example, equation (1) calculates the rate of change of impedance in dry skin for commercially available dry electrodes at 10 min = (32.75–183.04) kΩ/(10–0) min = –6.55 kΩ/min. For dry skin conditions, impedance showed a decreasing pattern for all three types of electrodes. In the case of tap water applied as an electrolyte, all the electrodes showed an increment impedance pattern. For saline water as the electrolyte, the impedance rate of change decreased; after a period, it became stable. Figures 5, 6, and 7 show the rate of change in impedance in the graphs as ‘rate of change in impedance (kΩ/min)’ versus ‘time (min)’ for three different electrodes: dry skin conditions, tap water applied as an electrolyte on the skin, and saline water used as an electrolyte on the skin. From these figures, we can have a closer observation of how different electrodes become stable in terms of impedance fluctuation for an hour-long measurement in different skin conditions.
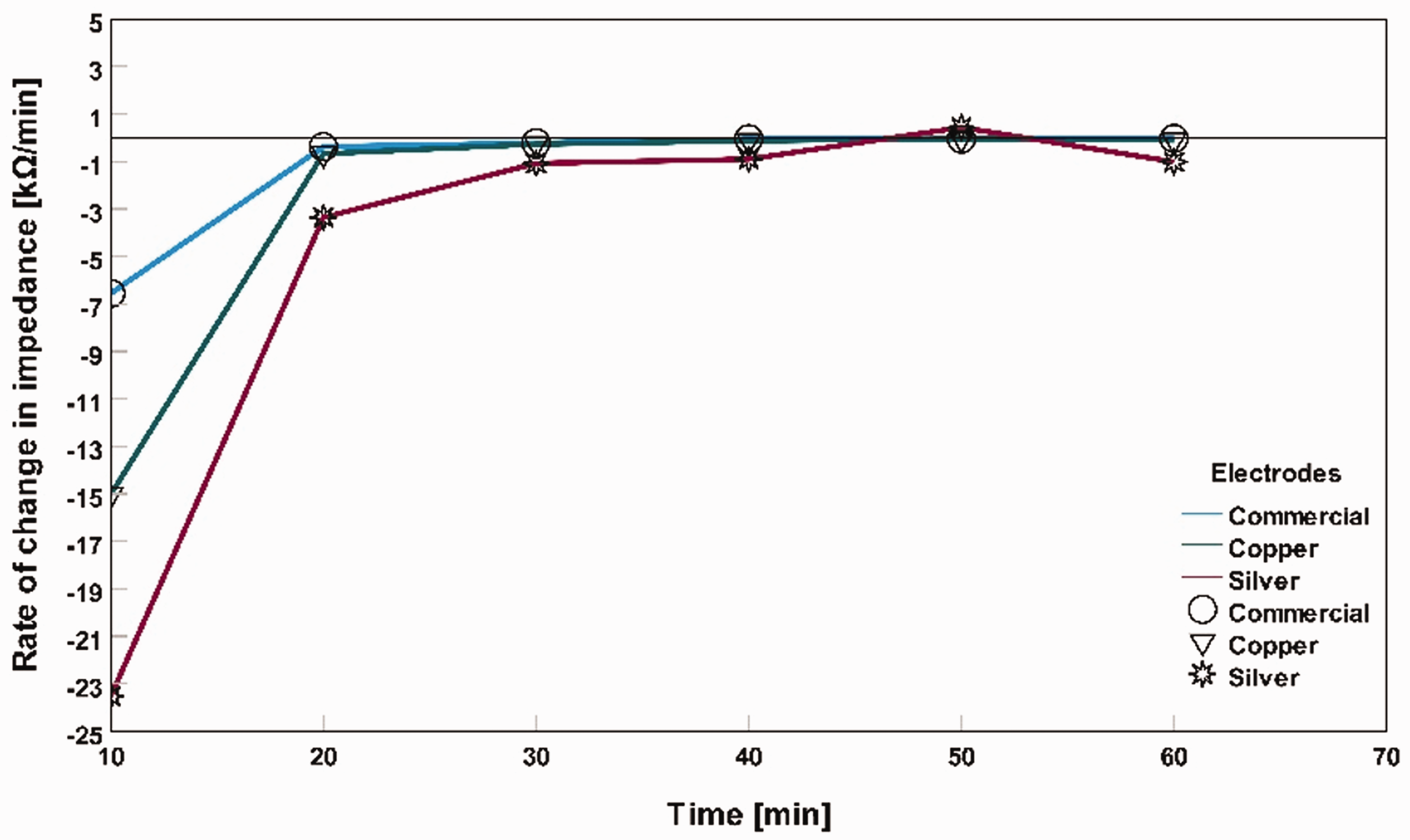
Rate of change in impedance on dry skin for different electrodes.
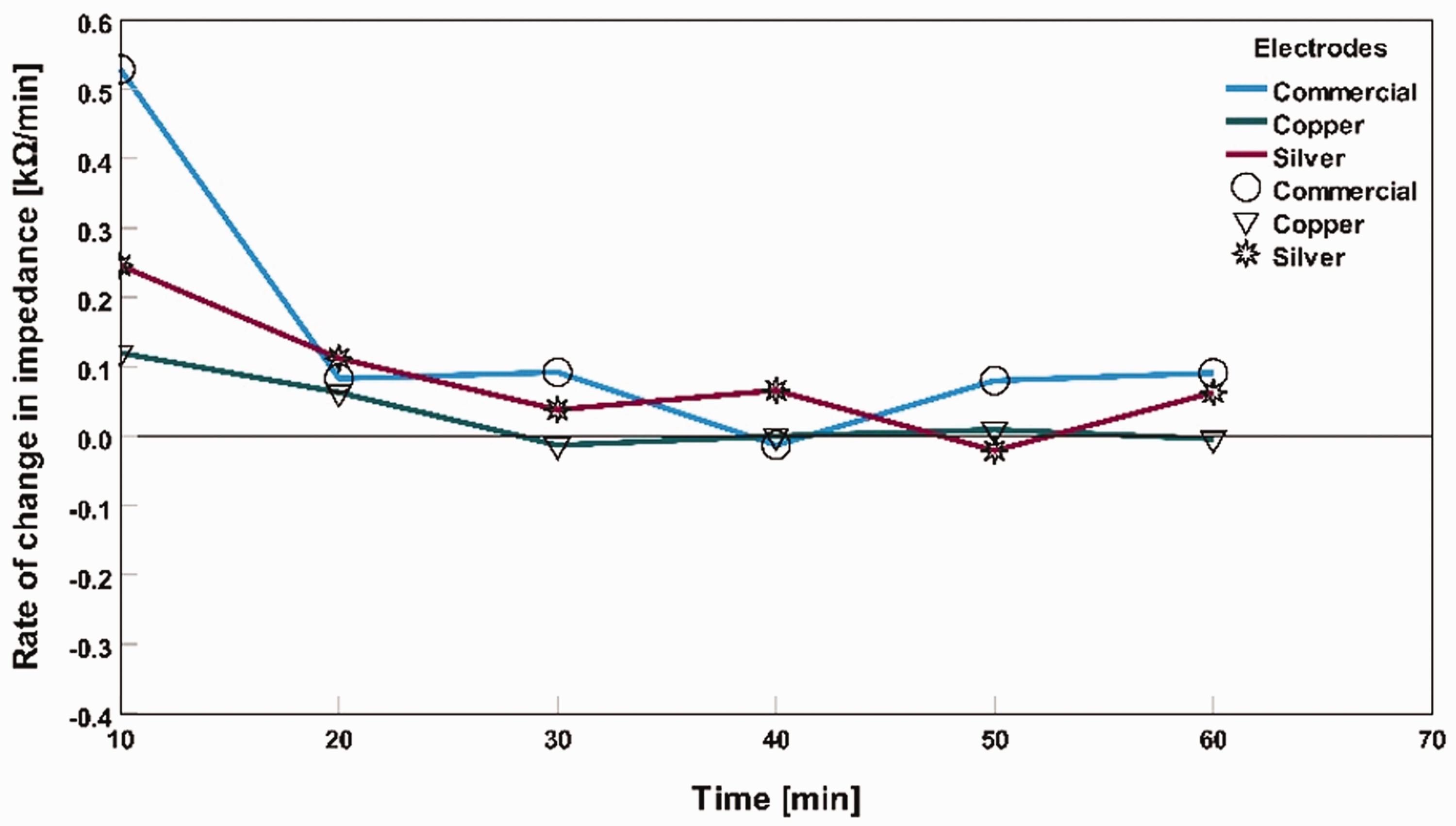
Rate of change in impedance with tap water as an electrolyte for different electrodes.
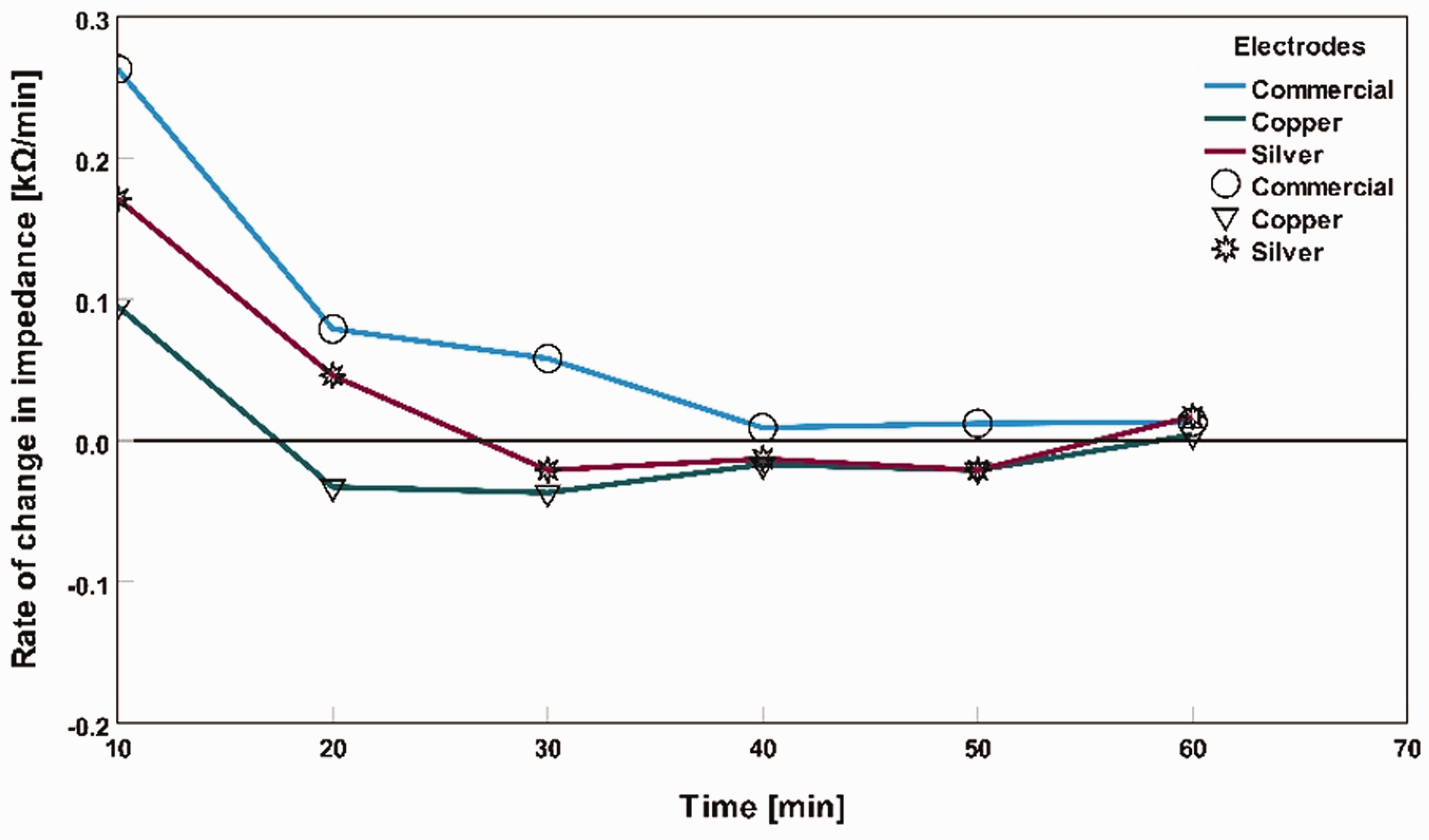
Rate of change in impedance with saline water as an electrolyte for different electrodes.
The recorded impedance measurement was analyzed by forming averages of all the participants’ measured impedance values for each point of time and plotting them in graphs at that point as ‘impedance (kΩ)’ versus ‘time (min)’ in Figures 8, 9 and 10. The error bars in graphs in Figures 8, 9 and 10 present the mean values’ standard error (SE). Error bars are formed by a 95% confidence interval (CI).
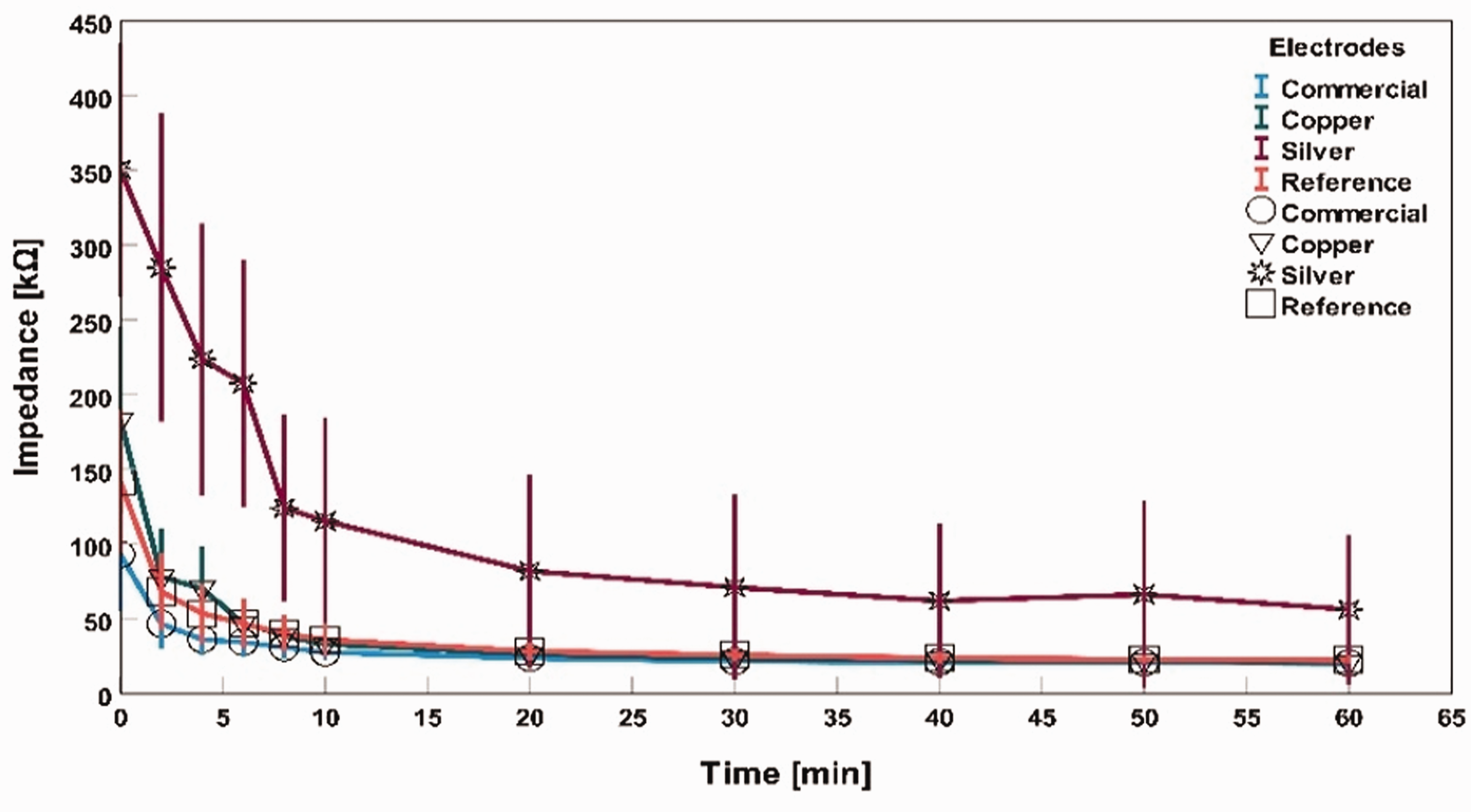
Impedance measurement without skin cleaning for different electrodes.
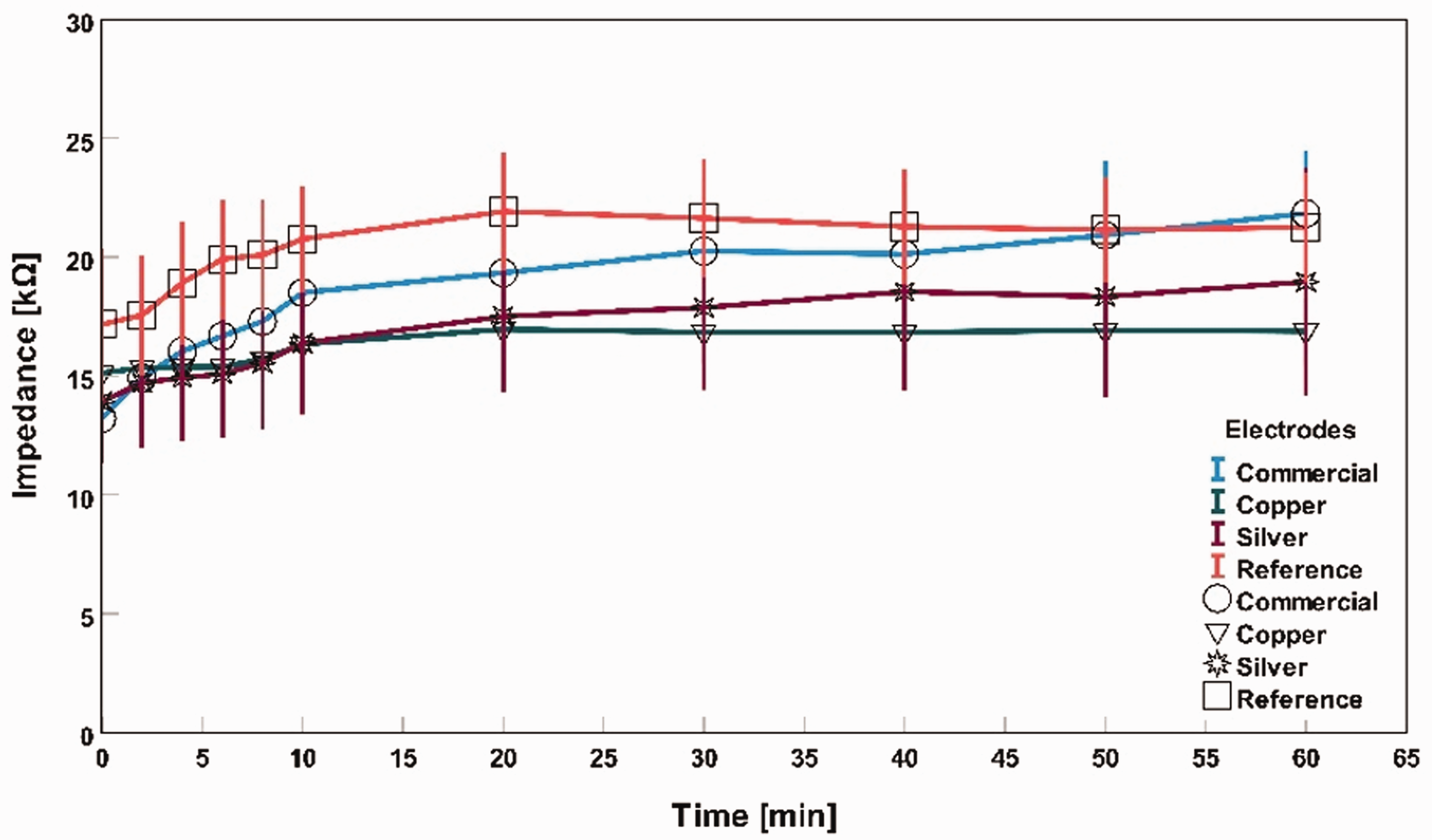
Impedance measurement with tap water as an electrolyte for different electrodes.
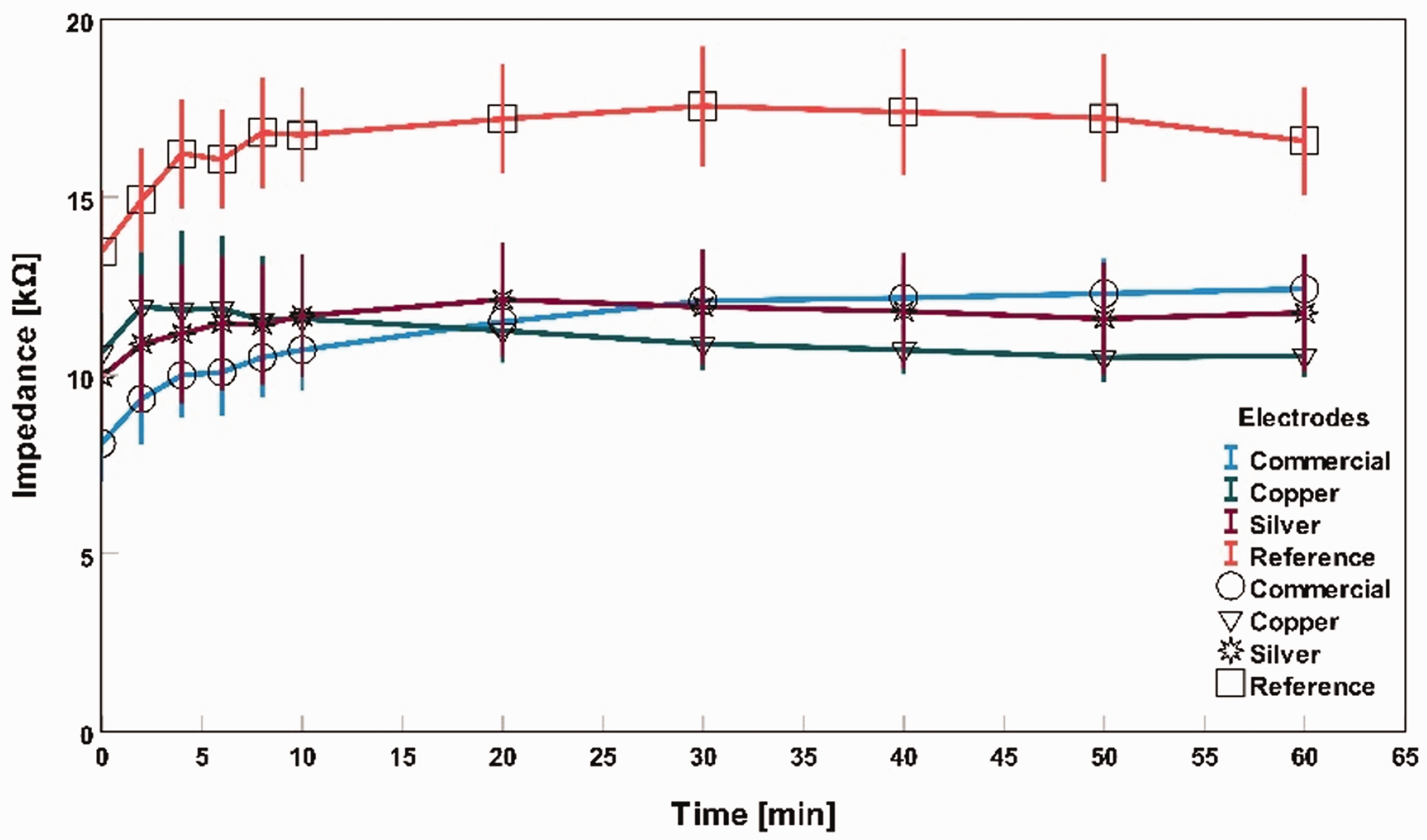
Impedance measurement with saline water as an electrolyte for different electrodes.
In Figure 8, all impedances were initially remarkably high for dry electrodes without any skin preparation. However, with time, the impedance started decreasing. For example, at 0 min, the average impedance of commercial, silver textile-based electrodes, and copper textile-based electrodes was 93 kΩ, 350 kΩ, and 183 kΩ, respectively. After 60 min, the average impedance became 20 kΩ, 56 kΩ, and 21 kΩ, respectively. The main reason for the skin impedance decrease is natural skin hydration, which acts as an electrolyte between the skin contact point and the electrode for this dry skin condition.
After applying tap water as an electrolyte between the dry electrode and the skin, the impedance values of all three types of electrodes were quite proximate because water particles decreased the skin’s impedance at the measurement points.
Figure 9 shows that all three electrodes of the commercial, silver-based textile electrode, and copper-based textile electrode impedance at 0 min were 13 kΩ, 14 kΩ, and 15 kΩ, respectively. When the water particle dried out over time, high impedance was observed for all three types of electrodes. After 60 min, the average impedance became 22 kΩ, 19 kΩ, and 17 kΩ for the commercial electrode, silver textile-based, and copper-based textile electrodes, respectively. Copper-based textile electrodes had a more stable impedance level over time than commercial and silver-based textile electrodes.
Saline water was used as an electrolyte between the skin and electrodes, and these electrolytes decreased the skin’s contact impedance at the measuring point for all three types of electrodes. A stable impedance level was observed throughout the hour-long measurement in Figure 10. Compared with commercial and silver-based textile electrodes, copper-based electrodes had the lowest impedance after 60 min. At 0 min, the average impedance of commercial, silver textile-based electrodes, and copper textile-based electrodes was 8 kΩ, 10 kΩ, and 11 kΩ, respectively. After 60 min, the average impedance became 12 kΩ, 12 kΩ, and 11 kΩ, respectively.
Figure 11 illustrates the variance in impedance for various skin conditions. When tap or saline water was used as the electrolyte, the difference between the commercial electrode, silver electro textile electrodes, and copper electro textile electrodes was close. Figure 11 also shows that all three materials exhibited significant impedance decrement when electrolytes were applied to dry skin over time.
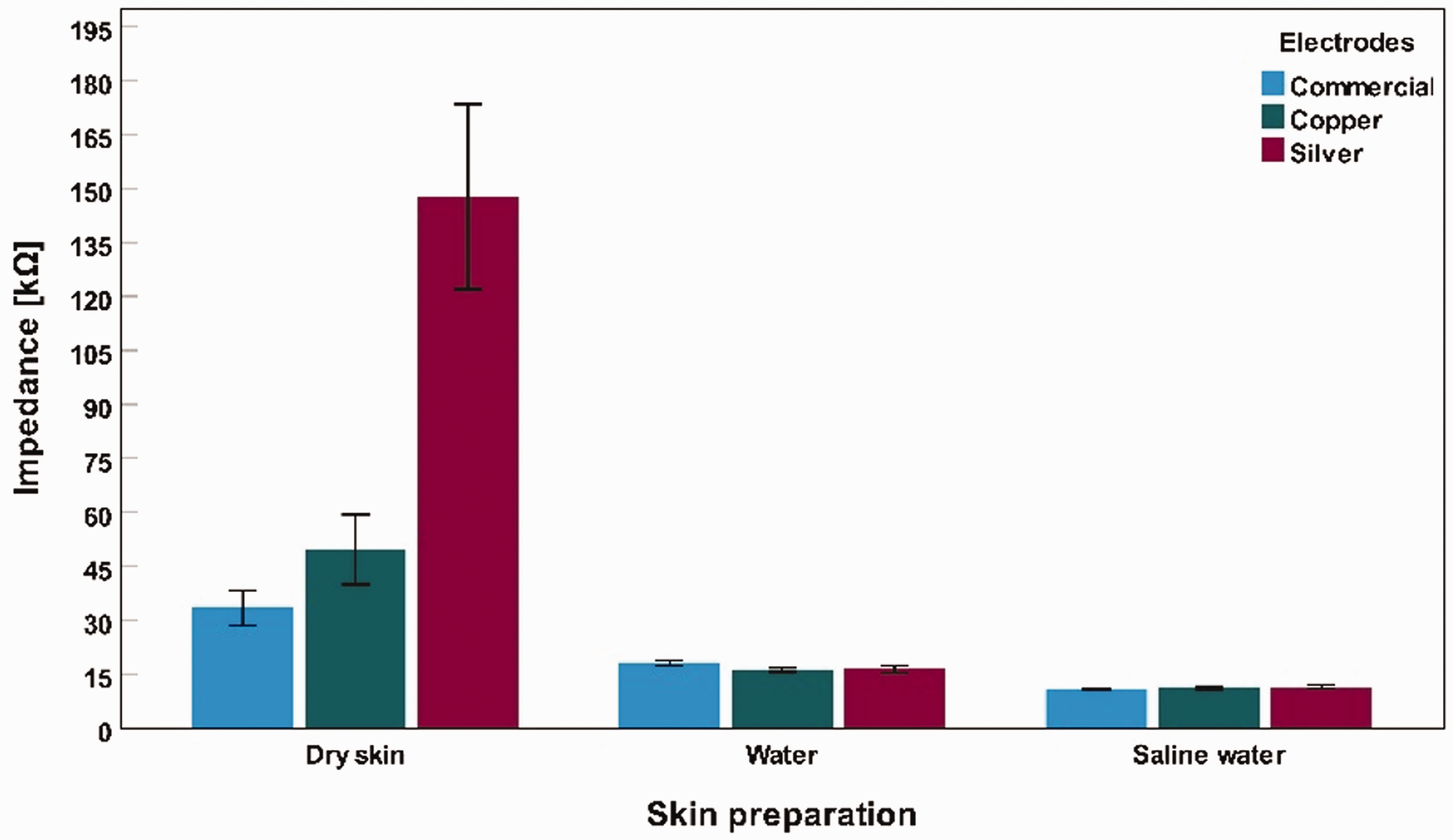
Impedance measurement in all three conditions for three distinct types of electrodes. The bars show the participants’ and time points’ impedance mean values. The error bars indicate the standard deviations of the mean values.
We observed that skin impedance for all three types of electrodes was close to 10 kΩ contact impedance when saline water was applied as the electrolyte, aligning with the water-based electrode from the other research. 46 We also observed that saline water as an electrolyte showed a better response to reduce contact impedance than tap water as an electrolyte, which was like other BCI-based research. 49
The silver conductive fabric has microcapillaries between fiber filaments, which helps to hold or wick water; however, copper-based electro textile does not hold water particles because it is a woven fabric with single-strand copper wires. 41 The silver-based textile electrodes absorbed the water particles, indicating an impedance rise after 60 min when tap water was applied as an electrolyte. Conversely, the copper-based textile electrodes remained low in impedance after 60 min when the tap water was applied as an electrolyte. The silver textile-based electrode holds saline water particles, which lowers the contact impedance; the impedance range became close to the copper-based textile electrodes.
According to the results, the performance of all the electrodes is similar in the case of water and saline water. However, there are differences when the measurements are done on dry skin. In practical use, the best situation would be to use textile-based electrodes, especially on dry skin, as this would remove the need for any pretreatments. It can be concluded that the copper-based textile electrodes were more suitable than the silver-based textile electrodes and comparable to commercial electrodes regarding impedance in all three skin conditions. An important next step is to study the effects of sweating, for example, due to a higher temperature environment, on the performance of these textile-based electrodes. Also, it is important to study how these electrodes can be used in a looser setup; for example, when attached to a hat that is not holding the electrodes so strictly against the skin.
Figures 12, 13 and 14 show the EEG brain wave responses from three different electrodes in three different skin conditions. These graphs show that the textile-based electrodes can show similar brain waves to the commercial dry electrodes when they are measured with the same setup. We do need signal analysis to analyze the signal further and to compare the EEG signal quality of the textile-based electrodes and commercial dry electrodes. However, in this paper, we only studied the possibility of using a textile-based electrode to replace a commercial Ag/AgCl dry electrode for long-term EEG signal acquisition. Thus, we give these results as a preliminary example to show the potential of the developed electrodes in EEG measurements. The next direction of this research is actual EEG measurements.
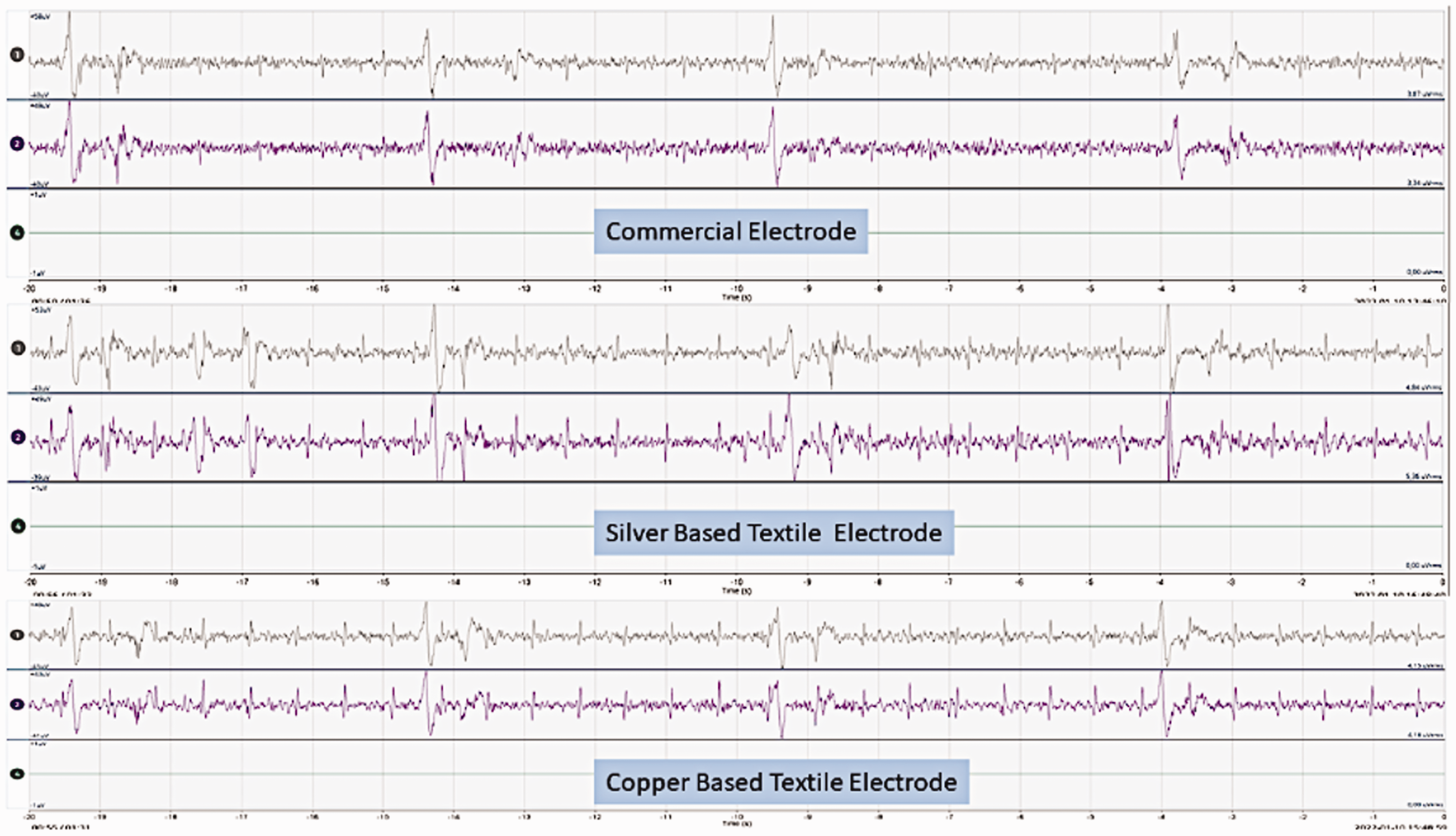
Electroencephalography (EEG) of three types of electrodes during dry skin conditions.
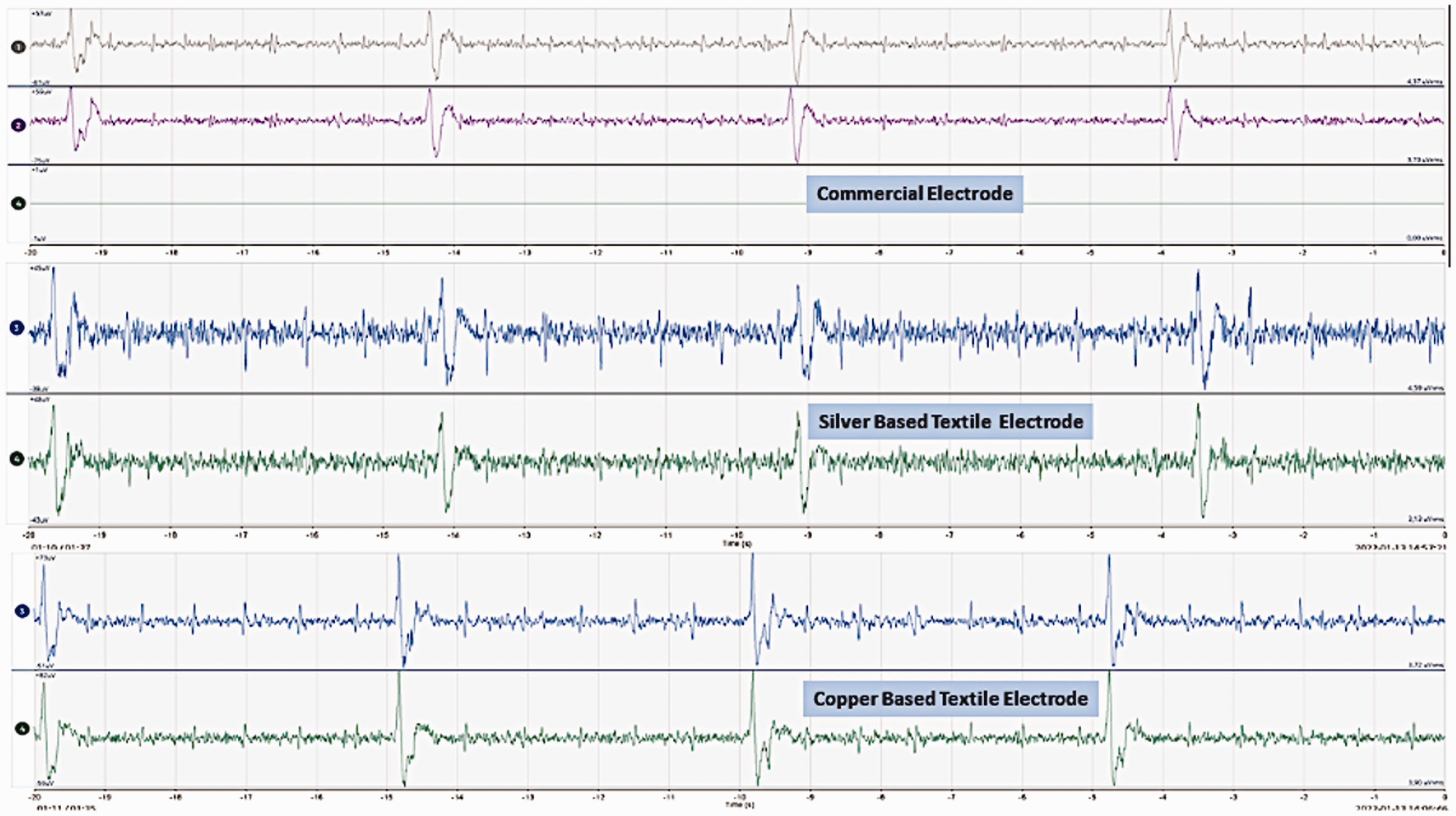
Electroencephalography (EEG) of three types of electrodes during tap water applied as electrolytes on the skin.
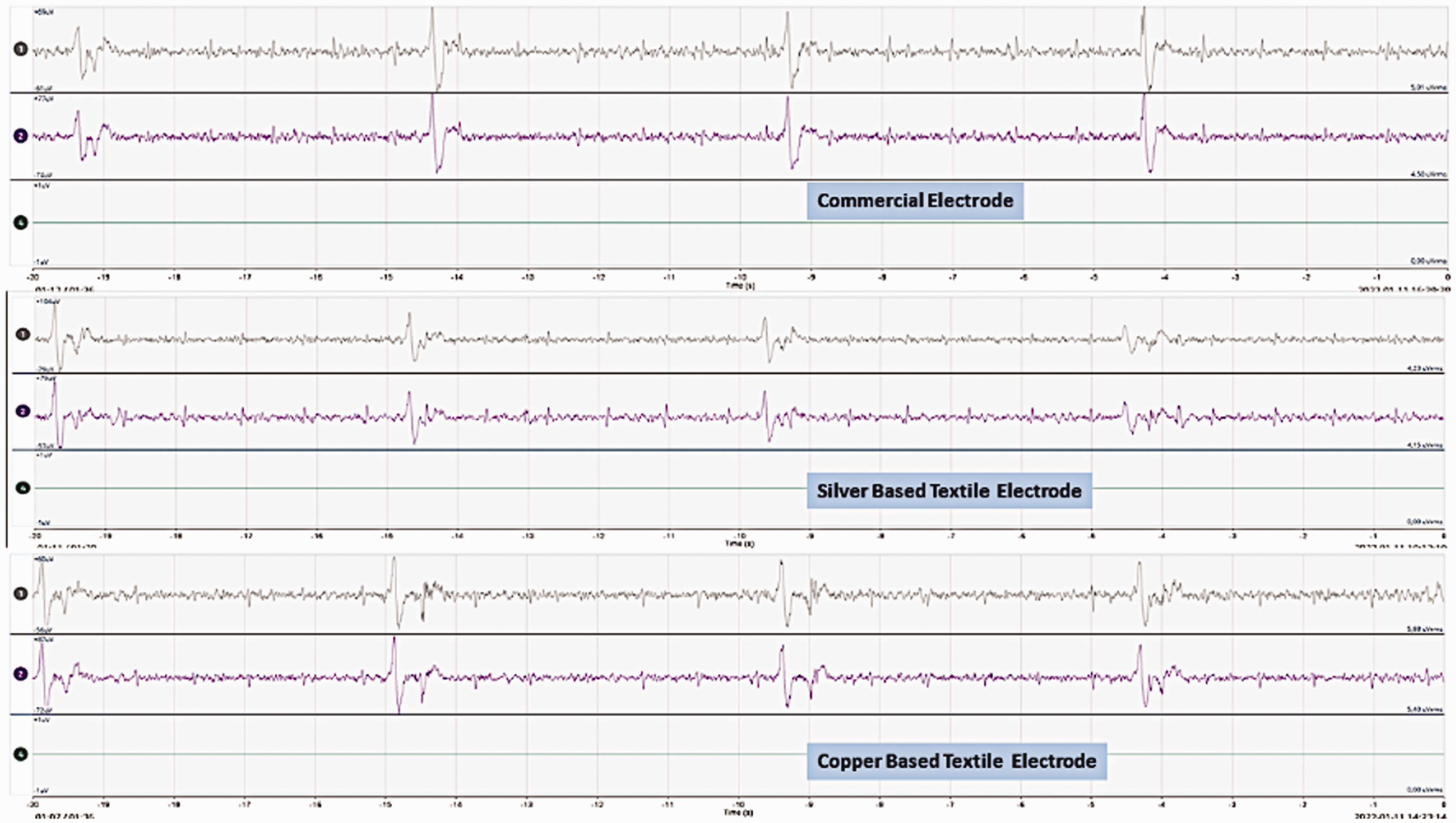
Electroencephalography (EEG) of three types of electrodes during saline water applied as electrolytes on the skin.
Conclusions
Textile-based electrodes are ideal for long-term health monitoring due to their comfort and adaptability to various conditions. Our study indicates that electrodes made from copper and silver-based electro textiles can be used in place of commercially available Ag/AgCl dry electrodes. All three types of electrodes’ skin impedance were tested for three skin conditions: dry skin, tap water applied as the electrolyte, and saline water applied as the electrolyte. Under dry skin conditions without skin preparation, copper textile-based electrode and commercial electrode skin impedance were 21 kΩ and 20 kΩ, respectively, which was almost the same level of impedance after decreasing from the initial impedance level. The skin impedance range at 0 min was 93 kΩ–351 kΩ and after 60 min it was 20–56 kΩ, for all three types of electrodes. Electrolytes of tap and saline water were applied to observe the effect and measure the skin impedance. When tap water was applied as an electrolyte, the skin contact impedance became less than 16 kΩ at 0 min; after 60 min, the impedance range was 17–22 kΩ. The copper textile electrode impedance was the lowest among all three electrodes after 60 min. Skin impedance was measured when saline water was applied to the skin at 0 min. The impedance range was 8–11 kΩ; after 60 min, the impedance range was 11–12 kΩ. When saline water was applied as the electrolyte, the copper-based textile electrode showed the lowest skin impedance of 11 kΩ among all three types of electrodes after 60 min. Therefore, although silver-based electro textile electrodes are unsuitable for dry skin because of their high skin impedance, they may be considered when the skin is electrolyzed with tap or saline water. Finally, the measured and recorded test results indicate that copper-based textiles show lower impedance values for all three skin conditions after a certain period than dry commercial and silver-based electro textile electrodes. Therefore, this copper-based textile electrode can be considered for long-term measurement considering skin impedance characteristics. Also, saline water is a better electrolyte than tap water. As for future work, electro textiles should be investigated further regarding data acquisition, durability, and signal quality to be more comparable with commercial dry electrodes.
ORCID iD
S M Musfequr Rahman https://orcid.org/0000-0002-7490-2344
Footnotes
Authors Note
The author Johanna Virkki is now affiliated with Faculty of Information Technology and Communication Sciences and Tampere Institute for Advanced Study, Tampere University, Finland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the European Regional Development Fund and the Ministry of Education and Culture, Finland.