Abstract
Healthcare associated infections (HAIs) are known as one of the major problems of the modern healthcare system, which result in additional cost and mortality. It has also been shown that pathogenic bacteria are mostly transferred via surfaces in healthcare settings. Therefore, antibacterial surfaces, which include fabrics and textiles, can be used in a healthcare environment to reduce the transfer of pathogenic bacteria, hence reducing HAIs. Silver nanoparticles have been shown to have broad spectrum antibacterial properties, and therefore they have been incorporated into fabrics to provide antibacterial functionality. Liquid flame spray (LFS) nanoparticle synthesis allows nanoparticles to be produced and deposited on surfaces at speeds up to and beyond 300 m/min. Herein, LFS is used to deposit silver nanoparticles onto two fabrics that are commonly used in the hospital environment with the aim of producing antibacterial fabrics. A thin plasma coating on top of the fabrics after silver deposition is used to improve nanoparticle adhesion. Fabrics coated with silver nanoparticles demonstrated antibacterial properties against Escherichia coli. Nanoparticle imaging and surface chemical characterization are performed using scanning electron microscopy and X-ray photoelectron spectroscopy.
The highlights of this research are as follows:
• high-speed synthesis and deposition of silver nanoparticles on fabrics;
• plasma coating onto fabrics with silver nanoparticles;
• antibacterial fabrics for potential use in healthcare environments.
Nanomaterials have been used in numerous applications over the years.1–3 In particular, silver nanoparticles have been of interest due to their broad spectrum antibacterial properties, which have been extensively documented. 4 As a result, they have been used in several consumer products. 5 Recently, roll-to-roll synthesis of nanoparticles has been demonstrated using liquid flame spray (LFS) technology.6–8 LFS is a flame pyrolysis technique that allows the simultaneous synthesis and deposition of noble metal and metal oxide nanoparticles in a continuous process. A precursor for the desired nanoparticles to be formed is injected into a high-temperature flame and the produced nanoparticles are deposited onto a substrate. This has been used to produce silver nanoparticles onto paper and glass surfaces.9–11 Furthermore, superhydrophobic paper boards have been produced using LFS.12,13 LFS produces no effluents in the nanoparticle production process and nanoparticle production speeds up to 300 m/min have been demonstrated. 14
Healthcare associated infections (HAIs) cause additional cost to patients as well as possible mortality. Antibacterial surfaces and fabrics can find numerous uses in the healthcare environment – from furniture and breathing devices to textiles for bedding, clothing and the surgery environment. The main objective herein is to minimize the transfer of harmful bacteria. For fabrics, many approaches have been proposed to achieve this.15–17 For example, a one-pot approach for producing antibacterial fabrics has been demonstrated, 18 where a fabric was immersed in a carboxymethyl chitosan solution containing silver using a pad-dry-cure process. Other methods include the use of various surface coatings, such as magnesium-based brucites, microwave assisted synthesis and immersion technique, among others.19–24 The incorporation of silver nanoparticles into fabrics for antibacterial properties has also been explored extensively.25–28
Plasma polymer coatings been used to impart functionalities such as superhydrophobicity and antistatic properties to fabrics.29–32 In addition, plasma coatings have been used as means of improving the adhesion of different materials to fabrics.33–37 Our previous work has demonstrated the antibacterial efficacy of LFS synthesized silver nanoparticles on paper and glass, and that the nanoparticle adhesion can be improved on glass surfaces with a plasma coating.9,10,38 In the current study, the LFS-generated antibacterial nanoparticles are deposited onto two fabrics that are commonly used in the hospital environment.
A thin plasma coating is used to improve the nanoparticle adhesion and antibacterial properties are determined.11,39 Scanning electron microscopy (SEM) is used for surface imaging and X-ray photoelectron spectroscopy (XPS) is used to determine the silver amount on the fabric surfaces.
Materials and methods
Silver deposition onto textiles
LFS was used for the deposition of silver nanoparticles onto the substrates. Figure 1 shows a schematic as well as an image of the experimental setup. A precursor containing silver nitrate (AgNO3, 99.9 + %, Alfa Aesar, DE) was injected into a flame through a nozzle to produce the nanoparticles. Hydrogen, oxygen and nitrogen with flow rates of 20/10/5 l/min were used as combustion gases. The precursor feed rate into the nozzle was 2 ml/min. Further details about the LFS deposition process are described in previous studies.40,41 Fabrics were attached to a carousel for the nanoparticle deposition. The nozzle was placed 20 cm away from the fabric surface. Passing the fabrics multiple times through the flame resulted in the deposition of different amounts of silver onto the fabric surface.
Schematics of the liquid flame spray setup used to deposit silver nanoparticles onto the fabrics.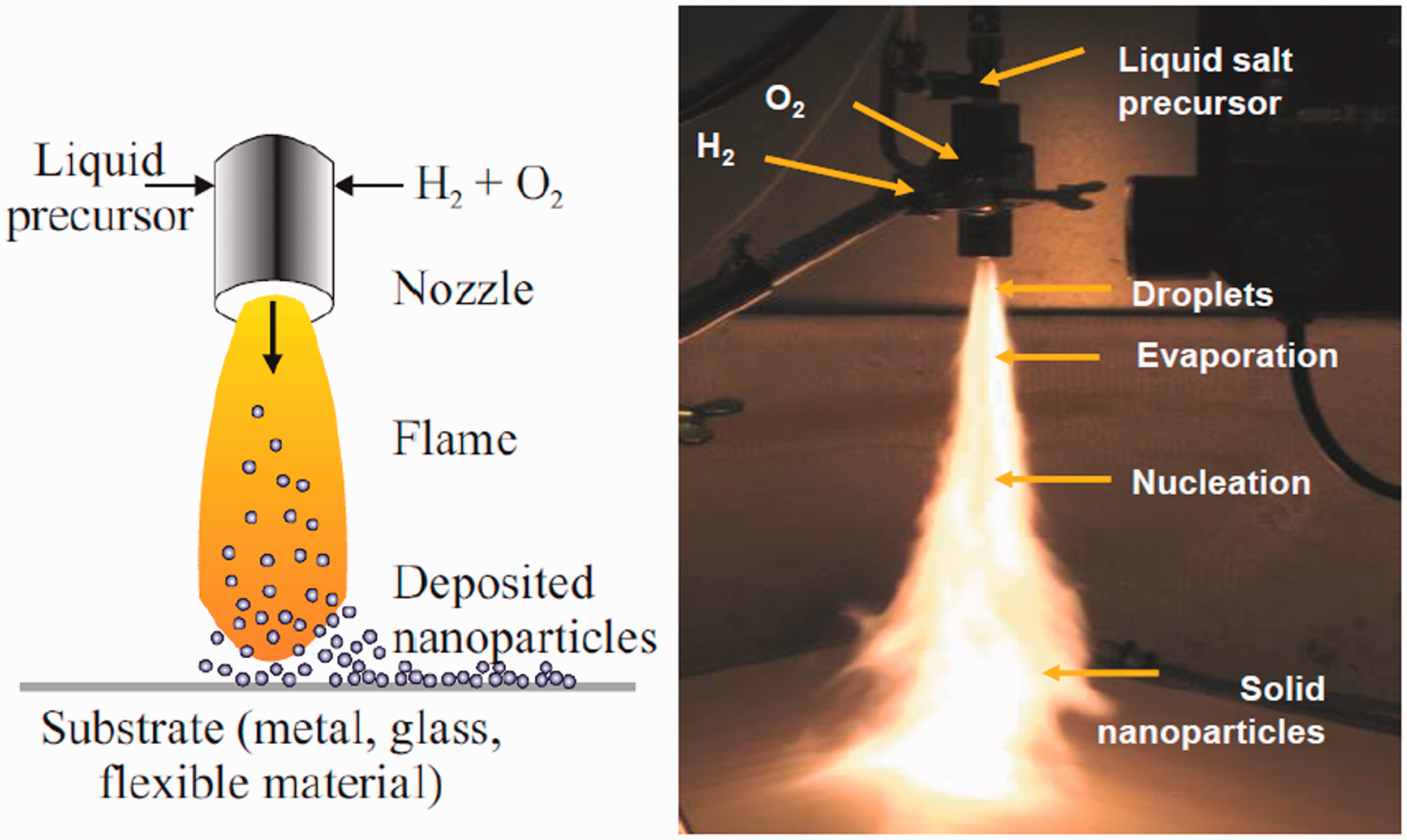
Plasma coating of textiles
A custom-built reactor made of a glass vessel attached to a double stage rotary pump was used for plasma deposition at RISE, Sweden. A 13.5 Hz radio frequency generator was connected to two copper bands that were wrapped around the glass vessel. Samples were wrapped around a perforated metal plate that was rotating in the chamber to allow maximum deposition. Pressure in the vacuum chamber was pumped below 10 mTorr before the coating begun. Hexamethyldisiloxane (HMDSO, > 98.5%, Fluka Steinheim, DE) was used as the coating polymer. 42 The two coating times used were 40 and 120 seconds, corresponding to two coating thicknesses.
Surface imaging of the coated fabrics
SEM was used to visualize the nanoparticles on the fabrics using a LEO Gemini 450 (FE-SEM, Carl Zeiss, DE). The samples were sputtered with a thin layer of platinum before imaging. An accelerating voltage of about 2.0 kV was used at a maximum magnification of up to 63 kX and a working distance of about 3.2 mm.
Surface chemical analysis using XPS
The chemical composition of sample surfaces was determined using a PHI Quantum 2000 XPS (Physical Electronics Instruments, US). Semi-quantitative elemental compositions was measured in relative atomic percentages. The samples were exposed to a monoenergetic Al source (50 W and 15 kV) through a 200 μm aperture with a pass energy of 187.85 eV. An XPS spectrum was obtained in the analysis software, and the chemical constituents were identified using the observed peak positions in the measured spectra. 43 The analysis software used was Multipack, and a reference carbon at a peak position of 285.0 eV was used for analysis. The elemental composition was obtained from three separate measurement locations on each sample.
Antibacterial testing
A previously developed ‘Touch’ test method was used to determine the antibacterial properties of the coated surfaces. 38 In the measurement procedure, bacteria culture was diluted in 0.9% NaCl. The resulting bacteria suspension was equivalent to 0.5 McFarland standard, approximately 1.5 × 108 colony-forming units (CFUs) per milliliter (CFU/ml). The sample surface was then inoculated with 50 μl of the new bacterial suspension, and incubated at room temperature for 24 h in an empty Petri dish. After incubation, the sample surface is pressed against a blood agar plate for 30 s and then incubated again at +37℃. The number of CFUs was determined on the next day. Gram-negative Escherichia coli (E. coli, ATCC 25922) and gram-positive Staphylococcus aureus (S aureus, ATCC 29213) were used as test organisms in the antibacterial tests.
Results
Silver nanoparticle deposition and plasma coating
Using the LFS deposition process, silver nanoparticles were deposited onto two fabrics, fabrics I and II. Both fabrics are commonly used in hospital environments, that is, fabric I is used as bedding covers (FOV Fabrics AB, SE), while fabric II is used by medical professionals as gowns in the operating room (One Med, ref 2311). Multiple passes of the sample through the flame results in a corresponding increase in the number of deposited nanoparticles. Two different nanoparticle amounts were deposited onto each fabric, corresponding to 10 and 20 passes, referred to as 10X and 20X in the following. Both samples experienced a slight discoloration of the surface after silver deposition. The color change was a result of nanoparticles on the surface. However, the color change in fabric I was not obvious due to its original dark green color. The deposited nanoparticles can cause unwanted color change in the surface of different fabrics. Therefore, it is important to consider this when choosing the substrate for nanoparticle deposition.
Fabric samples coated with only silver and with both silver and plasma are shown in Figure 2. A plasma polymer coating was deposited onto the nanoparticle coated fabrics. HMDSO was selected for the plasma coatings due to its hydrophobicity, low toxicity and transparent optical properties with good chemical and electrical stability.44–47 Two different plasma coating thicknesses were produced based on the deposition time: 40 s for a thickness of about 53 ± 5 nm and 120 s for 90 ± 3 nm. The plasma coating thicknesses were measured from atomic force microscopy (AFM) step height measurements on glass. The different thicknesses were specifically chosen in order to observe the effect of an increasing plasma layer on the antibacterial properties. However, the main objective of the plasma coating was to improve the nanoparticle adhesion, as demonstrated in our previous study on glass where contact mode AFM measurement was used to scratch the surface of samples to observe the loss of silver nanoparticles in a particular location.
11
The aesthetic properties of the samples were slightly altered, especially when the plasma coating thickness increased. It was clearly seen from the images that fabric I was a woven fabric. Fabric II appeared to be a mixture of different fibers, some of which were natural and others synthetic fibers.
Images of the fabrics showing the uncoated reference (Ref) and 10 times (10X) nanoparticle coated fabrics. Plasma coated samples are labeled with the number of times they are coated in the flame and the plasma coating times (10X_120s).
SEM analysis
Figure 3 shows SEM images of fabric surfaces with deposited silver nanoparticles. The nanoparticles appear to be spherical and uniformly distributed on the surfaces. For 10X samples, 40% of the sample area is covered with nanoparticles, whereas 42% area coverage is recorded for 20X samples. However, the area coverage may be dependent of the point of measurement, since more nanoparticles are expected around the center of the flame profile on the fabric. The average particle size is approximately 32 nm. After plasma coating the nanoparticles appeared to be a few nanometers larger, depending on the thickness of the plasma coating.
Silver nanoparticles deposited on to the fabrics before plasma coating (10X). Fabric I is shown in (a) and (b), and fabric II is shown in (c) and (d).
SEM images obtained from the cross-section of fabric I are shown in Figure 4. Cross-sections of the coated fabrics were obtained using broad ion beam cutting. Individual strands can be observed in the cross-section for fabric I. However, some melting of the sample, which is made from polyester, is evident due to heat generation during ion milling. The woven nature of fabric I allowed some pores to exist on the sample surface. SEM investigation of these pores suggest that nanoparticles had penetrated between some strands of the woven polyester fabric. However, closer analysis of the images showed that the penetration of nanoparticles was typically only beyond the top layer of strands. Thus, nanoparticles settled on the strands below the surface only when there was a direct line of sight. When there were no pores between the strands, no penetration of nanoparticles was observed. Similarly, nanoparticles settled on top of fabric II, and penetration was observed only when there was a direct path for nanoparticles to travel past the top layer fibers.
Scanning electron microscopy images from the cross-section showing silver nanoparticle penetration between fabric fibers. (a) and (b) Nanoparticles settling in a pore below the top layer when there was a direct line of sight. No nanoparticles are observed on the strands below the surface when there were no pores in the surface, as shown in (c) and (d).
XPS surface chemical analysis
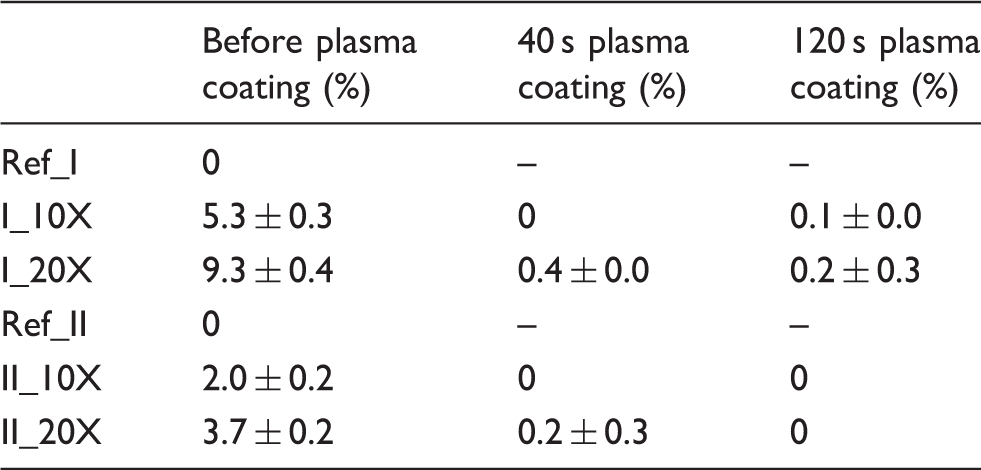
After nanoparticle deposition onto fabrics, surface chemical composition was determined. XPS results show that the main elemental composition consisted of carbon, oxygen, fluorine, sulfur, chlorine, silicon and silver. Carbon and oxygen were observed due to the chemical composition of the fabrics. Our previous studies of LFS nanoparticle deposition on paperboard has shown a carbonaceous layer on top of the nanoparticles. 48 This layer originates mainly from the volatile organic compounds, which are released from the paperboard substrate by the heat of the flame during the nanoparticle depositio. 49 The measured fluorine was about 35 ± 1% in fabric II and less than 1% in fabric I. Fluorochemical finishes have been used in the textile industry to impart water repellency, oil repellency and soil repellency. 50 Sulfur is used as a dye in the production of colorful textiles, whereas chlorine is sometimes used as a bleaching agent in textile production.51,52 Silicon was observed only after plasma coating as a result of the used HMDSO.
Silver amounts measured on the surface of fabrics in atomic percentage (%) and the corresponding standard deviations
Antibacterial properties
The newly developed ‘Touch’ test method that simulates the transfer of bacteria from one surface to another by touch was used for determining the antibacterial efficiency of the fabrics. Figure 5 shows the antibacterial results for both fabrics I and II obtained from three repeated measurements. Bacterial growth is evident in the reference samples that had no silver nanoparticles nor plasma coating. The growth of E. coli was inhibited in all the samples that had deposited silver nanoparticles. This was also the case even for samples with plasma coating on top of the nanoparticles. Since the antibacterial properties observed here are attributed to the release of silver, the plasma coating is either non-conformal, that is, it does not cover fully all the nanoparticles, or silver is released through the plasma coating or possible cracks in it. For gram-positive S. aureus, the coatings did not demonstrate antibacterial properties even for 20X coatings without any plasma coating on top. This is expected, as our previous results have confirmed that higher amounts of silver, beyond 20X coatings with LFS, are needed to create significant antibacterial properties against S. Aureus.
38
Plasma coatings are indicated by ‘40s’ and ‘120s’ in the sample labels.
Antibacterial results for fabrics I and II after 24 h incubation at room temperature. Bacterial growth in the graph is given as 1, 2 and 3, corresponding to 103–104 CFU, 104–105 CFU and >105 CFU, respectively. ‘Ref’ represents reference samples with no silver or plasma.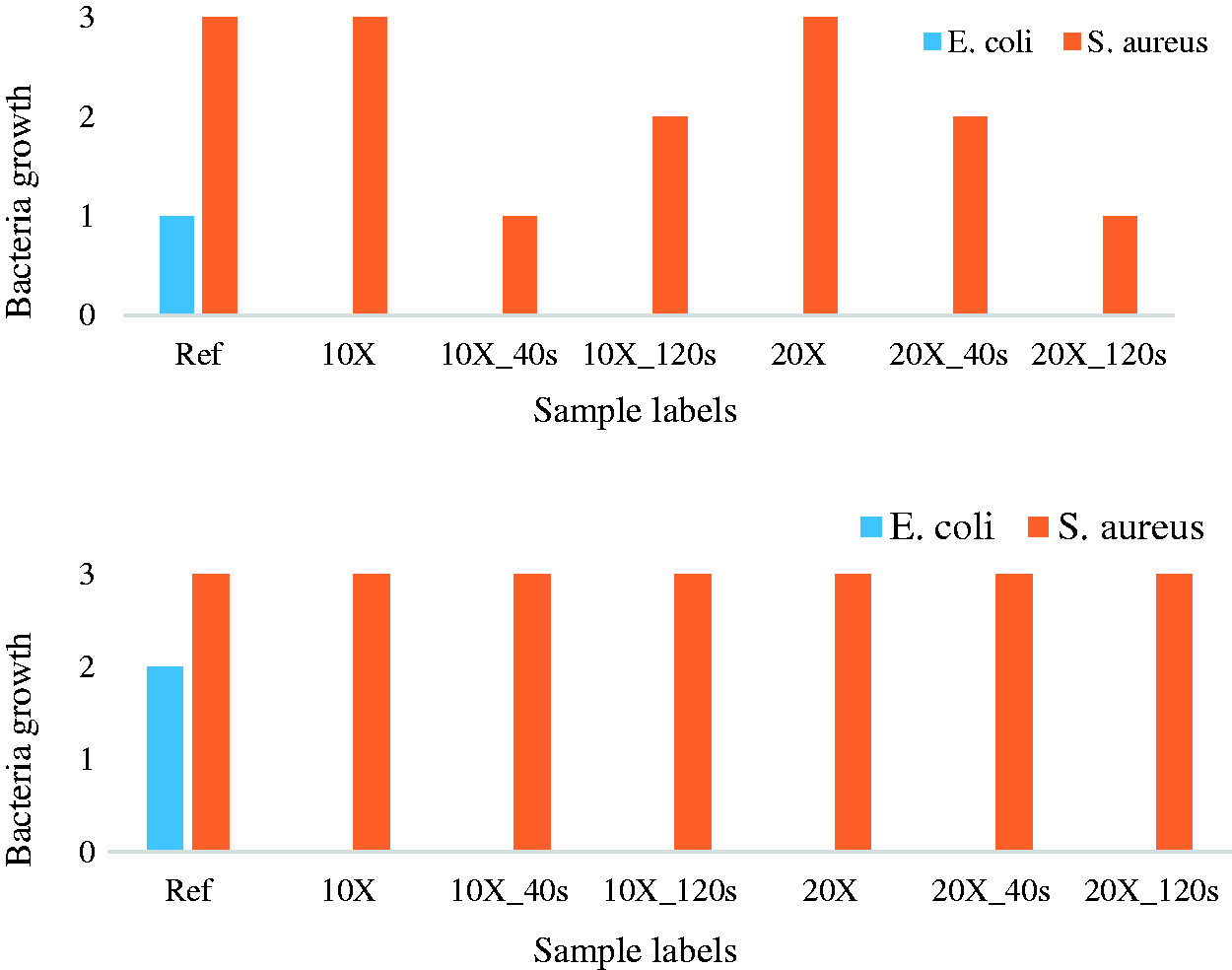
Conclusions
Fabrics and textiles that possess antimicrobial properties have been of interest over the years. Different substances have been used to produce these antimicrobial fabrics, including nanoparticles. In this study, silver nanoparticles are incorporated into the surface of two different fabrics that are used in the hospital environment. An aerosol deposition technique, which allows simultaneous production and deposition of nanoparticles onto different substrates, is used. The nanoparticle synthesis technique used is capable of roll-to-roll processing and can be scaled for industrial use. A plasma polymer layer is deposited on top of the samples after silver deposition to improve nanoparticle adhesion. The current work used vacuum plasma, but similar deposition is possible with existing roll-to-roll atmospheric plasma technology. The results show that antibacterial properties are obtained against E. coli in spite of the thin plasma coating on top. Antibacterial properties are attributed to the release of silver, but the exact mechanism requires further study. On the other hand, S. aureus was resistant to the coatings. Antibacterial properties were not observed for samples that had only silver nanoparticles on top. Previous studies, however, confirm that S. aureus is only susceptible at a high silver amount.
In summary, this study demonstrates a continuous high-speed process for producing antibacterial fabrics that can be used in different scenarios, including in a hospital environment. The exact mechanism for silver release form the fabrics as well as testing of these fabrics in a real-life environment are recommended as part of future studies. The proposed LFS process can also be used for disposable, single-use antimicrobial fabrics (non-wovens). Furthermore, washing durability in the case of intended reuse of the produced antibacterial fabrics needs further investigation. These issues will be addressed in a future communication.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academy of Finland under the project ‘Nanostructured large-area antibacterial surfaces (nLABS, Grant No. 275 475)’. JJS wishes to thank the UEF Faculty of Science and Forestry (Grant No. 579/2017) for financial support.
