Abstract
Zinc oxide is a critical industrial material with extensive applications in galvanization, ceramics, electronics, and renewable energy systems. Despite China’s vast zinc reserves (41 million tons), domestic production of primary zinc oxide from concentrates remains limited to 20%, with recycled materials contributing merely 11%. As a result, there is a heavy reliance on imported resources. This imbalance underscores the urgent need to optimize the utilization of secondary resources and adopt sustainable technologies. This review systematically examines recent advancements in zinc oxide smelting, enrichment, analytical characterization, and strategies for a circular economy. Modern hydrometallurgical techniques, such as high-temperature acid leaching and ammonia-ammonium carbonate systems, have achieved over 95% zinc recovery from low-grade ores. Innovations in pyrometallurgy, including microwave-assisted reduction and rotary kiln volatilization, have reduced energy consumption by approximately 30% while minimizing emissions. Advanced detection methods, such as X-ray fluorescence spectroscopy and combustion furnace-ion chromatography, enable precise monitoring of toxic elements, including lead, cadmium, and arsenic. Furthermore, circular economy approaches-such as slag geopolymerization and nano zinc oxide synthesis from industrial by-products demonstrate significant potential for waste valorization. By integrating interdisciplinary technologies, such as machine learning and biohydrometallurgy, this review outlines a potential roadmap toward a sustainable zinc industry, balancing economic growth with environmental responsibility.
1. Introduction
1.1. Presence of the zinc-containing materials
Chemical composition and typical zinc content of major zinc ores.
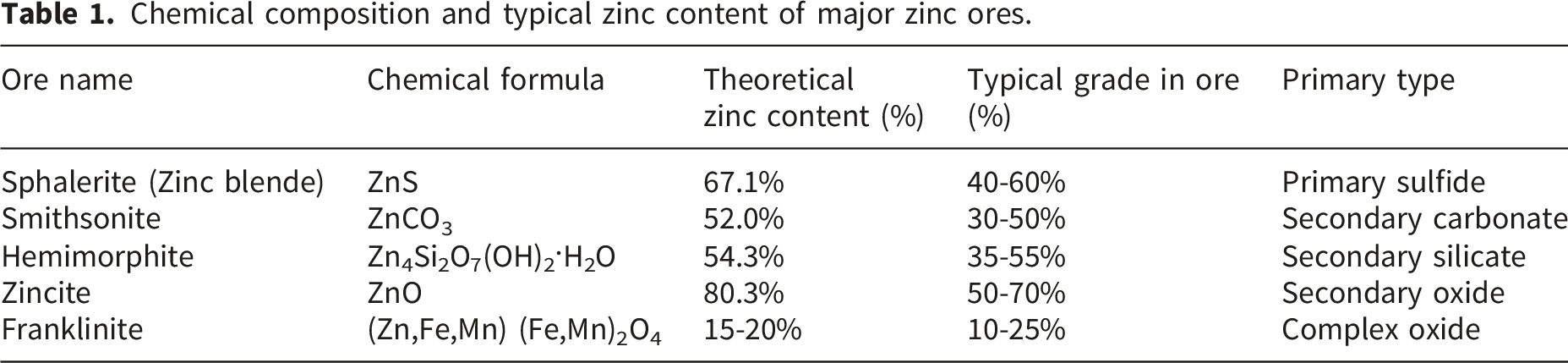
Zinc-containing materials, which are important non-ferrous metal raw materials, hold great potential for application in various industrial sectors. For example, zinc can form alloys with major metals, with brass being a commonly used alloy. Brass finds widespread applications in mechanical manufacturing, national defense, and printing and dyeing. Due to its excellent corrosion resistance, galvanization has become an effective method for protecting steel plates, representing one of the largest uses of zinc-containing materials, accounting for 48.1% of total consumption. Additionally, zinc is often used as an alternative material for making precision castings. 7 Zinc’s consumption is dominated by its use as a protective coating for steel. Galvanizing accounts for approximately 60% of the total global refined zinc consumption, underscoring its critical role in infrastructure and automotive industries. 8 This dominance also directly influences the types and volumes of zinc-containing scrap and residues generated, such as galvanizing dross and end-of-life galvanized steel.
1.2. Classification of zinc-containing materials
Sources and composition of zinc-containing industrial/secondary resources. 11

Meanwhile, the aggregate collected after the volatilization of zinc-containing materials treated by the fire method is enriched zinc oxide. In this enrichment process, the zinc-containing materials are mixed with anthracite in a certain proportion and fed into a rotary kiln at 1100 °C. The zinc compounds are reduced to elemental zinc, which is then oxidized to zinc oxide at the kiln’s outlet. The resulting zinc oxide is collected after cooling. 12 Solid wastes such as electric furnace steelmaking soot, steelmaking blast furnace soot, and electrogalvanizing slag, along with low-grade zinc ores, can be directly used as zinc oxide-enriched feedstock in the zinc smelting process. Compared with other primary ores, these materials are cost-effective and cause minimal environmental pollution. Therefore, their use helps mitigate the adverse environmental impacts of traditional zinc mining and promotes the efficient and safe utilization of waste, reducing over-reliance on primary mineral resources. 13
Overall, the rational utilization of zinc-containing materials is conducive to maintaining the clean production of the material smelting process, reducing the environmental pollution from the smelting industry at its source. It also holds great significance for the construction of an eco-friendly and resource-intensive industrial chain.
2. Smelting technology of zinc oxide materials
The rapid growth of today’s socio-economic landscape has led to a substantial increase in the demand for resources, particularly non-ferrous metals. Zinc concentrate, which contains a large number of metals, serves as a valuable resource, including various precious metals. In the zinc smelting process, it is crucial to both maximize zinc production and ensure the comprehensive utilization of associated non-ferrous metals. 14 This dual approach can improve the economic benefits of production enterprises and strengthen their competitiveness within similar industries. Additionally, it is imperative to reduce the emission of heavy metal pollutants to safeguard the environment. The leaching process, which is central to the hydrometallurgical recovery of zinc, could be quantitatively described by kinetic models. The leaching rate of zinc from secondary sources, such as secondary zinc oxide, using sulfuric acid often follows the equation: -ln (1-x) = k·tn. Where x is the zinc leaching efficiency at time t, k is the apparent rate constant. Studies have shown that the leaching of zinc oxide under certain conditions is controlled by diffusion through a product layer, with an apparent activation energy as low as 5.18 kJ/mol. Furthermore, the influence of acid concentration can be accounted for by the macroscopic kinetic equation: -ln (1-x) = A·exp (-Ea/RT)·cm·tn, where c is the initial acid concentration and m, n is the reaction order with respect to acid.15,16 The thermal decomposition of zinc carbonate (ZnCO3) or zinc hydroxide (Zn(OH)2) precursors is a fundamental step in producing high-purity ZnO. The overall decomposition reaction can be represented as follows: ZnCO3(s) → ZnO(s) + CO2(g); Zn(OH)2(s) → ZnO(s) + H2O(g); The kinetics of this solid-state decomposition can be complex. 17 The decomposition rate and the resulting ZnO properties (specific surface area) are highly dependent on the temperature and time profile. For instance, achieving a high yield (>95% decomposition) and high-activity ZnO (specific surface area >40 m2/g) requires precise control, such as maintaining a temperature of 300 °C for 240-300 minutes. 18 In pyrometallurgical routes, the chemical reactions governing the processes are well-established. The Indirect (French) Process involves the direct oxidation of vaporized metallic zinc: 2Zn(g) + O2(g) → 2ZnO(s), The Direct (American) Process, which utilizes zinc-containing ores or concentrates, involves a series of reactions including reduction and subsequent oxidation: ZnO(s) + CO(g) → Zn(g) + CO2(g); 2Zn(g) + O2(g) → 2ZnO(s). 19
Beyond primary production from ores, a substantial and increasingly important portion of zinc supply is obtained as a secondary product from various recycling streams and industrial processes. 20 The steel and iron industry is a major source of secondary zinc, particularly from the recycling of galvanized steel scrap. During the electric arc furnace (EAF) steelmaking process, zinc volatilizes and is captured in the baghouse as EAF dust, which typically contains 15-25% zinc and represents the most significant source of secondary zinc, accounting for a large portion of the zinc-bearing residues processed annually. 21 Similarly, the waste oxidizing sintering (WOS) process, specifically designed to treat EAF dust, is a primary route for recovering zinc. Other significant sources include: hot-dip galvanizing industries, which generate zinc-rich dross and ashes, contributing substantially to the secondary zinc stream. Copper and lead smelting, where zinc reports to flue dusts and slags due to its volatile nature, and could be concentrated to levels suitable for recovery. End-of-life product recycling, such as the processing of brass scrap and zinc-based alloys. Globally, it is estimated that secondary zinc production accounts for approximately 15-20% of the total refined zinc supply, underscoring its critical role in the circular economy of this metal. 22 Therefore, the efficient processing of these zinc-containing secondary materials is not only economically attractive but also essential for minimizing waste and reducing the environmental footprint of zinc production.
Traditional zinc oxide smelting methods primarily cover electric furnace zinc smelting and closed blast furnace zinc smelting. 23 The electric furnace process typically yields products with higher impurity levels and requires substantial consumption of water, electricity, and other energy sources, resulting in limited overall recovery efficiency. The closed blast furnace process is suitable for smelting lead-zinc mixed ores; although it enables the recovery of both lead and zinc, it causes serious environmental pollution. Typically, pyrometallurgical zinc smelting processes are associated with considerable smoke and dust emissions, high energy consumption, and low metal recovery rate. 24 Alternate strategies should be explored to address the significant losses of associated metals during smelting, as well as the ineffective enrichment of low-content elements and poor economic returns. Several novel technologies for the separation and recovery of valuable metals are outlined.
2.1. High-temperature and high-acid leaching
High-temperature and high-acid leaching can serve as an effective way to recover valuable metals. Initially, neutral leaching slag is obtained by treating zinc-containing raw materials in a neutral liquid. The neutral slag is then further processed under high-temperature and high-acid conditions, the rare metal extraction slag can be obtained through low-acid treatment. 16 While pyrometallurgy is associated with high gaseous emissions (e.g., SO2 and CO2), The high-pressure acid leaching shifts the environmental burden towards liquid waste generation and high energy consumption. Such as the high capital cost for corrosion-resistant autoclaves versus off-gas treatment systems in smelters. Under optimal conditions, the leaching rates of indium, zinc, thallium, cadmium, and cobalt can exceed 90%. However, this process is energy-intensive due to the high temperatures required, and the use of strong acids poses environmental risks, such as the release of acid mist, generation of heavy-metal-laden wastewater, and accumulation of hazardous solid residues.25–27 To mitigate these issues, recent research has shifted focus beyond acid neutralization and discharge toward closed-loop recycling strategies. For instance, regenerating and recycling leaching agents through solvent extraction or membrane separation technologies can significantly reduce chemical consumption. Simultaneously, resource recovery of neutralization residues-such as iron sulfate or gypsum-by repurposing them as additives in cement production represents a key pathway toward zero waste. Furthermore, optimizing steam utilization in autoclaves through energy integration serves as an effective mitigation strategy to lower operational costs and enhance environmental benefits. The detailed analysis of reference highlights that the formation of a Fe3O4-enriched layer can significantly impede matte/slag separation. 28 This phenomenon not only leads to direct metal losses (economic drawback) but also necessitates higher operating temperatures or longer settling times to overcome, which in turn increases energy consumption and associated greenhouse gas emissions (environmental drawback). Therefore, optimizing interfacial conditions is not merely a technical issue but a critical lever for balancing the economic viability and environmental footprint of the smelting process. Therefore, appropriate mitigation measures-such as acid recovery, effluent treatment, and energy-efficient reactor design are essential to minimize its environmental footprint.
2.2. One slag and two liquids in three paths
The term slag refers to the high-leaching residue produced by the entire leaching system, which contains a large concentration of valuable metals. The two liquids typically include a neutral liquid and a rare-metal-rich liquid. The neutral leaching solution primarily contains zinc, cadmium, and nickel, along with trace amounts of arsenic, copper, and cobalt, which can be transferred into the replacement solution through a replacement process and subsequently recycled in the replacement slag.29,30 Iron present in the extracted residue can be effectively removed via the hematite method, yielding iron-rich red coatings with fewer impurities that are suitable for secondary usage. However, this multi-step process, particularly the management of two separate liquid streams and the operation of the hematite method, is inherently water-intensive and contributes to higher overall energy consumption. 31 To address these challenges, future optimization efforts could explore the integration of water recycling systems and the adoption of energy-efficient technologies, such as advanced thermal management or process intensification techniques. These innovations aim to reduce the environmental footprint while maintaining the benefits of producing high-purity products. Further research is recommended to quantify the water and energy usage of this process and evaluate the feasibility of these proposed solutions.32,33
2.3. Cyclic cumulative enrichment of rare metals
Upon completion of the whole extraction process (solution pH = 5.1-5.3), thallium, antimony, and indium present in the extraction solution dissolve in water to form the hydroxide precipitates, which then enter the neutral leaching slag. This slag is subjected to high-temperature and high-acid treatment. During this stage, the M(OH)n precipitates formed in the early stage are redissolved into the solution. Simultaneously, undissolved metal ions in the zinc-containing materials also enter the leaching liquid. Therefore, rare metals undergo cumulative enrichment through repeated cycles with the continuous addition of fresh materials. 34 Finally, once the enrichment concentration of different metal ions reaches a recoverable threshold, zinc sulfite can be introduced into either a high-temperature, high-acid leaching solution or a neutral solution. Fe3+ may be reduced to Fe2+ following the dissolution of Fe from the neutral leaching solution. This process yields a residue enriched with rare metals and containing fewer impurities. This rare metal-enriched residue is then leached with a weak acid, resulting in a metal-rich solution containing rare metals that can be more efficiently recycled in later stages.
3. Type of zinc oxide enrichment
3.1. Zinc oxide enrichment of zinc leaching slag
Zinc oxide is typically produced through a wet smelting process, which involves the extraction method of converting zinc sulfide into zinc oxide, a compound that is easily dissolved by dilute sulfuric acid.
35
Conventional extraction methods include acid leaching, neutral leaching, and high-temperature acid leaching (Figure 1). After undergoing two stages of acid and neutral leaching, the selection of leaching conditions-primarily temperature or acidity facilitates the dissolution of zinc from baked zinc sand. The zinc content in leaching slag that remains undissolved is around 21%, mainly covering undissolved zinc sulfide, zinc ferrite, a small amount of insoluble zinc phosphate, zinc oxide, and zinc silicate. To recover the remaining zinc from the leaching slag, the rotary kiln fuming method is usually employed, where in the leaching slag is mixed with coking coal powder and subjected to sintering. During this process, lead, zinc, and cadmium are volatilized. The resulting flue gas contains not only zinc oxide powder but also other volatile heavy metals, such as lead and cadmium, posing a significant risk of air pollution if not properly controlled.36,37 Therefore, beyond the cooling and settling steps, efficient air pollution control devices (electrostatic precipitators or bag filters) are essential to capture these fine, heavy metal-containing particulates before the gas is released into the atmosphere.
38
Furthermore, the process generates wastewater that requires appropriate treatment to remove residual heavy metals and other contaminants to meet regulatory standards before discharge or reuse.39–41 The recovered zinc oxide powder can ultimately be collected for further usage (Figure 2). Conventional wet smelting and extraction process of zinc-containing materials.
36
Process of treating leaching slag by rotary kiln.
40


3.2. Zinc oxide enrichment from lead smoke furnace slag
Lead is rarely found as a single-metal deposit in nature and is often accompanied by zinc ores. The pyrometallurgical method is usually employed for lead smelting, with the blast roasting furnace being central to the lead smelting process.42,43 It was not until the 1990s that direct fire lead smelting integrated both traditional sintering roasting and reduction smelting processes within a single unit (Figure 3). The resulting slag typically contains 3%-20% zinc and 0.5%-5% lead, which is generally treated by pyrometallurgical equipment, such as a smoke chemical furnace. The primary approach in this treatment remains reduction and volatilization (Figure 4). It is important to note that the furnace gas generated during this pyrometallurgical treatment contains pollutants such as sulfur dioxide (SO2) and volatile heavy metal dust (lead, cadmium).
44
Consequently, this gas must be treated with advanced air pollution control devices (scrubbers, electrostatic precipitators) before release to mitigate atmospheric pollution. Similarly, the process water (or “pool water”) used in gas cooling and scrubbing systems can become contaminated with dissolved heavy metals and must undergo appropriate wastewater treatment (neutralization, precipitation) prior to discharge or recycling.
45
The integration of these treatment steps is essential for an environmentally sustainable operation. Roasting-reduction melting process using fire-based process. Schematic representation of the pyrochemical process of a smoke chemical furnace.
44


3.3. Zinc oxide enrichment from electric furnace ash residue
The steelmaking process in industrial electric furnaces usually relies on electric energy as the primary driving force. Arc heating is employed to smelt various steel raw materials. The smelting process is generally divided into three stages: melting, oxidation, and reduction (Figure 5). The smoke generated during electric furnace steelmaking is referred to as electric furnace ash. During the iron and steel regeneration process, zinc from the galvanized layer of iron and steel volatilizes in gaseous form and is subsequently collected as the flue gas passes through the dust collection system.
46
The resulting dust can be used in different applications depending on the type of zinc source. The principle of the rotary kiln used to control dust in steel mills is similar to that utilized in the treatment of leaching slag from zinc-containing raw materials via the wet method. This process also facilitates the reduction and volatilization of zinc compounds in the raw material, leading to their enrichment in the secondary soot.
47
Steelmaking process of electric furnace ash and slag.
3.4. Zinc oxide enrichment in gas ash (mud) slag
In the smelting process of a blast furnace, iron ore, coke, and solvent are constantly added to the top of the furnace. Simultaneously, a specified amount of hot air (1000-1300 °C) is introduced into the air inlet at the base of the furnace. Coal, oil, or gas is then introduced, initiating a reduction reaction in the iron ore to form molten iron, which is then discharged through the iron outlet. In addition, the coke and gangue from the iron ore, along with the ash from the spray, undergo sintering with limestone in the furnace, forming slag, which is then collected from the slag discharge (Figure 6). Blast furnace iron-making dust (also known as gas ash or gas mud) is regarded one of the main solid wastes generated by the steel industry. At the high temperatures in the blast furnace, metals such as zinc and lead existing in the iron ore are reduced to form steam. The dry, fine-grained dust produced by the ore and other solvents in the high-temperature zone is classified as gas mud.
48
With the continued growth in steel consumption in recent years, the output of gas mud has also increased, leading to a greater accumulation of valuable metals in it. Gas mud can be effectively treated using a rotary kiln, enabling the recovery and enrichment of high-purity zinc oxide. Flow chart of blast furnace smelting process.
Traditional smelting methods, such as electric furnace and closed blast furnace processes, often exhibit higher environmental burdens. For instance, mercury emission factors for the Imperial Smelting Process (ISP) can reach as high as 122±122 g Hg/t Zn, whereas advanced hydrometallurgical processes equipped with mercury removal devices can reduce this factor to approximately 5.7 g Hg/t Zn. 49 Furthermore, modern techniques incorporating comprehensive off-gas treatment (e.g., double-contact sulfuric acid plants and bag filters) can lower particulate matter emissions to below 10 mg/m3, which is significantly cleaner than uncontrolled traditional methods. In terms of resource efficiency, traditional pyrometallurgical routes often suffer from lower recovery rates. For example, the slag from traditional blast furnaces can contain as much as 8-9% zinc, leading to significant metal loss. In contrast, advanced treatment processes, such as the oxygen-enriched side-blown furnace smelting combined with fuming furnace technology, can efficiently process zinc leaching residues. This approach not only recovers valuable by-products like indium and silver but also reduces the final slag zinc content to very low levels, thereby significantly enhancing overall resource efficiency and reducing the volume of hazardous waste requiring disposal. 50
4. Analytical detection of zinc oxide enrichments
4.1. Inductively coupled plasma emission spectroscopy
Zinc-containing materials are often treated with crude zinc oxide. However, owing to technological limitations, a large amount of harmful elements such as lead, cadmium, and arsenic are inevitably mixed in the process. During the crude smelting of zinc oxide, the excessive presence of these toxic elements not only slows down the smelting process but also causes severe environmental pollution. Current methods for detecting metal content in by-products include titration, spectrophotometry, and atomic absorption spectroscopy. Recently, inductively coupled plasma (ICP) emission spectroscopy has gained widespread attention as a reliable technique for detecting metal content. Song et al. 51 established a method for accurately quantifying lead, cadmium, chromium, and arsenic in crude zinc oxide. Following sample dissolution, the dissolution analysis lines were examined, and the concentrations of the harmful metal elements were corrected accordingly. The ICP detection method is simple to operate, with a low detection limit and high precision, making it extensively used for the detection of lead, cadmium, arsenic, and other elements in zinc oxide.
4.2. Ethylenediaminetetraacetic acid detection method
Low-grade zinc oxide ore, smelting mortar, blast furnace gas mud, zinc-containing leaching slag, and other zinc oxide-enriched materials can be obtained through high-temperature volatilization, oxidation, and cooling using a rotary kiln or a smoke chemical furnace. However, due to variations in raw material sources and processing technologies, it is difficult to completely exclude the production of metallic zinc during the enrichment process. The presence of metallic zinc leads to the generation of a small amount of hydrogen during the leaching of the zinc oxide enrichment, which directly poses a significant environmental threat. To address this issue, the ethylenediaminetetraacetic acid (EDTA) titration method can be used to determine the elemental zinc content. Monocresol orange served as the indicator, while a sodium acetate-acetic acid buffer solution was used. The enrichment solution was then calibrated with EDTA. 52 Firstly, the content of water-soluble zinc and the ammonium chloride-water chloride system was measured without the atomic absorption spectral background. Subsequently, the zinc oxide content was determined. This method of calibrating zinc content in zinc materials provides a valuable reference for the comprehensive utilization of zinc materials. 53
4.3. X-ray fluorescence spectroscopy detection method
In the rotary kiln, the zinc-containing materials can be converted into crude zinc oxide. As a raw material, zinc-containing material is suitable for the production of zinc oxide. Its component content is typically assessed using semi-quantitative methods, such as chemical analysis or X-ray fluorescence (XRF) spectroscopy. Ytrebery et al. 54 developed a technique combining powder sheet pressing and XRF, which effectively determines the zinc content in zinc-enriched materials. The experimental results show that the zinc content determined by this method is consistent with the results obtained from the chemical method, with relative deviations ranging from 0.73% to 1.0%. This detection method provides rapid and reliable test results, making it a promising tool for monitoring the type and content of crude zinc oxide. Hijazi et al. 55 examined the zinc content in crude zinc oxide using XRF. First, two different groups of crude zinc oxide samples were selected, and the zinc oxide content was semi-quantitatively analyzed using XRF. By combining this with rietveld quantification, a precise calibration of the zinc content in crude zinc oxide was realized.
4.4. Integrated combustion furnace-ion chromatography detection method
Zinc materials are converted into zinc oxide through an enrichment process using a rotary kiln. Due to the complexity of the material source, the application of pyrometallurgical technology often leads to the co-enrichment of zinc oxide and halogens such as chlorine and fluorine. Halogens significantly influence the accumulation and purity of zinc oxide during thermal treatment. Specifically, halogens can react with zinc to form volatile compounds like zinc chloride (ZnCl2), which may evaporate before complete oxidation occurs, thereby reducing the yield of zinc oxide. Moreover, the presence of halogens can lead to the formation of secondary phases such as zinc hydroxychloride, which not only lowers the purity of the final product but also affects its crystalline structure and surface properties. Therefore, monitoring halogen levels is essential for optimizing process parameters and ensuring product quality. The detection of halogens in enriched products using combustion furnace-ion chromatography offers a reliable and efficient solution. As a novel analytical technique, it provides simple preparation, rapid operation, and accurate quantification. Importantly, it enables simultaneous monitoring of multiple halogens (e.g., fluorine, chlorine, bromine), demonstrating its broad detection capabilities. For instance, in an industrial zinc production facility, the implementation of this method allowed operators to detect elevated chlorine concentrations in the feed material early. By adjusting the roasting temperature and residence time accordingly, the plant reduced impurity levels by approximately 12% and improved zinc oxide recovery by 8%, leading to higher product purity and reduced energy consumption. This practical case illustrates how enhanced halogen monitoring can directly contribute to process efficiency and product quality. 56 Kone et al. 57 conducted extensive analyses and experimental investigations using this approach. Compared to conventional detection methods, their technique is more user-friendly and offers faster analysis. It realizes the synchronous detection of different halogens and dramatically reduces the detection cycle for zinc oxide enrichment. Based on their validations, a novel method for the rapid detection of halogens in enriched products using combustion furnace-ion chromatography was established.
4.5. Elemental analysis and detection method
Zinc sulfide, when processed in a rotary kiln, is converted into zinc oxide, which can be directly applied to ceramic raw materials or as a precursor for producing high-purity zinc.58–60 However, in the smelting process, organic carbon, sulfur-containing flue gases, and fuel toner may gather in the product, potentially compromising both the production process and the quality of ceramic materials. Fortunately, the Multi EA4000 Elemental Analyzer enables accurate detection of harmful elements such as carbon and sulfur in the product. This method offers rapid detection, high reliability, and a high degree of automation. Deng et al. 61 measured the total carbon and sulfur content in the enriched product and confirmed the method’s precision through their findings.
5. Comprehensive utilization of zinc oxide enrichments
5.1. Harmless treatment
In recent years, the continuous demand for zinc-containing materials has significantly outpaced the supply of zinc sulfide raw concentrates, resulting in a supply-demand imbalance. 62 In view of the possibility of zinc-containing materials being used as an alternative feedstock for zinc smelting, improving resource utilization and strengthening the disposal of smelting waste slag is critical. These issues have become significant obstacles to the sustainable development of enterprises. 21 Therefore, recycling zinc smelting slag and soot slag-produced in large quantities and currently underutilized-along with the optimization and improvement of existing processes could improve the resource recovery from zinc-containing smelting waste. In addition, this approach helps to avoid the adverse effects of impurities on zinc extraction during smelting.
Furthermore, during the iron and steel smelting process, zinc-containing raw materials accumulate on the furnace walls over time, altering the wall structure. This accumulation indirectly disrupts gas flow dynamics and can lead to considerable operational issues within the blast furnace. Therefore, increasing the safe and effective utilization of zinc-containing smelting slag from steel production is particularly critical. 63 Gao et al. 64 investigated the treatment of zinc-containing iron and steel smelting slag, considering the coexistence of multiple metals and proposing a comprehensive utilization scheme for such slag in China. They pointed out that microwave-ultrasonic intensive treatment could function as an effective method for utilizing iron and steel smelting slag.
5.2. Emerging treatment technologies for zinc-containing materials
Zinc-containing materials such as zinc sulfide, zinc ferric acid, and zinc silicate are challenging to dissolve simultaneously under alkaline conditions. This limitation highlights the inapplicability of the conventional ammonia-based zinc extraction process for these materials. In order to solve this problem, it is urgent to explore clean and efficient treatment technologies as a viable approach for effectively using these zinc-containing materials. 65 Recently, the unique and uniform heating capabilities of microwaves, along with their broad applicability, have led to their widespread use in metal recycling processes. 66
Besides, during the microwave reduction of metallurgical materials, the significant difference in the absorption characteristics between valuable minerals and impurities plays a critical role. The uniform heating provided by microwave ensures a consistent temperature increase throughout the multi-phase ore system, which promotes the generation of thermal stress at the interfaces between valuable minerals and impurities. This stress enhances the dissociation between them, facilitating the transformation of “wrapped” ores into a more manageable form while reducing energy consumption. Le et al. 67 investigated the behavior of zinc-iron spinel present in leaching slag under a reducing atmosphere. By optimizing experimental conditions, the majority of zinc can be successfully leached. In addition, ultrasonic treatment also plays an important role in enhancing the leaching efficiency of zinc-containing materials. Zheng et al. 68 studied low-grade zinc oxide using an ultrasonic process and revealed that ultrasonic irradiation markedly reduced the reaction time of ammonia-dipped zinc oxide. This reduction is beneficial to improve zinc recovery during leaching. These studies demonstrate that both microwave and ultrasonic technologies contribute to advancing the smelting processes of zinc-containing materials, enabling rapid and efficient zinc leaching.
6. Conclusion
With the advancement of ecological civilization, green and sustainable approaches to material processing have become a common challenge restricting enterprises from realizing economic growth and environmental protection. At the same time, the depletion of ore resources and the significant constraints on mining activities necessitate a shift in focus toward improving the recycling of zinc waste and developing more environmentally friendly, open-circuit, centralized treatment methods to achieve harmless disposal. Therefore, in the processing of zinc oxide materials, it is necessary to develop new equipment and technological processes through the interdisciplinary integration of efficient flotation techniques. In conjunction with current research technologies, Atomic Force Microscopy (AFM) can be used to examine the interaction between particles and bubbles, with the aim of acquiring detailed morphological information at nanometer-scale resolution. These environmentally friendly and convenient analytical techniques are expected to significantly promote the enrichment of zinc-containing materials. Furthermore, the introduction of novel chelating reagents has demonstrated enhanced flotation selectivity against gangue minerals such as silica and calcite.
The surface chemistry of zinc minerals dictates flotation efficiency, while the presence of dissolved ion species can either inhibit or promote collector adsorption. Future research should prioritize: (1) the development of eco-friendly flotation reagents with high biodegradability; (2) in situ characterization of mineral-reagent interactions using advanced spectroscopic and microscopic techniques; (3) process optimization through machine learning algorithms to predict flotation outcomes based on feed variability; and (4) pilot-scale validation of novel flowsheets integrating comminution, flotation, and hydrometallurgical steps. By adopting an integrated, innovative, and forward-thinking strategy in the roadmap-the industry could transform itself from a traditional resource processor into a leading enabler of a sustainable and technologically advanced circular economy.
Footnotes
Author contributions
Yucai Zhang: Methodology, Data curation, Investigation, Writing-original draft preparation. Zhaoyan Ma: Data curation. Xingjia Gu: Investigation. Shuping Jia: Data curation, Investigation, Writing-review and editing. Xiaoxin An: Methodology. Mengfan Niu: Data curation. Xianghao Zha: Conceptualization, Supervision, Writing-review & editing. Ruo He: Supervision, Writing-review & editing. Renpan Deng: Supervision. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was financially supported by the Kashi University On-campus Project (2024-2881), the National Natural Science Foundation of China (42567009), Universities Basic Operating Expenses for Scientific Research of Xinjiang Uygur Autonomous Region (XJEDU2025P078), Research Start-up Fee for High-level Talents of Kashi University (GCC2025ZK-006), Tianshan Innovation Team Plan of Xinjiang Uygur Autonomous Region (2023D14002), and the Interdisciplinary Research Program Project of Shihezi University (JCYJ202311).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that has been used is confidential.
