Abstract
Objective
Peripheral arterial disease (PAD) is a prevalent cardiovascular disorder that significantly impairs functional capacity and quality of life. Vascular inflammation is recognized as a key pathophysiological component of PAD. This cross-sectional study aimed to investigate the associations between a panel of novel, readily available hematological inflammatory indices and the presence of PAD in a nationally representative sample.
Methods
This is a cross-sectional study. This investigation utilized data from the National Health and Nutrition Examination Survey (NHANES) 1999 to 2004 cycles. PAD diagnosis was rigorously defined according to American Heart Association guidelines, with ankle-brachial index (ABI) values ≤0.9 obtained through standardized bilateral lower extremity doppler measurements and repeated verification protocols. Our analytical approach incorporated advanced statistical methodologies to elucidate complex relationships: Survey-weighted multivariable logistic regression analyses were conducted with sequential adjustment for demographic characteristics, clinical covariates, and traditional cardiovascular risk factors. The inflammatory biomarker panel included both composite indices - Systemic Inflammation Response Index (SIRI), Aggregate Index of Systemic Inflammation (AISI), Systemic Immune-Inflammation Index (SII), Neutrophil-to-HDL ratio (NHR), Monocyte-to-HDL ratio (MHR), Lymphocyte-to-HDL ratio (LHR), Platelet-to-HDL ratio (PHR) and Neutrophil-to-Lymphocyte ratio (NLR).
Results
Among 5,954 eligible participants, higher levels of SIRI, MHR, and NHR were significantly associated with an increased odds of prevalent PAD in fully adjusted models. Threshold analysis revealed nonlinear associations for MHR with PAD, with positive associations observed below identified inflection points. In comparative receiver operating characteristic (ROC) curve analysis, SIRI demonstrated modest discriminatory ability for PAD presence, though its predictive utility, like that of the other indices, was limited.
Conclusion
In this large, population-based cross-sectional study, several novel hematological inflammatory indices, particularly SIRI, MHR, and NHR, showed significant associations with prevalent PAD. These findings contribute to the understanding of the inflammatory landscape in PAD. However, their utility for clinical discrimination in a cross-sectional setting appears constrained, highlighting the need for further investigation into their potential roles.
1. Introduction
PAD is characterized by ischemia resulting from vascular insufficiency, primarily caused by atherosclerosis of the extremities, with a predominant involvement of the lower limbs. 1 Its clinical presentation spans a spectrum ranging from asymptomatic disease or intermittent claudication in early stages to rest pain accompanied by characteristic trophic changes—such as cutaneous atrophy, alopecia, and cyanosis—and markers of critical limb ischemia, including non-healing ulcers and gangrene, in advanced disease. With a global prevalence exceeding 200 million individuals, 2 PAD management is complicated by the fact that approximately two-thirds of affected patients remain asymptomatic, thereby delaying the timely initiation of preventive and therapeutic interventions.3,4
Accumulating evidence highlights the central role of systemic inflammatory pathways and dysregulated lipid metabolism in the pathogenesis of PAD. A chronic proinflammatory state promotes the progression of atherosclerosis through mechanisms involving endothelial dysfunction and plaque destabilization.5,6 Beyond its classical role in reverse cholesterol transport, high-density lipoprotein (HDL) exhibits multiple protective functions, including anti-inflammatory, antioxidant, antithrombotic, and immunomodulatory properties.7,8 Notably, hematologic parameters—such as neutrophil, monocyte, lymphocyte, and platelet counts—obtained from cost-effective and widely accessible complete blood counts, serve as reflections of systemic inflammation. This has prompted the development of novel hematological inflammatory indices derived from ratios involving HDL and complete blood cell counts, such as the NHR, MHR, LHR, PHR, SII, SIRI, AISI and NLR.9–12
Although previous studies have evaluated individual or selected combinations of these composite biomarkers in PAD populations, no comprehensive analysis has yet established the relationship between the full panel—NHR, MHR, LHR, PHR, SII, SIRI, AISI, and NLR—and the risk of PAD. Our study aims to address this gap by conducting a multi-parametric analysis of these novel hematological inflammatory indices within the context of PAD pathophysiology.
2. Materials and methods
2.1. Source of data and samples
This cross-sectional population-based study utilized a stratified, multistage probability cluster sampling design. Data were derived from the NHANES cycles spanning 1999 to 2004—a nationally representative epidemiological program that provides comprehensive demographic, dietary, and clinical risk factor information for the non-institutionalized U.S. population (https://www.cdc.gov/nchs/nhanes/). Participants first completed standardized household interviews, followed by detailed physical examinations conducted at mobile examination centers to assess cardiometabolic health. Venous blood samples were collected by certified phlebotomists and analyzed in accordance with established laboratory protocols, with results uniformly archived. The patient data were de-identified during the course of the NHANES study.
2.2. Population selection criteria
PAD is defined diagnostically by an ABI measurement of ≤0.9 in either lower extremity. ABI assessments were conducted following standardized protocols with participants resting in the supine position. The index was derived by dividing the mean systolic blood pressure measured at the ankle (either the posterior tibial or dorsalis pedis artery) by the mean systolic pressure obtained from the ipsilateral brachial artery. From the initial NHANES 1999–2004 cohort, we applied a three-tiered exclusion protocol: (1) absence of ABI measurements or values >1.5 (suggestive of non-compressible vessels), (2) incomplete biomarker or covariate data, and (3) restriction to adult participants aged 40 years or older.
2.3. Inflammatory indices
Hematologic parameters were quantified from EDTA-anticoagulated whole blood specimens using a Coulter® DxH 800 automated analyzer (Beckman Coulter, USA), which provided absolute counts of lymphocytes, platelets, monocytes, and neutrophils in standardized units (×103 cells/μL). Novel inflammatory indices were computationally derived as follows: NHR = neutrophil count/HDL; MHR = monocyte count/HDL; LHR = lymphocyte count/HDL; PHR = platelet count/HDL; SII = (platelet × neutrophil)/lymphocyte; SIRI = (neutrophil × monocyte)/lymphocyte; AISI = (neutrophil × platelet × monocyte)/lymphocyte; NLR = neutrophil/lymphocyte.
2.4. Statistical analyses
Statistical analyses were performed using EmpowerStats (X&Y Solutions, Boston MA) and R 4.4.1 (R Foundation) with survey package, rigorously accounting for the multistage probability sampling design through application of NHANES-recommended examination weights and strata/primary sampling unit variables per CDC analytic guidelines. Categorical variables were presented as weighted percentages, while continuous variables were expressed as mean ± standard error. The novel hematological inflammatory indices-PAD association was interrogated through three progressively adjusted survey-weighted multivariable logistic regression analyses models: Model 1: No covariates were adjusted. Model 2: Adjusted for Age, Sex, and Race. Model 3: Adjusted for Sex, Age, Race, C- reactive protein, Glycohemoglobin, Systolic pressure, Diastolic pressure, BMI, HDL, Smoking status, Hypercholesterolemia, Urinary Albumin-to-Creatinine Ratio (UACR). Smoking status was categorized based on self-reported cigarette consumption: Never smoker: <100 cigarettes smoked in lifetime. Former smoker: ≥100 cigarettes smoked in lifetime but currently quit. Current smoker: ≥100 cigarettes smoked in lifetime and currently smoking. Hypercholesterolemia was defined as a total cholesterol level ≥240 mg/dL or current use of lipid-lowering medication. Kidney function was assessed using the UACR, a sensitive marker of renal impairment. A UACR <300 mg/g was classified as normal or mildly impaired, while UACR ≥300 mg/g indicated significant kidney dysfunction. Nonlinear relationships were handled using Generalized Additive Model (GAM) fitting. Using the log-likelihood ratio test, we compared segmented regression models (two-segment linear regression models fitted to each interval) with non-segmented or unilinear models. This allowed us to further investigate threshold effects. To locate breakpoints, we used a two-step recursive technique. To further analyze the association between novel inflammatory markers and PAD, we conducted an analysis using the svyROC package. The svyROC package is designed to handle complex survey data, enabling the estimation of receiver operating characteristic (ROC) curves, the area under the curve (AUC), and optimal cutoff points for individual classifications, while accounting for complex sampling designs. The 95% confidence intervals for the AUC were calculated using the bootstrap resampling method. All inferential tests adopted two-tailed P value < 0.05 significance threshold. In the R analysis, the svydesign function is employed with the following parameter specifications: The id parameter is set to the SDMVPSU variable from the NHANES, representing the Primary Sampling Units (PSUs). The strata parameter is assigned the SDMVSTRA value from the NHANES, indicating the stratification variable. The weights parameter is configured with the appropriate weighting variable. The nest parameter is set to TRUE to signify that PSUs are uniquely nested within strata. Since PAD is a binary outcome variable, the family parameter is specified as quasibinomial, with the link parameter set to logit, corresponding to the use of a quasi-binomial distribution. Regarding the selection of weights, in accordance with NHANES guidelines, the weight of the smallest subgroup encompassing all variables included in the analysis is designated as the final study weight. The calculation follows these formulas: For the 1999–2002 cycles, the weight is computed as (2/(2 + 1)) × WTMEC4YR. For the 2003–2004 cycles, the weight is computed as (1/(2 + 1)) × WTMEC2YR. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki, as revised in 2024.
The reporting of this study conforms to STROBE guidelines. 13
3. Result
3.1. Association novel hematological inflammatory indices between and PAD
A total of 5,954 eligible individuals out of 31,126 participated in the study, 48.29% were male and 51.71% were female. Mexican Americans comprised 22.00% of the total population. The prevalence of PAD was 1.49%. A rigorous screening process was conducted according to the predefined criteria, and the number of ineligible participants excluded at each stage is presented in Figure 1. The differences between the PAD and non-PAD populations are shown in Table 1, with the novel hematological inflammatory indices being predominantly higher in the PAD population. Multivariable regression analyses delineated dose-response relationships between novel hematological inflammatory indices and PAD across three hierarchical models (Table 2). The fully adjusted model (Model 3) revealed significant associations with PAD for SIRI (OR = 1.108; 95% CI: 1.009, 1.216), MHR (OR = 1.152; 95% CI: 1.009, 1.315), and NHR (OR = 1.217; 95% CI: 1.060, 1.421). This figure provided a detailed overview of the characteristics of 5,954 eligible participants, who were divided into two groups based on the presence or absence of PAD. Among them, 89 participants were assigned to the PAD group, and 5,865 participants were assigned to the non-PAD group. Population characteristics stratified by PAD. Associations between inflammatory biomarkers with PAD. Model 1: No covariates were adjusted. Model 2: Adjusted for Age, Sex, and Race. Model 3: Adjusted for Sex, Age, Race, C- reactive protein, Glycohemoglobin, Systolic pressure, Diastolic pressure, BMI, HDL, Smoking status, Hypercholesterolemia, UACR. All OR correspond to the risk ratio associated with a one-standard-deviation increase.
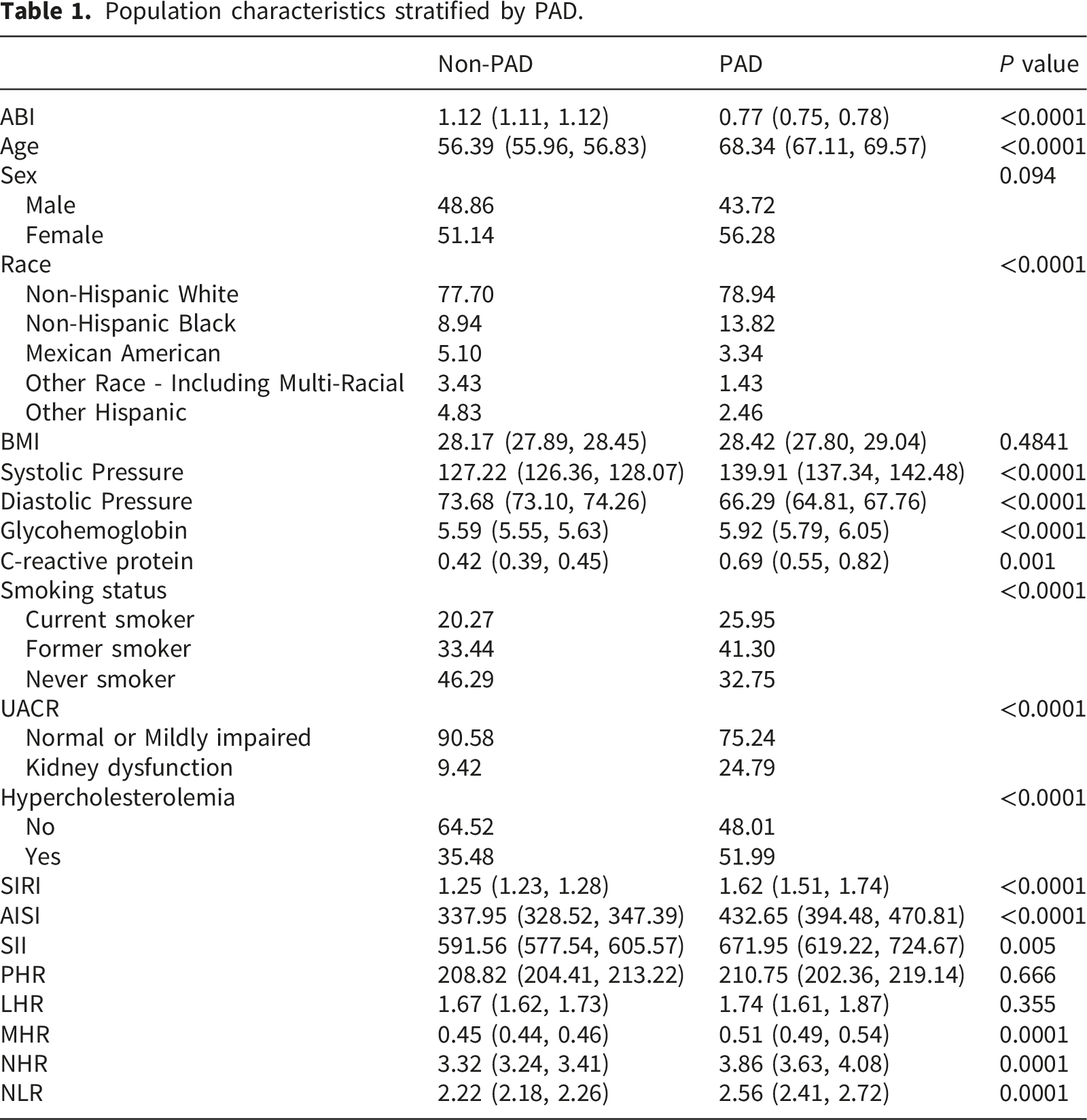

GAM and smoothed curve fitting showed that the relationship between MHR and PAD was nonlinear, with fully adjusted breakpoint values of 0.485. SIRI and NHR exhibited a linear relationship with PAD (Figure 2, Table 3). In the nonlinear relationship between MHR and PAD, we also found a saturation effect. When MHR was less than 0.485, MHR was positively correlated with the prevalence of PAD (OR=1.659; 95% CI:1.140, 2.414). On the right side of the breakpoints, the correlation was not significant (OR=1.033; 95% CI: 0.877, 1.216). Smooth curve fitting for inflammatory biomarkers with ABI. Threshold effect analysis of inflammatory biomarkers on PAD using a two-piecewise linear regression model in Model 3. All OR correspond to the risk ratio associated with a one-standard-deviation increase.
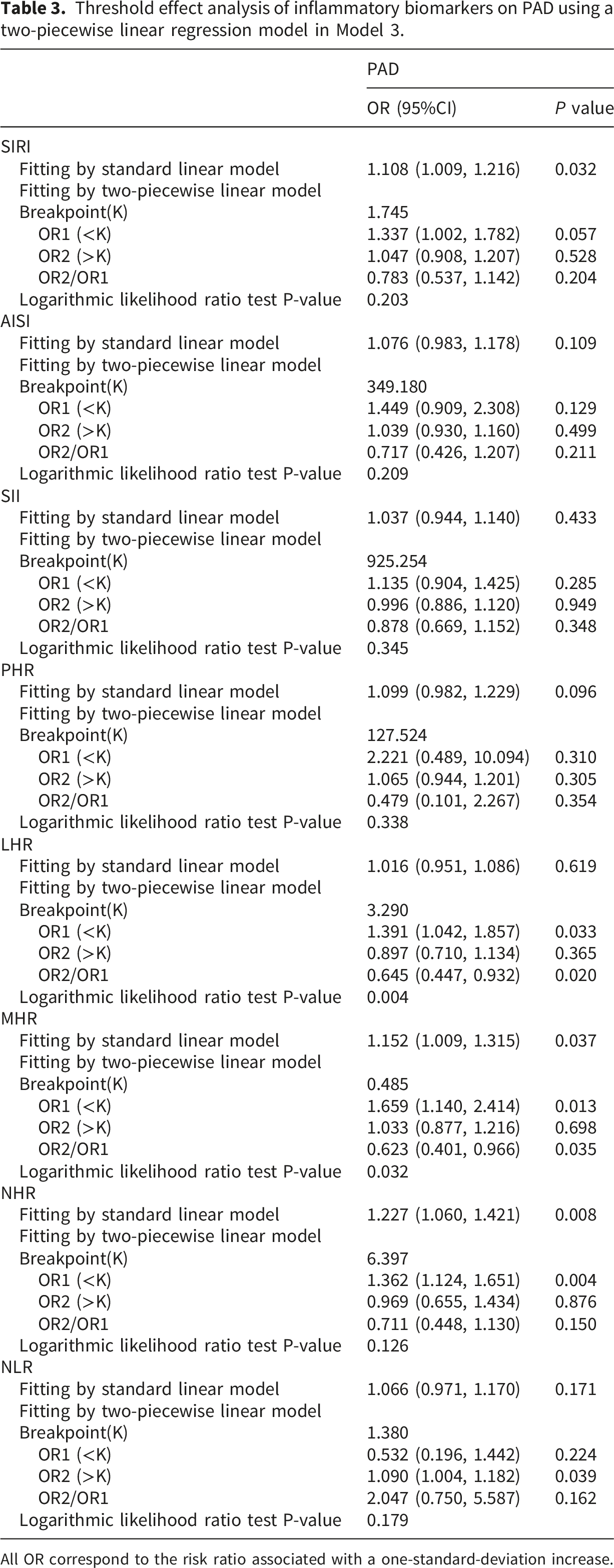
3.2. ROC analysis
Comparison of AUC values between SIRI and other inflammatory biomarkers.

4. Discussion
This cross-sectional study, which included 5,954 adult participants, demonstrated significant associations between certain novel hematological inflammatory indices and prevalent PAD. However, their standalone diagnostic utility appears limited, as evidenced by AUC values below 0.70 in ROC analysis. Therefore, while these indices are not suitable as definitive diagnostic tools, they may serve as complementary and pathophysiologically informative markers within a broader clinical assessment framework. Further experimental and prospective clinical studies are required to validate their potential utility and to elucidate their precise role in the pathophysiology of PAD.
Russell Ross 14 first proposed the hypothesis that atherosclerosis is an inflammatory disease in 1999 by observing circulating monocytes infiltrating developing fatty streaks, and since then several studies have explored the role of various immune cells in the development of atherosclerosis.15,16 Neutrophils and lymphocytes are involved in the regulation of innate and adaptive immunity. 17 Neutrophils are the most common leukocytes and are associated with the initiation and regulation of immune processes. Neutrophil elastase secreted by neutrophils is involved in the regulation of chronic inflammation as reported by Talukdar et al. 18 Increased reactive oxygen species (ROS) and neutrophil activity are associated with the development of PAD. Lymphocytes are involved in regulating adaptive and innate immunity by working closely with other cell types and displaying protective or regulatory properties. Platelets can be activated by a variety of inflammatory factors such as P-selectin, epinephrine, glutamate, 5-hydroxytryptamine, dopamine, and cytokines. Platelets are considered a non-specific first-line marker of inflammation and may modulate endothelial and leukocyte function during inflammation by binding to cells. Activated platelets release pro-inflammatory molecules such as P-selectin, CD40L, IL-1, glutamate, and serotonin and modulate endothelial cell function during the development of PAD. 19
Although these inflammation-associated cells are strongly associated with the development of arterial plaque, unlike other results, 20 our findings indicate that novel hematological inflammatory indices exhibit limited predictive capacity for PAD. On one hand, MHR demonstrate a nonlinear relationship with PAD, losing predictive value for the condition above certain thresholds. On the other hand, AUC value of SIRI and NHR falls below 0.7, indicating its predictive capability for PAD is constrained. Therefore, we believe that developing new, convenient inflammatory indices to predict PAD is an urgent priority.
However, our study has some limitations: 1. Since it was a retrospective study, a causal relationship could not be established, and this finding remains to be validated in prospective studies. 2. Only one blood test was performed in each case, and circulating cells have a short lifespan, so serial testing is a better option. 3. Calculating novel hematological inflammatory indices was not difficult, but platelet, lymphocyte, monocyte and neutrophil counts were not uncommonly missing from the database, which may cause selection bias.
Footnotes
Ethical considerations
This study utilized data from the National Health and Nutrition Examination Survey (NHANES), a publicly accessible resource released under an open license permitting unrestricted reuse. All data employed in this research are fully de-identified and contain no personally identifiable information. The use of NHANES data is strictly confined to statistical analysis and reporting purposes. Any attempt to re-identify individuals is strictly forbidden and would contravene the confidentiality protections mandated by federal law. Accordingly, no additional approval was required to access or use the data for this study.
Author contributions
Yuchen Zhu and Peng Zhou were responsible for R language analysis and data organization. Yuchen Zhu, Bingjie Zhu, and Yiming Huang drafted the initial manuscript and prepared the figures and tables. Peng Zhou, Chao Yang, and Qin Li conceived the study design and reviewed the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by grants from the National Natural Science Foundation of China (No. 82270520).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
