Abstract
Objective
To systematically review and quantitatively synthesize in vivo evidence on extracellular vesicle (EV)–based therapies for regenerative endodontic procedures, focusing on mineralization and angiogenesis outcomes.
Methods
We conducted a systematic review and meta-analysis of in vivo preclinical dentoalveolar/regenerative endodontic models comparing EV-based interventions (exosomes/microvesicles/apoptotic bodies), alone or with scaffolds, versus control conditions. Searches were performed in Web of Science Core Collection and Scopus from inception to 31 December 2025, with reference screening. Random-effects meta-analyses pooled standardized mean differences (SMDs) for mineralization and angiogenesis; heterogeneity was assessed using I2. Risk of bias was evaluated using the Systematic Review Centre for Laboratory animal Experimentation tool.
Results
Twenty-one studies met inclusion criteria; seven were included in the mineralization meta-analysis and five in the angiogenesis meta-analysis. EV-based therapies significantly increased mineralization versus controls (SMD = 6.43; 95% confidence interval (CI) [3.13–9.73]; I2 = 91%) and angiogenesis (SMD = 7.89; 95% CI [3.94–11.85]; I2 = 82%). Subgroup analyses suggested stronger effects for EVs derived from dental pulp stem cells, stem cells from human exfoliated deciduous teeth, and stem cells from apical papilla. Risk of bias was predominantly unclear due to limited reporting of randomization and blinding. Considerable heterogeneity, small numbers of pooled studies, and variability in EV isolation, characterization, dosing, and outcome assessment limit generalizability and translation.
Conclusions
EV-based therapies enhance mineralization and angiogenesis in preclinical regenerative endodontic models, with dental stem cell–derived EVs showing the greatest apparent potential. However, effect sizes should be interpreted cautiously given very high heterogeneity and methodological/reporting limitations.
Introduction
Regenerative endodontic procedures (REPs) are designed to restore the damaged pulp–dentin complex, yet traditional approaches such as scaffolds and cell transplantation still face significant limitations—immune responses, potential tumorigenicity, and constrained cell survival in vivo. 1 In contrast, extracellular vesicles (EVs)—membrane-bound nanoparticles released by cells—offer a promising acellular strategy.2,3 EVs function as paracrine mediators, delivering proteins, lipids, and microRNAs to target cells, thus promoting regenerative processes with reduced immunogenicity and improved stability compared to whole-cell therapies. 4
Preclinical in vivo studies have highlighted the reparative power of EVs derived from dental stem cell sources. 5 Lu et al. 6 demonstrated that EVs from stem cells of human exfoliated deciduous teeth (SHEDs), when incorporated into gelatin methacryloyl (GelMA) hydrogels, significantly enhanced odontogenic differentiation of dental pulp stem cells (DPSCs). Similarly, Wang et al. 7 showed that EVs from Schwann cells supported pulp regeneration via the stromal cell–derived factor 1 (SDF-1)/ C-X-C chemokine receptor type 4 (CXCR4) signaling axis in a large-animal model.
Moreover, systematic reviews have underscored the potential of engineered EVs and scaffold-based delivery platforms to improve EV retention and efficacy in dental tissues.8,9,10 For instance, combination strategies pairing EVs with biomaterial scaffolds have been shown to amplify mineralization and vascularization in dentoalveolar regeneration models. 11 Given the biological and anatomical continuity of the dentoalveolar complex, preclinical models of periodontal or gingival regeneration were considered relevant when they assessed regenerative outcomes directly applicable to pulp–dentin or endodontic tissue repair.
Despite these encouraging advances, the literature lacks a comprehensive quantitative synthesis focused on in vivo EV applications in REPs. Existing reviews are predominantly descriptive or include in vitro findings, without stratifying results by EV source or specific regenerative outcomes. To address this gap, we conducted a meta-analysis evaluating in vivo EV-based regenerative outcomes in endodontic models—focusing on both mineralization and angiogenesis. This analysis aims to clarify the comparative impact of different EV sources, highlight methodological shortcomings, and inform the future clinical translation of EV therapies in endodontics.
Methods
Search strategy
We designed a comprehensive and reproducible literature search to identify in vivo studies examining EV therapy in REPs. This systematic review and meta-analysis was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. The protocol was aligned with PRISMA-S guidelines. Electronic searches were conducted in Web of Science Core Collection and Scopus from database inception to 15 July 2025. These databases were selected because they provide broad multidisciplinary coverage and index the vast majority of high-impact preclinical and biomaterials research relevant to regenerative endodontics. In addition, manual screening of reference lists and citation tracking of included studies was performed to identify any potentially relevant articles not captured by database searches. To capture all relevant studies, we combined indexed and free-text terms related to regenerative endodontics and EVs, using Boolean operators. The detailed search strategy is provided in Supplemental Table S1. This article is registered with PROSPERO (Centre for Reviews and Dissemination 420251107328). The search was updated on 31 December 2025 using the same databases and search strategy; no additional eligible in vivo studies were identified.
Search strings were adapted to each database's syntax and controlled vocabulary. No filters were applied for publication date or language to maximize coverage. Results were exported with full search strategies, date of conduct, number of hits per database, and stored in Covidence for screening and record-keeping. Iterative adjustments ensured capture of key known studies. The search was last updated on 31 December 2025.
Eligibility criteria
We included studies based on clearly defined eligibility criteria refined through the Population, Intervention, Comparison, Outcome framework, following PRISMA 2020 guidelines. Only in vivo preclinical models were eligible, provided they assessed regenerative endodontic or dental tissue engineering procedures. Interventions required the administration of EV-based therapies, including exosomes, apoptotic bodies, or microvesicles, either alone or delivered via scaffold/carrier systems. Studies were included if they featured a concurrent control group receiving sham treatment, vehicle, or standard materials, and reported quantitative regenerative outcomes with sufficient statistical detail—specifically postintervention means, standard deviations (SDs), and sample sizes—for mineralization and/or angiogenesis. Studies using periodontal, gingival, or dentoalveolar defect models were included only when outcomes directly reflected pulp-related angiogenesis, mineralization, or dentoalveolar regeneration relevant to REPs.
We only considered original full-text articles published in peer-reviewed journals; in vitro/ex vivo studies, case reports, reviews, editorials, protocols, and conference abstracts were excluded. No restrictions were imposed on language or publication date to ensure a comprehensive and unbiased literature capture. Studies were excluded if they lacked in vivo experimentation, combined EVs with other cell-based therapies without separately reporting EV effects, failed to provide necessary statistical data for meta-analysis, or used immature tooth models without quantitative regenerative outcomes. Two independent reviewers screened titles, abstracts, and full texts, resolving disagreements through discussion or consultation with a third reviewer. This rigorous methodology ensured that only robust in vivo EV-therapy studies with quantifiable regenerative endpoints were included in our qualitative synthesis, with appropriate subsets advanced for meta-analyses focused on mineralization and angiogenesis.
Data extraction and quality assessment
Data extraction was independently performed by two reviewers who piloted a standardized form to ensure consistency and reduce errors. Extracted variables included: study and publication details (author, year, species, and tooth model), EV characteristics (cell source, isolation and characterization methods), delivery approaches (dosing, scaffold, route), regenerative assay outcomes (quantitative measures of mineralization and angiogenesis), and comparator details. For studies contributing to the meta-analysis, mean values, SDs, sample sizes, and relevant statistical metrics were recorded. When data were ambiguous or absent, we contacted authors directly to request missing information.
Methodological quality and risk of bias were evaluated using the systematic review centre for laboratory animal experimentation (SYRCLE) risk-of-bias tool, tailored to animal intervention studies. This tool assesses 10 domains—including selection bias (random sequence generation, allocation concealment), performance bias (blinding of caregivers), detection bias (blinding of outcome assessors), attrition bias, reporting bias, and other biases. Two reviewers independently rated each study as having low, high, or unclear risk of bias. Any disagreements were resolved through consensus, with a third reviewer arbitrating unresolved cases.
We recorded these assessments and used them to evaluate the strength of evidence and interpret heterogeneity in outcomes. Studies with high risk in key domains were flagged and considered in sensitivity analyses to assess their impact on pooled effect estimates. This rigorous process was undertaken to ensure transparency, reproducibility, and reliability in our synthesis of in vivo EV-based regenerative studies. For each included in vivo study, information on ethical approval (name of the ethics committee, approval number, and approval date) was extracted when reported; when such information was not provided in the original publication, this was recorded as “not reported.”
Outcome definitions and hierarchies
We predefined two primary outcomes—mineralization and angiogenesis—operationalized using common quantitative metrics to ensure consistency across studies. For mineralization, we accepted measurements such as percentage of mineralized tissue area, linear thickness (µm or mm), and volumetric bone parameters like bone volume fraction (BV/TV), bone mineral density, or bone volume (BV). The hierarchy was as follows: (1) percent area of mineralized tissue, (2) thickness, and (3) volumetric metrics, in alignment with prior regenerative tissue meta-analyses. If multiple mineralization metrics were reported in a single study, the highest-priority measure was selected to prevent bias and ensure comparability.
For angiogenesis, we included outcomes reflecting vascular formation and function, such as counts of cluster of differentiation (CD)31/CD34-positive blood vessels, vascular endothelial growth factor (VEGF) levels, or similar endothelial markers. When studies reported multiple angiogenesis measures, we averaged effect sizes to preserve statistical independence while capturing overall angiogenic potential.
This structured approach—prioritizing a predefined hierarchy and managing multiple measures—ensured comparability across diverse experimental models and clear interpretation in quantitative synthesis.
Statistical analysis and data synthesis
We conducted two parallel meta-analyses following Cochrane standards to evaluate continuous outcomes for mineralization and angiogenesis from preclinical studies using EV-based therapies. Eligible studies compared treated versus control groups and reported postintervention means, SDs, and sample sizes. Mineralization outcomes included measures such as percent area of mineralized tissue, thickness (µm/mm), or volumetric ratios (BV/TV), while angiogenesis outcomes encompassed CD34/CD31 expression, vessel counts, and VEGF quantification at sites like dental pulp, gingiva, Matrigel plugs, and skin wounds.
We conducted a meta-analysis following Cochrane standards, focusing on continuous mineralization and angiogenesis outcomes across EVs–based dentoalveolar regeneration studies. For mineralization, when multiple outcomes were reported within a study, we applied a predefined hierarchy (1: percent area, 2: thickness, 3: volumetric ratios) and included only the highest-priority measure. For angiogenesis, studies typically reported a single effect; for those with multiple, effect estimates were averaged to maintain statistical independence.
We pooled SMDs using a random-effects model with inverse-variance weighting. Heterogeneity was quantified using the I2 statistic, where values above 50% indicated substantial variability. 12 Due to the small number of included studies (<10), we employed the Hartung-Knapp-Sidik-Jonkman (HKSJ) adjustment—or its modified version—to obtain more conservative confidence intervals under heterogeneity. 13 This approach mitigates inflated type I error rates associated with DerSimonian-Laird estimators in meta-analyses with few studies. 13
We generated a forest plot in R using the meta::metagen() and meta::forest() functions to visualize individual SMDs, pooled effect, and heterogeneity statistics. Sensitivity analysis involved leave-one-out procedures, whereby each study was sequentially omitted to assess the robustness of pooled results and their influence on I2. Subgroup analyses, based on source of stem cells, were conducted to explore potential sources of heterogeneity.
This rigorous methodology ensured that our synthesized estimate of mineralization effects was statistically sound, appropriately weighted for variance, and transparent in assessing heterogeneity and robustness.
All standardized mean difference (SMD) calculations were independently rechecked by two authors using extracted postintervention means, SDs, and sample sizes. The magnitude of SMDs observed reflects the combination of large between-group differences and small within-group variance, which is common in tightly controlled preclinical animal experiments. To enhance transparency and reproducibility, the extracted raw data and step-by-step SMD calculations for all meta-analyzed outcomes are provided in Supplemental Tables S2 and S3.
Results
Study selection
The study selection process followed the PRISMA 2020 framework to ensure transparent and systematic reporting. A total of 119 records were identified through database searches of Web of Science Core Collection and Scopus and 27 records from manual reference screening. After removing 39 duplicate records, 80 titles and abstracts underwent level-1 screening against predefined inclusion and exclusion criteria. At this stage, 54 records were excluded for reasons such as being non-in vivo, lacking EV therapy, or not addressing regenerative endodontic outcomes. An updated search covering the period from July to December 2025 did not yield any additional studies meeting the inclusion criteria.
The remaining 26 full-text articles were retrieved and assessed in detail for eligibility. Of these, five articles were excluded, most commonly due to insufficient outcome reporting or non-EV interventions.
Ultimately, 21 in vivo EV-therapy studies met all inclusion criteria and were carried forward into qualitative synthesis (Table 1). Among these, seven studies also reported sufficient data for inclusion in the quantitative meta-analysis of mineralization outcomes, and five were included in the meta-analysis of angiogenesis outcomes. A PRISMA flow diagram summarizing this selection process is provided in Figure 1.

PRISMA flow diagram depicting the selection process of in vivo extracellular vesicle therapy studies in regenerative endodontics, from initial database search to final inclusion in qualitative and quantitative syntheses
Characteristics of 21 in vivo studies investigating extracellular vesicle–based therapies in regenerative endodontic and dental tissue engineering models.
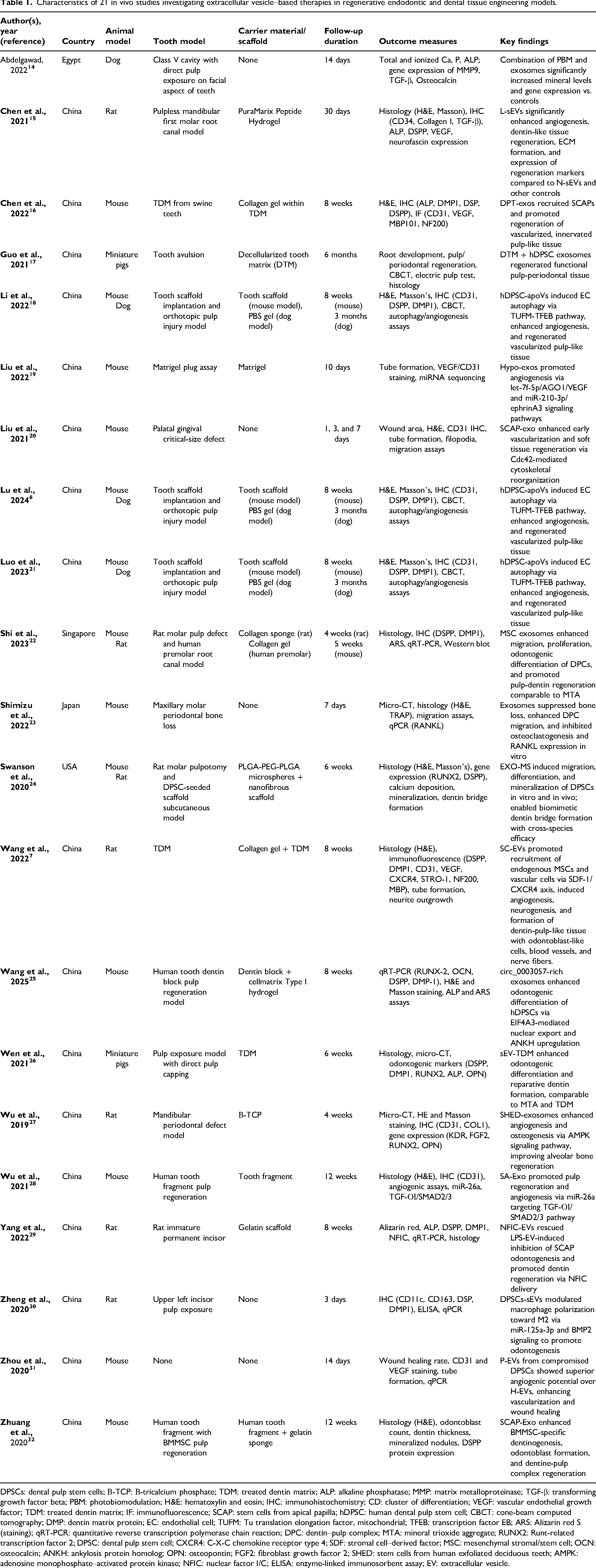
DPSCs: dental pulp stem cells; Β-TCP: Β-tricalcium phosphate; TDM: treated dentin matrix; ALP: alkaline phosphatase; MMP: matrix metalloproteinase; TGF-β: transforming growth factor beta; PBM: photobiomodulation; H&E: hematoxylin and eosin; IHC: immunohistochemistry; CD: cluster of differentiation; VEGF: vascular endothelial growth factor; TDM: treated dentin matrix; IF: immunofluorescence; SCAP: stem cells from apical papilla; hDPSC: human dental pulp stem cell; CBCT: cone-beam computed tomography; DMP: dentin matrix protein; EC: endothelial cell; TUFM: Tu translation elongation factor, mitochondrial; TFEB: transcription factor EB; ARS: Alizarin red S (staining); qRT-PCR: quantitative reverse transcription polymerase chain reaction; DPC: dentin–pulp complex; MTA: mineral trioxide aggregate; RUNX2: Runt-related transcription factor 2; DPSC: dental pulp stem cell; CXCR4: C-X-C chemokine receptor type 4; SDF: stromal cell–derived factor; MSC: mesenchymal stromal/stem cell; OCN: osteocalcin; ANKH: ankylosis protein homolog; OPN: osteopontin; FGF2: fibroblast growth factor 2; SHED: stem cells from human exfoliated deciduous teeth; AMPK: adenosine monophosphate–activated protein kinase; NFIC: nuclear factor I/C; ELISA: enzyme-linked immunosorbent assay; EV: extracellular vesicle.
Characteristics and methodological features of in vivo EV therapy studies
Table 1 summarizes 21 in vivo studies investigating EV–based therapies in regenerative endodontic and dental tissue engineering models. Animal subjects ranged from mice, rats, miniature pigs, and dogs, with one study also including a human premolar model. Models incorporated pulp exposure or injury, tooth avulsion, periodontal defects, and critical-size defects. Carrier materials varied—most studies used collagen-based scaffolds, treated dentin matrix (TDM), decellularized tooth matrix, hydrogels, or β-TCP, while several applied EVs without a scaffold. Follow-up periods varied widely, from three days up to six months. Outcome measures included mineralization markers, histological and immunohistochemical analyses, micro-CT, gene and protein expression assays, tube formation and angiogenesis tests, and functional assays such as electric pulp testing. Across studies, EV treatments consistently enhanced angiogenesis, odontogenic or osteogenic differentiation, vascularized pulp-like tissue formation, dentin bridge development, and reduced bone or periodontal tissue loss, in many cases rivaling or exceeding the performance of traditional controls such as mineral trioxide aggregate or TDM.
Detailed characteristics of EV sources, isolation, characterization, dosing, and delivery methods are summarized in Supplemental Table S4. EV sources included DPSCs, SHED, stem cells from apical papilla (SCAP), dental pulp cells (DPCs), Schwann cells, hESC-derived mesenchymal stromal/stem cells (MSCs), and other engineered cells. Isolation methods were predominantly ultracentrifugation, with others using size-exclusion or commercial kits. Most studies characterized EVs through transmission electron microscopy, nanoparticle tracking analysis or dynamic light scattering, western blotting for exosomal markers, and in some cases flow cytometry or immunogold labeling. Dosing strategies ranged from micrograms to milligrams of protein content or particle count, typically administered via scaffold implants, gels, injections into pulp defects, or topical application. Reporting of ethical approval varied across studies; while several articles specified the approving ethics committee and approval identifiers, others did not report ethics approval details, which was documented accordingly (Supplemental Table S5).
Meta-analysis of outcomes
Mineralization outcomes
A total of seven studies were included in the meta-analysis of EV–based mineralization outcomes (Figure 2). Using a random-effects model with inverse-variance weighting and HKSJ adjustment, the pooled SMD was 6.43 (95% CI [3.13–9.73], p = .0001), indicating a significant overall enhancement in mineralization in treated groups compared to controls. However, substantial heterogeneity was observed (Q = 78.71, p < .0001; I2 = 91%), reflecting considerable variation across studies. Subgroup analyses by stem cell source of EVs revealed notable differences: SHED-based studies showed an SMD of 7.75 (95% CI [−3.34–18.85]), hESC-derived MSCs yielded a consistent SMD of 6.71 (95% CI [4.53–8.90], I2 = 0%), and DPSC-based studies indicated a moderate effect size (SMD 4.82, 95% CI [2.41–7.22], I2 = 0%). In contrast, results for DPCs were nonsignificant (SMD 0.48, 95% CI [−0.47–1.42]), while a single SCAP-based study demonstrated a large effect (SMD 14.13, 95% CI [9.20–19.07]). Overall, these findings indicate a statistically significant improvement in mineralization with EV therapies; however, the magnitude should be interpreted cautiously given very high heterogeneity and the preclinical nature of the evidence.

Forest plot of standardized mean differences in mineralization outcomes between extracellular vesicles derived from different stem cell sources and control groups.
Angiogenesis outcomes
In a meta-analysis of five preclinical studies, EV-based therapies produced a marked increase in angiogenesis (Figure 3). The pooled SMD was 7.89 (95% CI [3.94–11.85], p < .0001), indicating a robust overall treatment effect across diverse angiogenesis metrics, including CD34/CD31 expression, vessel formation, and VEGF levels. Significant heterogeneity was detected (Cochran's Q = 39.71, p < .0001; I2 = 82%), likely attributable to variations in animal species, tissue sites, EV sources, and assay methods.
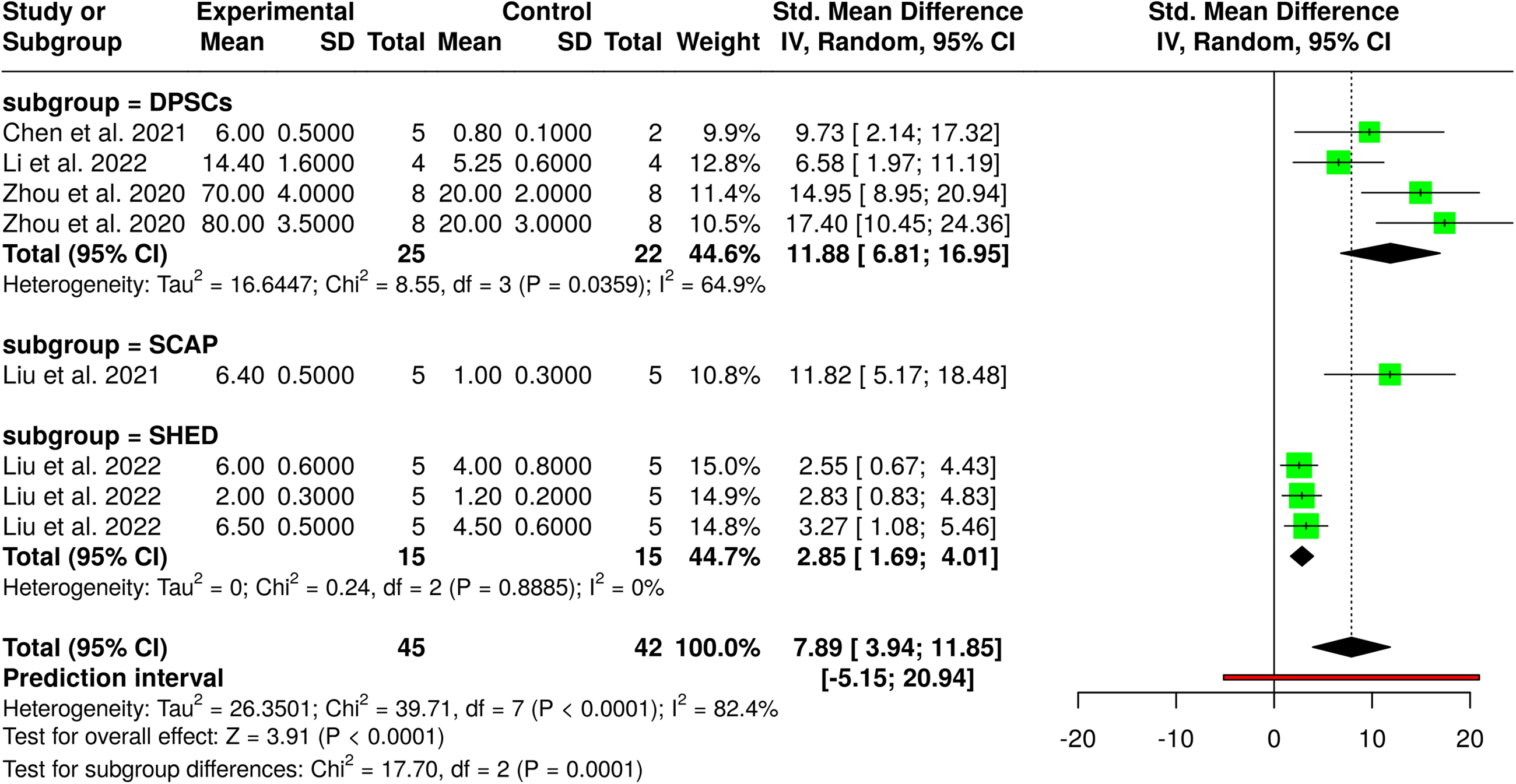
Forest plot of standardized mean differences in angiogenesis outcomes following EV-based treatments versus control.
Subgroup analysis by EV origin revealed distinct effects: DPSC-derived EVs (three studies) demonstrated substantial angiogenic effects (SMD = 11.88, 95% CI [6.81–16.95]; I2 = 65%), SCAP-derived EVs (one study) showed a similar effect (SMD = 11.82, 95% CI [5.17–18.48]), while SHED-derived EVs (three measures) produced a moderate yet consistent effect (SMD = 2.85, 95% CI [1.69–4.01]; I2 = 0%). Despite the high overall heterogeneity, the uniform positive direction and substantial magnitude of effects support the pro-angiogenic potential of EV therapies in preclinical models, while the very high heterogeneity limits the precision and generalizability of the pooled magnitude.
Risk of bias
Risk of bias was assessed for all 21 included studies using the SYRCLE tool, which evaluates six domains of bias specific to animal studies (Figure 4). Selection bias was rated as “Low” in only five studies, while the remaining studies were marked as “Unclear” due to insufficient reporting of random sequence generation and allocation methods. Performance bias, which includes random housing and caregiver blinding, was consistently rated “Low” across all studies, reflecting standardized experimental conditions and consistent housing practices. However, detection bias, which evaluates blinding of outcome assessors and random outcome measurement, was rated “Unclear” in all cases, as no study reported sufficient details in this regard. Attrition bias and reporting bias were also predominantly rated as “Unclear” due to the absence of information on incomplete data handling or the presence of study protocols. A few studies received “Low” risk in the “Other bias” domain due to well-controlled experimental designs or proper statistical analysis. Overall, the risk of bias was rated as “Unclear” for all studies, highlighting a general lack of methodological transparency in reporting randomization, blinding, and data management procedures.

Risk-of-bias assessment of included in vivo studies using the SYRCLE tool. The left panel presents a summary of domain-level judgments across all studies, while the right panel displays individual study-level risk-of-bias evaluations across six domains: selection, performance, detection, attrition, reporting, and other bias.
Discussion
The results of this meta-analysis align with and expand upon a growing body of evidence supporting the therapeutic utility of EVs in regenerative endodontics. Notably, recent studies by Lu et al. 6 and Wang et al. 25 corroborate the regenerative potential of DPSC-derived and Schwann cell-derived EVs, respectively, emphasizing their roles in angiogenesis and mineralized tissue formation. Lu et al. demonstrated that functional EVs from SHEDs loaded onto GelMA scaffolds promoted odontogenic differentiation of DPSCs, consistent with our observation of favorable mineralization outcomes using stem cell-derived EVs. Similarly, Wang et al. reported that Schwann cell–derived EVs facilitated pulp regeneration through endogenous stem cell recruitment via the SDF-1/CXCR4 axis, supporting our subgroup findings of EV-mediated angiogenic enhancement.
The very large effect sizes observed in this meta-analysis should be interpreted within the context of preclinical research. Therefore, the pooled estimates primarily support the direction of benefit (favoring EV-based interventions) rather than a clinically interpretable magnitude of effect. In animal models, SMDs are often inflated relative to clinical studies due to limited biological variability, homogeneous experimental conditions, and highly sensitive outcome measures. Consequently, these SMDs should not be interpreted as directly translatable to expected clinical effect magnitudes, but rather as indicators of strong biological efficacy under controlled experimental conditions.
Additionally, the findings of Shi et al. 22 reinforce the regenerative effects of EVs from mesenchymal stromal cells, showing their ability to stimulate DPC migration, proliferation, and dentin–pulp complex formation. These results are in harmony with the biological outcomes summarized in our review and confirm the broader regenerative mechanisms that EVs exert in vivo.
Comparative evaluation with Swanson et al. 24 highlights the advantage of EV-incorporated scaffolds in sustaining odontogenic cues and facilitating dentin bridge formation. The observed dentinogenesis in both subcutaneous and orthotopic models supports our inclusion of scaffold-based delivery systems in many studies, further affirming the utility of EV–biomaterial hybrids in tissue engineering. This approach also echoes findings from Zhuang et al., 32 where SCAP-derived EVs were able to promote dentine–pulp complex regeneration when delivered via gelatin sponges in subcutaneous models.
In the context of angiogenesis, our observations mirror those of Liu et al., 19 who showed that exosomes derived from hypoxia-conditioned SHEDs improved angiogenesis through the transfer of let-7f-5p and miR-210-3p. This mechanism aligns with the high angiogenic effect sizes identified in SHED-based subgroups. Additionally, Wu et al. 28 provided mechanistic insight into EV-mediated miR-26a transfer that activated the transforming growth factor beta (TGF-β)/SMAD2/3 signaling axis, promoting vascularization and pulp regeneration—complementing our meta-analytic findings across studies utilizing SHED- and DPSC-derived EVs.
Disparities between our pooled effect sizes and those implied in individual studies, such as Zhou et al. 31 and Guo et al., 17 likely reflect differences in study design, follow-up duration, and EV characterization. For instance, Guo et al. reported functional regeneration of pulp-periodontal tissue in avulsed teeth using exosome-laden decellularized matrices in a large-animal model, emphasizing the translational potential of EVs. Yet, due to the lack of quantitative outcome data compatible with meta-analysis, such studies were excluded from our pooled analysis. Their absence may slightly underestimate the overall regenerative efficacy observed in real-world models.
Furthermore, the consistent pro-regenerative effects across multiple studies—despite variations in EV source, isolation method, and delivery—underscore the robustness of EVs as a biological platform. However, substantial variability was observed in EV isolation approaches (e.g. differential ultracentrifugation, precipitation-based kits, size-exclusion methods), dosing strategies, and reporting metrics, with EV quantities expressed inconsistently as protein mass, particle number, or concentration per volume. This lack of standardization complicates cross-study comparison, contributes to heterogeneity in effect estimates, and poses challenges for reproducibility and translational interpretation. However, as highlighted in Shimizu et al., 23 the absence of standardized reporting on experimental blinding and randomization continues to be a limitation across preclinical EV studies. This issue was also evident in our SYRCLE bias assessments and reinforces the need for improved adherence to methodological guidelines such as Animal Research: Reporting of In Vivo Experiments (ARRIVE) 2.0.
Taken together, our findings are in broad agreement with recent literature, demonstrating that EV-based therapies—particularly those derived from DPSCs, SHEDs, and SCAPs—exhibit promising potential to enhance angiogenic and odontogenic regeneration in vivo. Yet, variation in outcomes by EV source, model system, and delivery strategy highlights the need for standardized protocols and well-powered comparative studies to guide future clinical translation.
Translational considerations
Despite the robust regenerative effects observed in preclinical models, several translational challenges must be addressed before EV-based therapies can be applied clinically. First, dose scaling remains unresolved, as EV doses in animal studies are commonly reported in micrograms or particle counts without standardized normalization to body weight, defect volume, or tissue surface area, complicating extrapolation to human applications. Second, delivery strategies are highly variable and often scaffold-dependent; while hydrogels, collagen matrices, and treated dentin matrices improve local retention in small-animal models, their clinical feasibility, handling properties, and regulatory classification require careful evaluation. Third, the stability, biodistribution, and off-target effects of locally delivered EVs in humans remain insufficiently characterized. Finally, large-scale clinical translation will require Good Manufacturing Practice (GMP)-compliant EV production, standardized isolation and characterization protocols, batch-to-batch consistency, and validated potency assays. Collectively, these factors highlight that although EV-based therapies show strong biological efficacy in vivo, substantial methodological and regulatory hurdles must be overcome before routine clinical implementation in regenerative endodontics.
Limitations
This meta-analysis has several limitations that should be acknowledged. First, despite applying rigorous inclusion criteria, the number of studies eligible for quantitative synthesis was relatively small, limiting the statistical power of subgroup and sensitivity analyses. Second, high heterogeneity was observed across studies, which may be attributed to differences in animal species, experimental models, EV sources, dosing regimens, delivery scaffolds, and outcome assessment methods. Specifically, heterogeneity may arise from the use of different animal species (mice, rats, dogs, and miniature pigs), diverse defect and implantation models (direct pulp exposure, root canal regeneration, periodontal defects, subcutaneous implantation, and Matrigel plug assays), and wide variation in follow-up durations ranging from a few days to several months. Additional variability is introduced by scaffold dependency, as studies employed different carrier systems, as well as by heterogeneous outcome assessment methods, including histological scoring, immunohistochemistry, micro-CT metrics, gene/protein expression assays, and angiogenesis-specific functional tests. Together, these factors substantially contribute to between-study variability and limit the comparability of pooled effect sizes. Third, many included studies lacked sufficient methodological transparency—particularly in randomization, allocation concealment, and blinding—resulting in an overall unclear risk of bias based on the SYRCLE tool. Importantly, unclear reporting of random sequence generation and blinding of outcome assessment may have led to inflated effect estimates in some studies, particularly for subjective or semiquantitative outcomes such as histological scoring and immunohistochemical analyses. In addition, variability in methodological rigor across studies is likely to have contributed to the substantial between-study heterogeneity observed in the meta-analyses. These sources of bias reduce confidence in the precision of pooled effect sizes and further support cautious interpretation of the magnitude of observed effects. Fourth, the absence of standardized EV isolation, characterization, and dosing protocols across studies represents a major limitation. Included studies employed diverse isolation techniques, variable characterization panels, and heterogeneous dosing metrics, often without reporting purity or yield in a comparable manner. These inconsistencies hinder reproducibility, limit direct comparison of effect sizes across studies, and represent a significant barrier to the clinical translation of EV-based therapies. Finally, publication bias could not be formally assessed due to the small number of studies per outcome, potentially overestimating treatment effects. Although comprehensive manual reference screening was performed, the literature search was limited to Web of Science and Scopus, and relevant studies indexed exclusively in other databases may have been missed. Additionally, the magnitude of pooled SMDs may be inflated by small sample sizes and low within-group variance inherent to preclinical models, further limiting direct clinical generalizability.
A major limitation of this meta-analysis is the presence of very high statistical heterogeneity, with I2 values exceeding 80% for both mineralization and angiogenesis outcomes. Such heterogeneity substantially limits the generalizability of the pooled effect estimates and indicates that the magnitude of treatment effects varies widely across studies. This variability likely reflects differences in animal species, experimental models, follow-up durations, EV sources, dosing strategies, scaffold or carrier use, and outcome assessment methods. Consequently, the pooled results should be interpreted as reflecting overall directional efficacy rather than precise quantitative effect sizes applicable across all preclinical contexts.
Strengths
Despite these limitations, this study has several notable strengths. To our knowledge, it is the first meta-analysis to focus exclusively on in vivo applications of EVs in regenerative endodontics. By adhering to PRISMA and Cochrane guidelines and incorporating hierarchical outcome definitions, we ensured methodological rigor and minimized selective reporting. The use of advanced meta-analytic techniques, such as the HKSJ adjustment, allowed us to account for between-study heterogeneity with improved statistical accuracy. Additionally, subgroup analyses by EV source provided new insights into the comparative regenerative potential of EVs derived from different stem cell types, which is critical for future therapeutic selection.
Future directions
Future research should prioritize methodological standardization to improve study quality and reproducibility. This includes adopting ARRIVE 2.0 reporting guidelines, using validated EV isolation and characterization protocols, and implementing blinded outcome assessments. Comparative studies that directly evaluate different EV sources, doses, and delivery systems in clinically relevant animal models are needed to identify optimal therapeutic strategies. Furthermore, long-term in vivo studies assessing not only regenerative outcomes but also safety, immune responses, and integration with host tissue will be essential. The field would also benefit from omics-based approaches to characterize EV cargo and elucidate mechanisms underlying their pro-regenerative effects. Finally, progressing toward well-designed clinical trials will require scalable, GMP-compliant EV production and regulatory frameworks tailored for acellular regenerative products.
Conclusion
This systematic review and meta-analysis provide preclinical evidence supporting the efficacy of EV therapies in promoting mineralization and angiogenesis in REPs. While findings were consistent across various EV sources—particularly DPSCs, SHED, and SCAP—outcome magnitude varied by stem cell origin and delivery method. Despite current methodological shortcomings and heterogeneity, the consistent pro-regenerative trends affirm the promise of EVs as a cell-free therapeutic strategy. To accelerate clinical translation, future preclinical studies must adopt standardized methodologies and reporting, and ultimately, well-controlled human trials are warranted to validate the clinical utility of EV-based therapies in endodontics.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261433133 - Supplemental material for Extracellular vesicle therapies in regenerative endodontic procedures: A systematic review and meta-analysis of in vivo studies
Supplemental material, sj-docx-1-sci-10.1177_00368504261433133 for Extracellular vesicle therapies in regenerative endodontic procedures: A systematic review and meta-analysis of in vivo studies by Madina A Kurmanalina, Nader Tanideh, Amin Tamadon, Aruzhan M Aitmukhanbetova, Nadiar M Mussin, Maryam Mardani, Mahdi Mahdipour, Shahrokh Zare and Ramazon Safarzoda Sharoffidin in Science Progress
Footnotes
Acknowledgments
The authors acknowledge the use of AI–based tools for language editing and grammar refinement only. No AI tools were used for data analysis, study design, or interpretation of results. The authors take full responsibility for the content of the manuscript.
Author contributions
Conceptualization: Madina A. Kurmanalina and Amin Tamadon; methodology: Amin Tamadon and Nader Tanideh; protocol registration: Amin Tamadon; investigation: Madina A. Kurmanalina and Aruzhan M. Aitmukhanbetova; data curation: Madina A. Kurmanalina and Aruzhan M. Aitmukhanbetova; formal analysis: Amin Tamadon; visualization: Madina A. Kurmanalina and Amin Tamadon; writing—original draft: Madina A. Kurmanalina and Amin Tamadon; writing—review and editing: Amin Tamadon, Nader Tanideh, Shahrokh Zare, Aruzhan M. Aitmukhanbetova, Mahdi Mahdipour, and Maryam Mardani; supervision: Nader Tanideh and Shahrokh Zare; resources: Nadiar M. Mussin and Mahdi Mahdipour; project administration: Amin Tamadon; and funding acquisition: Nader Tanideh.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research is financially supported by Shiraz University of Medical Sciences (Grant No. IR.SUMS.REC.1404.227).
Conflicts of interest
Authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data availability statement
Data is available and presented in study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
