Abstract
Zika virus (ZIKV) is a flavivirus of significant epidemiological importance, utilizing various transmission strategies and infecting “immune privileged tissues” during both the pre- and postnatal periods. One such transmission method may involve extracellular vesicles (EVs). EVs can travel long distances without degrading, carrying complex messages that trigger different responses in recipient cells. They can easily cross specialized tissue barriers, such as the placental barrier and the blood-brain barrier, which protects the central nervous system. It is known that some viruses can hijack and exploit the EVs biogenesis machinery to package regulatory elements, viral segments, and even complete viral genomes. This allows them to evade the immune system, amplify their tropism, and enhance their spread. ZIKV likely uses EVs produced by infected cells to insert its genomic RNA or parts of it. This mechanism can ensure viral entry and infection of the nervous tissue, partly explaining its broad viral tropism and silent persistence in various tissues and organs for months. This narrative review summarizes the main features of ZIKV and EVs, highlighting the most recent evidence on the involvement and effects of EVs during ZIKV infection. It also discusses the possibility of EVs acting as carriers of ZIKV through the nervous tissue.
Introduction
This review is guided by the scale for the assessment of narrative review articles (SANRA). 1
Zika virus (ZIKV) is a flavivirus of significant epidemiological importance due to its transmission strategies and its capacity to infect “immune-privileged tissues” such as testes, eyes, placenta, and the nervous tissue, particularly in children during pre and post-natal stages. 2 Fetal infection with ZIKV causes various neurological consequences collectively known as “congenital zika syndrome” (CZS), with microcephaly being one of the most evident complications. 3 However, it is now known that most children exposed to ZIKV in-utero are born with a normal cranial size, although neurological alterations can be developed after birth as sequelae of the infection. 4
The latter reinforces the need to study the different possible strategies for ZIKV entry into the nervous tissue. Some hypothesis suggests that the spread of ZIKV through the hematogenous route and the efficient infection of microvascular brain endothelial cells (MBECs) allow viral migration through the tissue parenchyma. 5 Other studies suggest viral entry into the nervous tissue through infected immune cells in a Trojan horse mechanism, where the infected cells carry the virus to immune-privileged tissues like the testicles and brain. 6
The extracellular vesicles (EVs) secreted by different cells have the capacity to travel long distances without degrading. In some viral infections, they transport parts or complete viral genomes, viral proteins, and other regulative molecules, which can affect the physiology of tissue-resident cells, without the need for assemble virions. 7 Once inside the tissue, the virus and possibly the EVs can modulate various cell functions, affecting the nervous system resident cells, such as neurons, astrocytes, pericytes, microglia and even the MBECs.
Given the importance of EVs as communication and signaling elements, produced under both physiological and pathological conditions, and their dual nature that can either aid or counteract viral spread and disease progression, we compiled a narrative review that summarizes the key processes of ZIKV infection and EVs biogenesis, analyzes the evidence regarding the involvement of EVs during ZIKV infection, highlights the similarities between EVs and viruses, and discusses the possibility that EVs might act as carriers of ZIKV through its entry into the nervous tissue.
Key insights into ZIKV biology and pathogenesis
ZIKV (family flaviviridae) is an enveloped virus with an icosahedral shape and a single-stranded RNA genome of 10.8 kb, containing an unique open reading frame (ORF).8,9 Once inside the cell, the viral genome is directly translated into a polyprotein that is processed by both host and viral proteases into three structural proteins named capsid (C), membrane (prM/M), and envelope (E), along with seven non-structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B y NS5), that are essential for genome replication, polyprotein processing, and manipulation of host cell processes that benefit viral infection,2,10,11 Figure 1.

ZIKV transmission primarily occurs through the bites of female mosquitoes of the genus Aedes, typically Aedes aegypti and Aedes albopictus.8,12 Other forms of transmission include blood transfusion, sexual contact (accounting for 3% of reported cases), and the transplacental or vertical route (mother-to-fetus), estimated to occur in 20–30% of infected pregnant women13–15 (the Suppl Table 1 shows a comparison of ZIKV and other vertical transmitted viruses). The impact of the timing of infection on fetal transmission remains difficult to establish; however, infections during the first trimester of pregnancy are associated with a higher risk of inducing congenital damage. 16
A variety of in vivo models have been employed to investigate ZIKV vertical transmission, each offering unique insights into maternal-fetal dynamics and pathogenesis. For example, immunocompetent C57BL/6J mice infected intravenously with Puerto Rican ZIKV (PRVABC59) exhibit placental damage and fetal growth restriction but lack direct vertical transmission. 17 Immunodeficient Ifnar1-/- and AG6 mice, infected via different pathways, reveal ZIKV's ability to cross the maternal-fetal barrier and cause fetal abnormalities.18,19 Notably, vaginal exposure models using Ifnar1-/- mice suggest direct transmission from the vaginal tract, underscoring the importance of the route of infection. 20 Additionally, Atg16l1-deficient mice demonstrate that autophagy modulation can limit ZIKV placental infection, reducing fetal impact. 21 These models collectively highlight strain-specific and infection routes differences, along with immune impacts, and placental invasion mechanisms that contribute to our understanding of ZIKV's vertical transmission and fetal susceptibility (Suppl Table 2).
Importantly, about 80% of ZIKV cases are asymptomatic. Typically, the virus causes a self-limiting mild illness, associated with fever, rash, joint pain, and conjunctivitis. Symptoms such as diarrhea, abdominal pain, anorexia, and myalgia have also been reported. 14 Complicated symptoms include myelitis and meningoencephalitis, 22 and in some cases, Guillain-Barré syndrome (GBS), an acute peripheral neuropathy that causes ascending paralysis.23,24 The relationship between ZIKV and microcephaly was confirmed after the virus was isolated from the amniotic fluid of microcephalic fetuses and from the brains of deceased fetuses with evident anomalies in their nervous systems. 23 Additionally, RNA and viral antigens were detected in placentas from spontaneous abortions. 14 These findings were crucial in defining CZS.
Mechanisms of ZIKV entry into the central nervous system (CNS)
The mechanisms by which ZIKV enters and replicates within the pre- and postnatal brain remain poorly understood. It has been hypothesized that ZIKV may exploit the endothelial tissue separating the bloodstream from the CNS, facilitating viral spread in fetuses during the first trimester of gestation. However, it remains unclear how ZIKV infiltrates tissues with mature, selective, and relatively impermeable endothelium, such as the cerebrovascular system, and how it efficiently persists within these tissues without compromising the integrity of the mature blood-brain barrier (BBB). 25
A study using type I interferon knockout mice (Ifnar1tm1.2Ees) and AG129 mice infected subcutaneously with various ZIKV strains, demonstrated that viral presence in cells of the choroid plexus and meninges was detectable from the third day post-infection (dpi). Pericytes were identified as the primary infected cells. By the fourth dpi, viruses were detected in multiple areas of the cerebral cortex, yet infection was not observed in the endothelial cells (EC) of capillaries, even up to 6 dpi. These findings suggest that ZIKV may infiltrate nervous tissue via its circulation, potentially exploiting the blood-cerebrospinal fluid (B-CSF) barrier rather than affecting the BBB. 26
Other hypotheses suggest that monocytes may serve as efficient carriers of the ZIKV due to their high capacity for transmigration across endothelial barriers, such as the BBB. 20 Indeed, CD14 + and CD16 + monocytes and dendritic cells (DCs) appear to be primary targets for ZIKV infection, allowing these cells to infiltrate tissues and act as “Trojan horses,” transporting the virus to immune-privileged sites such as the placenta, testes, and brain. 6 In vitro, ex vivo, and in vivo assays have confirmed that ZIKV infection promotes monocyte transmigration through endothelial barriers, thereby facilitating viral dissemination. 20
Different studies suggest that ZIKV can traverse the BBB by infecting MBECs. Although infected MBECs undergo activation, the infection does not compromise their viability, nor does it induce increased barrier permeability. This suggests that MBECs may release the virus through their basolateral surfaces via a transcytosis mechanism that preserves BBB integrity. 5 However, persistent viral replication and the ensuing host inflammatory response could potentially disrupt this barrier over time, with variability depending on the ZIKV strain involved. 27
Another option for virus entry into nervous tissue lies in the entry of ZIKV indirectly, through other mechanisms. One of them may be through EVs, in which part of the genome, viral proteins or other contents can be packaged, which directly affect the physiology of the cells.
EVs and viruses, a love and hate story
All you need to know about EVs: key insights
According to their biogenesis, EVs are classified into three main groups: I) apoptotic bodies (500 nm - 2 µm), coming from cell disintegration after an apoptosis process, II) ectosomes or microvesicles -MV- (100–1000 nm), originated by the budding of the plasma membrane (PM), and III) exosomes (30–200 nm), coming from an endocytic process and the formation of endosomes or multivesiculated bodies (MVB). 32
In general, EVs biogenesis processes begin when the contents or cargo programmed for packaging within EVs are directed to the site of production, either in the PM (for MV) or in the limiting membrane of intraluminal vesicles (ILVs) of MVB (for exosomes). 33 Cargo are then enriched and accumulate in the forming vesicles through a gradual mechanism of clustering and budding, followed by their fission and release. It is important to mention that cargo are the first regulators of EVs formation, these being mainly dependent on the cell type and their physio-pathological state. 33 Figures 2 and 3 summarize the EVs biogenesis process for exosomes and MV respectively.
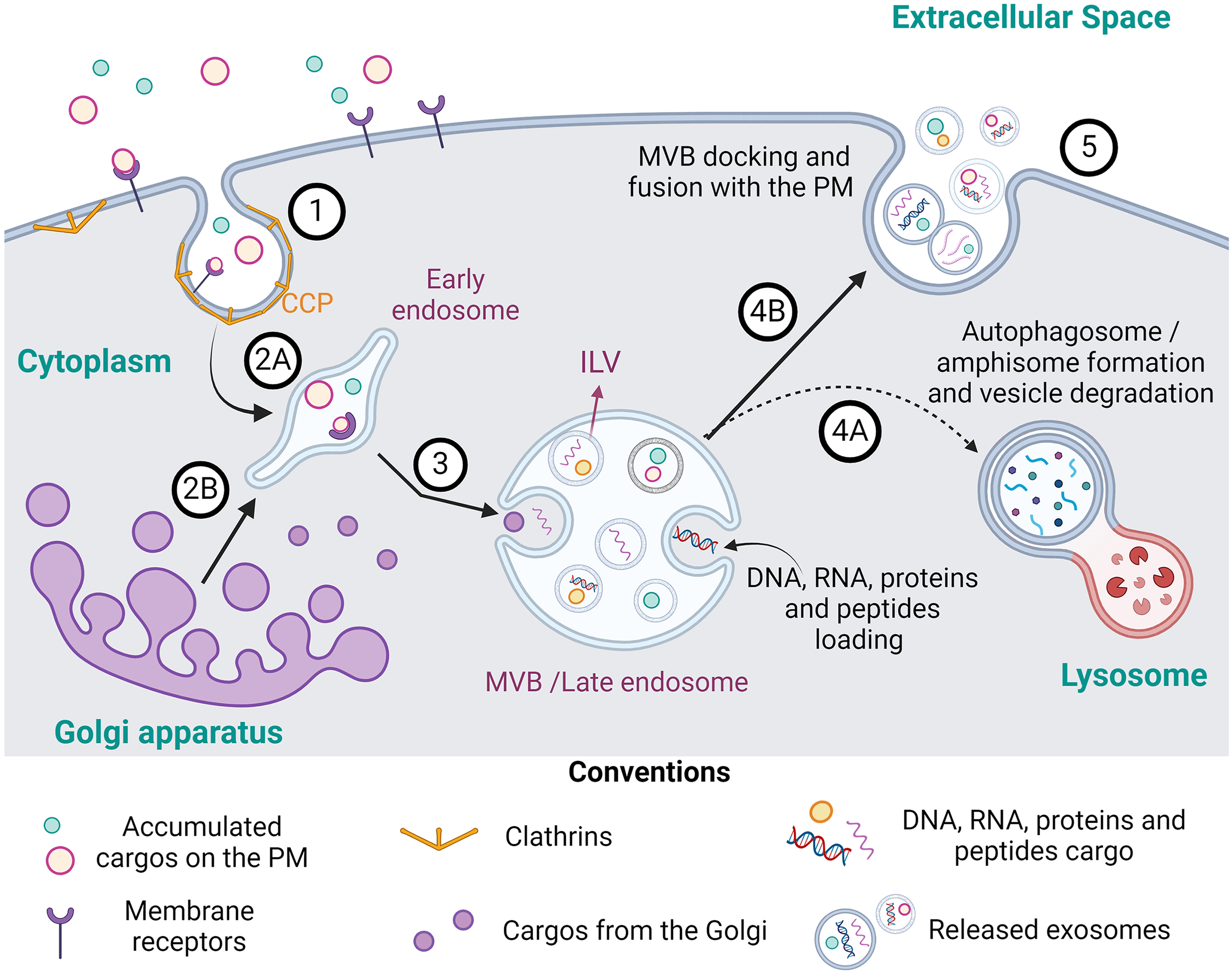

Regardless of whether they are MV or exosomes, EVs generally contain two classes of molecules: constitutive and cargo. Both types of molecules depend on the type of cell from which they are derived. Constitutive molecules regulate the localization, orientation, and uptake of EVs by recipient cells. In contrast, cargo molecules also depend on the physiological or pathological conditions of the cell that produces them. 40 Figure 4 summarizes the most common constitutive and cargo molecules reported.

Viruses as part of the cargo of EVs from infected cells
Due to their structural and functional similarities, viruses share several characteristics with EVs:
Form of entry into the cell: Both EVs and viruses bind to the plasma membranes of recipient cells to enter through fusion or endocytosis, triggering specific reactions in these cells.
45
Transport of genetic material: EVs, like viruses, can transport genetic material that modulates the functions of recipient cells and may facilitate viral replication.
46
Biogenesis: Some viruses utilize proteins and/or the ESCRT complex during their replication cycle.
47
First, how is it possible for viruses (or parts of them) to be loaded into EVs?
Second, why is this convenient for the viruses?
The EVs as Agents of Viral Spread
Thus, EVs, which normally stimulate physiological responses in recipient cells, can be “hijacked” by viruses to negatively regulate immune responses and increase susceptibility to infection, cellular death, or differentiation of recipient cells.33,48 Below, we will discuss some key aspects of the virus-EVs relationship.
As a result of the hijacking of their production machinery, EVs secreted by infected cells can carry viral particles or genetic material and transfer them to uninfected cells. 48 Consequently, EVs appear to play a significant role in viral propagation. This mechanism provides viruses with a means for escaping immune surveillance.47,48 For example, EVs from mosquito cells (line C6/36) infected with DENV-2 carry the complete viral genome, which retains the ability to infect human skin keratinocytes and EC.50,51 A similar phenomenon occurs with the hepatitis C virus (HCV), where productive infection can be established from EVs containing the complete viral genome. 50 This indicates that the assembly of viral particles is not always necessary for the infection of susceptible cells. 47
The encapsulation of viruses within EVs shields viral proteins, allowing viruses to persist and spread despite neutralizing antibodies. This packaging increases viral resistance to environmental stressors (like detergents, temperature and UV radiation), enabling EVs to act as unique pathogenic units through an “en-bloc” transmission mechanism. 52
Likewise, EVs can increase the susceptibility of cells to infection by transporting host receptors that facilitate virus entry into cells that normally do not express these receptors or do so at low levels. For example, EVs from SARS-CoV-2-infected cells transfer the viral receptor ACE2 and the CD9 tetraspanin to uninfected EC. CD9 facilitates the aggregation of ACE2 on the cell surface of recipient cells, enhancing their sensitivity to infection by this virus, 53 (Suppl table 3). Similarly, the HIV-1 viral receptor CCR5 is carried on MV and transferred to EC that do not endogenously express it, thereby favoring infection. 47 Additionally, the HIV-1 Nef protein can be carried in EVs, increasing viral spread by making recipient cells more susceptible to the virus. It can also induce senescence or cell death in CD4+ T lymphocytes or decrease their incorporation into T cell EVs, preventing the binding of virions to vesicular CD4 and increasing the amount of circulating virus available to infect other cells.48,54
Finally, another property of viral particle transport in EVs lies in the modulation of viral tropism. For example, some viruses alter the lipid and protein composition of EVs to enhance their binding and adsorption to susceptible cells. This has been observed in monocytes infected with ZIKV, where the secretion of EVs enriched with phosphatidylserine (PS) was documented. 55 Furthermore, the “pseudoenvelopment” of non-enveloped viruses by host cell membranes may broaden their tropism, as EVs surface proteins can direct viruses to specific recipient cells. This receptor-independent mechanism of viral entry has been proposed for both hepatitis B and C viruses (HBV, HCV) 47
• Modulation of the Immune Response: A Function of EVs from Infected and Uninfected Cells
Uninfected cells release EVs that can protect other cells from viruses by sequestering viral particles, thereby preventing attachment and entry. For instance, saliva-derived EVs (saEVs) inhibit ZIKV infection by blocking viral binding to target cells, potentially representing an oral innate immune defense mechanism, 56 (Suppl Table 4). Similarly, EVs from semen and vaginal secretions can block HIV-1 infection in vitro. 57
On the other hand, EVs from virus-infected cells, might contain viral and host factors, that play a role in immune responses; some authors have postulated that EVs from infected cells carry IFN intermediates or IFN-stimulated genes (ISGs), that inhibit viral replication and spread in recipient cells. 48 For example, the EVs from HSV-1 infected cells carry the stimulating interferon genes protein (STING) and other factors that activate ISG15 and ISG56, and inflammatory genes like IL-6 and IL-1β, inhibiting viral replication. Another case was reported for influenza A virus (IAV)-infected cells, which release EVs that deliver the miRNA hsa-miR-1975, triggering IFN-dependent antiviral responses in recipient cells 58 (Suppl Table 3).
Furthermore, EVs from infected cells may transport IFN-induced transmembrane proteins (IFITMs), which restrict viral entry to target cells (as seen in HCV) and bolster adaptive immunity. For example, EVs from IAV-infected cells carry IFITMs that stimulate the secretion of cytokines like IL-6 and TNF-α, while those from DENV-infected cells carry IFITM2 proteins that prevent DENV entry. 47 Our group also reported that EVs from macrophages infected with DENV-2, exert a protective effect on endothelial integrity, at least during the initial stages of infection 59 (Suppl Table 3). This is of great importance, as the secretion of proinflammatory cytokines and chemokines stimulated by EVs in recipient cells leads to the induction of the adaptive immune response and the recruitment of immune cells. 47
EVs as possible carriers of ZIKV and their role in ZIKV pathogenesis
ZIKV can disrupt developing nervous tissue, but to accomplish this, ZIKV must reach the tissue, meaning that virus from the infected mother needs to cross two important physiological barriers: the placental barrier (PB) and the BBB.
The placenta's main structural and functional units are the chorionic villi, composed of specialized epithelial cells known as trophoblasts, which serve as the primary defense against pathogens attempting to cross the blood-placenta barrier. 60 Evidence regarding the involvement of EVs in placental alteration indicates that following ZIKV infection, macrophages (permissive to the infection) produce large quantities of EVs, which are transported to the human placenta. These EVs induce the production of pro-inflammatory cytokines by trophoblasts, recruiting additional immune cells, thereby promoting inflammation, a crucial determinant of ZIKV pathogenicity. 61 Additionally, it has been reported that EVs from ZIKV-infected trophoblasts contain microRNAs (miRs) from the chromosome group 19 (C19MC miRNAs) that function as antiviral agents. These miRs, such as miR-517–3p, miR16B-5p, and miR512–3p, can be transferred to non-placental cells, conferring protection and upregulating autophagy in target cells, independent of interferon pathways 62 (Suppl Table 4). However, currently, many clinically relevant interactions between EVs and ZIKV remain unknown, and research is ongoing. 63
On the other hand, the BBB separates the lumen of blood vessels from the brain parenchyma, facilitated by highly regulated tight junctions between the EC of the brain microvasculature, which restrict paracellular permeability (gate function). These junctions also regulate the lateral diffusion of lipids and proteins between the apical and basolateral surfaces of the EC membrane (fence function). 64 Furthermore, the proper functioning and homeostasis of the BBB are regulated by astrocytes, pericytes, neurons, and microglia, along with the factors secreted by these cells, which are key in maintaining BBB stability. 65 Disruption of this stability increases EC permeability, a hallmark of CNS infection. Despite this, some reports indicate that ZIKV infection does not alter BBB permeability, suggesting that an endocytic transport system is required for the virus to cross this barrier. 60
Due to their characteristics, EVs can easily cross the BBB and PB, and serve as cargo shuttles for bidirectional communication between the CNS and the periphery, 60 so it is possible that ZIKV uses EVs to ensure its dispersion and entry into the nervous tissue. This was demonstrated by the work of York et al., 66 where infection with ZIKV modified the biogenesis and cargo of EVs, generating EVs of different sizes and densities that may be involved in various processes of viral pathogenesis and that could be directed to different cellular targets, with tetraspanin CD63 being essential for the release of EVs with infectious cargo (Suppl Table 4).
Similarly, in an in vitro BBB model established with EC from the human cell line hcMEC/D3, it was evident that EVs from ZIKV-infected cells, transported and delivered viral elements such as viral RNA and the NS1 and E proteins. These viral components altered EC stability by promoting the rearrangement of the inter-endothelial junction protein VE-cadherin, 67 (Suppl Table 4). Interestingly, a study by Zhao et al. showed that EVs derived from ZIKV-infected umbilical cord vein ECs (HUVECs) do not mediate the infection of susceptible cells despite carrying large amounts of E protein and viral RNA. In fact, the viral E protein was found distributed on the membrane surface of the EVs, where E-specific neutralizing antibodies are “distracted,” diminishing the effect of ZIKV infection and controlling the immune reaction. 68 (Suppl Table 4).
Conversely, the study by Zhou et al. 69 demonstrated that ZIKV infection promotes the release of EVs from neurons, which have the capacity to infect new cells. This suggests that EVs can not only increase the spreading capacity of the virus but also have the potential to alter neuronal survival and physiology by activating the expression of neuronal Sphingomyelinase (nSMase)-2/SMPD3, which regulates the production of exosomes. This activation is likely the basis of the neurological manifestations observed in brains exposed to ZIKV during embryonic development. 70 Regarding astrocytes, it has been shown that ZIKV infection is more efficient in single cultures of human fetal astrocytes compared to cultures of neurons or neuroprogenitor cells (NPCs) derived from the same fetal tissues, also generating a significant increase in the production of EVs. 71 (Suppl Table 4).
Other studies have reported that EVs from infected mosquito cells can modify the behavior of uninfected cells through the transmission of viral elements such as the E protein, which could be involved in the pathogenic mechanisms associated with the progression to severe forms of the disease.72,73 This is of great interest since in vitro evidence suggests that the ZIKV E protein is a sufficient stimulus to initiate a process of endothelial hyperpermeability by interacting with the Msfd2a transporter (selectively expressed by EC), promoting its degradation through the cellular ubiquitin-proteasome system. 74
Despite this, evidence of the participation of EVs from nervous tissue cells in the pathogenesis of ZIKV remains limited. Therefore, more complex models involving the different cell types that make up the BBB are needed to evaluate the role of EVs, not only in the infection of cells but also in their involvement in the pathogenesis of ZIKV in nervous tissue, particularly during advanced differentiation states such as those occurring during postnatal development.
Despite all the above-mentioned information, we acknowledge certain limitations of this review, primarily due to the limited published evidence evaluating: i) the experimental role of EVs in facilitating viral entry into nervous tissue, ii) the specific contents of EVs derived from the different infected cells, iii) the mechanisms through which EVs interact with target cells and release their cargo, and iv) their possible role in the pathogenesis and dysfunction of various nervous tissue cell types during infection.
Conclusions
Due to their nature, structure, and origin, EVs have the ability to cross the protective barriers of an organism. There is evidence that it is likely that EVs produced by cells infected with ZIKV transport active molecules that favor some of the viral processes, including their entry and spread in the nervous tissue, preventing their visibility to the immune system. Given that transport of the complete and assembled virus within the EVs is not necessary, it is likely that part of the nervous tissue damage evidenced in children exposed to ZIKV at different times of tissue development is a combined effect of different variables in which the viral and regulatory elements carried by EVs produced by infected cells are included. This is why it is necessary to establish highly complex in vivo and in vitro models that allow in-depth evaluation of the role of EVs from different types of infected cells in the pathogenesis and more specifically in the neuropathogenesis of ZIKV.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241312073 - Supplemental material for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis?
Supplemental material, sj-docx-1-sci-10.1177_00368504241312073 for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis? by María-Angélica Calderón-Peláez, Jaime E. Castellanos and Myriam L. Velandia-Romero in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504241312073 - Supplemental material for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis?
Supplemental material, sj-docx-2-sci-10.1177_00368504241312073 for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis? by María-Angélica Calderón-Peláez, Jaime E. Castellanos and Myriam L. Velandia-Romero in Science Progress
Supplemental Material
sj-docx-3-sci-10.1177_00368504241312073 - Supplemental material for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis?
Supplemental material, sj-docx-3-sci-10.1177_00368504241312073 for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis? by María-Angélica Calderón-Peláez, Jaime E. Castellanos and Myriam L. Velandia-Romero in Science Progress
Supplemental Material
sj-docx-4-sci-10.1177_00368504241312073 - Supplemental material for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis?
Supplemental material, sj-docx-4-sci-10.1177_00368504241312073 for Extracellular vesicles in ZIKV infection: Carriers and facilitators of viral pathogenesis? by María-Angélica Calderón-Peláez, Jaime E. Castellanos and Myriam L. Velandia-Romero in Science Progress
Footnotes
Acknowledgements
Vice-chancellor of research of Universidad El Bosque for the support and finance of MACP doctorate (2020–2022) in Universidad Nacional de Colombia. The doctorate was also supported by the Minciencias/Colfuturo scholarship for National Doctorate programs No. 9092 (resolution No. 0608 of June 21st, 2022).
Author contributions
Conceptualization: MACP, MLVR. Writing-original draft preparation: MACP, MLVR. Writing review and editing: MACP, MLVR, JEC. Visualization: MACP, MLVR. Project administration: MLVR; Funding acquisition: MLVR, JEC.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Not applicable
Funding
This work was supported by the Ministerio de Ciencia, Tecnología e Innovación de Colombia (Minciencias), and Universidad El Bosque under Grant number 130884467149 Contract 431–2020.
Ministerio de Ciencia, Tecnología e Innovación de Colombia (Minciencias), Universidad El Bosque, (grant number 130884467149 Contract 431-2020.).
Consent to participate
Not applicable
Consent for publication
Not applicable
Data availability
The authors confirm that all data supporting this review are available within the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
