Abstract
Infective endocarditis (IE) remains a life-threatening condition with high global morbidity and mortality, demanding more effective strategies for early detection and management. Precision medicine offers a transformative approach, starting with comprehensive risk profiling that combines genetic predisposition, preexisting comorbidities, and lifestyle factors such as intravenous drug use and oral hygiene. This paradigm extends to point-of-care diagnostics and culminates in targeted, patient-specific treatment strategies. To explore its potential in IE, we systematically searched PubMed, Scopus, and Embase, identifying and synthesizing key literature into a narrative review. Our findings highlight precision medicine as a promising framework to enhance timely diagnosis and support individualized therapeutic decision-making in IE.
Keywords
Introduction
Sir William Osler first described the condition known as infective endocarditis (IE) in the 1890s. It was first proposed as a cohesive theory, according to which susceptible patients experienced “fungal” growth in their valves that subsequently “transferred to distant parts of the body.” 1 With an estimated incidence ranging from 1.5 to 11.6 cases per 100,000 population and a mortality rate of approximately 25%. The incidence of IE is notably higher in individuals with cardiovascular implantable electronic devices, such as pacemakers and implanted cardioverter-defibrillators, as well as in those with prosthetic heart valves. 2 Rheumatic heart disease, which predominantly affects the mitral valve, has been recognized as a significant risk factor for the development of IE, and more recently injection drug use (IDU) has emerged as an important risk factor. 3
The majority of IE cases are caused by Gram-positive bacteria, particularly species of
Host risk factors are a crucial issue in IE development. 5 Current efforts should focus on the comprehensive characterization of both host and microorganism's factors, utilizing the principles of precision medicine. This approach would enable the identification of specific risk factors and guide the development of targeted interventions, potentially improving outcomes for patients at risk of IE. Understanding the interplay between host susceptibility and microbial virulence can lead to more personalized and effective preventive and therapeutic strategies. 6 Recent advancements in this field have been identified, underscoring the significance of individualized patient's management and highlighting potential future developments. We evaluated the application of precision medicine at the point of care (POC) for patients with IE in critical care settings and propose a precision medicine–based diagnostic algorithm through a narrative review after evaluation of relevant academic sources.
This narrative review aims to examine how precision medicine principles can be translated into bedside and POC applications in IE, integrating pathophysiological insights, advanced diagnostics, and individualized therapeutic strategies to support real-time clinical decision-making.
Methods
This review employs a structured literature analysis approach, guided by the Scale for the Assessment of Narrative Review Articles. 7 Through a systematic evaluation of relevant academic sources, it aims to provide a comprehensive and evidence-based overview of current research on IE. The review presents an in-depth discussion of pathophysiology, risk factors, diagnostic strategies, current management practices, and future research directions, with a particular emphasis on the growing relevance of precision medicine.
A comprehensive literature search was conducted using the electronic databases PubMed/MEDLINE, Excerpta Medica Database, and Scopus. The search covered publications from January 2000 to December 2025, reflecting contemporary diagnostic methods, microbiological advances, and individualized therapeutic strategies relevant to precision medicine. Search terms included combinations of: IE, precision medicine, personalized medicine, and individualized medicine. Reference lists of key articles were also reviewed to identify additional relevant publications.
Eligible sources were peer-reviewed articles written in English, including clinical practice guidelines, randomized controlled trials, prospective and retrospective cohort studies, and high-quality narrative and systematic reviews. Studies were included if they addressed clinical, microbiological, diagnostic, prognostic, or therapeutic aspects of IE with relevance to individualized or precision-based approaches. Exclusion criteria comprised case reports with limited generalizability, non–peer-reviewed publications, editorials without substantive data synthesis, and studies not directly related to endocarditis or precision medicine.
The literature analysis followed a structured process that included definition of the research objective, identification and selection of relevant studies, thematic categorization, extraction of key concepts, and comparative synthesis of findings. Emphasis was placed on integrating physiological, microbiological, clinical, and surgical perspectives, while highlighting how advances in genomics, biomarker profiling, imaging, and risk stratification contribute to precision medicine in IE.
Discussion
Precision medicine in infectious diseases
Precision medicine in infectious diseases is founded on an individualized, data-driven approach to clinical decision-making that integrates pathogen-specific characteristics, host factors, immune response, and individualized risk prediction. Its primary objective is to optimize therapeutic effectiveness, refine prognostic accuracy, and strengthen preventive strategies by tailoring interventions to biologically and clinically distinct patient profiles. 8
A central pillar of this approach is the integrated assessment of three core domains: precise characterization of the causative pathogen, comprehensive evaluation of host immune status, and analysis of host–pathogen interactions.9,10 Together, these elements allow a more accurate understanding of disease heterogeneity, enable targeted therapeutic strategies, and support improved prediction of outcomes and complications. 3
Technological innovation constitutes another fundamental pillar, with advances in molecular diagnostics, antimicrobial resistance profiling, and pathogen genomics enabling rapid and accurate etiological identification. 11 These tools are increasingly complemented by multiomics platforms—including genomics, transcriptomics, proteomics, and metabolomics—which collectively provide a systems-level biological framework for risk stratification and personalized therapeutic decision-making across diverse clinical settings.
Despite potential clinical benefits, the translation of precision medicine into routine infectious disease practice remains limited. 12 Persistent barriers include resource constraints, high costs, insufficient bioinformatics infrastructure, fragmented integration between clinical care and laboratory technologies, and the absence of robust regulatory and public health frameworks. Addressing these challenges requires specialized centers with interdisciplinary expertise and integrated clinical–microbiological–technological models capable of operationalizing precision medicine in complex infectious diseases.
Pillars of precision medicine in IE
Pathogen, host, and host–pathogen interactions
Precision medicine in IE is founded on the integrated assessment of three core pillars: the pathogen, the host, and their dynamic interaction. Precise pathogen characterization is essential, as microorganisms with specific virulence determinants—such as adhesins, biofilm-forming capacity, and adaptive resistance mechanisms—can colonize damaged endocardial surfaces and persist within vegetations, with organisms like
Pillars of precision medicine in infective endocarditis.
The pathogen: Virulence, persistence, and resistance
Diagnosing IE can be particularly challenging due to the wide variety of pathogens involved and the limitations of conventional blood cultures, which may yield negative results in 15% to 60% of cases.
17
POC molecular diagnostics in the context of IE typically employ Polymerase chain reaction (PCR), isothermal amplification methods, or microfluidic-based assays, allowing for the detection of fastidious organisms such as
Additionally, some emerging platforms integrate multiplex PCR panels that can detect both bacterial and fungal DNA, which is critical in prosthetic valve endocarditis or immunocompromised patients. The use of these strategies has achieved diagnostic rates of up to 92% in
Multiple studies have highlighted the importance of specific bacterial genotypes in the context of complex infections, particularly in
Subsequent investigations have confirmed that specific sequence types, including spa-CC008 (MLST CC8) and related lineages, are significantly associated with metastatic complications, persistent bacteremia, and an increased risk of abscess formation and septic embolization. These associations underscore the role of bacterial genetic background in shaping clinical outcomes. 23
The genetic characteristics linked to persistent
The host: Immune response and susceptibility
Precision medicine seeks to customize diagnostics and treatment based on an individual's unique genetic, environmental, and lifestyle characteristics. 24 This approach emphasizes not only tailored treatment but also risk assessment, prevention, and proactive health strategies. In the context of IE, a precision medicine approach begins with a thorough risk assessment to identify individuals at higher risk of developing IE. This assessment considers genetic predispositions, existing health conditions, and lifestyle factors, such as IDU or oral hygiene practices. By understanding these risk factors, healthcare providers can identify vulnerable patients and tailor prevention and treatment strategies, accordingly, minimizing the risk of disease onset.25,26
Host response also plays a critical role in disease expression. Transcriptomic profiling has demonstrated that distinct host endotypes during IE are significantly associated with clinical outcomes. Differential gene expression patterns influence inflammatory responses, mortality risk, and treatment response, underscoring the relevance of host biology in precision medicine. 27 Several studies have shown that specific transcriptomic endotypes in IE—commonly classified into distinct molecular subgroups EE1/EE2—are associated with differential mortality and clinical trajectories. These endotypes reflect heterogeneous immune activation states and inflammatory pathways that contribute to disease severity. 28 Similar observations have been reported in sepsis, where transcriptomic phenotypes (such as α, β, γ, and δ) are associated with differential treatment responses and individualized management strategies. These molecular profiles reflect variations in endothelial injury, immune dysregulation, and multidimensional host-response pathways, reinforcing the role of transcriptomics as a key pillar of precision medicine in severe infectious diseases. 29
On the other hand, genetic polymorphisms affecting the innate immune response contribute to individual susceptibility to IE. Variants involving cytokines and Toll-like receptors (TLRs) show the strongest associations. Evidence from genetic studies in patients with IE highlights polymorphisms in cytokine-related genes, with certain heterozygous genotypes being more frequent among affected individuals. 27
In particular, TLR polymorphisms—such as those involving TLR2 and TLR5—have been strongly associated with increased susceptibility to IE, sometimes in combination with other genetic variants. Additional studies report associations between polymorphisms in interleukin genes (including IL-1β and IL-12B) and altered risk or severity of disease. 30 Collectively, these findings suggest that functional genetic variations influencing innate immunity, including cytokine signaling, TLR pathways, and complement activation, play a relevant role in modulating host response to infection and the pathogenesis of IE. 31
Another key consideration is the spectrum of predisposing conditions consistently reported in the literature. Patients with native valve disease and those with prosthetic cardiac material present distinct risk profiles for IE. IE risk varies according to valve status and the presence of intracardiac devices. Native-valve disease is mainly associated with underlying structural abnormalities and conditions predisposing to transient bacteremia, whereas prosthetic valves and cardiac implantable electronic devices confer a higher lifelong risk due to foreign material favoring bacterial adherence and biofilm formation. These patients require closer surveillance and, in selected situations, prophylactic antibiotic measures (Table 1).6,32,33
Risk factors for infective endocarditis (IE).

IE associated with IDU predominantly affects the right heart, with
Accurate diagnosis of IE is facilitated by the 2023 Duke—International Society for Cardiovascular Infectious Diseases criteria, which incorporate clinical, microbiological, and echocardiographic assessments. 38 These diagnostic advancements were primarily driven by modifications in the major imaging and microbiological criteria.
Host–pathogen interaction: From pathophysiology to precision therapeutics
A comprehensive approach to the understanding, diagnosis, and management of IE requires the integration of diverse pathophysiological, medical, surgical, and supportive strategies to optimize patient outcomes. 5 Given the complexity of the disease, effective management often demands a collaborative, multidisciplinary effort. An IE team (IET) should include specialists such as an infectious disease specialist, microbiologist, radiologist, neurologist, cardiovascular surgeon, critical care specialist, physiotherapy/rehabilitation, psychiatrist and cardiologist (Figure 2). This team-based approach is essential for ensuring that all aspects of the disease are addressed, from accurate diagnosis to effective management and comprehensive patient care. IET should also integrate medical, mental health, and social determinants of care, aligning clinical management with psychosocial support. This team-based approach fosters coordinated decision-making, patient engagement, and treatment adherence, thereby supporting comprehensive and individualized care. 39

Infective endocarditis team (IET).
By focusing on personalized treatment plans, precision medicine adapts to each patient's distinct biological and genetic profile, considering variations that affect responses to medications and therapies. 40 By aligning antibiotic choices with the patient's genetic makeup, healthcare providers can improve disease management, especially for patients with known drug resistance or previous adverse reactions to certain antibiotics. Furthermore, coupling POC diagnostics with rapid antibiotic susceptibility testing or resistance gene detection supports precision medicine strategies 8 (Figure 3).
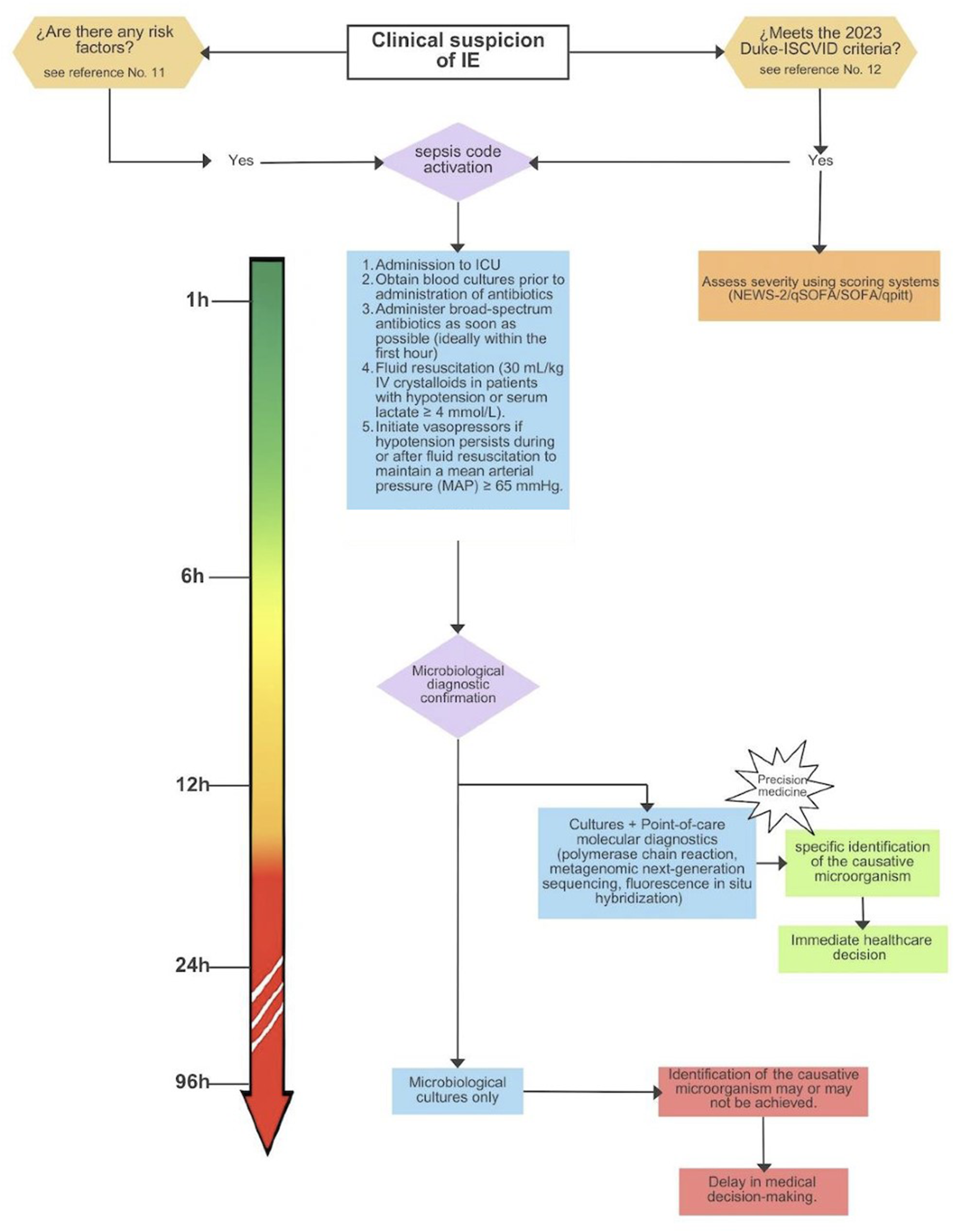
Precision medicine algorithm for infective endocarditis.
Understanding an individual's drug metabolism further supports personalized treatment, helping avoid medications that could cause harmful side effects. Pharmacogenomic insights enable accurate dosage adjustments according to metabolic rates, ensuring effective therapeutic levels without risking toxicity. 41 Leveraging artificial intelligence (AI) and genomic technologies, precision medicine can analyze intricate datasets, enabling targeted and individualized treatment strategies. AI facilitates identifying risk-prone subpopulations and predicting therapeutic responses. 42
Preventive strategies in IE leverage the comprehensive insights of precision medicine to address the multifaceted nature of the disease. Recognizing the complex interplay of genetic and lifestyle factors allows for targeted interventions, such as administering prophylactic antibiotics to high-risk patients undergoing dental procedures or recommending lifestyle modifications to reduce risk factors. 43 These preventive approaches are crucial in lowering the incidence of IE among at-risk populations.
Optimizing antimicrobial therapy requires an integrated understanding of pharmacokinetic and pharmacodynamic principles, which is essential for the comprehensive management of patients with IE within the framework of precision medicine. Individualized dosing strategies based on serum concentration monitoring aim to maximize therapeutic efficacy while minimizing toxicity. 44
Among these strategies, vancomycin dosing guided by area-under-the-curve/minimum inhibitory concentration (MIC) monitoring—rather than through levels alone—has gained relevance, as it improves efficacy and reduces nephrotoxicity. Similarly, for β-lactam antibiotics, maintaining plasma concentrations above the MIC for a sufficient proportion of the dosing interval, often through prolonged or continuous infusions, is a key pharmacodynamic objective. High-dose regimens, including daily doses exceeding conventional thresholds, may be required in selected cases such as infections caused by difficult-to-treat pathogens, with evidence supporting synergistic combinations (e.g. β-lactams with carbapenems) in specific scenarios. 45
Other agents, such as daptomycin, play an important role in prolonged treatments, particularly in cases of persistent bacteremia when surgical management is not feasible. 46 However, despite increasing pharmacological evidence, clinical consensus remains limited. Future progress will depend on larger studies and broader availability of individualized therapeutic drug monitoring to ensure optimized, patient-specific antimicrobial regimens in IE.
Surgical intervention may be indicated for patients who experience significant valve damage or have persistent infection despite antibiotic treatment.
6
Surgical options include valve repair or replacement and the debridement of infected tissues. These procedures are performed early, a concept that may appear conflicting; according to the American College of Cardiology/American Heart Association guidelines, early surgery is defined as an intervention during the initial hospitalization and before the completion of a full course of appropriate antibiotic therapy. The American Association for Thoracic Surgery guidelines state that once an indication for surgery is established, the procedure should be performed within a few days—either as an emergency (within 48 h) or as soon as clinically feasible.
47
Risk stratification in IE requires systematic assessment of valve type, anatomical extent of infection, and patient-specific factors by a multidisciplinary endocarditis team. Existing risk scores, largely derived from observational data, incorporate prosthetic valve involvement and paravalvular extension but lack universal validation.
48
Clinical decision-making therefore remains driven by infection localization, frailty, and comorbidities, as no IE-specific risk model is routinely applied in practice. Surgical indications in IE are driven by cardiac dysfunction, uncontrolled infection, embolic risk, and neurological status. Early surgery is strongly indicated for heart failure due to valve dysfunction, infection by
Finally, long-term monitoring is essential to maintain the efficacy of treatment in IE patients. Regular follow-ups allow healthcare providers to track patient progress and adjust treatments as needed in response to changes in health or new findings in precision medicine. 50 This continuous assessment ensures that treatment remains responsive and effective, promoting optimal patient outcomes over time.
A holistic approach to managing IE emphasizes the importance of IET, thorough new and real time diagnostic methods, tailored antibiotic regimens, potential surgical interventions, innovative adjunctive therapies, patient education, and ongoing follow-up. This integrated strategy not only aims to treat the infection effectively but also seeks to prevent complications and enhance the patient's overall quality of life.39,51
Present and future perspectives in precision medicine
Infectious diseases rank among the top 10 global public health concerns. According to the World Health Organization (WHO), these diseases are responsible for millions of deaths annually. This can lead to severe health outcomes, including significant morbidity and mortality, alongside economic impacts across the globe. 52
Precision public health (PPH) has become a critical tool in addressing these challenges. It aims to deliver the right intervention to the right population at the right time. Advances in technology, such as access to individual and population-level genetic risk data, socioeconomic insights, have significantly enhanced the precision of public health efforts, allowing interventions to be better targeted and delivered in more acceptable and impactful formats. 53
A large percentage of IE cases are attributed to odontogenic organisms, indicating that preventive investments in dental care could not only reduce the incidence of IE but also prove to be a cost-effective public health strategy. 54 PPH efforts must integrate real-time big data analysis, utilizing AI for risk classification within populations. This approach forms the cornerstone of precision medicine in the management of IE, enabling targeted interventions and improving outcomes by tailoring strategies to specific risk profiles. 55
For host-based assays to succeed in resource-limited areas, they must align with the WHO's ASSURED criteria: affordability, sensitivity, specificity, ease of use, rapid results, equipment-free operation, and accessibility for the most underserved populations. 56 Although these criteria may seem incompatible with precision medicine in low-resource settings, recent advances in technology are transforming this outlook. Many new POC assays emphasize low-cost, equipment-free models capable of multianalyte testing with high sensitivity and specificity. While initial costs may be higher than traditional diagnostics available in these regions, these assays hold promise for substantial long-term cost savings. 57
Machine learning (ML) can play a role as a clinical decision-support tool, offering high precision in the context of IE. It enables more accurate predictions and supports symbolic reasoning, as well as patient triage for those requiring surgical intervention. 58 Multiple studies have demonstrated the strong and robust prognostic capacity of ML models. For example, supervised learning algorithms have been developed to predict high-risk outcomes. Some of these models are capable of identifying key clinical variables and have shown significant effects in specific clinical settings, such as cardiac, renal, pulmonary, and theoretical triage scenarios. 59
Other studies have also demonstrated the development of different models, such as gradient boosting techniques. In recent cohorts, these models have been used to predict early mortality in both intraoperative and postoperative periods. 60 Important variables incorporated into these models include platelet count, serum albumin levels, and the size of vegetations. Similarly, several factors can be calculated to estimate embolic risk using variables such as the presence and size of vegetations. These studies show promising results and demonstrate improved prognostic capacity. 61 However, it is important to acknowledge that ML still has limitations, particularly in terms of interpretability, ethical considerations, and sample size. Therefore, within the framework of precision medicine, patient-specific characteristics must be carefully evaluated and taken into account when interpreting and applying these models. 58
Future studies on IE should focus on enhancing risk-based diagnostics, personalizing treatment, and improving clinical outcomes. Rapid molecular tests are now central to timely and accurate diagnosis, helping to identify pathogens in cases where cultures lack sensitivity or specificity. However, larger and more in-depth studies are essential to ensure proper interpretation of these tests. As research on the immune system and systemic responses to sepsis progresses, the need to tailor treatments to individual patient factors—such as risk profiles, genetics, and comorbidities—becomes increasingly clear. Finally, ensuring easy bedside access to new technologies and real-time data, is critical for effective decision-making. Figure 4 summarizes the role of precision medicine in IE.

Precision medicine and infective endocarditis.
Limitations
The limitations of this study include several factors. First, as a narrative review, this study is inherently subject to selection bias and does not follow the rigid methodology of systematic reviews. However, literature was selected based on relevance, scientific quality, and contribution to the conceptual framework of precision medicine in IE. Second, our study focused only in IE, as precision medicine is increasingly centered on microbial and imaging technologies, requiring more comprehensive hospital-based data specifically in non-IE patients. Finally, we acknowledge that our institution benefits from a multidisciplinary team that facilitates the implementation of precision medicine for all patients—an approach that may not be universally available in other countries.
Conclusion
Precision medicine provides a unifying framework that connects pathophysiology, diagnostics, therapeutics, and public health strategies in the management of IE. A personalized medicine strategy in IE is essential for optimizing timely diagnosis and management in critically ill patients, as it integrates all aspects of a patient's life into their care. An individualized approach improves understanding of patient-specific conditions, prioritizes diagnostic strategies, guides targeted antibiotic therapy according to comorbidities, determines decisions regarding surgical intervention, and addresses antibiotic resistance—ultimately enhancing clinical outcomes in critical care settings while leveraging advances in healthcare technology.
Footnotes
Acknowledgments
The authors thank the Fundación Clínica Shaio. ChatGPT 5.0 has been used for language corrections.
Authors’ contributions
Conceptualization: JOCB; data curation: JOCB, LMCG, and PACO; formal analysis: JOCB, LMCG, and PACO; methodology: JOCB, LMCG, and PACO; figures design; JOCB, PACO, and LMCG; supervision: JOCB and RBB; validation: JOCB, RBB, LMCG, and PACO; writing—original draft: JOCB, RBB, LMCG, and PACO; writing—final manuscript: JOCB, RBB, LMCG, and PACO.
Funding
The authors received no financial support for the research and/or authorship. Open access funding was provided by Fundación Clínica Shaio, Bogotá-Colombia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
