Abstract
Waardenburg syndrome (WS) is a rare genetic disorder characterized by congenital sensorineural hearing loss and pigmentary abnormalities, accounting for 2–5% of congenital deafness. While molecular testing is the diagnostic gold standard, clinical recognition remains crucial in low-resource or conflict-affected environments where specialized services are unavailable. We report a Syrian male in his early 20s who presented to the otorhinolaryngology clinic seeking exemption from military service, citing long-standing right-sided hearing loss. The patient and his family had never pursued medical evaluation for his pigmentary features or hearing problem. Examination revealed a white forelock, heterochromia iridis, synophrys, broad nasal root, and dystopia canthorum (W Index 2.2). Pure-tone audiometry demonstrated severe unilateral sensorineural hearing loss. Systematic neurological, ophthalmological, and musculoskeletal assessments were normal. Due to the lack of access to genetic testing, a clinical diagnosis of WS type I was made, and the patient was referred for genetic counseling. This case highlights diagnostic challenges in conflict-affected, resource-limited settings. Despite striking phenotypic features, the patient remained undiagnosed until adulthood. Missed opportunities included the absence of childhood hearing screening, delayed recognition of pigmentary signs, and a lack of educational or psychosocial support. Literature indicates that phenotypic diagnosis is reliable when multiple major criteria are present, yet diagnostic delays significantly affect quality of life. This report underscores the importance of timely recognition of WS in low-resource contexts. Strengthening primary care awareness, implementing basic audiological and pigmentary screening, and integrating psychosocial support may help mitigate diagnostic delays and improve long-term outcomes for patients with rare genetic disorders.
Keywords
Introduction
Waardenburg syndrome (WS) is a collection of inherited disorders characterized by congenital hearing loss and distinctive physical features, affecting roughly 1 in 40,000 individuals. It is responsible for 2–5% of all cases of congenital deafness.1,2 Four clinical subtypes have been described, with type I (WS1) first outlined by Waardenburg in 1951 and distinguished by dystopia canthorum.1–4 Although molecular confirmation is ideal, WS can often be reliably diagnosed based on phenotypic criteria. In low-resource settings, where genetic testing and specialized services are unavailable, recognition of clinical features becomes the cornerstone of diagnosis.1,2 We report a Syrian male in his early 20s who was incidentally diagnosed with Waardenburg syndrome. This case is notable for the uncommon presentation of unilateral sensorineural hearing loss in Waardenburg syndrome type I and for the reliance on clinical assessment in the absence of genetic testing, which remains inaccessible in many resource-limited settings. Moreover, the patient's low level of health awareness contributed to delayed medical evaluation despite long-standing symptoms. Together, these elements underline the clinical and educational value of this report and highlight the broader challenges of diagnosing rare genetic disorders in environments with constrained diagnostic resources. The reporting of this study conforms to CARE guidelines. 5
Case presentation
A Syrian male in his early 20s presented to the otorhinolaryngology clinic in Yusuf al-Azma Hospital in mid-2023 seeking a medical exemption from compulsory military service, reporting long-standing right-sided hearing loss. His main motivation for consultation was administrative, and neither he nor his family had previously considered his hearing problem or pigmentary features as medical conditions warranting evaluation.
According to his history, the hearing loss had been present since childhood, but no formal audiological assessment had ever been performed. There was no history of otitis media, trauma, or exposure to ototoxic medications. His parents confirmed that developmental milestones were otherwise normal, and the patient completed basic schooling. No prior interventions or hearing devices had been used.
Family history revealed no consanguinity, and no relatives were reported to have pigmentary abnormalities or hearing loss. This information was based solely on the patient's account.
During the patient's evaluation (Figure 1), he had a white forelock at the front of his hair, along with heterochromia iridis (the left iris was light blue, the right iris was a mix of blue and hazel).

Clinical photograph demonstrating the characteristic phenotypic features: white forelock, heterochromia iridum, synophrys, dystopia canthorum, and broad nasal root.
He also had synophrys, a broad and high nasal root, and the W index was 2.2 (consistent with dystopia canthorum).
Rinne's test was positive in both ears, while Weber's test showed lateralization to the left, suggesting sensorineural hearing loss in the right ear. The patient confirmed that he was an only child, his parents were non-consanguineous, and no family members exhibited similar phenotypic features.
Pure-tone audiometry (Figure 2) revealed severe sensorineural hearing loss in the right ear (threshold > 50 dB).

Right ear pure tone audiometry showing sensorineural hearing loss (threshold > 50 dB).
Due to the inability to perform genetic testing in Syria and the patient's financial constraints in seeking testing abroad, a clinical diagnosis of Waardenburg syndrome was made. A series of consultations was requested:
- Dermatology consultation showed no skin abnormalities. - Gastroenterology consultation revealed no pathological findings. - Radiological and ultrasound imaging showed no abnormalities.
Unfortunately, no medical intervention, such as hearing aid fitting, was undertaken, as the patient declined this option. The refusal may have been influenced by financial constraints limiting his ability to afford the device, as well as by contextual concerns related to his social environment, including fear of losing perceived sympathy or accommodations associated with his condition during military service.
Discussion
In 1951, Dutch ophthalmologist Petrus Johannes Waardenburg first identified a rare genetic condition now known as Waardenburg syndrome (WS). 6 This disorder occurs in roughly 1 in 40,000 individuals and accounts for about 3% of congenital deafness cases. It affects all ethnicities equally and shows no gender bias. 7 Diagnosis relies on specific criteria, categorized into major and minor indicators (Table 1). A definitive WS diagnosis requires either two major criteria or one major criterion plus two minor features. 1
Diagnostic criteria of Waardenburg syndrome.
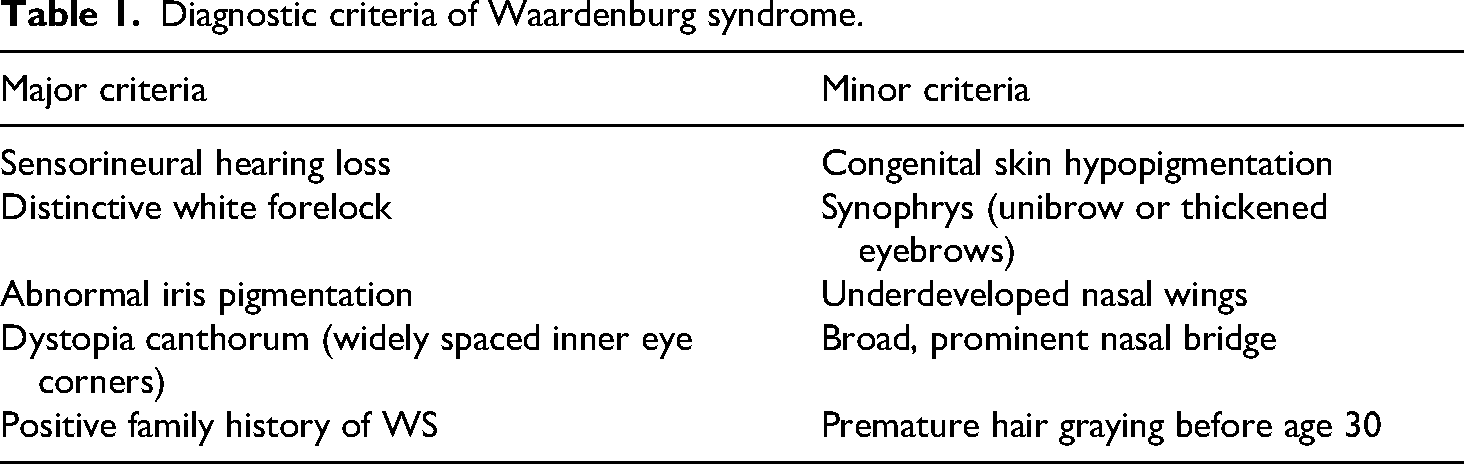
According to the diagnostic criteria proposed by Waardenburg, there are four major criteria in this case: dystopia canthorum, white forelock, heterochromia iridum, and sensory neural hearing loss. There are two minor criteria: a broad nasal root and synophrys. It is worth mentioning that having an affected relative as a diagnostic criterion may also be met, especially since the medical and family history relies solely on the patient's account without verification due to their refusal to cooperate or undergo examination. This is particularly relevant given that some cases may present with mild or unnoticed symptoms. 8
The syndrome is classified into four main types, each with unique clinical presentations (Table 2):
Classification of Waardenburg syndrome.

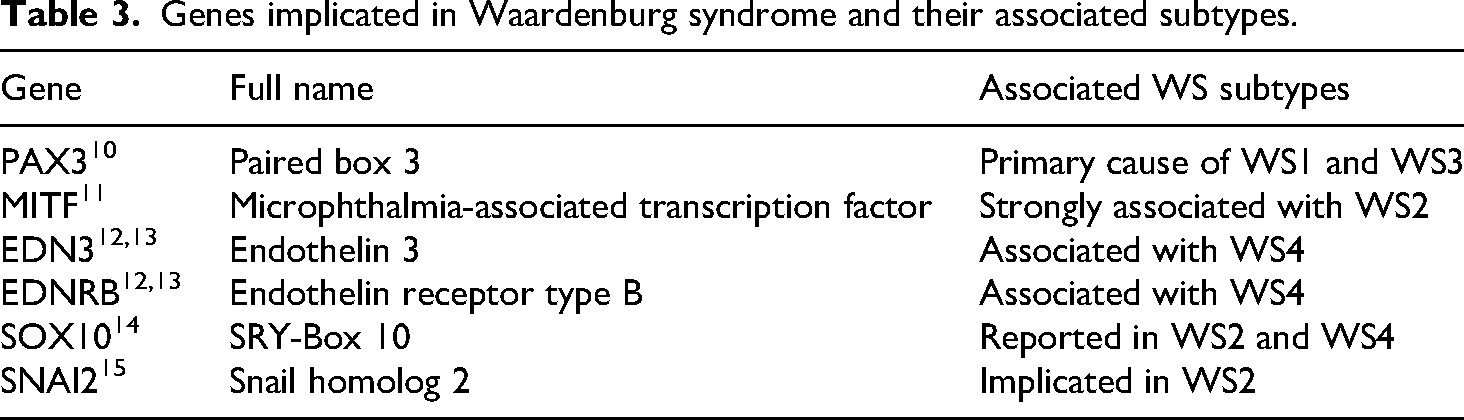
WS1 and WS2 represent the most common subtypes, whereas WS3 and WS4 are considerably rarer; the condition arises from mutations in six key genes (Table 3) 9 :
Genes implicated in Waardenburg syndrome and their associated subtypes.
Congenital deafness is a common feature in some patients with Waardenburg syndrome (WS). This hearing deficit typically stems from malformations or the complete absence of the organ of Corti. The associated hearing loss is sensorineural, present from birth, and generally stable in severity, ranging from mild to complete deafness.1,8 While bilateral involvement is more common, unilateral hearing impairment can occur in rare instances. Studies indicate that congenital sensorineural hearing loss (SNHL) is more prevalent in WS type 2 compared to WS type 1. 6 In this case, our patient was type 1 with unilateral hearing loss, indicating that every case is unique in its features. 16 The systematic review by Song et al. 17 provides one of the most comprehensive evaluations of hearing loss patterns in Waardenburg syndrome, noting that bilateral sensorineural hearing loss overwhelmingly predominates across all subtypes. Unilateral involvement, although documented, is considered uncommon and prone to under-recognition. Our case aligns with this minority pattern and illustrates the practical implications of unilateral presentation in a real-world, resource-limited environment. Because normal hearing was preserved in the contralateral ear, the functional impact was less evident to the patient and his family. This contributed to significantly delayed care-seeking, a pattern consistent with observations from the systematic review that atypical or subtle auditory manifestations often go undetected in early stages. Notably, while the review describes instances of delayed diagnosis, these typically occur within well-resourced systems with established screening programs. Our case extends this discussion by showing how structural barriers, absence of neonatal hearing screening, lack of audiology and genetics services, and low structural community health literacy, can exacerbate diagnostic delays far beyond what is reported in the literature, in this instance persisting into adulthood. WS1 and WS2 represent the most prevalent forms of Waardenburg syndrome (WS) and frequently exhibit overlapping clinical features. 6 The key diagnostic distinction between them lies in the presence of dystopia canthorum, a hallmark feature of WS1 that is consistently absent in WS2. 18 The Waardenburg (W) index serves as an anthropometric measurement derived from interpupillary distance and both inner and outer canthal measurements. Values exceeding 1.95 indicate positive dystopia canthorum. 6 In our study, the patient demonstrated W indices above this threshold (W index = 2.2). Type 3 WS presents with clinical features identical to type 1 but includes additional musculoskeletal limb anomalies, potentially including cleft palate, limb hypoplasia, and syndactyly. Type 4, alternatively termed Shah-Waardenburg syndrome, is characteristically associated with Hirschsprung disease, with aganglionic mega colon representing its hallmark feature. 18 Notably, the patient did not exhibit musculoskeletal or gastrointestinal abnormalities. WS typically follows an autosomal dominant inheritance pattern, with most affected individuals having at least one parent with the condition. However, sporadic cases may occur through de novo mutations. 7 The occurrence of this rare disorder in the studied patient, if his relatives are not affected, aligns with research suggesting that WS can arise spontaneously in individuals without familial history, likely due to unexplained genetic mutations. 16
Delayed diagnosis has direct implications for quality of life. Early recognition of hearing loss and pigmentary signs could have facilitated timely audiological rehabilitation and educational support, potentially improving the patient's communication skills, school performance, and social integration. Missed opportunities included the absence of prior hearing screening, lack of referral for genetic counseling, and failure to address psychosocial needs. Similar reports from other low-resource regions emphasize that diagnostic delays are common and often result in preventable disability. 6 Delayed diagnosis in this case reflects the combined impact of structural healthcare limitations and patient-related factors commonly encountered in conflict-affected, resource-limited environments. The absence of neonatal hearing screening, limited access to audiological and genetic services, and the lack of structured referral pathways significantly reduced opportunities for early identification despite the presence of clear pigmentary and craniofacial features. At the patient level, long-standing unilateral hearing loss and visible phenotypic differences were normalized within the family context and not perceived as medical concerns requiring evaluation, illustrating how low health awareness and sociocultural perceptions may further delay care-seeking. Because functional hearing was preserved in the contralateral ear, the perceived impact on daily life was limited, which likely contributed to the absence of early medical attention. As a result, the patient remained undiagnosed until adulthood and sought evaluation only for administrative reasons related to military exemption rather than health concerns. Such delays may have important consequences for communication development, educational support, psychosocial integration, and timely access to rehabilitative interventions. This case, therefore, highlights how atypical clinical presentations, combined with systemic healthcare barriers and reduced community health literacy, can substantially prolong diagnostic timelines beyond those reported in well-resourced healthcare systems. Another challenge raised by this case is the reliance on phenotypic diagnosis without molecular confirmation. While literature supports the validity of clinical criteria, phenotypic overlap between WS subtypes and variability in expression raise concerns about under- or misdiagnosis, especially in sporadic cases. This highlights the importance of improving local diagnostic infrastructure, including audiology, ophthalmology, and, where possible, molecular genetics.1,8,18 These observations emphasize the importance of improving early clinical recognition of congenital syndromes in low-resource settings. Strengthening basic clinical awareness and maintaining a high index of suspicion when characteristic phenotypic features coexist with auditory symptoms may help reduce diagnostic delays and their long-term functional consequences.
Conclusion
This case demonstrates the consequences of delayed diagnosis of Waardenburg syndrome type I in a resource-limited setting. Despite evident pigmentary features and long-standing hearing loss, the condition remained unrecognized into adulthood, reflecting limited health awareness, restricted diagnostic resources, and sociocultural barriers to care. Strengthening clinical screening and community awareness is essential to reduce diagnostic delays and improve outcomes in similar contexts.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261430043 - Supplemental material for Delayed diagnosis of Waardenburg syndrome type 1 in a Syrian adult: Challenges and lessons from resource-limited settings, a case report and literature review
Supplemental material, sj-docx-1-sci-10.1177_00368504261430043 for Delayed diagnosis of Waardenburg syndrome type 1 in a Syrian adult: Challenges and lessons from resource-limited settings, a case report and literature review by Bilal Hasan, Zulfiqar Hamdan and Gharam Sliman in Science Progress
Footnotes
Acknowledgments
Not applicable.
Ethical consideration
Ethics approval was not applicable for this study, as our institution's IRB committee does not mandate approval for reporting individual cases or case series; we have de-identified all patient details.
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor-in-chief of this journal on request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
