Abstract
Objective
Alterations in gut microbiota have been reported in patients with aortic valve stenosis (AVS), yet the impact of haemodynamic restoration following transcatheter aortic valve implantation (TAVI) on microbiota composition remains unclear. This study protocol describes a prospective cohort investigation designed to examine changes in gut microbiota and related metabolic markers after TAVI.
Methods
‘GUT-TAVI’ is a single-centre, prospective observational cohort study enrolling 40 adults with severe AVS undergoing TAVI. Stool samples will be collected at two timepoints (1 month to 1 day pre-TAVI and 3 months post-TAVI) for 16S-rRNA sequencing. Serum trimethylamine N-oxide (TMAO), standard biochemical markers, echocardiographic parameters, and dietary adherence scores will also be assessed. The primary endpoint is the change in gut microbiota composition following TAVI. Secondary analyses will examine associations between microbiota changes, haemodynamic parameters, and TMAO levels while accounting for potential confounders.
Results
As a protocol, no results are yet available. Planned analyses include alpha- and beta-diversity comparisons, multi-variable modelling, sub-group analyses, and sensitivity analyses addressing antibiotic exposure and procedural variability.
Conclusions
This study may provide preliminary insights into how haemodynamic improvement after TAVI is associated with changes in gut microbiota and metabolic function. Findings may help inform future, larger-scale studies investigating the gut–heart axis in cardiovascular diseases.
Objective
The interplay between aortic valve stenosis (AVS) and the gastrointestinal system was first recognised in 1958, when the condition was linked to gastrointestinal bleeding from angiodysplasias – later termed Heyde's syndrome. 1 The underlying pathophysiology is thought to involve an acquired deficiency of von Willebrand factor, resulting from shear stress-induced degradation as blood passes through the stenotic aortic valve, which increases susceptibility to bleeding. Previous studies 2 have also investigated the role of transcatheter aortic valve implantation (TAVI) in Heyde's syndrome.
Interestingly, TAVI in addition to restoring haemodynamic function, may modulate the composition and activity of the gut microbiome through several mechanisms. Enhanced cardiac output following the procedure improves splanchnic perfusion, alleviating congestion and reducing intestinal mucosal oedema. 3 These changes may, in turn, restore intestinal barrier integrity and promote favourable alterations in microbial composition. Until now, only two publications4,5 and one registered clinical trial (NCT06021535) have addressed gut microbiota measurements by 16S-rRNA sequencing in the context of AVS (Table 1). The latter is an ongoing randomised observational clinical trial designed to compare the gut microbiome of healthy individuals with that of patients diagnosed with AVS.
Published studies about the use of 16S-rRNA in aortic stenosis.
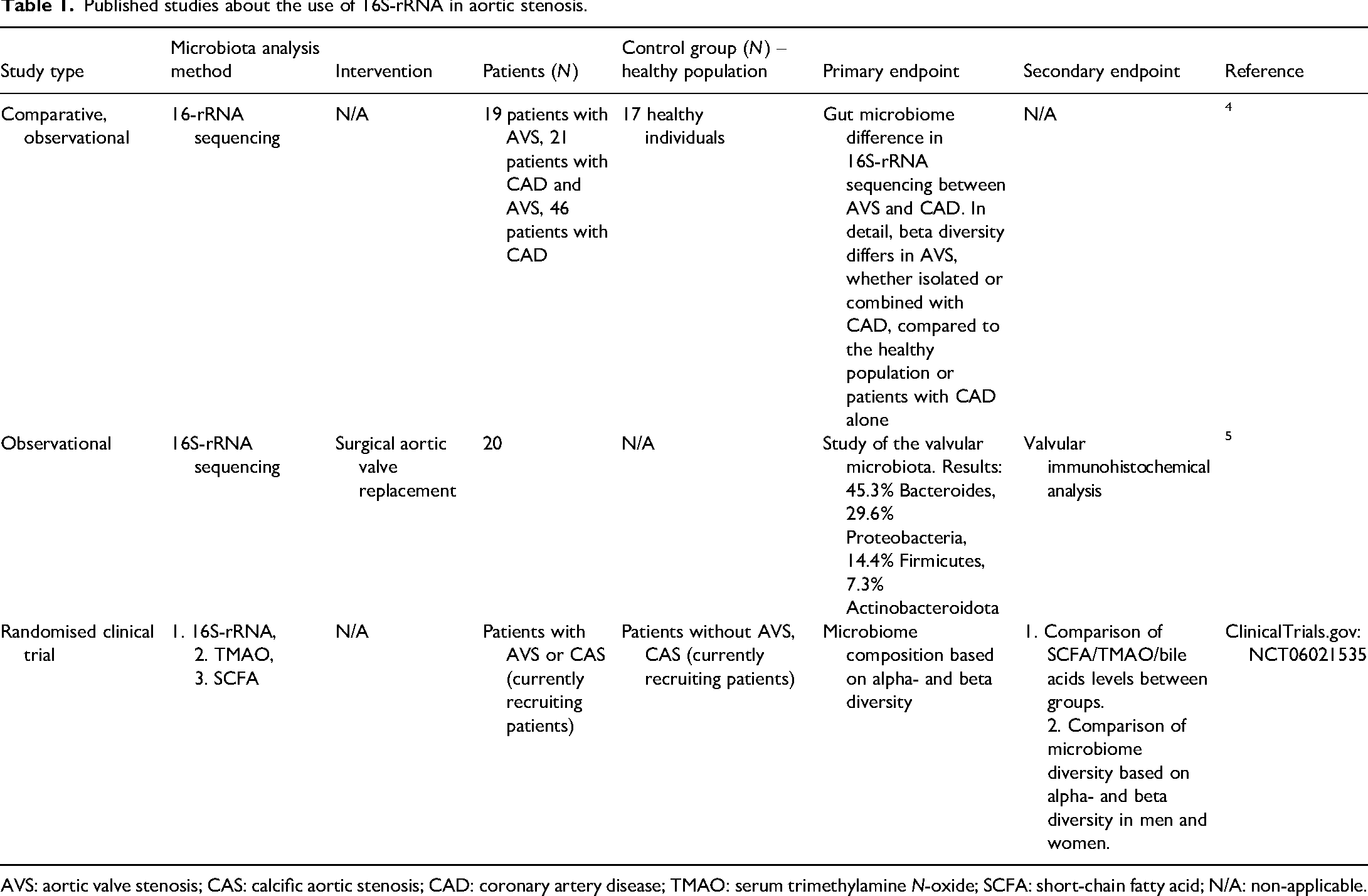
AVS: aortic valve stenosis; CAS: calcific aortic stenosis; CAD: coronary artery disease; TMAO: serum trimethylamine N-oxide; SCFA: short-chain fatty acid; N/A: non-applicable.
Liu et al. 4 recruited 19 patients with aortic stenosis, 21 patients with coronary artery disease (CAD) and AVS and 46 patients with CAD only and evaluated their stool microbiota via 16S-rRNA sequencing. In the observational study by Curini et al., 5 the valvular microbiota was analysed via immunohistochemistry among 20 surgically excised aortic valves. Yet, there is no clinical evidence of the impact of TAVI on intestinal microbiota in real-life settings.
Based on the above, we aim to address whether the haemodynamic improvements post a successful TAVI could affect human gut microbiota composition via 16S-rRNA sequencing.
Methods
Study design
‘GUT-TAVI’ is a single-centre, prospective observational cohort study enrolling patients undergoing TAVI for AVS according to the 2025 European Society of Cardiology (ESC) guidelines, 6 that will take place at the first department of cardiology of the National and Kapodistrian University of Athens at Hippocration Hospital, a tertiary General Hospital. The study aims to recruit a total of 40 participants and it followed the Equator guidelines 7 based on the STROBE checklist. 8
Study endpoints
The primary endpoint is within-participant change in gut microbiota community composition from baseline (pre-TAVI) to 3 months post-TAVI, assessed by 16S-rRNA sequencing and operationalised using Bray–Curtis beta diversity 9 (primary distance metric) with a paired/permutation-based comparison.
Key secondary endpoints include (i) within-participant change in Shannon alpha diversity from baseline to 3 months post-TAVI and (ii) within-participant change in serum trimethylamine N-oxide (TMAO) levels from baseline to 3 months post-TAVI.
Additional secondary/exploratory endpoints include associations between microbiome changes (e.g. beta-diversity shifts and/or alpha-diversity changes) and (i) baseline AVS severity and (ii) post-TAVI valve haemodynamics.
Study population and sample size
All consecutive adult patients who are candidates for TAVI at Hippocration General Hospital of Athens, Greece, during the period from September 2025 to September 2028, will be screened for eligibility and invited to participate in the study. Enrolment will be continuous throughout this period and will reflect the routine clinical volume of the centre. Written informed consent will be obtained from all participants prior to inclusion.
This study is designed as a prospective, single-arm, paired (baseline versus 3-month) investigation of gut microbiome changes following TAVI. The primary endpoint is within-participant change in gut microbiota community composition at 3 months, operationalised using Bray–Curtis dissimilarity 9 and tested using permutational multi-variate analysis of variance (PERMANOVA) with permutations constrained within participant to reflect the paired/repeated-measures design. 10 Published data directly characterising within-person Bray–Curtis dissimilarity change following TAVI are sparse, and variability estimates required for formal a priori power calculations are not well established; therefore, the present study is designed as a feasibility- and estimation-oriented cohort, consistent with established approaches to planning distance-based microbiome endpoints. 11
We selected a fixed target of 40 enrolled participants as a pragmatic, feasibility-driven sample size that is (i) operationally achievable within a single-centre TAVI workflow and (ii) consistent with prior cardiovascular and valve-related microbiome studies that have detected community-level compositional differences using beta diversity/ordination and distance-based testing in small-to-moderate cohorts. For instance, a 16S-rRNA study in heart failure analysed 12 patients versus 12 age-matched controls and reported distinct community composition in both unweighted and weighted UniFrac analyses. 12 A shotgun meta-genomic study in symptomatic atherosclerosis included 12 patients versus 13 controls and demonstrated group separation based on microbial species abundance with accompanying taxonomic/functional differences. 13 In valve-related phenotypes, cross-sectional 16S-rRNA studies have reported significant beta-diversity differences between AVS and healthy controls (30 versus 30; Adonis, p = 0.001), 14 and significant beta-diversity differences across cardiac valve calcification, CAD, and control groups. 4 These studies support feasibility in similarly sized cardiovascular cohorts, while acknowledging that data on within-person microbiome change after TAVI remain limited.
After approximately the first 10–15 participants complete the 3-month follow-up and sequencing are finalised, a pre-specified feasibility and estimation review will assess recruitment rate, completeness of paired sampling, and laboratory/sequencing success, and will summarise observed within-participant distance characteristics. These interim summaries will be used solely for operational refinement and sensitivity/precision reporting (including simulation-based detectability summaries) and will not involve interim hypothesis testing or changes to the pre-specified primary endpoint or primary analysis plan. 11
Inclusion criteria
Adults who fulfil the criteria for TAVI and who sign the relevant consent form will be included in the study. To minimise heterogeneity among selected patients with peri- or post-procedural complications including para-valvular leaks and endocarditis who require extensive courses of antibiotics or subsequent interventions, a sub-group analysis will be performed for patients with sub-optimal TAVI results.
Exclusion criteria
Patients will be excluded if they present any condition or intervention that may independently affect gut microbiota composition. These include:
Use of antibiotics, systemic corticosteroids, antivirals, probiotics, bile acid sequestrants, or new medications within 1 month prior to enrolment. History of inflammatory bowel disease. End-stage renal disease requiring dialysis. End-stage chronic liver disease. Acute infection: patients with aortic stenosis due to rheumatic fever or infectious endocarditis will be excluded. Active cancer under treatment. Psychiatric illness impairing ability to consent. Substance or alcohol abuse.
Additionally, participants missing a stool sample or withdrawing consent at either timepoint will be excluded from analyses. The inclusion and exclusion criteria are also summarised in Table 2.
Inclusion and exclusion criteria.

TAVI: transcatheter aortic valve implantation.
Participants’ baseline demographic data
Demographic and anthropometric parameters will be included only in the screening phase including the following: sex, age, weight, height, and body mass index. Social history will also be documented about smoking, alcohol, and substance use. Comorbidities and the medical history are also part of the screening phase. Diagnoses including hypertension, diabetes, dyslipidaemia, vascular disease, heart failure, stroke, transient ischaemic attack (TIA), and pulmonary embolism will be included in the study file.
Study's laboratory variables
16S-rRNA will be processed in stool samples during every visit and the changes in gut microbiota composition post-TAVI will be the primary outcome. Participants will follow hygiene protocols and use provided sterile containers for stool collection. The latter will be stored frozen at −80 °C to preserve microbial DNA. In the laboratory, an aliquot of stool will be homogenised and subjected to DNA extraction using bead-beating combined with chemical lysis to ensure efficient disruption of bacterial cell walls, followed by inhibitor-removal steps and silica column purification. The purified DNA will then serve as the template for 16S-rRNA gene amplification by polymerase chain reaction (PCR). The bacterial 16S-rRNA gene will be amplified using primers targeting commonly used hyper-variable regions (V3–V4), which provide suitable taxonomic resolution for community profiling in human gut microbiota studies. 15 Amplicons will be sequenced using an Illumina-based next-generation sequencing platform. 16 Raw sequence data will undergo quality filtering, trimming, and de-noising using established bioinformatics pipelines.17,18 These workflows allow the removal of sequencing errors and chimera detection, producing high-resolution amplicon sequence variants. Taxonomic assignment will be performed against a curated reference database that entails standard resources for microbial classification. 19 Alpha- and beta-diversity metrics, as well as relative abundance analyses, will be generated using QIIME2 and validated statistical packages in R, following the published best practices in microbiome data processing.17,20
The biomarker TMAO in the venous blood sample will be monitored during every visit and its correlation with the gut microbiota will be considered a secondary outcome. Additional biochemical values in venous blood sample will be included in the study including: complete blood count, liver enzymes (serum glutamic-oxaloacetic transaminase, serum glutamic-pyruvic transaminase, alkaline phosphatase, gamma-glutamyl transferase, renal function (urea, creatinine), electrolytes (K+, Na+, Ca2+, Mg2+), N-terminal pro-B-type natriuretic peptide, high-sensitivity C-reactive protein (hsCRP), and estimated glomerular filtration rate using the Cockcroft–Gault equation.
Biostatistical models
Statistical analyses will be performed after completion of 16S-rRNA sequencing and generation of the feature table and distance matrices, in collaboration with the study statistician. The primary microbiome analysis will assess within-participant change from baseline (pre-TAVI) to 3 months post-TAVI. Both alpha and beta diversity will be evaluated. Unless otherwise specified, statistical tests will be two-sided with a significance level of 0.05.
The primary microbiome analysis will evaluate within-participant change in community composition from baseline to 3 months using Bray–Curtis beta diversity 9 with paired PERMANOVA (permutations constrained within participant). Key secondary analyses will evaluate within-participant change in Shannon alpha diversity 21 and serum TMAO. Additional diversity indices and alternative distance metrics (e.g. UniFrac) will be treated as secondary/sensitivity analyses, and multiplicity will be addressed for secondary/exploratory testing as described below.
Alpha diversity
Alpha diversity reflects within-sample community characteristics such as the number of different species (richness) and the evenness of distribution of individual organisms among those species. 22 Alpha diversity will be summarised using pre-specified indices, including the Shannon index 21 and Simpson index. 23 The Shannon index accounts for both richness and evenness within a sample; higher values indicate greater diversity and a more even distribution of species. This index is sensitive to less abundant taxa. The Simpson index focuses on the probability that two randomly selected individuals from a sample belong to the same species (second-order diversity).
The primary alpha-diversity analysis will evaluate within-participant change in Shannon index from baseline to 3 months post-TAVI (key secondary microbiome endpoint). This will be assessed using (i) a paired test (paired t-test when approximately normally distributed, otherwise Wilcoxon signed-rank test) and, where covariate adjustment is required or incomplete follow-up occurs, (ii) a linear mixed-effects model with time (baseline vs. 3 months) as a fixed effect and participant as a random effect. Simpson diversity and any additional alpha indices will be analysed as secondary/sensitivity outcomes using analogous paired methods. If clinically relevant covariates are included (e.g. major peri-procedural complications, recent antibiotic exposure), they will be incorporated as fixed effects with careful restriction of model complexity given the expected sample size.
Alpha-diversity indices provide a summary of within-sample diversity but do not capture taxon-specific shifts; therefore, alpha-diversity results will be interpreted as complementary descriptors rather than a complete characterisation of compositional change.
Beta diversity
Beta diversity quantifies between-sample compositional dissimilarity. Dissimilarity will be calculated using the pre-specified primary metric (Bray–Curtis). 9 Weighted UniFrac and unweighted UniFrac 24 will be evaluated as sensitivity analyses. Ordination (e.g. principal coordinates analysis) will be used for visualisation.
The primary beta-diversity hypothesis test (primary microbiome endpoint) will assess whether overall community composition differs between baseline and 3 months post-TAVI using PERMANOVA 10 applied to the distance matrix, with permutations constrained within participant to account for the paired/repeated-measures design. The PERMANOVA effect size (e.g. R2) will be reported alongside p-values.
Because permutation-based multi-variate methods can be influenced by group differences in dispersion, homogeneity of multi-variate dispersion will be assessed (e.g. permutational analysis of multivariate dispersions) and considered when interpreting PERMANOVA findings. Analysis of similarities 25 may be performed as an additional secondary/sensitivity analysis; if performed, it will be interpreted cautiously and in conjunction with dispersion results.
Serum TMAO
Serum TMAO levels at baseline and 3 months post-TAVI will be summarised descriptively. Given typical right-skewness, TMAO will be analysed on the log-transformed scale (or using a robust alternative if transformation is inadequate). The within-participant change will be tested using paired methods and/or a linear mixed-effects model with participant as a random effect and time as a fixed effect, with pre-specified covariates included only where justified and parsimonious.
Association analyses
Associations between microbiome changes and (i) baseline AVS severity and (ii) post-TAVI valve haemodynamics will be explored using regression-based approaches appropriate to the data structure. Microbiome change will be summarised using pre-specified features (e.g. change in Shannon; principal coordinate scores derived from Bray–Curtis; and/or PERMANOVA-based effect summaries). Models will be kept parsimonious given the expected sample size, with attention to collinearity and overfitting.
Multiplicity and reporting
The study's primary microbiome inference will be based on the pre-specified beta-diversity analysis (Bray–Curtis and paired PERMANOVA). Shannon alpha-diversity 21 changes and serum TMAO changes are pre-specified key secondary endpoints. Other diversity metrics and distance metrics will be treated as secondary/sensitivity analyses. If taxon-level differential abundance testing is undertaken, multiplicity will be controlled using an appropriate false-discovery rate approach, and results will be reported with effect sizes and confidence intervals where feasible.
Echocardiography parameters
The echocardiography parameters of interest will be monitored and reviewed during every visit. The following echocardiographic parameters will be recorded: left ventricular ejection fraction (LVEF%), left ventricular end-diastolic diameter (LVEDD), inter-ventricular septal thickness in diastole (IVSd), left ventricular posterior wall thickness in diastole (LVPWd), maximum aortic flow velocity (AoVmax), mean pressure gradient (PGmean), change in mean pressure gradient (ΔPGmean), maximum pressure gradient (PGmax), aortic valve area (AVA), Doppler velocity index (DVI), and stroke volume index (SVI). Echocardiographic measurements will be performed and reviewed by the same senior imaging team to reduce inter-observer variability.
Dietary questionnaires
At each visit, patients will complete a dietary questionnaire derived from the validated Mediterranean diet questionnaire 26 (Supplemental file 1).
Adherence to the Mediterranean diet will be evaluated using 14 validated questions. This tool assesses dietary habits including the use of olive oil as the main culinary fat (≥4 tablespoons/day), daily intake of vegetables (≥2 servings, including at least one portion raw or as a salad) and fruit (≥3 units/day), and limited consumption of red meat, hamburgers, or processed meats (<1 serving/day) and butter, margarine, or cream (<1 serving/day). Additional items capture the frequency of sweetened or carbonated beverages (<1/day), wine (≥7 glasses/week), legumes (≥3 servings/week), fish or shellfish (≥3 servings/week), commercial sweets or pastries (<3/week), and nuts (≥3 servings/week). The questionnaire also records preference for lean meats such as chicken, turkey, or rabbit over red or processed meats, and the frequency of consuming vegetables, pasta, rice, or other dishes prepared with sofrito (≥2 servings/week).
Each item in the questionnaire is scored 1 point when the pre-defined criterion is met and 0 points otherwise, yielding a total score from 0 to 14. For descriptive purposes, adherence will be categorised into low (0–5), moderate (6–9), and high (10–14) adherence, consistent with published PREDIMED (primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts)-based classifications. 27 In the analyses, the score will be treated as a continuous covariate. This score will be included in multi-variable models examining associations between gut microbiota composition, diversity metrics, and post-TAVI clinical parameters.
Study timepoints
Data will be collected at 2 timepoints (1 month to 1 day pre-TAVI, and 3 months post-TAVI), along with dietary questionnaires to account for potential confounding factors. Microbial composition will be characterised using alpha diversity, beta diversity, and taxonomic abundance measures derived from 16S-rRNA sequencing. For each participant, changes in microbial metrics between pre- and post-TAVI samples will be compared with the corresponding changes in plasma TMAO concentration and key echocardiographic parameters. Associations will be assessed using multi-variable regression models adjusting for relevant clinical covariates (e.g. age, sex, renal function, and Mediterranean diet adherence). This approach will identify whether shifts in gut microbiota are related to biochemical or echocardiographic changes after TAVI. The study's flow chart is shown in Figure 1.

Study flowchart.
Phase 1: 1 month to 1 day before TAVI (T1)
Phase 1 will include screening and baseline data collection (1 month to 1 day before TAVI). Participants will be assessed for eligibility based on inclusion/exclusion criteria. Informed consent will be obtained, and participants will agree to telephone follow-up.
Participants will collect a stool sample using a standardised kit for 16S-rRNA sequencing. A food-frequency questionnaire will be completed to account for dietary influences on microbiota. This questionnaire will be repeated at each sample collection. Samples will be labelled with study ID, sample sequence (A for phase 1 and B for phase 2), date of collection, and will be returned to the hospital promptly. No identifiable information will be recorded on the specimen. Apart from the stool samples, the rest of the biochemical variables will be tested (including TMAO levels and basic biochemical profile), along with the echocardiography parameters. All patients’ details will be de-identified.
Transcatheter aortic valve implantation (TAVI)
The study's intervention (TAVI) date will be documented between the timepoints T1 and T2. As per the ESC guidelines, 28 antibiotic prophylaxis prior to TAVI initiation should be considered to eliminate the risk of prosthetic valve endocarditis. In this scenario, Enterococcus is the commonest species. 29 Hence, the patients will receive a short course of antibiotics including either a single 2.2 g intravenous dose of amoxicillin/clavulanic acid given within 120 min before vascular access (ideally within 60 min) or a single 3 g dose of intravenous ampicillin. In β-lactam-allergic patients, vancomycin 15 mg/kg or teicoplanin 9–12 mg/kg intravenously is recommended. 28
Phase 2: follow-up (3 months after TAVI) (T2)
All the previous assessments will be repeated 3 months post-procedure. If antibiotics were taken within this period, the follow-up assessment will be delayed by 1 month (i.e. 4 months post-TAVI). The selection of the 3-month timepoint was based on the study by De La Serre et al., 30 in which changes in Bacteroides within the gut microbiota became evident after 8–12 weeks. In the sub-group analysis of patients with sub-optimal TAVI outcomes (significant para-valvular leak or major procedural complications), post-TAVI changes compared to pre-TAVI in alpha-diversity indices (i.e. Shannon, Simpson index21,23), serum TMAO concentrations, and echocardiographic parameters will be assessed using paired tests (paired t-test or Wilcoxon signed-rank test, as appropriate), and changes in beta diversity will be evaluated using PERMANOVA 10 on Bray–Curtis 9 or UniFrac 24 distance matrices with timepoint as a factor. The study's comparison timepoints are demonstrated in Figure 2.
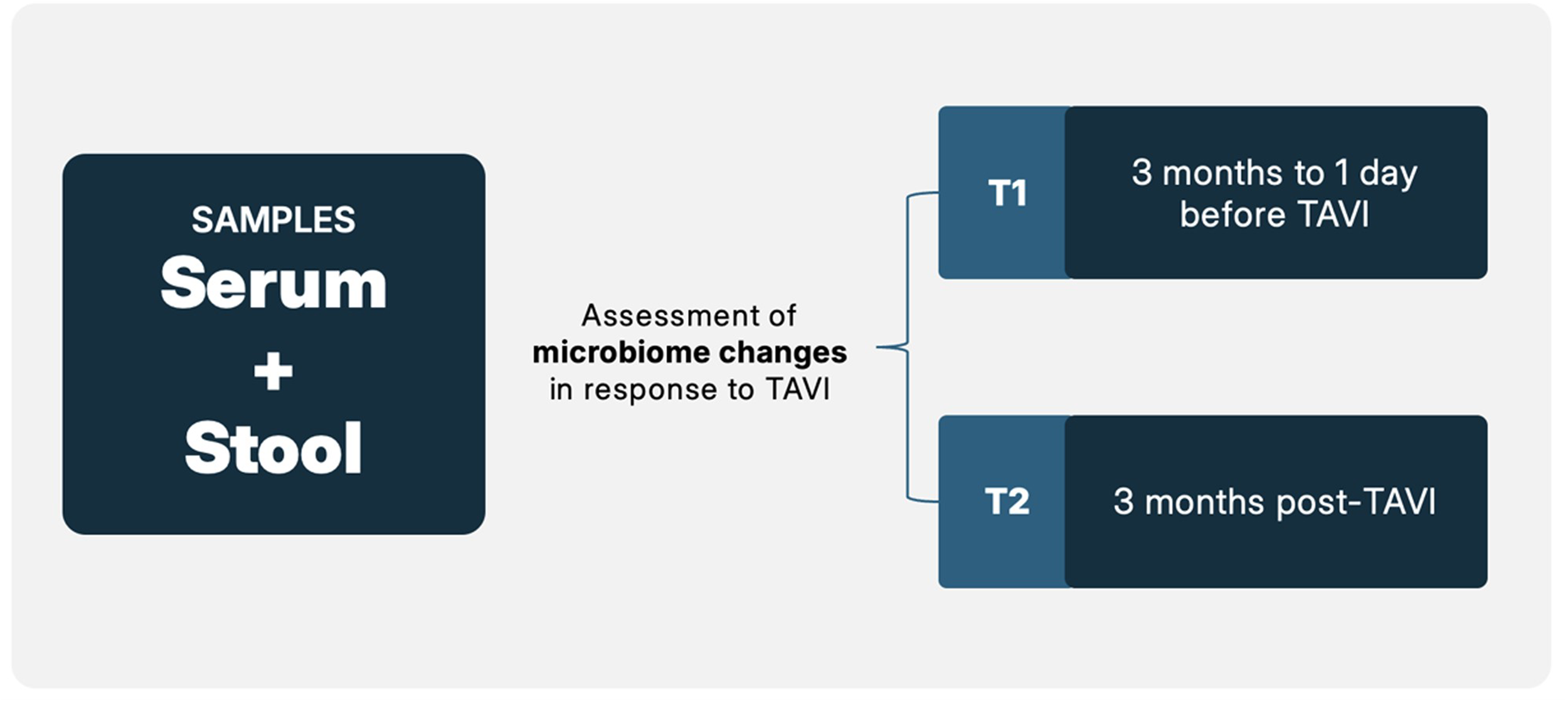
Timepoints and comparisons.
Ethics
The study has been approved by the Hospital's Ethics Committee. The institutional review board approval number is 92/03-09-2025 and permission was given on 3/9/2025 by Hippocration General Hospital Scientific Committee. Our study will be conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024.
Discussion
This article presents the rationale and design of a single-centre, prospective observational study aimed to evaluate the impact of TAVI on gut microbiota. By integrating 16S-rRNA sequencing with measurement of the metabolite TMAO, the study will also assess the indirect effects of TAVI on gut microbial function.
In our previous recent review 31 we sought to shed light on the relationship between metabolites and valvular heart disease. In the present study, we focus specifically on aortic stenosis. The latter is the most prevalent valvulopathy worldwide and the third most common cardiovascular condition in developed countries after hypertension and CAD, and is associated with considerable morbidity and mortality.6,32
TAVI is a minimally invasive therapeutic option for severe AVS, offering haemodynamic restoration in patients who are often at high surgical risk. 6 The disease itself is characterised by progressive thickening and calcification of the valve leaflets, resulting in restricted mobility, narrowing of the valve orifice, increased left ventricular afterload, and compensatory myocardial hypertrophy. The ESC guidelines 6 for the management of valvular heart disease outline the indications and contraindications for TAVI based on surgical risk, patient age, anatomical characteristics, and comorbidities. Given the growing understanding of the ‘gut–heart’ axis, 3 exploring the relationship between TAVI and gut microbiome composition may yield novel insights into disease mechanisms and therapeutic opportunities. TAVI was chosen as the intervention given the substantial haemodynamic improvements that occur following valve replacement. Hence, the gut microbial balance will be positively influenced by increasing splanchnic perfusion and reducing intestinal congestion.
The gut microbiome, comprising trillions of micro-organisms, is a dynamic ecosystem that exerts wide-ranging effects on systemic physiology through complex mechanisms. 33 The isolation of specific pathogens within the intestinal tract is feasible only in cases where tissue specimens are obtained following colectomy or gastrectomy. 34 This is possible in experimental animal models or in patients with a specific surgical indication. In the general population, the isolation of microbial strains from stool samples provides an indirect representation of the gut microbiota, without the ability to investigate the microbial composition of individual segments of the intestinal lumen. One of the principal methods for identifying intestinal micro-organisms is 16S-rRNA sequencing in stool samples. This approach, which involves amplicon sequencing of the 16S-rRNA ribosomal sub-unit, 35 is relatively cost-effective and straightforward to implement. It provides taxonomic resolution at the genus level and estimates relative abundance within a microbiome sample by targeting a specific genetic sequence encoding the prokaryotic ribosomal sub-unit. While this gene region is highly conserved among bacteria, it also contains multiple hyper-variable regions that are unique to each micro-organism. 36
Beyond the direct isolation and culture of intestinal strains, certain plasma biomarkers in humans have been studied and correlated with gut dysbiosis. One of the most prominent is TMAO, an indirect biomarker that exerts effects through its metabolites. Elevated TMAO levels have been associated with an increased cardiovascular risk in meta-analyses37,38 through mechanisms such as the formation of foam cells. 39 However, other studies have reported no such association between cardiovascular risk and this biomarker. 40
Prior research has also assessed the role of TAVI in the context of Heyde's syndrome. Goltstein et al. 2 sought to further investigate the role of TAVI in the treatment of angiodysplasias. They recruited patients with anaemia and a negative baseline endoscopic evaluation (gastroscopy and colonoscopy) prior to undergoing small bowel capsule endoscopy. Patients diagnosed with angiodysplasia subsequently underwent TAVI. A repeat capsule endoscopy was performed 6 months after the procedure. The number of angiodysplasias decreased from 6.4 (absolute number) to 2 (p = 0.04), while typical angiodysplasias disappeared completely in 6 patients. These results were reflected in a reduced need for blood transfusions: five of the six patients who had previously been transfusion-dependent maintained stable haemoglobin levels. Despite the small patient sample and the unclear association with the microbiome, there is a clear need for further investigation into the ‘gut–heart’ connection.
While AVS was previously considered a chronic degenerative process of the valve, current evidence supports a theory involving active participation of valvular interstitial cells and osteogenesis. 41 Within this context, Curini et al. 5 investigated in 2023 the aortic valve microbiome in 20 patients with severe aortic stenosis who underwent surgical valve replacement. During surgery, each excised aortic valve was divided into two portions: one was immediately processed for immunohistochemical analysis, while the other was frozen at −80 °C for DNA analysis. The researchers found that 76% of the isolated T lymphocytes were CD4-positive, 19% were CD8-positive, and the remainder were unclassified. Regarding the valvular microbiota, 17 phyla were identified. Bacteroides was the most abundant (45.3%), followed by Proteobacteria (29.6%, considered pro-inflammatory), Firmicutes (14.4%), Actinobacteroidota (7.3%), and Euryarchaeota (1.6%).
Our present protocol possesses several notable strengths. The use of distinct sampling timepoints enables comparisons of microbiota composition, thereby allowing the detection of variations. This approach will provide, for the first time, insights into the potential association between gut microbiota alterations over a 3-month period. Such analysis may reveal correlations between disease severity and the relative abundance or depletion of specific genera. Furthermore, by comparing gut microbiota profiles before and after TAVI, the study will evaluate the intervention's impact over a 3–4-month period, elucidating procedure-related effects and the potential impact of haemodynamic alterations due to any degree of failure of TAVI therapy.
The concurrent assessment of 16S-rRNA sequencing and TMAO levels, both pre- and post-TAVI, represents an innovative approach for characterising the gut microbiota and its metabolic activity in this patient population. Upon study completion, the main results will address whether TAVI is associated with measurable changes in gut microbiota composition and diversity at 3 months, and whether these changes correlate with improvements in haemodynamic parameters, biochemical markers, and TMAO levels. By characterising microbiota and metabolite changes before and after TAVI and relating these to echocardiographic markers of reverse cardiac remodelling, this study may identify microbial or metabolic features associated with improved or impaired recovery. Such insights could contribute to more refined risk stratification and may ultimately inform targeted interventions such as personalised dietary optimisation, microbiota-directed therapies, or enhanced monitoring approaches to support post-TAVI patient management. It also may potentially provide a deeper mechanistic understanding of how extra-cardiac factors influence cardiac remodelling.
However, we acknowledge that our patients, in addition to the brief course of pre-TAVI prophylactic antibiotics, may have been exposed to further antibiotic treatments before sampling. It is notable that the study population primarily comprises elderly patients with multiple comorbidities, which inherently increases their risk of infection. Nevertheless, all participants will receive the same short pre-TAVI antibiotic regimen, whereas those who experience complications requiring prolonged hospitalisation or additional antibiotic therapy will be evaluated as a separate sub-group. As previously indicated, if patients require a second course of antibiotics after TAVI, phase 2 sampling will be deferred by 1 month to minimise potential confounding from antibiotic-related perturbations of the gut microbiota. In the absence of broad, high-quality studies specifically addressing this scenario, our hypothesis is informed by systematic reviews 42 suggesting that gut microbial composition typically begins to recover within approximately 2–4 weeks following antibiotic exposure. De La Cochetière et al. 43 showed that the dominant species were not statistically different in 4 and 8 weeks post-oral amoxicillin when tested by 16S-rRNA, while meta-genomic analysis by Palleja et al. 44 indicated a return of microbial species to baseline levels within 1.5 months.
Interestingly, intravenously administered antibiotics tested in patients who underwent cardiac surgery led to quicker gut microbiota homoeostasis compared to oral as they required approximately 2 weeks of withdrawal, while the bacterial genera of Bacteroides, Faecalibacterium, Blautia, Subdoligranulum, and Phascolarctobacterium were stable. Yet, Lactobacillus, Streptococcus, and Roseburia abundance was still affected. 45 Studies in mice colonised with human faecal microbiota and subsequently exposed to antibiotics have shown that a low-fibre diet delays microbial recovery. 46 The latter accentuates the involvement of environmental factors and importance of Mediterranean diet adherence. 26
Apart from the antibiotic exposure, other limitations may include the small sample size (40 patients) which will represent our patient population but may limit the statistical power to detect modest changes in microbiota composition. Thirdly, the single-centre design may limit external validity, as the findings may reflect the characteristics and clinical practices of one tertiary hospital. The magnitude of this limitation is moderate, as TAVI populations across centres are broadly comparable. Finally, diet, lifestyle, and environmental influences, although partially captured by the Mediterranean diet questionnaire, cannot be fully controlled. Residual confounding may persist, with unpredictable direction depending on unmeasured behaviours.
Despite these limitations, the study is designed to provide robust exploratory evidence on the temporal relationship between TAVI and gut microbiota dynamics, offering a foundation for larger multi-centre investigations.
A deeper understanding of the relationship between TAVI and the gut microbiome may open new avenues for personalised therapeutic interventions (precision medicine) by probiotics, prebiotics, or targeted pharmacological agents.
Conclusions
This study protocol outlines a prospective, cohort, single-centre investigation into the impact of TAVI on gut microbiota composition and function in patients with AVS. By combining 16S-rRNA sequencing with serum TMAO measurements at multiple timepoints, the study aims to capture both direct microbial shifts and associated metabolic changes following the intervention. This will be the first study to longitudinally explore the gut–heart axis in the context of TAVI, integrating microbiome profiling with cardiovascular parameters. Given its observational design and limited sample size, any findings will be exploratory and hypothesis-generating. Findings from this work may inform future, larger-scale trials and support the development of precision medicine strategies targeting the gut microbiome in cardiovascular diseases.
Supplemental Material
sj-doc-1-sci-10.1177_00368504261426427 - Supplemental material for Transcatheter aortic valve implantation and gut microbiota: Rationale and design of the ‘GUT-TAVI’ cohort study
Supplemental material, sj-doc-1-sci-10.1177_00368504261426427 for Transcatheter aortic valve implantation and gut microbiota: Rationale and design of the ‘GUT-TAVI’ cohort study by Magdalini Manti, Kyriakos Dimitriadis, Eirini Dri, Kyriaki Mavromoustakou, Angeliki Vakka, Georgios Koutsopoulos, Panagiotis Tsioufis, Themistoklis Katsimichas, Spyridon Siakavellas, Maria Gazouli and Konstantinos Tsioufis in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504261426427 - Supplemental material for Transcatheter aortic valve implantation and gut microbiota: Rationale and design of the ‘GUT-TAVI’ cohort study
Supplemental material, sj-docx-2-sci-10.1177_00368504261426427 for Transcatheter aortic valve implantation and gut microbiota: Rationale and design of the ‘GUT-TAVI’ cohort study by Magdalini Manti, Kyriakos Dimitriadis, Eirini Dri, Kyriaki Mavromoustakou, Angeliki Vakka, Georgios Koutsopoulos, Panagiotis Tsioufis, Themistoklis Katsimichas, Spyridon Siakavellas, Maria Gazouli and Konstantinos Tsioufis in Science Progress
Footnotes
Abbreviations
Author contributions
MM: conceptualisation, investigation, methodology, visualisation, and writing (original draft, review, and editing); KD: supervision and writing (original draft, review, and editing); ED: methodology and supervision; KM: methodology; AV: methodology; GK: methodology; PT: methodology and writing (original draft, review, and editing); TK: methodology; SS: methodology; MG: methodology; KT: conceptualisation, investigation, methodology, visualisation, and writing (original draft, review, and editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The study is currently in progress and data are not yet available.
Trial registration
ClinicalTrials.gov identifier: NCT07052071.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
