Abstract
Chlormequat is a quaternary ammonium salt used as a plant growth-regulating agent. It is structurally similar to choline, acting at muscarinic and nicotinic receptors. Herein, we present the case of suicidal poisoning with an unknown dose of chlormequat by a 45-year-old woman. Initially, carbofuran was suspected of ingestion. Following ingestion, circulatory arrest occurred. After hospital admission, gastric lavage was performed, and activated charcoal was administered. Initial toxicological analysis identified mild alcohol intoxication, while high-resolution mass spectrometry later discovered a high concentration of chlormequat in the blood and urine. Continuous hemodialysis was indicated to accelerate chlormequat elimination. Antioxidant and Cytosorb therapy were applied to reduce oxidative stress and inflammation. Laboratory results further showed that chlormequat is a mild acetylcholinesterase inhibitor, revealing an additional mechanism of toxicity. However, despite all initial efforts, deep coma persisted with clinical signs of areflexia above C1 and malignant brain edema described on the CT scan. The patient died on day 3 of the ICU stay in palliative care. The authors present complete laboratory and toxicological results, including toxicokinetic data, oxidative stress parameters, acetylcholinesterase, and butyrylcholinesterase activity. In conclusion, modern analytical methods are necessary to provide more effective targeted treatment and could help uncover previously unknown mechanisms of toxicity.
Introduction
Chlormequat ([(2-chloroethyl)trimethylammonium chloride]) is a quaternary ammonium compound widely utilized in agriculture. It blocks the cyclases, including copalyl-diphosphate synthase and ent-kaurene synthase, effectively inhibiting the early steps of gibberellin synthesis. Through this mechanism, chlormequat regulates the growth characteristics of many plants to increase crop yield. 1 Chlormequat is also structurally similar to choline (Figure 1). It has been reported as a direct agonist of muscarinic and nicotinic acetylcholine receptors. 2 Acute chlormequat poisoning resembles a cholinergic crisis manifested by symptoms such as miosis, increased salivation, vomiting and diarrhea, diaphoresis, visual disturbance, confusion, seizures, trismus or muscle weakness, pulmonary edema with associated hypoxia, hypertension, or bradycardia with hypotension, and dysrhythmias. If fatal, death usually occurs within an hour after ingestion.3–10
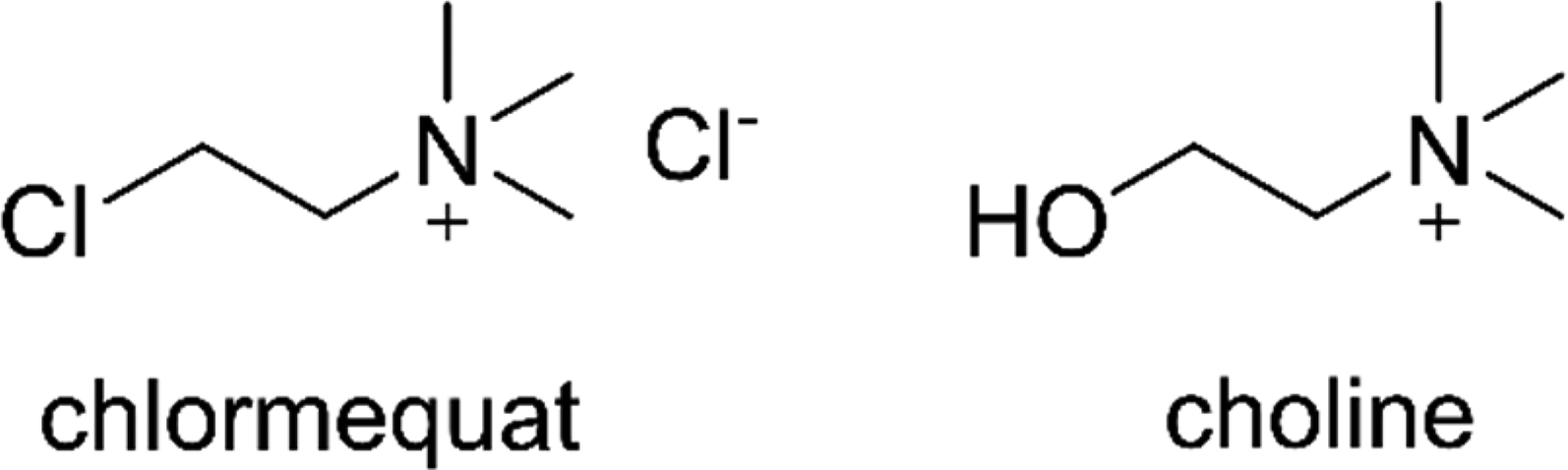
Chlormequat chloride and choline structure.
Despite its easy availability, few cases of chlormequat poisoning have been reported. The first case of chlormequat poisoning was published in 1989. 5 Since then, we have recorded another 13 cases in the scientific literature, one being after accidental inhalation, one after self-injection, and the others after chlormequat per oral administration. The inhalation case did not provide any specifics. 7 The victim of the self-injection case was a pig breeder who used chlormequat illegally to euthanize animals. 4 The remaining cases included mainly intentional ingestions. Except for two cases, all had fatal outcomes.3–10
This case report provides additional insight into the clinical presentation of this rare intoxication, together with data regarding the human toxicity of chlormequat.
Patient information
A mid-40s’ woman from a farmer family deliberately ingested an unknown quantity of unknown compound mixed with Beefeater Pink Strawberry-flavored gin. She was found with “no signs of life,” lasting by estimation from 0 to 20 minutes. After 3 minutes of telephone-assisted cardiopulmonary resuscitation, she received advanced life support according to ERC guidelines 2021 for cardiac arrest with the first rhythm of asystole. 11 Spontaneous circulation returned after the 2nd adrenaline dose at 7 minutes. According to information from family members, carbofuran intoxication was suspected. Thus, due to severe hypotension and bradycardia, 2 mg of atropine were administered via intraosseous (IO) access, resulting in a rise of blood pressure lasting 10 to 15 minutes. Pre-hospital care also included rocuronium (50 mg, IO) for tracheal intubation.
According to the family, the patient was not being treated for any illness and was not taking any medications.
Clinical findings
On admission (ICU at the Department of Anesthesiology, Resuscitation and Intensive Medicine at University Hospital Hradec Kralove in May 2023), the patient was comatose (GSC 3), artificially ventilated, pulsation was centrally palpable (53/minute), blood pressure was 75/35 mmHg, oxygen saturation was 92%, normal-sized isocoric pupils were unresponsive to light, toxic odor came from the oral cavity, the periphery was warm (36.5 °C), and erythema all over the body and profuse sweating were present with no signs of trauma. The reporting of this study conforms to CARE guidelines. 12
Diagnostic assessment
Toxicology analysis
High-resolution mass spectrometer identified chlormequat (Figure 2) and atropine in both blood and urine samples. Carbofuran was not detected, despite being initially suspected as the poisoning agent based on communication with the victim´s father. At admission, serum ethanol concentration was 26.0 mmol/l (1.2 g/kg). Detailed methodology of toxicological analysis is discussed in the Supplemental material.

Chlormequat concentration in blood and urine samples.
Blood count and biochemistry
At admission, the blood count showed leukocytosis (20.1 × 109/l). Blood biochemistry revealed hyperglycemia (18.8 mmol/l), combined respiratory and metabolic acidosis (pH 7.07, PaCO2 6.48 kPa, BE −17.3 mmol/l, bicarbonate ion 12 mmol/l, lactate 9.7 mmol/l, O2 saturation 89.9%) and mineral imbalance, including mild hyponatremia (134 mmol/l), hypermagnesemia (1.19 mmol/l), and hyperphosphatemia (2.45 mmol/l). Furthermore, blood biochemistry showed an increase in ALT (3.34 µkat/l), AST (4.71 µkat/l), GMT (1.36 µkat/l), and CK-MB (1.89 µkat/l), which indicated liver and myocardial damage. Plasma butyrylcholinesterase (BChE) activity or initial CRP was within the physiological range. However, CRP showed a delayed increase. Protein, glucose, erythrocytes, leukocytes, and epithelial cells were present in urine. Detailed findings, their time course, and laboratory lower and upper physiological limits are depicted in Supplementary Tables S1–S6. Hyperglycemia was treated with insulin, and acidosis with bicarbonate.
Other laboratory assessments
In blood collected on admission, we found reduced erythrocyte acetylcholinesterase (AChE) activity and increased malondialdehyde concentration and superoxide dismutase (SOD) activity in plasma (for methodology, see Supplemental material). While MDA remained increased during the first day after admission, erythrocyte AChE and SOD returned to the control range within 1 hour after the admission (Figure 3).

Erythrocyte acetylcholinesterase and plasma superoxide dismutase activity and malondialdehyde concentration.
Finally, we assessed the inhibitory efficacy of chlormequat on human recombinant (hr) AChE and hrBChE in vitro to confirm its selective action toward human AChE (for methodology, see Supplemental material). Chlormequat at 1 mM significantly reduced hrAChE activity by 12% and 11% at 1 and 5 minutes after exposure, respectively. At 10 mM, hrAChE activity decreased by 26%, 27%, 21%, and 16% after 1, 5, 10, and 15 minutes, respectively. Chlormequat at concentrations ranging from 0.001 to 10 mM did not affect hrBChE activity (Figure 4).

Chlormequat inhibition potency to hrAChE and hrBChE. *Significantly different compared to control (p ≤ 0.05).
Therapeutic intervention
As mentioned above, the patient received a standard pre-hospital care and treatment. Blood pressure was supported by another two atropine boluses (2 mg each; each bolus resulted in a 10-minute rise in blood pressure). The patient was initially sedated with a combination of sufentanil and propofol to achieve a sedation level of SAS 1 (according to the Sedation Agitation Scale). Diazepam was added for neuroprotection. However, sedation was relatively quickly discontinued on the second day, due to areflexia above C1. Further circulatory support included noradrenalin (initially 2.5 mg/h), atropine (1 mg/h for 3 h), crystalloids (initially 1000 ml, followed by 100 ml/h), and albumin (20%, 200 ml) were started. The dose of noradrenaline was titrated to a MAP (mean arterial pressure) of 70 to 80 mmHg. Vasopressin was also administered at a dose of 1.8 IU/h. During the first 24 hours, both vasopressors were tapered until the patient was without circulatory support. Gastric lavage was performed. A 300 to 350 ml of pinkish strong-smelling fluid was aspirated, and activated charcoal suspension (40 g, Carbosorb®) was administered. After determining the poison, atropine was discontinued, and antioxidant therapy started, including acetylcysteine (150 mg/kg over 60 minutes, followed by 50 mg/kg over the next 4 hours, and finally 10 mg/kg over the next 16 hours) and thiamine, first day 1500 mg and second day 200 mg (total dose 1700 mg). She was also given intravenous omeprazole 40 mg. The antiarrhythmic drug amiodarone was also administered on the first day at a dose of 900 mg/24 hours. The initial metabolic acidosis was corrected with bicarbonate, 100 ml 8.4%. The continuous hemodialysis using the multiFiltrate Ci–Ca system (Fresenius Medical Care, Bad Homburg, Germany) was started with a Cytosorb capsule (from 4th to 37th hours after admission; Cytosorbents Inc., Princeton, NJ, USA) through a femoral catheter (12 F, Teleflex). Heparin was administered at 500–1000 IU/h during hemodialysis and from the second day onwards she was given nadroparin 0.4 ml once a day for protection against deep vein thrombosis.
Therapeutic hypothermia was applied with a target temperature of 35.5 °C achieved cca 2.5 hours after the admission, and lasted the following 24 hours (Supplementary Table S7). Using bedside echocardiography, left ventricular ejection fraction was estimated at 40%. Except for bradycardia, ECG did not reveal any pathology. Bedside EEG revealed gradual development of epileptic activity (periodic high-amplitude spikes) without clinically present convulsions on the first day, which was treated with combined anti-epileptic therapy (diazepam 10 mg, clonazepam 1 mg, levetiracetam 1 g/day, valproate 1600 mg/day). CT scan of the brain was performed on the second day after admission, showing global post-ischemic edematous changes (more pronounced supratentorially); the quadrigeminal cistern was markedly reduced but present.
Forensic autopsy
On the third day after admission, the patient remained comatose (GCS 3) with absent reflexes above C1 and isoelectric EEG. After communication with the family, palliative care was started, and the patient died cca 56 hours after admission. Given the nature of the case, the forensic autopsy followed at the Department of Forensic Medicine (Faculty of Medicine and University Hospital in Hradec Kralove) revealing miosis on external examination. Dissection revealed only non-specific changes, referring to acute circulatory failure with respiratory paralysis, acute dilatation of the cardiac compartments, acute congestion of internal organs, variable presence of petechiae under the serous membranes, and the presence of brain and lung edema. Histopathological samples were not taken. Brain edema could be fully explained by hypoxic impairment during circulatory arrest. However, the contribution of chlormequat exposure cannot be completely excluded.
Discussion
Chlormequat is a colorless-to-white crystalline substance or clear-to-light yellowish color liquid with a fish-like odor.3,13 If chosen as a suicide tool, victims may mask its smell with an alcoholic beverage. High alcohol intake can significantly affect the toxicokinetic and toxicodynamic parameters of chlormequat poisoning. However, no scientific studies have evaluated the simultaneous action of both agents, and clinical reports have provided only two other cases with little information. Boumrah et al. reported a fatal poisoning of a 45-year-old male who intentionally self-injected himself with a solution containing chlormequat. 4 The detected post-mortem ethanol blood concentration was 1.16 g/l. In the same year, Vijitharan et al. described a case of a 50-year-old male who ingested chlormequat solution under the influence of alcohol. 8 Information on ethanol blood concentration was not addressed. Further research seems necessary to illuminate possible toxicological interactions between both compounds.
Chlormequat toxicity has been associated with cholinomimetic action. According to in vitro experiments, chlormequat acts as at least a partial agonist of nicotinic receptors and a weak and unselective agonist of muscarinic receptors. 2 In our patient, we observed a transient inhibition of AChE activity in erythrocytes. The finding was subsequently confirmed by in vitro experiments. Compared to the 3- to 12-hour interval, the value at admission suggests approximately 40% reduction of the enzyme activity, corresponding to mild AChE inhibition. 14 The results indicate that chlormequat is a weak AChE inhibitor at high concentrations.
The dose causing acute toxicity in humans is not known. Chlormequat toxicity has been extensively studied in animals. Experiments in mice, rats, hamsters, and a monkey study established an oral LD50 at 215 to 1070 mg/kg. Rabbits, cats, and dogs are more sensitive, with LD50 ranging from 7 to 81 mg/kg.2,15 In humans, fatalities have occurred after ingesting a mouthful (cca 40–80 ml) to 200 ml of chlormequat solutions.6–9 Nonetheless, it is challenging to derive exact quantities if the product is not specified due to varying chlormequat chloride concentrations in commercially available formulas (from 345 to 720 g/l).6,7
The signs and symptoms of chlormequat poisoning in our patient, including hypotension, desaturation, acidosis, mineral imbalance, hyperglycemia, erythema, hypothermia, and myocardial damage, were consistent with findings in other reported cases, cholinergic crisis, or resuscitation. In vivo experiments further showed that chlormequat can induce renal and liver damage. Both organs display the highest chlormequat concentrations after a single-dose oral administration. 2 In kidneys, Schulz et al. described massive protein excretion in the proximal segment of the renal tubules of sheep administered with a single dose of 200 mg/kg. 16 The presence of erythrocytes and epithelial cells and the high compound concentrations in our patient's urine suggest urinary tract damage. Nevertheless, this cannot be stated with certainty without a prior medical history and clinical record to exclude the most common diagnoses associated with the finding, such as urinary tract infections. In the liver, chlormequat can cause acute damage and diffuse fatty degeneration,16,17 which has been associated with oxidative stress and pro-inflammatory state. 18 Kang et al. 19 also showed that chlormequat induces lipid accumulation and promotes the activation of the mTOR/SREBP1 signaling pathway while inhibiting AMPK activation in both in vivo rat livers and in vitro HepG2 cells, suggesting the possible underlying mechanism. To mitigate liver toxicity and oxidative stress in our case, the therapy was amended with acetylcysteine and thiamine. Additionally, hemodialysis was adjusted with the Cytosorb capsule to downregulate the associated immune responses.
Chlormequat treatment is symptomatic, as no specific antidote exists. The management focuses on monitoring and correcting dysrhythmias, hypotension, and other metabolic or mineral imbalances. In the presented case, symptom-based therapy using conventional drugs led to rapid stabilization of circulation even after unwitnessed cardiac arrest. Prompt gastric lavage followed by activated charcoal suspension is another essential procedure for managing poison ingestions. Experiments in rats showed that chlormequat is almost completely absorbed from the gastrointestinal tract with the maximum blood level at about 2 h2, indicating a short therapeutic window. Treatment of the poisoning may also include elimination-enhancing procedures. Two studies provide chlormequat toxicokinetic data.6,10 Both showed that chlormequat undergoes biphasic elimination with a fast initial phase (half-life of 4.6–5.5 hours) followed by a slower second phase (half-life of 16.2–88.6 hours). Patients survived in both studies. However, their highest recorded chlormequat concentrations in blood were only 5.1 and 14.2 mg/l, being 147 and 53 times lower than in our patient. To accelerate chlormequat elimination, our patient was treated with continuous hemodialysis. Over the sampling interval of 46 hours following admission, chlormequat concentration in the blood followed a similar biphasic decline with the half-lives of the initial and second phase estimated at 4.3 and 9.3 hours, respectively, suggesting that hemodialysis accelerated chlormequat elimination.
Atropine, a treatment for cholinergic crises, is generally not recommended for chlormequat poisoning.6–8,10 It may accelerate chlormequat absorption from the gastrointestinal tract. 20 After chlormequat intoxication, atropine is also suspected to increase the susceptibility of myocardial muscle to dysrhythmia. Nisse et al. mentioned a 46-year-old male receiving atropine (2 mg) due to severe bronchorrhea who suddenly developed ventricular fibrillation. 7 Harter et al. reported that atropine (0.5 mg, i.v.) may have precipitated atrial fibrillation with rapid ventricular response. 6 In our patient, 6 mg of atropine in the pre-hospital and early hospital phase, followed by 3 mg in a 3-hour infusion after admission, did not induce any relevant adverse effects. Though short-lasting, the effect of initial boluses on the circulatory system provided sufficient time to initiate more effective methods of circulatory support.
Conclusion
In this report, we present the case of a 45-year-old female who committed suicide through the ingestion of chlormequat, a plant growth regulator—a method considered highly unusual and extremely rare. Due to its unpleasant fish-like odor, chlormequat may be ingested with alcohol, as observed in our case—a combination that may further alter its toxicological parameters. Chlormequat primary toxic effect in humans stems from its muscarinic and nicotinic receptor activity. Additionally, we corroborated its anti-AChE effect, which represents a secondary mechanism of toxicity in the case of high-dose poisonings. Currently, no antidote exists for chlormequat poisoning. Therefore, as with any ingestion poisoning, early intervention remains critical, with gastric lavage and activated charcoal playing key roles. Additionally, continuous hemodialysis can be considered to accelerate chlormequat elimination.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251386979 - Supplemental material for A case report of fatal chlormequat poisoning: Identification of potential additional mechanism of toxicity
Supplemental material, sj-docx-1-sci-10.1177_00368504251386979 for A case report of fatal chlormequat poisoning: Identification of potential additional mechanism of toxicity by Michal Čečrle, Martin Mžik, Alžběta Dlabková, Lubica Múčková, Martina Hrabinová, Lenka Fikejzlova, Jakub Opravil, Nela Váňová, Petr Hejna, Štěpánka Pohlová Kučerová, David Astapenko, Martin Jakl, Jaroslav Chladek, Jana Žďárová Karaová and Jaroslav Pejchal in Science Progress
Footnotes
ORCID iDs
Informed consent
Written informed consent was obtained from the patient's family for publication of this case report. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Funding
This work has been supported by the Ministry of Defence of the Czech Republic by the “Long Term Organization Development Plan 1011” – Healthcare Challenges of WMD II of the Military Faculty of Medicine Hradec Kralove, University of Defence, Czech Republic (DZRO-FVZ22-ZHN II) and by the Ministry of Health of the Czech Republic – conceptual development of research organization (UHHK, 00179906).
Declaration of conflicting interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
Data will be made available at the corresponding author on a reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
