Abstract
Objective
Alzheimer's disease (AD) is characterized by amyloid-beta (Aβ1–42) aggregation, oxidative stress, and neuronal loss, necessitating novel therapeutic agents to mitigate these pathological hallmarks.
Method
This study investigates the neuroprotective and antioxidant properties of newly synthesized borenium (compounds 1–4) and borinium (compounds 5–8) derivatives in an in vitro AD model using differentiated SH-SY5Y neuroblastoma cells exposed to Aβ1–42. Furthermore, automated Total Antioxidant Capacity assays were conducted using commercially available kits on culture media collected from cell cultures following 24 h of incubation. 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide and lactate dehydrogenase tests were done to determine the cytotoxicity of boron compounds after application to the exposed cell lines of Aβ1-42.
Results
It was determined that the boron compounds applied to the cell lines at different concentrations did not show any neurotoxic effect at a concentration of 50 μM in 24 h of incubation. All boron compounds were determined to have antioxidant properties. It was found that the borinium compounds are much more neuroprotective than the borenium.
Conclusion
These findings highlight the therapeutic potential of borenium and borinium compounds as neuroprotective and antioxidant agents for AD, warranting further mechanistic and in vivo studies.
Introduction
Alzheimer's disease (AD) the most prevalent neurodegenerative disorder, affects approximately 11 million individuals annually, with healthcare costs projected to impose a significant global burden by 2050.1,2 Predominantly sporadic (>95% of cases), with rare familial forms, AD is characterized by progressive cognitive decline, memory impairment, and behavioral changes driven by complex pathological mechanisms.3,4 Central to AD pathogenesis is the accumulation of amyloid-beta (Aβ) peptides, particularly Aβ1-42, a 36–43 amino acid fragment derived from the proteolytic cleavage of amyloid precursor protein (APP) by β- and γ-secretases.5–7 The amyloid hypothesis posits that Aβ1-42 aggregation into extracellular senile plaques initiates neurotoxic cascades, including synaptic dysfunction, neuronal apoptosis, and oxidative stress. 8 Misfolded tau proteins are stored in neurofibril balls inside the cell, which can lead to memory loss, confusion, personality and cognitive decline over time.9,10 Since Aß peptides are the main component of senile plaques, it is important to know the structure of Aß and understand its biochemical properties to understand the mechanisms of AD.11,12 Although there are many ongoing studies on this subject, an effective treatment method and effective molecular compounds have not yet been found. 13 In this study, we investigated the effect of borenium and borinium compounds that we have recently synthesized in our laboratory on Aß peptides and examined whether they will be active compounds. The new synthesis is called cationic forms of boron; borinium and borenium. This is also because it is easily surrounded by electron donor groups due to its low coordination number and empty orbitals. Borenium cations have two sigma bonds and three coordination numbers and cannot coordinate the neutral ligand by electron transfer to the empty p orbitals of boron. Despite the electron deficiency caused by the empty orbitals of boron, the activity of the donor ligand increases with electron donation.14,15 Our laboratory has synthesized four borenium (compounds 1–4) and four borinium (compounds 5–8) derivatives, the molecular structures of which are presented in Figure 1.

Formulas of borenium (1, 2, 3, 4) and borinium (5, 6, 7, 8) compounds.
Previous studies have demonstrated the safety of these compounds, showing no genotoxicity (via sister chromatid exchange and micronucleus tests) or cytotoxicity at low concentrations, suggesting their potential for therapeutic applications. 16 When we look at the literature, it has been reported in various studies that boron is a neuroprotective agent and a micronutrient necessary for the preservation of brain health and function, and that it prevents β-amyloid aggregation in the brain by forming borate esters with elemetin proteins, which suggests that effective treatments for AD can be developed through research. 17 Additionally, boron compounds, such as boric acid and boron nitride nanoparticles, have been shown to mitigate oxidative damage and neurotoxicity in various models, including aluminum-induced neurotoxicity and nicotine-induced lung cell toxicity. 18 When we look at the studies conducted in recent years, the role of oxidative processes in neurodegeneration shows that the important role of antioxidants in the treatment of AD.
Antioxidants we know such as glutathione, astaxanthin, ascorbyl palmitate, catalase and molecular hydrogen play important roles in AD. This suggests that oxidative stress or damage plays a role in the development of AD through different pathways.19–21 Therefore, there is a need for new compounds that can reduce the extent of oxidative damage that may be effective against AD. By employing cell viability assays 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide (MTT), lactate dehydrogenase (LDH) and total antioxidant capacity (TAC) measurements, we aim to evaluate the efficacy of these compounds in counteracting Aβ1-42-mediated neurotoxicity and oxidative stress. 22 This study addresses the gap in effective AD therapeutics by exploring the therapeutic potential of these chemically distinct boron derivatives, providing a foundation for their further development as pharmacological agents in neurodegenerative disease management.
Materials and methods
Cell culture and differentiation
Human SH-SY5Y neuroblastoma cells (ATCC, CRL-2266, USA) were cultured in Dulbecco's Modified Eagle's Medium (Gibco, USA) supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBS, Gibco, USA) and 1% (v/v) penicillin/streptomycin (100 U/mL penicillin, 100 μg/mL streptomycin, Sigma-Aldrich, USA). Cells were maintained at 37 °C in a humidified atmosphere containing 5% CO2 and 95% air. Upon reaching 80% confluence, cells were rinsed with phosphate-buffered saline. To induce differentiation, all-trans-retinoic acid (all-trans-RA) at a concentration of 10 M (Sigma Aldrich®) was added to a fresh medium containing 5% FBS, and cells were cultured accordingly. To establish an in vitro model of, differentiated cells were exposed to varying concentrations of Aβ1-42 obtained from Sigma Aldrich®. Cell viability assays were conducted after 24 h to determine the half-maximal inhibitory concentration (IC50) for Aβ1-42. Subsequently, boron derivatives at a concentration of 50 μM were administered to differentiated cells to assess their potential protective effects against Aβ1-42 toxicity over 24 h. Positive controls were treated with 1% Triton×(SigmaVR, USA). All experiments were performed in triplicate, with three independent biological replicates.
MTT cell viability assay
Cells were seeded at a density of 2 × 10⁴ cells/well in 96-well plates (Corning, USA) and incubated for 24 h at 37 °C with 5% CO₂ to ensure cell adhesion which were incubated at 37 °C with 5% CO2 for 24 h. Subsequently, the cells were treated with boron derivatives at a concentration of 50 μM, either individually or in combination with Aβ1-42 (IC50). Following 24 h of incubation, the MTT assay was performed using MTT Cell Growth Assay Kits (Merck Millipore). Briefly, 10 μL of MTT solution was added to each well and incubated for 4 h. Subsequently, 100 μL of acid-isopropanol (isopropanol with 0.04 N HCl) was added to dissolve any formazan crystals formed, and the optical density was measured at 570 nm using a microplate reader.23,24 Each condition was tested in triplicate wells across three independent experiments.
LDH cytotoxicity assay
Cells were seeded at a density of 2 × 10⁴ cells/well, they were incubated at 37 °C with 5% CO2 for 24 h. Subsequently, the cells were treated with boron derivatives at a concentration of 50 μM, both individually and in combination with Aβ1-42 (IC50). Following 24 h of incubation, the LDH assay was conducted using LDH Cytotoxicity Assay kits. Absorbance was measured at 450 nm using a microplate reader. 25 Each condition was tested in triplicate wells across three independent experiments.
TAC analysis
Automatic total antioxidant capacity assays were performed using a commercially available kit (Rel Assay Diagnostics VR, Gaziantep, Turkey) according to the manufacturer's description. 26 Measurements from cellular samples were carried out due to the manufacturer's recommended procedure. 27 Ascorbic acid (25 μM) was used as a positive control. Each condition was assessed in triplicate across three independent experiments.
Statistical analysis
SPSS 18.0 program was used to perform statistical analysis. Differences between means were determined using one way analysis of variance, followed by Duncan's post hoc tests, when appropriate. Alpha levels for significance were set at p < .05. All experiments were conducted with three replicates.
Results
Cytotoxicity assessment of borenium and borinium compounds
To evaluate the cytotoxic effects of novel borenium (compounds 1–4) and borinium (compounds 5–8) derivatives, differentiated SH-SY5Y neuroblastoma cells were treated with each compound at 50 μM for 24 h in the presence or absence of Aβ1–42. Cell viability was assessed using MTT and LDH assays, with results summarized in Table 1. In the absence of Aβ1-42, all borenium and borinium compounds exhibited no significant cytotoxicity compared to untreated controls (100% viability, p > .05). MTT assay results showed cell viability ranging from 93.9 ± 6.1% (borinium No. 5) to 99.2 ± 6.2% (borenium No. 3), and LDH assay results confirmed low cytotoxicity, with viability ranging from 92.8 ± 9.3% (borenium No. 4) to 96.4 ± 6.8% (borinium No. 8). In contrast, Aβ1-42 alone significantly reduced cell viability to 47.3 ± 6.9% (MTT) and 50.3 ± 6.9% (LDH) compared to controls (p < .05), while the positive control (1% Triton X-100) reduced viability to 12.1 ± 4.3% MTT and 18.5 ± 3.5% LDH.
The neurotoxic and oxidative effects of boron derivatives (50 μM) on Aβ1-42-induced Alzheimer disease model in vitro.
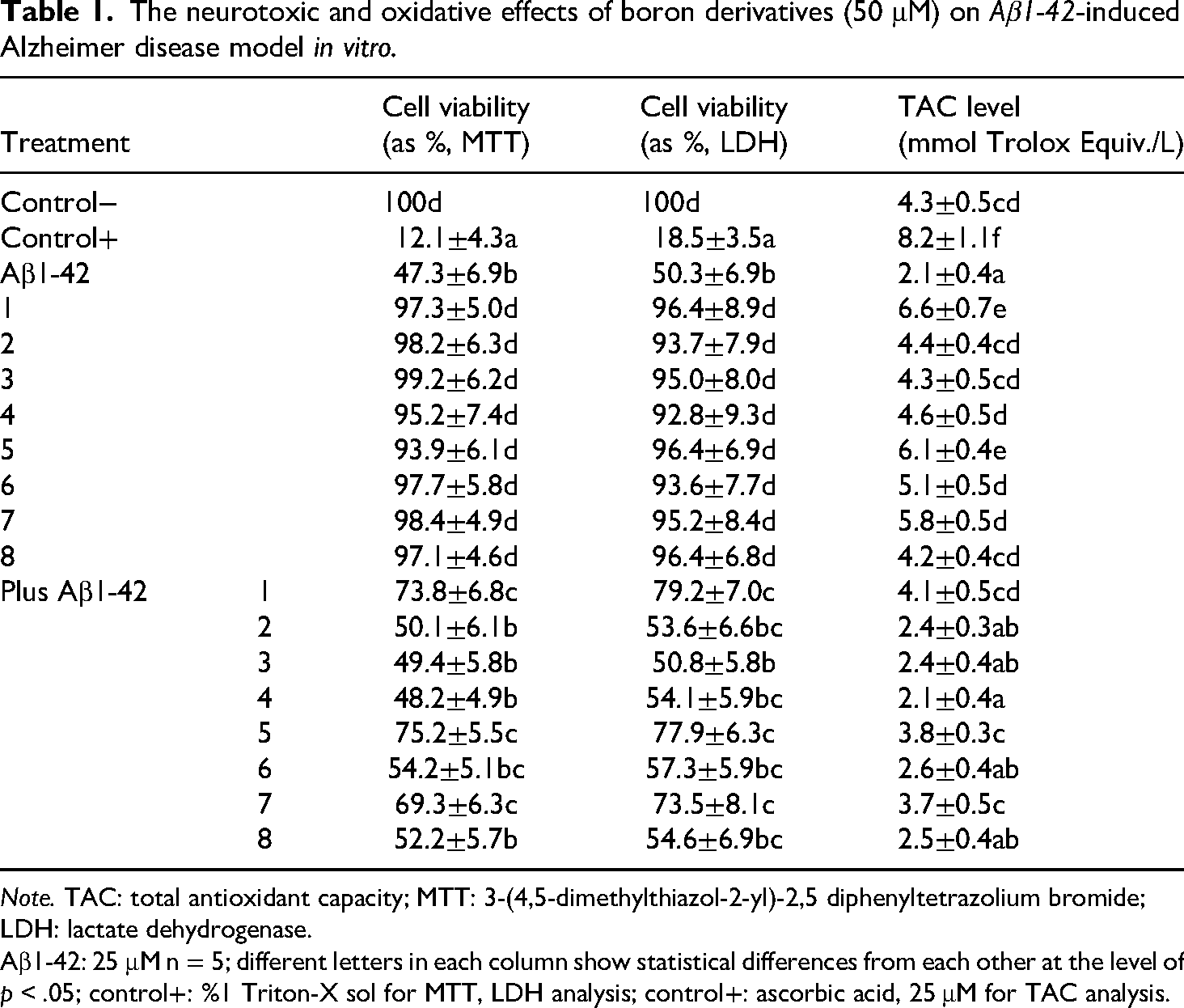
Note. TAC: total antioxidant capacity; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide; LDH: lactate dehydrogenase.
Aβ1-42: 25 μM n = 5; different letters in each column show statistical differences from each other at the level of p < .05; control+: %1 Triton-X sol for MTT, LDH analysis; control+: ascorbic acid, 25 μM for TAC analysis.
Neuroprotective effects of borenium and borinium compounds
In cells cotreated with Aβ1-42 (25 μM) and boron compounds (50 μM), borenium No. 1, borinium No. 5, and borinium No. 7 demonstrated significant neuroprotective effects (p < .05). Specifically, MTT assay results indicated that borenium No. 1, borinium No. 5, and borinium No. 7 restored cell viability to 73.8 ± 6.8%, 75.2 ± 5.5%, and 69.3 ± 6.3%, respectively, compared to Aβ1-42 alone (47.3 ± 6.9%). Similarly, LDH assay results showed reduced cytotoxicity, with viability values of 79.2 ± 7.0% (borenium No. 1), 77.9 ± 6.3% (borinium No. 5), and 73.5 ± 8.1% (borinium No. 7), compared to Aβ1-42 alone (50.3 ± 6.9%). Other compounds (borenium Nos. 2–4 and borinium Nos. 6, 8) showed limited protective effects, with viability values not significantly different from Aβ1-42 alone (p > .05), ranging from 48.2 ± 4.9% to 54.2 ± 5.1% MTT and 50.8 ± 5.8% to 57.3 ± 5.9% LDH. Specifically, cell viability assays revealed no significant decrease in cell viability compared to untreated controls, indicating the safety of these compounds at the tested concentration. Furthermore, among the evaluated compounds, borenium No. 1, borinium No. 5, and borinium No. 7 exhibited notably superior neuroprotective effects compared to other compounds.
Antioxidant capacity of borenium and borinium compounds
The TAC level of borenium and borinium compounds were assessed in vitro AD model induced by Aβ1-42 exposure as shown that Table 1. In untreated controls, TAC was 4.3 ± 0.5 mmol Trolox equivalent/L, while Aβ1-42 treatment significantly reduced TAC to 2.1 ± 0.4 mmol Trolox equivalent/L (p < .05). The positive control (25 μM ascorbic acid) yielded a TAC of 8.2 ± 1.1 mmol Trolox equivalent/L. In the absence of Aβ1-42, borenium No. 1, borinium No. 5, and borinium No. 7 significantly increased TAC to 6.6 ± 0.7, 6.1 ± 0.4, and 5.8 ± 0.5 mmol Trolox equivalent/L, respectively (p < .05), indicating robust antioxidant activity. Other compounds showed moderate TAC levels (4.2 ± 0.4 to 5.1 ± 0.5 mmol Trolox equivalent/L), comparable to controls (p > .05). In the presence of Aβ1-42, borenium No. 1, borinium No. 5, and borinium No. 7 partially restored TAC to 4.1 ± 0.5, 3.8 ± 0.3, and 3.7 ± 0.5 mmol Trolox equivalent/L, respectively (p < .05), while other compounds showed minimal improvement (2.1 ± 0.4 to 2.6 ± 0.4 mmol Trolox equivalent/L, p > .05). These findings highlight the superior antioxidant capacity of borinium compounds, particularly Nos. 1, 5 and 7, in counteracting Aβ1-42-induced oxidative stress.
Discussion
Boron, an element present in both organic and inorganic complexes, has garnered increasing attention for its potential therapeutic effects on health. 28 While the physiological impacts of boron have been under investigation for some time, our recent work has focused on newly synthesized borenium and borinium compounds.15,29 Since borenium and borinium compounds are newly synthesized compounds, studies on the different biological and physical properties of these compounds have been evaluated in our previous studies 30 and their neurotoxic and oxidative properties were studied in this article. In a novel approach, this study assessed cytological and biochemical alterations induced by these compounds such as cell viability as LDH and oxidative damage levels. The investigation elucidated the neurotoxic and oxidative damage profiles of these newly synthesized compounds, providing valuable insights into their potential therapeutic utility. The exploration of boron compounds in biomedical research is hindered, in part, by limited investigation into their toxicity, which poses challenges in both animal models and extrapolation to human physiology, due to the need for prolonged and extensive exposure to ascertain acute or chronic effects. 31 High doses of boron compounds have demonstrated toxicity across various species, particularly affecting developmental and reproductive organs.
However, existing studies indicate that boron compounds do not exhibit mutagenic, genotoxic, or carcinogenic effects. 32 Notably, in a 2011 study conducted in Turkey, the neuroprotective potential of boron was examined, wherein rats subjected to aluminum chloride-induced brain injury were treated with boric acid, revealing a reversal of damage effects at a determined dosage of 3.25 mg/2000 kcal/day. 33 Moreover, Boron Neutron Capture Therapy is employed therapeutically for glioblastoma and melanoma brain tumors. Another study found that new boron-containing carbamate compounds exhibited stronger inhibitory activity compared to the AChE inhibitor tacrine, which could be used as potential new agents for the treatment of AD and cancer. 34 Our study explored the cytotoxic and neuroprotective effects of borenium and borinium compounds in an in vitro AD model induced by Aβ1-42 exposure. This indicates that the tested compounds maintain cell viability and do not elicit adverse cytotoxic effects under the experimental conditions. This nonneurotoxic property is a critical prerequisite for potential therapeutic agents in neurodegenerative diseases, including AD. Among the compounds evaluated, borenium No. 1, borinium No. 5, and borinium No. 7 showed superior neuroprotective effects, suggesting a selective advantage of these compounds in mitigating Aβ1-42-induced neurotoxicity. The distinct neuroprotective potential may be attributed to their specific chemical structures, which could facilitate interactions with key cellular targets involved in neuroprotection. Similarly, in a parallel study, assessments were conducted to discern the cytotoxic effects of newly synthesized borenium and borinium compounds on human peripheral lymphocytes using LDH and MTT assays.19,35 Results revealed that borenium compounds exhibited no cytotoxicity even at elevated concentrations, while borinium compounds demonstrated an absence of cytotoxic effects at lower concentrations. These findings highlight the potential of borenium and borinium compounds as promising candidates for neuroprotective interventions, with borenium compounds showing particular promise due to their lack of cytotoxicity across a range of concentrations. Oxidative stress, characterized by an imbalance between the production of reactive oxygen species (ROS) and the cellular antioxidant defense mechanisms, has been implicated as a significant contributor to the pathogenesis and progression of AD. 36 Accumulating evidence suggests that oxidative damage to biomolecules, including lipids, proteins, and nucleic acids, plays a pivotal role in the neurodegenerative processes observed in AD.36,37 Furthermore, oxidative stress has been linked to the aggregation and deposition of Aβ peptides, a hallmark pathological feature of AD, as well as tau protein hyperphosphorylation, synaptic dysfunction, and neuronal loss. 38 Additionally, the antioxidant capacity of these compounds was assessed by measuring their ability to counteract oxidative stress in the AD model.
Boron compounds have been demonstrated to possess antioxidant properties through various mechanisms, including scavenging free radicals, chelating metal ions, and modulating enzymatic activity involved in oxidative stress pathways. 39 Recent studies have observed that administration of taurine and boron to brain tissue limits protein carbonyls and serum levels of advanced oxidation protein products in an experimental streptozotocin-induced AD model. 40 Furthermore, Ozdemir et al. reported that boron (boric acid) significantly reduced oxidative stress markers in this AD model in rats. 41 The another research has shown that boric acid, a commonly studied boron compound, exhibits antioxidant effects by enhancing the activity of endogenous antioxidant enzymes such as superoxide dismutase and catalase (CAT), thereby mitigating oxidative damage in cellular and animal models. 42
Additionally, boron-containing polyphenolic compounds such as boric acid derivatives have been found to exhibit antioxidant activity by scavenging ROS and inhibiting lipid peroxidation. 43 In a separate investigation, the efficacy of sodium borate decahydrate and boric acid compounds against Aß toxicity was examined, revealing experimental evidence of their capacity to enhance cellular survival rates. 44 Furthermore, boric acid has been recognized for its potential in safeguarding DNA from oxidative damage induced by aflatoxin B1. 45 These results are consistent with studies demonstrating that boron derivatives, including boric acid, mitigate oxidative damage by scavenging ROS, inhibiting lipid peroxidation, and protecting DNA. 46 Moreover, boron compounds have demonstrated the ability to mitigate heavy metal (e.g. Cd, Hg, Bi, Pb) toxicity through their antioxidant properties.47,48 Moreover, recent investigations have focused on the development of boron-based antioxidants, including borenium and borinium compounds, which hold promise for therapeutic applications. 49 These compounds have demonstrated significant neuroprotective effects against oxidative stress-induced damage in vitro, highlighting their potential utility in mitigating oxidative stress-related pathologies such as neurodegenerative diseases. 50
Conclusions
This study demonstrates the significant neuroprotective and antioxidant potential of novel borenium and borinium compounds in an in vitro AD model induced by Aβ1-42 toxicity. Borenium No. 1, borinium No. 5, and borinium No. 7 emerged as lead candidates, effectively mitigating Aβ1-42-induced cytotoxicity and oxidative stress, with borinium compounds exhibiting superior antioxidant capacity. These findings highlight their dual role in preserving neuronal integrity and counteracting oxidative damage, addressing two core pathological features of AD. The nonneurotoxic profile of these compounds at 50 μM further supports their safety for potential therapeutic applications. However, further studies are essential to elucidate their precise mechanisms of action, optimize their pharmacokinetic properties, and validate their efficacy in preclinical and clinical settings. These findings provide a complete foundation for the development of boron-based therapeutics as innovative strategies for AD and other neurodegenerative disorders.
Footnotes
Acknowledgments
The authors thanks Prof Dr Hasan Türkez and Prof Dr Halil İbrahim Uğraş. This study was presented at the VII. Polymers and Materials.
Ethical approval
It is not necessary to obtain ethical approval.
Author contributions
SB, ÖÖT, and BK conceived the study, participated in the statistical analysis and drafted the manuscript. All author have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
