Abstract
Objective
The diagnosis of Ménière's disease (MD) and delayed endolymphatic hydrops (DEH) is challenging due to overlapping symptoms. This study aimed to retrospectively evaluate the diagnostic performance of gadolinium-enhanced delayed inner ear magnetic resonance imaging (MRI) for visualizing endolymphatic hydrops (EH) in patients with MD and DEH.
Methods
This retrospective study included 191 patients with unilateral MD (n = 164) or DEH (n = 27), stratified into definite MD (DMD, n = 129), probable MD (PMD, n = 35), and DEH subgroups. All patients underwent intratympanic administration of gadolinium followed by 3.0-T MRI using 3D-FLAIR and 3D-real-IR sequences. Clinical data, including pure-tone audiometry (PTA), speech recognition score (SRS), electrocochleography (ECochG), and caloric testing, were analyzed to assess correlations between EH and auditory–vestibular function.
Results
EH was detected in 85.2% (23/27) of clinically diagnosed DEH patients. Detection rates were 96.1% in DMD cases and 28.6% in PMD cases. There was no significant difference in cochlear or vestibular EH between the DMD and DEH groups. However, the DMD group exhibited a significantly higher incidence and severity of EH than the PMD group (P < 0.05). In DMD patients, the degree of EH was positively correlated with PTA, clinical stage, and disease course (P < 0.05), and negatively correlated with SRS (P < 0.05). The degree of vestibular EH was also positively correlated with caloric test abnormalities (Spearman's ρ = 0.350, P < 0.001).
Conclusion
Gadolinium-enhanced delayed MRI of the inner ear provides objective evidence of EH and improves the diagnostic accuracy for MD and DEH. Its incorporation into standardized diagnostic protocols may further enhance clinical decision-making.
Keywords
Introduction
Ménière's disease (MD) is an inner ear disorder characterized by recurrent rotational vertigo, fluctuating hearing loss, tinnitus, and aural fullness. In 2020, the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) updated its clinical practice guidelines, establishing diagnostic criteria for definite MD (DMD) and probable MD (PMD). 1 Although the precise pathogenesis of MD remains under investigation, endolymphatic hydrops (EH)—an excessive accumulation of endolymph—is widely recognized as its core pathological feature. The pathophysiology of EH is thought to involve disrupted endolymph homeostasis, either due to overproduction or impaired absorption, leading to increased volume and pressure within the endolymphatic space. 2 This pressure may distort the membranous labyrinth and cause micro-ruptures, allowing potassium-rich endolymph to mix with perilymph. Such ionic imbalance can induce hair cell depolarization block and trigger acute neuro-otologic symptoms. 3 This foundational concept is supported by temporal bone analyses, such as the retrospective review by Foster et al., 4 which demonstrated that all patients with typical MD symptoms exhibited EH.
Delayed endolymphatic hydrops (DEH), first described by Schuknecht in 1978, 5 is characterized by the delayed development of secondary EH in an ear with pre-existing profound sensorineural hearing loss (SNHL). The diagnostic criteria proposed by the Japanese Society for Equilibrium Research (JSER) are widely used, 6 classifying DEH into ipsilateral (IDEH) and contralateral (CDEH) types depending on whether vertigo or fluctuating hearing loss occurs in the same or opposite ear as the initial SNHL. It is accepted that secondary EH directly underlies these delayed-onset symptoms. 7 Clinically, MD typically presents with spontaneous episodic vertigo lasting 20 min to 12 h, usually accompanied by concurrent auditory symptoms, whereas DEH is distinguished by vertigo attacks that arise years after profound hearing loss, with or without fluctuating auditory symptoms in the contralateral ear.
Because the clinical manifestations of MD and DEH overlap extensively with each other and with other vestibular disorders, diagnosis based solely on subjective history and pure-tone audiometry (PTA) is unreliable. This highlights the need for an objective biomarker. In vivo visualization of EH became feasible with advances in magnetic resonance imaging (MRI).8,9 In 2007, Nakashima et al. demonstrated that a 3.0-T three-dimensional fluid-attenuated inversion recovery (3D-FLAIR) sequence could clearly depict EH in humans.10,11 Subsequently, a three-dimensional real inversion recovery (3D-real-IR) sequence further improved contrast resolution between the endolymphatic space and surrounding bone structures. 12 The principle of this technique relies on the differential distribution of gadolinium (Gd)-based contrast agents. Following intratympanic injection, Gd selectively enters the perilymphatic space but not the endolymph due to the blood–labyrinth barrier. On delayed imaging, typically 24 h after injection, perilymph appears bright on T1-weighted sequences such as 3D-FLAIR and 3D-real-IR, while the endolymph remains dark. EH is identified as an abnormal enlargement of this non-enhancing endolymphatic space relative to the enhancing perilymphatic space. 13 At present, Gd-enhanced delayed MRI is the only available method that can directly and non-invasively visualize EH.
The purpose of this study was to retrospectively evaluate the diagnostic performance of delayed inner ear MRI in patients with unilateral MD and DEH, and to explore correlations between the severity of EH and clinical symptoms as well as auditory–vestibular function.
Materials and methods
This study was designed and reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 14
Study population and ethical approval
A total of 191 patients diagnosed with unilateral MD (n = 164) or DEH (n = 27) at Tangdu Hospital between September 2022 and September 2024 were consecutively enrolled in this retrospective study. Eligibility criteria included fulfillment of the 2020 AAO-HNS diagnostic criteria for MD 15 or the 1987 JSER criteria for DEH, 16 and completion of intratympanic gadolinium (Gd) injection followed by delayed MRI. Exclusion criteria were: (1) bilateral MD; (2) insufficient Gd uptake in the inner ear; (3) middle or inner ear infection or malformation; (4) history of head trauma; and (5) cerebellopontine angle lesions. The patient selection process, including exclusions and reasons, is illustrated in a flowchart (Results section).
This study was approved by the Institutional Review Board of Tangdu Hospital, Fourth Military Medical University (No. 202311-23). The requirement for written informed consent was waived owing to the retrospective design. All procedures adhered to the ethical principles of the Declaration of Helsinki (2013 revision). Patient data were de-identified prior to analysis.
Data collection and variable definitions
Clinical, audiological, and vestibular data were retrieved from electronic medical records.
Clinical variables
Disease duration (“course”) was defined as the interval in years between symptom onset and MRI examination. Clinical staging of MD followed the 1995 AAO-HNS criteria, which classify disease severity into four stages based on the PTA of the affected ear: stage I (≤25 dB HL), stage II (26–40 dB HL), stage III (41–70 dB HL), and stage IV (>70 dB HL). 17 The affected side was determined by concordant patient-reported symptoms and audiometric findings.
Audiovestibular variables
Audiometry included air-conduction thresholds at 0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz. PTA was calculated as the mean threshold at 0.5, 1, and 2 kHz. Speech recognition score (SRS) was obtained using standardized monosyllabic word lists presented at 20–30 dB above the PTA threshold.
Vestibular function was assessed using caloric testing and electrocochleography (ECochG). For caloric testing, the maximum slow-phase velocity (SPVmax) of nystagmus was measured after biauricular bithermal air stimulation (50°C and 24°C). Canal paresis ≥25% was considered abnormal. 18 For ECochG, the amplitudes of the summating potential (SP) and action potential (AP) evoked by click stimuli were recorded, and an SP/AP ratio ≥0.38 was considered positive. 19
MRI acquisition and analysis
Intratympanic Gd injection was routinely performed as part of the diagnostic protocol for suspected EH. Under local anesthesia, 1 mL of a 1:7 dilution of gadobenate dimeglumine in sterile water was administered into the middle ear cavity. Patients remained in the lateral decubitus position for 30 min, and the contralateral ear underwent the same procedure.
MRI was performed 24 h later using a 3.0T system (Philips). The protocol included T2WI-DRIVE, post-contrast 3D-FLAIR, and post-contrast 3D-real-IR sequences. Detailed parameters are listed in Table 1.
MRI acquisition parameters.

Note: MRI: magnetic resonance imaging; 3D-FLAIR: three-dimensional fluid-attenuated inversion recovery; 3D-real-IR: three-dimensional real inversion recovery; TR: repetition time; TE: echo time; TI: inversion time; ETL: echo train length; TA: acquisition time; min:s: minutes:seconds.
Image evaluation was independently conducted by two radiologists blinded to clinical information. The severity of cochlear EH (C-EH) and vestibular EH (V-EH) was graded according to the criteria proposed by Bernaerts et al. 20
Statistical analysis
All statistical analyses were performed using SPSS software (version 26.0; IBM Corp., Armonk, NY, USA). Continuous variables were summarized as mean ± standard deviation (SD) when normally distributed or as median with interquartile range (IQR) when non-normally distributed. Categorical variables were expressed as counts and percentages. Intergroup comparisons of continuous variables (DMD, PMD, DEH) were conducted using the Kruskal–Wallis H test. When significant differences were detected, post hoc pairwise comparisons were performed with the Dunn–Bonferroni correction. Categorical variables were compared using the chi-square test or Fisher's exact test, as appropriate. Correlations between EH grades and clinical parameters were assessed using Spearman's rank correlation coefficients. Inter-rater reliability for MRI grading was evaluated using Cohen's kappa statistic. A two-tailed P value < 0.05 was considered statistically significant.
Results
A total of 191 patients were consecutively enrolled, including 129 with DMD, 35 with PMD, and 27 with clinically diagnosed DEH. The patient selection process and MRI-based reclassification are illustrated in Figure 1.

Patient selection and MRI-based reclassification flowchart.
Baseline characteristics and MRI-based diagnostic reclassification of DEH patients
A total of 27 patients were initially classified as DEH based on clinical criteria (12 IDEH and 15 CDEH). Gadolinium-enhanced delayed MRI of the inner ear revealed diagnostic discrepancies compared with the clinical classification. Based on MRI findings, patients were reclassified into four groups: eight ipsilateral type, eight contralateral type, seven bilateral type, and four with no EH. For example, five patients clinically diagnosed as CDEH were reclassified as bilateral, and two patients with IDEH were reclassified as bilateral.
Overall, 13 of 27 patients (48.15%) had a discrepancy between MRI and clinical diagnosis, and DEH was excluded in four patients (14.81%) due to the absence of EH on MRI. Thus, the MRI-confirmed DEH group consisted of 23 patients. This reclassification underscores the importance of imaging confirmation in DEH diagnostic algorithms. Individual patient characteristics and discrepancies are summarized in Table 2.
Clinical data for DEH patients.

Note: “Unrecordable” indicates that the waveform or response could not be measured.
Symbols: + represents “present”; − represents “absent.”
Grading: (I–III), degree of endolymphatic hydrops.
Criteria for positive results: caloric test, asymmetry ratio ≥25%; ECochG, SP/AP ratio ≥0.38.
DEH: delayed endolymphatic hydrops; MRI: magnetic resonance imaging; C-EH: cochlear EH; V-EH: vestibular EH; Bil: bilateral type; Contra: contralateral type; Ipsi: ipsilateral type; EH: endolymphatic hydrops; ECochG: electrocochleography; NA: not applicable.
Demographics and audiovestibular characteristics
The DMD group comprised 129 patients (60 males, 69 females). MRI revealed EH in 96.1% (124/129) of these patients. Audiological testing showed a median PTA threshold of 59.0 dB (IQR 41.0–70.0), with 44.64% (50/112) achieving an SRS of 100%, while 23.21% (26/112) had an SRS below 50%. ECochG results were positive in 36.52% (42/115), negative in 40.00% (46/115), and unrecordable in 23.48% (27/115). Among the 124 patients who underwent caloric testing, 65 (52.42%) demonstrated semicircular canal dysfunction in the affected ear.
The PMD group consisted of 35 patients (14 males, 21 females). MRI detected EH in only 28.6% (10/35) of patients. Speech audiometry results were uniformly normal, with 100% SRS in all 29 tested patients. ECochG yielded only one positive result (3.12% of 32 patients). In caloric testing, 9 of 31 patients (29.03%) showed semicircular canal dysfunction.
The final MRI-confirmed DEH group included 23 patients (11 males, 12 females). MRI confirmed EH in all patients (100%). Of the 22 patients who underwent speech audiometry, 72.73% (16/22) demonstrated decreased ipsilateral SRS. ECochG results were negative in all patients. Caloric testing (n = 20) showed that 14 patients (70.00%) had ipsilateral semicircular canal dysfunction.
A comparison of clinical data across the three groups revealed significant differences in onset age, PTA threshold, low-, medium-, and high-frequency PTA, PTA threshold difference between the two ears, SRS, and SP/AP amplitude ratio of ECochG (P < 0.001), while no significant difference was found in sex, course, or caloric test results (P > 0.05). Specifically, the mean PTA in the DMD group was 59.0 ± 15.2 dB, significantly higher than the 14.0 ± 3.5 dB in the PMD group (P < 0.001). Further comparisons are summarized in Table 3.
Clinical characteristics of DMD, PMD, and DEH patients.

Note: Values are presented as mean ± standard deviation (SD) for continuous variables and as n (%) for categorical variables. P-values for continuous variables were calculated using the Kruskal-Wallis H test, and those for categorical variables were calculated using the chi-square test or Fisher's exact test, as appropriate. Superscript letters (a, b, c) indicate the results of post-hoc pairwise comparisons with Dunn–Bonferroni correction; groups sharing the same letter are not significantly different (P > 0.05), whereas groups with different letters are significantly different (P < 0.05).
DMD: definite Ménière's disease; PMD: probable Ménière's disease; DEH: delayed endolymphatic hydrops; PTA: pure-tone audiometry; SRS: speech recognition score; ECochG: electrocochleography; SP: summating potential; AP: action potential; abn.: abnormal.
MRI findings, EH grading, and inter-rater agreement
To evaluate inter-rater reliability, Kappa values were used to assess the agreement between the results of two experienced radiologists. The kappa value was 0.852 (excellent) for the assessment of C-EH and 0.816 (excellent) for the assessment of V-EH. In cases of disagreement, the final grade was determined by a third otolaryngologist after repeated evaluation of these MR images.
In the affected ears of the DMD group, 69.8% (90/129) of patients had grade II C-EH, and 84.5% (109/129) had grade II or III V-EH. In contrast, in the PMD group, the majority of patients had normal findings, with only 5.8% (2/35) showing C-EH and 25.7% (9/35) showing V-EH. In the DEH group, 47.8% (11/23) of patients had grade II C-EH, and 78.2% (18/23) had grade II or III V-EH. Both DMD and DEH groups had significantly higher percentages and more severe grades of C-EH and V-EH than the PMD group (P < 0.001). In the contralateral ear, the percentage of EH in the DEH group (30.4%) was significantly higher than that in the DMD group (9.3%) and the PMD group (5.7%). The detailed distributions of EH grades are summarized in Table 4.
Distribution of cochlear (C-EH) and vestibular endolymphatic hydrops (V-EH) grades in patients with DMD, PMD, and DEH.

Note: Values are presented as n (%). P-values were calculated using chi-square test or Fisher's exact test, with post-hoc pairwise comparisons corrected by the Dunn–Bonferroni method. Superscript letters (a, b, c) indicate that groups sharing the same letter are not significantly different (P > 0.05), whereas different letters indicate significant differences (P < 0.05).
C-EH: cochlear endolymphatic hydrops; V-EH: vestibular endolymphatic hydrops; DMD: definite Ménière's disease; PMD: probable Ménière's disease; DEH: delayed endolymphatic hydrops; EH: endolymphatic hydrops.
Representative MRI images of bilateral DEH are shown in Figure 2. These images illustrate the characteristic features of grade II C-EH and grade III V-EH in both 3D-FLAIR and 3D-real-IR sequences.
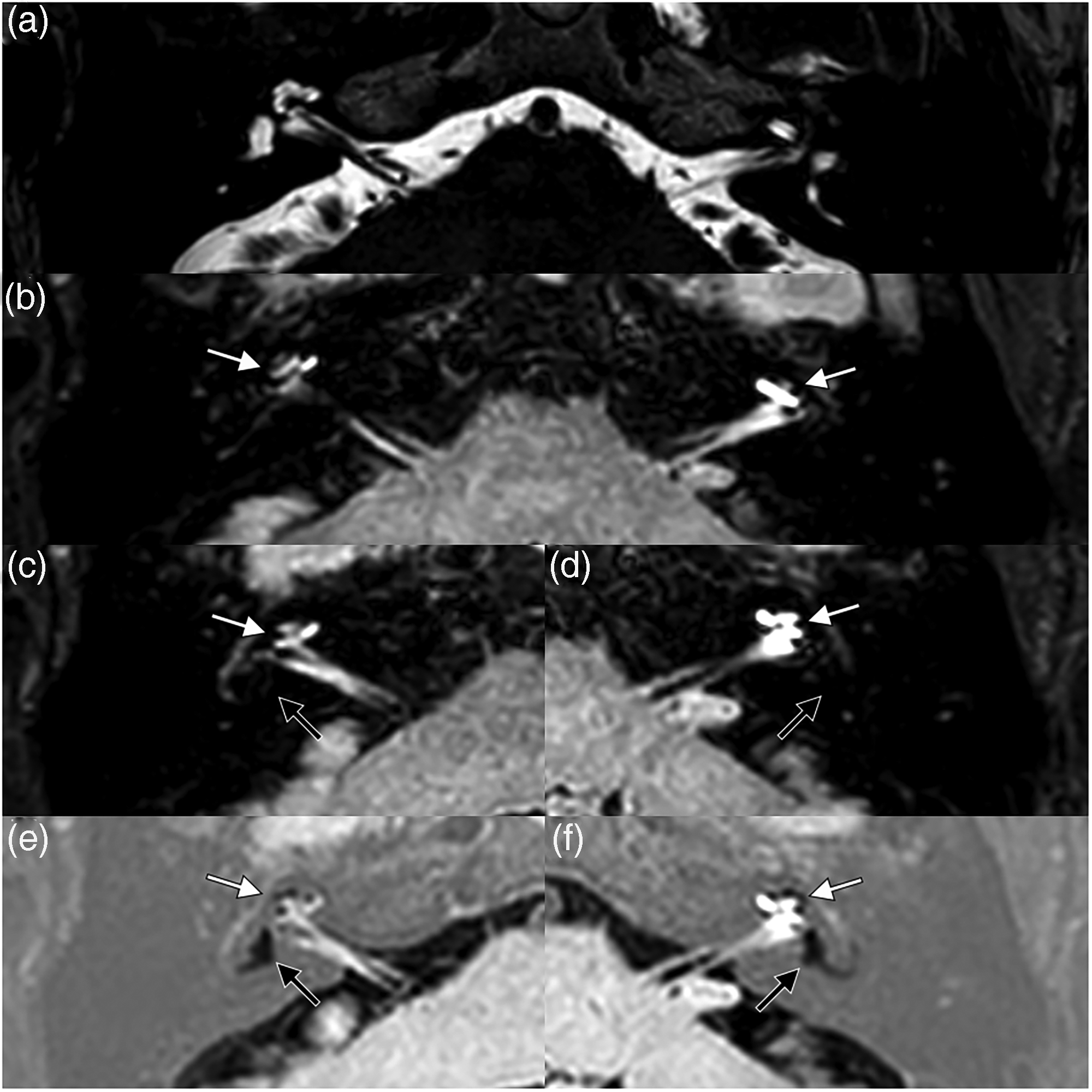
Representative MRI of bilateral delayed endolymphatic hydrops in a 57-year-old man. (a) Axial T2WI-DRIVE MRI shows symmetrical internal auditory canals without abnormalities. (b) Axial 3D-FLAIR MRI demonstrating bilateral cochlear hydrops; white arrows indicate scala vestibuli obliteration by the distended cochlear duct (C-EH grade II). (c, d) Axial 3D-FLAIR MRI of the right and left vestibules; black arrows indicate effacement of the perilymphatic space and complete loss of the bony vestibular outline (V-EH grade III). (e, f) Corresponding 3D-real-IR images confirm bilateral grade II C-EH (white arrows) and grade III V-EH (black arrows).
3D-real-IR generally exhibits better performance than 3D-FLAIR in distinguishing the endolymphatic space, perilymphatic space, and perilabyrinthine bone, although it is less effective when gadolinium concentration in the perilymph is insufficient.12,14 Therefore, the combined application of both sequences can provide more reliable diagnostic information.
Correlations between EH grade and clinical features
In the DMD group, the severity of both C-EH and V-EH was positively correlated with disease course, clinical stage, and PTA thresholds across all frequencies, while being negatively correlated with SRS. Additionally, the severity of V-EH was positively correlated with caloric test abnormalities (ρ = 0.350, P < 0.001).
In the PMD group, V-EH severity showed a significant positive correlation with low-frequency PTA (ρ = 0.439, P = 0.008), whereas no other significant associations were observed.
A detailed summary of these correlation analyses is presented in Table 5.
Correlation between the degree of C-EH or V-EH and clinical feature in patients with DMD, PMD and DEH.

Note: Values are presented as correlation coefficients (ρ) with corresponding P-values.
C-EH: cochlear endolymphatic hydrops; V-EH: vestibular endolymphatic hydrops; DMD: definite Ménière's disease; PMD: probable Ménière's disease; DEH: delayed endolymphatic hydrops; PTA: pure tone audiometry; ECochG: electrocochleography; SRS: speech recognition score; NA: not applicable (clinical stages not assessed in DEH).
Discussion
This retrospective study demonstrated that gadolinium-enhanced delayed inner ear MRI can identify EH in the majority of patients with MD and DEH, and revealed frequent discrepancies between clinical and MRI-based diagnosis in DEH. These findings suggest that imaging confirmation may improve diagnostic accuracy and provide additional information for disease subtyping.
Correlation between EH and clinical features in the DMD group
In the DMD group, the severity of both C-EH and V-EH showed a positive correlation with PTA thresholds across all frequencies, with the strongest association in the low-frequency range, consistent with previous pathophysiological models of basilar membrane compliance changes.21–23 This finding supports the concept that EH progression is closely related to low-frequency auditory dysfunction. V-EH also demonstrated correlation with PTA, which may reflect the hydrodynamic continuum between cochlear and vestibular compartments. 24 The correlation with disease course was also significant, aligning with earlier reports, 25 although variability in disease progression across individuals has been described. 26 In contrast, ECochG parameters showed dissociation from EH severity, suggesting that a positive SP/AP ratio may reflect hair cell dysfunction or membrane displacement rather than EH itself. 27 Caloric responses correlated specifically with V-EH, in line with prior studies,28,29 supporting the hypothesis that vestibular hydrops alters semicircular canal mechanics by reducing endolymph convection or causing utriculosaccular herniation. 30
Clinical significance of EH in the PMD group
PMD is generally regarded as an early stage in the spectrum of MD. In our study, V-EH correlated significantly with low-frequency PTA despite preserved mean thresholds, consistent with the concept of early vestibular involvement preceding cochlear hydrops. 31 The relatively low detection rate of EH on MRI in PMD compared with functional tests highlights the limited sensitivity of imaging in incipient disease. Histopathological data have confirmed preferential vestibular and apical cochlear involvement in early MD, 32 explaining low-frequency hearing loss via Reissner's membrane distension and ionic disequilibrium. The dissociation between structural (MRI) and functional (caloric test) findings underscores the importance of multimodal evaluation in suspected PMD.
Diagnostic reclassification in the DEH group
Our results showed that 85.2% of clinically diagnosed DEH patients demonstrated EH on MRI, but 14.8% did not, indicating diagnostic discrepancies. Notably, 30.4% of cases were reclassified as bilateral EH. This finding aligns with reports that delayed MRI frequently revises clinical DEH classification.33,34 Although auditory and vestibular functional tests often failed to localize EH in DEH, imaging provided objective confirmation, emphasizing the limitations of symptom-based diagnosis. These results support the view that MRI-based reclassification may reshape the diagnostic framework for DEH in the current imaging era.
Comparisons across DMD, PMD, and DEH groups
Our intergroup comparisons suggest that DMD is typically characterized by low-frequency hearing loss, while DEH presents with deficits across all frequencies, indicating different patterns of pathophysiology despite similar EH severity on MRI. PMD, by contrast, demonstrated milder and less frequent EH, consistent with its role as a prodromal stage of MD. 35 These observations suggest that EH imaging alone is insufficient to distinguish disease entities and should be integrated with clinical context.
Technical considerations: intratympanic vs. intravenous gadolinium
The choice of contrast administration is an important methodological issue. Intratympanic gadolinium achieves high local concentration, providing superior sensitivity for EH detection, as reflected in our study results. However, it is invasive, requires otologic expertise, and carries risks of tympanic membrane trauma or transient vertigo. 36 Intravenous gadolinium, while less invasive and more acceptable to patients, often provides lower inner ear contrast, especially in early or unilateral disease. 37 Therefore, intratympanic injection may offer greater diagnostic value in research or selected clinical settings, but intravenous administration remains more practical for routine use.
Potential false negatives and age-related EH
False negatives remain a concern in delayed MRI. Inadequate diffusion of gadolinium due to anatomic barriers, leakage through the tympanic puncture or eustachian tube, or suboptimal imaging parameters may all contribute. 38 In addition, age-related changes in inner ear fluid dynamics may influence EH expression and MRI detection. 39 This factor should be considered when interpreting imaging results in older patients.
Limitations
Several limitations should be noted. First, the retrospective design limited data completeness and precluded standardized assessment during different phases of vertigo attacks. Second, the sample size was relatively small, which may restrict statistical power. Third, only symptomatic patients were included; therefore, the prevalence and clinical implications of asymptomatic EH could not be addressed. Fourth, intratympanic gadolinium represents an off-label use; although approved by our ethics committee, prospective multicenter trials are needed to further validate its safety and diagnostic utility.
Conclusion
This study suggests that gadolinium-enhanced delayed MRI of the inner ear may provide objective evidence of EH and improve diagnostic accuracy in MD and DEH, particularly by reclassifying atypical or bilateral cases. Intratympanic gadolinium administration offered high sensitivity for EH detection but requires careful consideration of its invasiveness and off-label nature. While our findings underscore the potential of delayed MRI as an adjunct to clinical assessment, they should be interpreted with caution due to the retrospective design, small sample size, and risk of false negatives. Future large-scale, prospective studies are warranted to validate these results and to explore the role of MRI in detecting asymptomatic EH and refining disease classification.
Footnotes
Acknowledgements
The authors thank the staff of the Department of Otolaryngology, Tangdu Hospital, Fourth Military Medical University, for their assistance in patient management and MRI examinations.
Ethical approval
This retrospective study was approved by the Institutional Review Board of Tangdu Hospital, Fourth Military Medical University (No. 202311-23). The requirement for informed consent was waived because of the retrospective design. The study was conducted in accordance with the principles of the Declaration of Helsinki (1975, as revised in 2024).
Consent to participate
All patient data were de-identified prior to analysis. The off-label use of intratympanic gadolinium was specifically reviewed and approved by the Institutional Review Board.
Author contributions
Study conception and design: Lianjun Lu and Guangbin Cui. Material preparation and data collection: Zi Wang, Chengcheng Liu, Wei Gao, and Jianing Guo. Data analysis and interpretation: Yong Jing, Pengwei Ma, Rui Liang, and Hongkai Mei. Drafting of the manuscript: Chengcheng Liu and Lianjun Lu. Critical revision of the manuscript: Wen Wang and Guangbin Cui. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Discipline Innovation Development Plan Project of Tangdu Hospital (2021LCYJ017). The funding body had no role in study design, data collection, data analysis, data interpretation, or manuscript preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in the published article. Additional de-identified data are available from the corresponding author upon reasonable request.
