Abstract
Objective
To investigate the role and mechanism of long noncoding RNA LINKA (LncRNA LINKA) in hyperoxia-induced acute lung injury (HALI), specifically focusing on its impact on the GPNMB (glycoprotein nonmetastatic B protein)/HIF-1α (hypoxia-inducible factor 1-alpha) signaling pathway of apoptosis.
Methods
An experimental animal study was conducted using specific pathogen-free (SPF) male C57BL/6 mice and GPNMB knockout (KO) mice. Lung injury was assessed by measuring total protein in bronchoalveolar lavage fluid (BALF), lung wet/dry weight (W/D) ratio, serum levels of inflammatory (tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β)) and oxidative stress (malondialdehyde (MDA) and superoxide dismutase (SOD)) mediators, histopathological scoring (hematoxylin and eosin staining), apoptosis rate (terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay), and expression levels of GPNMB/HIF-1α pathway proteins (p-GPNMB, phosphorylated leucine-rich repeat kinase 2 (p-LRRK2), p-HIF-1α) and apoptosis regulators (BCL2-associated X protein (Bax), B-cell lymphoma 2 (Bcl-2)) via western blotting.
Results
Overexpression of LncRNA LINKA significantly mitigated HALI in mice, evidenced by: reduced total protein in BALF; decreased lung W/D ratio (indicating attenuated pulmonary edema); lower serum levels of TNF-α, IL-1β, and MDA, and higher SOD; improved lung histopathology with reduced inflammatory cell infiltration and alveolar septal thickening; and decreased apoptosis rate. Conversely, silencing LncRNA LINKA exacerbated HALI. Mechanistically, LncRNA LINKA overexpression increased phosphorylation of GPNMB, LRRK2, and HIF-1α, upregulated antiapoptotic Bcl-2, and downregulated pro-apoptotic Bax. Crucially, these protective effects and pathway activation were abolished in GPNMB KO mice.
Conclusions
LncRNA LINKA may protect against HALI by activating the GPNMB/HIF-1α signaling pathway, leading to suppressed apoptosis and inflammation. This identifies LncRNA LINKA as a potential therapeutic target for HALI.
Introduction
Oxygen toxicity remains a concern, especially for the lungs. 1 In current clinical practice, hyperoxia has been used to treat hypoxemia in critically ill adult patients. 2 However, the use of hyperoxia beyond the body's adaptive capacity will lead to lung damage, which is called hyperoxia-induced acute lung injury (HALI). 3 In patients with COVID-19 and volunteers exposed to hyperoxia, hyperoxia can cause diffuse destruction of the alveolar-capillary barrier, alveolar hemorrhage, vascular leakage, inflammatory infiltration, and pulmonary fibrosis.4,5 The oxygen toxicity caused by the use of inhaled high concentrations of oxygen in previous research has been confirmed in animal models, and the above phenomenon has also been observed from the histology.6–9 These studies indicate that there are similarities between the responses to oxygen toxicity in animal models and clinical patients.
At present, the treatment of HALI is still mainly symptomatic and supportive treatment, and there is a lack of specific targeted therapy. Therefore, clarifying the relevant pathogenic mechanism is expected to provide a basis for the treatment of HALI. Although the pathogenic mechanism of HALI is still unclear, more and more studies have shown that the apoptosis of alveolar epithelial cells is closely related to the occurrence and development of HALI. 10 Our previous study also confirmed that inhibiting the apoptosis of alveolar epithelial cells can significantly improve HALI. 11 In 2024, we discovered that the suppression of glycoprotein nonmetastatic B protein (GPNMB) gene expression could significantly inhibit mitochondrial-mediated apoptosis and alleviate lung injury, suggesting that GPNMB may be a potential target for apoptosis in HALI. 12 However, the specific pathways involved remain unclear.
In light of the potential role of GPNMB in cell apoptosis of HALI, we further explored the upstream factors that may regulate GPNMB or its apoptotic pathways. In this process, we focused on the long noncoding RNA LINKA (LncRNA LINKA). Also known as LINC01139, LncRNA LINKA is highly expressed in tumors and confirmed to be involved in the regulation of apoptosis.13–15 In nonsmall cell lung cancer, downregulation of LncRNA LINKA expression in A549 cells can significantly reduce cell proliferation and migration and promote cell apoptosis. 13 This suggests that LncRNA LINKA has antiapoptotic properties. It is noteworthy that the research conducted by Lin et al. demonstrated that LncRNA LINKA may play a role in antiapoptosis by recruiting GPNMB and phosphorylation of hypoxia-inducible factor 1-alpha (HIF-1α). 16 This indicates that LncRNA LINKA may influence the apoptotic pathway by regulating GPNMB.
However, in HALI, the relationship between LncRNA LINKA and GPNMB/HIF-1α signaling pathway is still unclear. This study aims to observe the changes in the GPNMB/HIF-1α signaling pathway by regulating the expression level of LncRNA LINKA, so as to explore new targets for the clinical treatment of HALI.
Materials and methods
Animal and animal model
The specific pathogen-free (SPF) healthy male C57BL/6 mice (18–22 g, 6–8 weeks old) were purchased from Laboratory Animal Center of Zunyi Medical University (Animal License No. SYXK, Qian 2021-0004), and GPNMB knockout mice were purchased from Cyagen Biosciences Inc, bred and characterized in our laboratory, all raised in the SPF laboratory of the Biological Research Center of Zunyi Medical University. Animals were acclimatized for 7 days pre-experiment, and maintained at 18°C to 22°C, a relative humidity of 50% to 60% and a 12-hour light/dark cycle with free access to mouse chow and water. All animal experiments strictly adhered to the 3R principles (Replacement, Reduction, Refinement) and were approved by the Animal Ethics Committee of the Zunyi Medical University, China (permit#: ZMU21-2202-070). Every effort was made to minimize animal numbers and suffering. Animals were immediately euthanized if exhibiting: > 20% weight loss within 48 hours, or severe lethargy or inability to access food/water. All procedures followed the ARRIVE 2.0. 17
Our experiments consist of two main parts: the verification of the protective effect of LncRNA LINKA on HALI and the importance of GPNMB as an intermediate regulator. In the first part of the experiment, 30 healthy male C57BL/6J mice were randomly divided into five groups: Control group, Hyperoxia group, Hyperoxia + OE LncRNA LINKA group, Hyperoxia + si LncRNA LINKA group and Hyperoxia + Empty vector group, with six mice in each group. In the second part of the experiment, a total of 18 healthy male C57BL/6 mice and 18 healthy male GPNMB KO (knock out) mice were randomly divided into 6 groups, Control + GPNMB wild type (WT) group, Hyperoxia + GPNMB WT group, Hyperoxia + OE LncRNA LINKA + GPNMB WT group, Control + GPNMB KO group, Hyperoxia + GPNMB KO group, Hyperoxia + OE LncRNA LINKA + GPNMB KO group, with six mice in each group. Mice in the Hyperoxia + overexpression (OE) LncRNA LINKA group, Hyperoxia + si LncRNA LINKA group, Hyperoxia + OE LncRNA LINKA + GPNMB WT group, Hyperoxia + OE LncRNA LINKA + GPNMB KO group and Hyperoxia + Empty vector group were intra-tracheally injected with 40μL adeno-associated virus vector after we anesthetize the mice with pentobarbital (50 mg/kg), while mice in the other groups received an equal volume of normal saline via intra-tracheal intubation. Under laryngoscopic guidance, the epiglottis is lifted, and a venous catheter is inserted into the trachea. Concurrently, the adeno-associated virus is administered via syringe. After treatment, the mice were normally fed for 3 weeks. Keep mice in hyperoxia (CO2 < 0.5%, O2 > 90%) or normo-oxygen chambers for 48 hours to establish a disease model. 6 The “Guide for the Care and Use of Laboratory Animals, 8th Edition” was followed and no deaths occurred among the mice during the experiment. 18 Euthanasia was performed via cardiac puncture under anesthesia using sodium pentobarbital (150 mg/kg). The chest will be opened to obtain bilateral lung tissue samples for testing purposes. The death of the mice was confirmed upon the absence of a heartbeat for a continuous duration of 5 minutes under direct visualization of the thoracic cavity.
Reagents
The fluorescence microscope and an ordinary light microscope were purchased from Leica, USA. The microplate reader was purchased from Bio-Rad Company, USA. Electrophoresis apparatus was purchased from Bio-Rad, USA; Bio-Rad ChemiDoc MP gel imaging System was purchased from Bio-Rad (USA); terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) kit (C10617) was purchased from Thermo Fisher Scientific company, USA; ELISA kits for tumor necrosis factor-α (TNF-α, E-EL-M3063), interleukin-1β (IL-1β, E-EL-M0037), malondialdehyde (MDA, E-EL-0060), superoxide dismutase (SOD, E-EL-H6188), human matrix metalloproteinase-2 (MMP-2, E-EL-M0780), and human matrix metalloproteinase-9 (MMP-9, E-EL-M3052) were purchased from Elabscience Biotechnology Co., Ltd, China. The Bicinchoninic Acid (BCA) kit (PC0020) was purchased from Beijing Solarbio Science & Technology Co., Ltd, China. Overexpression and silencing of LncRNA LINKA adeno-associated viruses were purchased from Hanbio Biotechnology Co.Ltd, China. (The sequence of overexpressed LncRNA LINKA: 5 ‘-TGG ATA AAT GAG CTG T-3’; the sequence of si LncRNA LINKA: 5 ‘-UGU CUA AGG UGG AGA UUA C-3’); antibodies of phosphorylated GPNMB (p-GPNMB), phosphorylated HIF-1α (p-HIF-1α), phosphorylated leucine-rich repeat kinase 2 (p-LRRK2), BCL2-associated X protein (Bax) (2772), B-cell lymphoma 2 (Bcl-2) (3498) and secondary antibody (7074) were purchased from Cell Signaling Technology Company, USA and YenZym Antibodies, LLC.)
Detection of total protein levels in the bronchoalveolar lavage fluid
After anesthesia, the mice in each group were fixed on the operating table in the supine position, with the nearby hair removed. The front of the neck skin was cut longitudinally, the subcutaneous tissue and muscle were separated, and the main trachea was fully exposed. Whole lung lavage was performed by injecting 1 ml normal saline into the trachea using a syringe and after 30 s, the BALF was pumped back. Repeat the above steps three times and then collect the BALF, centrifuging the supernatant. The total protein concentration in BALF was determined according to the BCA kit.
Detection of levels of inflammatory mediators in serum
After anesthesia, the approximately 1 mL carotid blood of experimental mice in each group was collected, and the levels of TNF-α, IL-1β, MDA, SOD, MMP-2, and MMP-9 in serum were detected according to the instructions of the ELISA kit.
Wet/dry ratio of lung
After anesthesia and euthanasia, the upper lobe of the left lung of mice was removed, and the excess water was wiped off by absorbent paper, weighed as wet mass. Then the left lung was dried at 60°C for 48 hours and weighed as dry mass, and wet/dry ratio (W/D) was calculated.
Hematoxylin and eosin staining and pathological scoring of lung tissue
After anesthesia and euthanasia, with the excess water wiped off by absorbent paper, the lower lobe of the left lung of mice was removed, cut into small pieces (1 mm), fixed in 4% paraformaldehyde, embedded and sectioned, stained with hematoxylin and eosin (H&E), and finally observed under a light microscope for pathological scoring. 14
Detection of apoptotic cells with TUNEL
After the mice were anesthetized and euthanasia, the right upper lobe of the lung was removed, and the operation steps were according to the instructions of the TUNEL kit. Finally, the slices were sealed with a 4',6-diamidino-2-phenylindole-containing antifluorescence quenching agent and photographed under a fluorescence microscope.
Western blotting
The mice were anesthetized and fixed on the operating table. The right lower lobe of the lung was removed, and the total protein was extracted after adding the lysate. BCA was used for protein quantification, followed by electrophoresis, membrane transfer and blocking. The primary antibody of p-GPNMB, p-HIF-1α, p-LRRK2, Bax and Bcl-2 was added and incubated overnight, and the corresponding secondary antibody was incubated at room temperature for 2 hours. The gel imaging system was exposed and developed, and the Image was processed by Image J software. The gray value of the target band was used as the relative protein expression, and β-actin was used as an internal reference.
Statistical analysis
In each experiment, under each group and condition, three to six mice or repeated samples were used. Representative images are presented for the western blotting experiments. Each experiment was independently repeated between three to six times. The results are expressed as the mean ± standard error of the mean (SEM) derived from at least three independent experiments. The experimental groups in question were groups 5 and 6, respectively. Data analysis was performed using one-way analysis of variance (ANOVA), followed by the least significant difference test. The Holm-Sidak method was applied to adjust for multiple comparisons. SPSS.29.0 was used for statistical analysis. Graphpad Prism 10.0 was used for statistical graphics.
Results
LncRNA LINKA reduced the total protein concentration in BALF in HALI
The total protein concentration in BALF of the experimental mice in each group was detected by using Bicinchoninic Acid Assay. It can be clearly observed that the total protein concentration in BALF of mice significantly increases after experiencing hyperoxia stimulation. In hyperoxia groups, the overexpression of LncRNA LINKA can effectively reduce the total protein concentration in BALF of the HALI mouse model. Similarly, silencing LncRNA LINKA significantly inhibited this process (Figure 1).

LncRNA LINKA reduced the total protein in BALF and the levels of inflammatory and oxidative stress mediators in serum, improve pulmonary exudation and reduce pulmonary edema. (a) Total protein concentration in BALF of different groups (n = 6); (b) W/T ratio of different groups; (c) levels of TNF-α in different groups (n = 6); (d) levels of IL-1β in different groups (n = 6); (e) levels of MDA in different groups (n = 6); (f) levels of SOD in different groups (n = 6); (g) levels of MMP-2 in different groups (n = 6); and (h) levels of MMP-9 in different groups (n = 6). *
LncRNA LINKA improves pulmonary exudation and reduce pulmonary edema in HALI
The alveolar-capillary leakage was evaluated by the W/D ratio of lung tissue, and the results showed higher values in the hyperoxic groups, indicating pulmonary edema and fluid exudation from the lungs. Similarly, the overexpression of LncRNA LINKA can improve lung tissue capillary leakage and reduce edema in HALI, and silencing LncRNA LINKA can increase lung water leakage (Figure 1).
LncRNA LINKA reduced the levels of inflammatory and oxidative stress mediators in serum
We used the ELISA kits to detect changes in the levels of relevant substances in the bodies of mice. The results indicated that, compared to the control group, the levels of pro-inflammatory factors (TNF-α and IL-1β) in the hyperoxia groups of mice increased, while oxidative stress damage occurred with an increase in the level of oxidized molecule MDA and a decrease in the level of antioxidant molecule SOD, suggesting that hyperoxia exposure may lead to inflammatory responses and oxidative stress damage in mice. Our research also found that the overexpression of LncRNA LINKA can partially reverse the above change, alleviating inflammation and oxidative stress damage in HALI mice, thereby protecting them. Silencing this gene inhibited the protective effect of LncRNA LINKA. The above results indicate that LncRNA LINKA may have a protective effect on HALI mice, and this effect is achieved by reducing inflammation and oxidative stress damage.
In addition, we also found that the levels of downstream MMP-2 19 and MMP-9 20 of HIF-1α are negatively correlated with the levels of LncRNA LINKA, suggesting a certain correlation between the two (Figure 1).
LncRNA LINKA can alleviate lung tissue injury and inhibit cell apoptosis
The pathological changes of lung tissue were observed by light microscope, and the results showed that LncRNA LINKA could inhibit the infiltration of inflammatory cells in the alveolar septum and alveolar space, improve vascular permeability, and inhibit the thickening of the alveolar septum. Silencing LncRNA LINKA could reverse the above effects. Compared with the Control group, the pathological score of lung tissue and the apoptosis rate in the Hyperoxia group increased. Compared with the Hyperoxia group, the pathological score of lung tissue and the apoptosis rate in the Hyperoxia + OE LncRNA LINKA group decreased. Compared with the Hyperoxia + OE LncRNA LINKA group. The pathological score of lung tissue and the apoptosis rate in the Hyperoxia + si LncRNA LINKA group increased. These results showed that the overexpression of LncRNA LINKA significantly reduced inflammatory cell infiltration, improved alveolar structure, and inhibited cell apoptosis, while LncRNA LINKA silencing had no such effects (Figure 2).

LncRNA LINKA can alleviate lung tissue injury and inhibit cell apoptosis. (a) TUNEL assay was used to detect the apoptosis of lung tissue (bar = 20μm). (b) The morphological changes of lung tissue in each group (bar = 100μm). (c) Pathological scores of lung tissue in each group (n = 3). (d) The apoptosis rate of lung tissue in each group (n = 6). *
Effect of LncRNA LINKA on the expression of GPNMB/HIF-1α signaling pathway-related proteins in lung tissues
Detection of GPNMB/HIF-1α signaling pathway related proteins by western blot showed (Figure 3) that the apoptosis level in mice subjected to high oxygen treatment significantly increased (with a decrease in the antiapoptotic protein Bcl-2 and an increase in the pro-apoptotic protein bax), accompanied by changes in proteins related to the GPNMB/HIF-1α signaling pathway (p-GPNMB, p-LRRK2, and p-HIF-1α). Overexpression of LncRNA LINKA can significantly reduce the apoptosis level in mice and activate the GPNMB/HIF-1α signaling pathway, whereas silencing LncRNA LINKA inhibits this process.
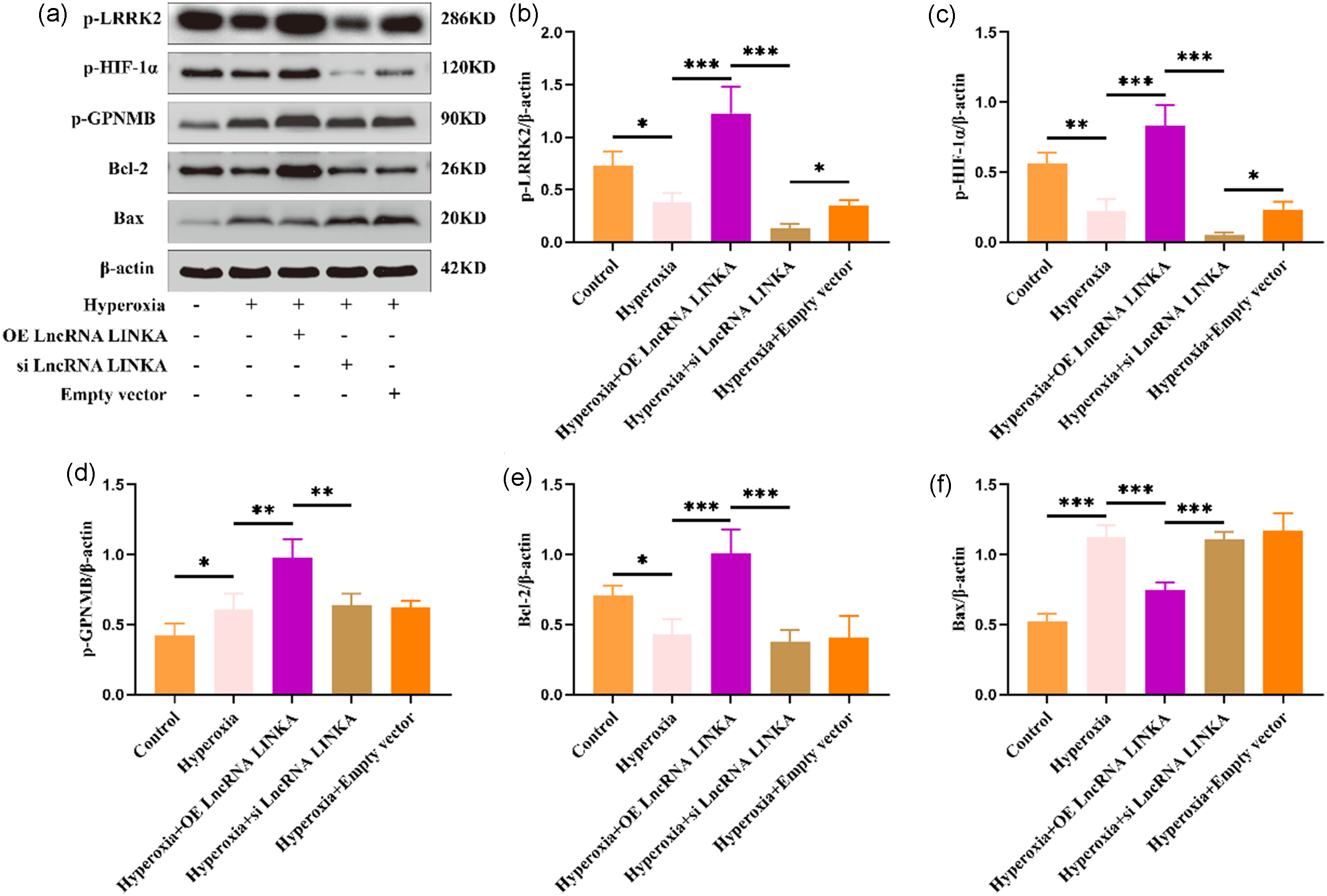
Expression of GPNMB/HIF-1α signaling pathway-related proteins in each group (n = 3). *
In summary, LncRNA LINKA can influence the levels of GPNMB/HIF-1α signaling pathway and apoptosis-related proteins in the lung tissue of HALI mice.
Knockdown of GPNMB gene blocks LncRNA LINKA activation of GPNMB/HIF-1α signaling pathway and promotes apoptosis
A HALI model was established using GPNMB wild-type mice and GPNMB knockout mice, and GPNMB/HIF-1α signaling pathway and apoptosis-related proteins were detected by western blotting. The results showed that overexpression of the LncRNA LINKA promoted the expression of proteins related to the GPNMB/HIF-1α signaling pathway (p-GPNMB, p-LRRK2, and p-HIF-1α) and inhibited apoptotic protein levels (upregulation of Bcl-2 expression and downregulation of Bax expression). However, the knockdown of GPNMB reversed the effects of LncRNA LINKA mentioned above (downregulation of p-LRRK2, p-HIF-1α, and Bcl-2 expression, and upregulation of Bax expression). The above suggests that LncRNA LINKA inhibits apoptosis levels through GPNMB and activates the HIF-1α pathway simultaneously (Figure 4).

Expression of GPNMB/HIF-1α signaling pathway-related proteins and apoptosis-related proteins in each group (n = 3). *
Discussion
Hyperoxic oxygen therapy is often used in critically ill patients in ambulances, emergency rooms, and hospitals; however, there is increasing evidence that excessive exposure to oxygen may lead to adverse consequences. 21 Mechanical ventilation and hyperoxic oxygen therapy are the cornerstone of the treatment of hypoxia and acute respiratory distress syndrome (ARDS), which are themselves harmful. 22 ARDS caused by COVID-19 requires prolonged exposure to hyperoxia, which is more than twice as long as that of other viral pneumonia.23,24 A high-quality meta-analysis of more than 16,000 critically ill patients found that exposure to free oxygen concentration was associated with a dose-dependent increase in the risk of short-term and long-term mortality. 25 Although existing evidence shows that hyperoxic oxygen therapy may cause potential iatrogenic injury, serious exposure to a hyperoxic environment is inevitable in order to maintain the life of critically ill patients, so more attention should be paid to the correct response and treatment of HALI in clinical work. At present, the treatment of HALI is still mainly based on prevention and symptomatic and supportive treatment, and there is no effective means. Effective strategies are urgently needed to improve the prognosis of patients and reduce mortality. There is no unified conclusion on the pathogenic mechanism of HALI, and there are many relevant theories, including oxidative stress, apoptosis, autophagy, ferroptosis, etc., which were found in our previous studies.6,26,27 Therefore, clarifying its pathogenic mechanism is expected to provide a theoretical basis for its clinical treatment.
LncRNA LINKA, also known as LINC01139, is located on chromosome 1q43 and consists of two exons spanning 5634 nucleotides. 28 LncRNA LINKA is highly expressed in solid and liquid tumor samples, including breast cancer, glioma, nonsmall cell lung cancer, etc. The mechanisms involved include cell proliferation, apoptosis, epithelial-mesenchymal transition (EMT), cell invasion and migration, and glycolysis.13,29,30 Malkeki et al. 13 found that downregulation of LncRNA LINKA expression level in nonsmall cell lung cancer can promote cell apoptosis. Yang et al. 14 In the diabetic nephropathy model, overexpression of LncRNA LINKA can inhibit podocyte apoptosis, showing antiapoptotic properties. The above studies suggest that LncRNA LINKA has different biological effects in different tissues, but there is limited evidence for LncRNA LINKA in HALI. In the present study, by gene regulation of LncRNA LINKA expression, we found that upregulation of LncRNA LINKA could reduce pulmonary edema in HALI mice, improve alveolar structure and local inflammatory cell infiltration, inhibit systemic inflammatory response and apoptosis, and silence LncRNA LINKA expression could aggravate lung injury. LncRNA LINKA was shown to play a protective role in HALI. Lin et al. 16 found that the antiapoptotic mechanism of LncRNA LINKA may be related to the HIF-1α signaling pathway. The study found that LncRNA LINKA could induce the phosphorylation of GPNMB, LRRK2, and HIF-1α. GPNMB can recruit and maintain the stability of p-HIF-1α. Chen et al. 31 further confirmed that LncRNA LINKA significantly stabilizes the HIF-1α protein and forms a positive feedback loop between LncRNA LINKA and the HIF-1α to regulate the inflammatory microenvironment and amplify the HIF-1α signaling cascade. However, the relationship between LncRNA LINKA and HIF-1α signaling pathway in HALI remains unclear. GPNMB is an endogenous type 1 transmembrane glycoprotein encoded by the GPNMB gene located at the 7p15 locus, which is post-transcriptionally localized to the cell membrane with 12 glycosylation sites, a PKD structural domain and an integrative recognition sequence, and can be cleaved by the metalloprotease ADAM10, releasing a soluble fragment that then binds to multiple receptors and triggers cellular responses. 32 GPNMB is known to have different functions on different types of cells, and in diabetic retinopathy, GPNMB promotes angiogenesis. 33 Available studies have shown that GPNMB also has anti-inflammatory effects. 34 HIFs) are a family of nuclear transcription factors that serve as master regulators of the adaptive response to hypoxia, including HIF-1, HIF-2, and HIF-3, controlling the transcription of many genes involved in metabolism, angiogenesis, erythropoiesis, and other adaptations to hypoxia. 35 HIF-1 is composed of HIF-1α and HIF-1β. HIF-1α, the most prominent isoform involved in the pathogenesis of inflammatory lung injury, binds to the DNA sequence of its target promoter, leading to the activation of more than 200 genes, including inflammation and apoptosis.36,37 HIF-1α shows two sides in biology, as well as in lung injury. Zhong et al. 36 found in their study on lipopolysaccharide (LPS)-induced acute lung injury (ALI) that myeloid cell receptor one could activate inflammasome 3, release inflammatory mediators and aggravate lung injury by activating HIF-1α/glycolysis pathway. Shang et al. 38 used Dachengqi decoction to inhibit the HIF-1α signaling pathway in the LPS-induced ALI model, which could reduce pulmonary edema, improve oxygenation, and inhibit the release of inflammatory mediators, thereby alleviating ALI. HIF-1α is directly involved in the regulation of a variety of pro-inflammatory proteins through the NF-κB pathway, including C-reactive protein, MDA and TNF-α. 39 The above studies indicate that activation of the HIF-1α signaling pathway can promote inflammatory response and aggravate lung injury. However, Shi et al. 40 found that dexmedetomidine can maintain mitochondrial homeostasis by activating HIF-1α/heme oxygenase-1 signaling pathway, thereby improving LPS-induced acute lung injury in vivo and in vitro. Li et al. 41 also confirmed that heme oxygenase-1 could inhibit Golgi stress and oxidative stress by activating HIF-1α signaling pathway, thus reducing LPS-induced acute lung injury. In addition, in HALI, hyperoxia can inhibit HIF-1α/vascular endothelial growth factor signaling pathway to induce apoptosis of type II alveolar epithelial cells and promote neonatal bronchoalveolar dysplasia. 37 The above studies suggest that activation of HIF-1α signaling pathway can alleviate lung injury by inhibiting inflammatory response and apoptosis. As mentioned above, it has been confirmed that LncRNA LINKA plays an antiapoptotic role, possibly by recruiting GPNMB to phosphorylate HIF-1α to maintain its stability. 16 In this study, overexpression of LncRNA LINKA could inhibit the release of inflammatory mediators, alleviate oxidative stress and lung injury, and increase the phosphorylation of GPNMB, LRRK2, HIF-1α, and antiapoptotic protein Bcl-2, while decreasing level of pro-apoptotic protein Bax to inhibit cell apoptosis. Silencing LncRNA LINKA resulted in the opposite results. When we knocked out GPNMB, the group with overexpression of LncRNA LINKA did not show the above effects, and the phosphorylation levels of LRRK2 and HIF-1α were all downregulated, and the levels of pro-apoptotic proteins were upregulated.
Meanwhile, our research findings also reveal two viable therapeutic strategies to improve HALI:
Targeted LncRNA LINKA Gene Therapy: The protective effect of LncRNA LINKA upregulation including alleviation of pulmonary edema, cell apoptosis, and inflammation, makes LncRNA LINKA a new molecular target. In therapy, adeno-associated virus (AAV) vectors can deliver the LncRNA LINKA gene to enhance its expression, similar to AAV-mediated delivery of coagulation factors in the treatment of hemophilia to reduce bleeding events.
42
New applications of drugs targeting the GPNMB/HIF-1α axis: Our experiments demonstrate that stabilizing phosphorylated HIF-1α through this cascade reaction can significantly improve HALI outcomes. Therefore, repurposing existing HIF-1α regulators like some herbal medicine to activate this pathway represents a promising strategy for clinical intervention in HALI.
43
However, this study has certain limitations. Firstly, since the causal relationship between LncRNA LINKA and HALI improvement is inferred primarily through gene overexpression or silencing experiments, the absence of HIF-1α gene regulation experiments prevents the necessity of HIF-1α in this pathway from being proven. Specifically, we cannot rule out the possibility that LncRNA LINKA's protective effects may involve HIF-1α-independent mechanisms. Secondly, while GPNMB knockout eliminates the effect of LncRNA LINKA, no related experiments on GPNMB gene overexpression in si LncRNA LINKA mice have been conducted. This absence excludes a definitive conclusion regarding whether the recruitment of GPNMB is essential for HIF-1α activation in our model. Thirdly, given the known sexual dimorphism in HALI responses, 44 the role of LncRNA LINKA in HALI in female mice remains uncertain. Further work is needed to refine the following aspects: (1) HIF-1α inhibition/rescue studies; (2) conditional overexpression of GPNMB; and (3) gender-stratified HALI modeling.
In summary, we have clarified the important role of LncRNA LINKA in the HALI mouse model. Overexpression of LncRNA LINKA can alleviate lung injury by reducing pulmonary edema, improving lung structure, inhibiting inflammatory response and apoptosis, and the mechanism may be related to the activation of GPNMB/HIF-1α signaling pathway (Figure 5).

LncRNA LINKA protects against hyperoxic acute lung injury via GPNMB/HIF-1α signaling pathway. GPNMB/HIF-1α: glycoprotein nonmetastatic B protein/hypoxia-inducible factor 1-alpha.
Conclusion
LncRNA LINKA may improve hyperoxic acute lung injury by regulating the GPNMB/HIF-1α signaling pathway, inhibiting cell apoptosis and inflammatory response.
Footnotes
Ethical approval
This study was approved by the Animal Ethics Committee of the Zunyi Medical University, China (permit#: ZMU21-2202-070).
Author's contribution
QD and LB performed the experiments and wrote the article. SQ, JZ, and HM performed the experiments. XL and KY revised the article. LC designed the study and reviewed the article. All authors read and approved the final manuscript as submitted.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Foundation of Zunyi Science and Technology and Big Data Bureau, Science and Technology Program of Guizhou Province, Scientific Research Projects of Kweichow Moutai Hospital (grant number No. (2023) 199, No. (2023) 221, ZK-2022-660, ZK-2023-544, No.MTyk 2022-17, No.MTyk 2022-24, No.MTyk2024-32).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of AI use
We employed [Deepseek] to enhance the grammatical accuracy and readability of the manuscript. All AI-generated suggestions were critically reviewed and approved by us, who retain full responsibility for the final content.
