Abstract
Objectives
Platelet–neutrophil aggregates (PNA) bridge inflammatory processes with the hemostatic system. The goal of this study was the comparative evaluation of PNAs in healthy individuals vaccinated against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), septic patients, coronavirus disease 2019 (COVID-19) patients, and a pre-pandemic control group from 2019.
Methods
This observational study involved prospective blood sample collection from vaccinated individuals at separate times after immunization with Pfizer-BioNTech mRNA vaccine, together with the retrospective analysis of laboratory data from COVID-19, sepsis, and control subjects. PNA ratios and anti-S-protein antibody levels were also measured.
Results
Following primary SARS-CoV-2 vaccination, PNA levels increased significantly, showing a gradual decline over six months while remaining elevated. Booster vaccinations did not elevate PNA levels. In COVID-19 patients PNA ratios were the highest, while in the patients with sepsis remained comparable to those of the control group. A moderate correlation was found between PNA and anti-S-protein levels after primary immunization (rho = 0.31, p = 0.0071), but no correlation was observed post-booster. Strong associations between PNA and platelet counts occurred in COVID-19 and sepsis groups, while leukocyte and neutrophil counts showed no significant correlations with PNA.
Conclusions
The results of this study may support the possibility of using PNA ratio assessment for the evaluation of the immune response.
Keywords
Introduction
Platelets are crucial for hemostasis and coagulation, interacting with leukocytes and endothelial cells in vascular processes. Lacking extensive protein synthesis capabilities, they release pre-formed molecules from granules upon activation, influencing various physiological and pathological states including thrombosis, inflammation and atherothrombosis. 1
Platelets play a significant role in immunity and inflammation through activation and cytokine release, forming heteroaggregates with granulocytes, monocytes, and lymphocytes. 2 Platelet activation is mediated through various receptors, including GPIb/IX/V, GPIa/IIa, integrins (GPIIb/IIIa), 3 and receptors for thrombin (PAR-1, PAR-4), 4 ADP (P2Y1, P2Y12), 5 thromboxane A2 (TxA2), 5 P-selectin, and tyrosine kinase receptors. 6 Activated platelets release molecules that modulate immune responses and redirect leukocytes to inflammation sites. 7
The number of platelet–leukocyte heteroaggregates, particularly platelet–neutrophil aggregates (PNAs), increases during inflammation, linking hemostasis and immune responses. Platelets carry antimicrobial peptides and enhance leukocyte functions, 8 while neutrophils, form PNAs that further activate them. 9 PNA formation involves platelet activation by stimuli like vascular injury, release of mediators (ADP, TXA2), and expression of adhesion molecules (P-selectin, GPIIb/IIIa). 10 Neutrophils bind platelets via PSGL-1, stabilized by Mac-1 binding to fibrinogen or vWF on platelets. Soluble mediators like PF4 and thrombin enhance PNA formation. 11 PNAs also promote neutrophil extracellular trap formation (NETosis), driven by platelet-derived mediators. NETs act as scaffolds for thrombus propagation and pathogen capture, thus PNAs improving pathogen clearance through combined mechanisms. 12 Dysregulated PNA formation can lead to excessive thrombosis in conditions like sepsis and ARDS due to the overproduction of NETs and increased platelet activation. 13 NETosis is critical in coronavirus disease 2019 (COVID-19), contributing to a hypercoagulable state and severe complications.14,15,16 PNAs amplify this by enhancing NET formation and thromboinflammation, creating a cycle of immunothrombosis and endothelial dysfunction.17,18,19,20 Similarly, in sepsis, PNAs and NETs drive inflammation, thrombosis, and organ damage. 21 In both COVID-19 and sepsis, NETs promote thrombosis by various mechanisms, leading to microvascular occlusions. 22
Vaccination against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has been shown to reduce COVID-19-associated platelet and neutrophil activation, and correlate with decreased infection-associated thrombotic events and mortality.23,24 Platelets can internalize SARS-CoV-2, leading to cell death and vesicle release, a process inhibitable by monoclonal antibodies against the Spike protein.25,26
This study aimed to comparatively evaluate PNAs in SARS-CoV-2 vaccinated healthy individuals (including a booster group), septic patients, COVID-19 patients, and a pre-pandemic control group from 2019. Secondarily, to analyze the potential significance of PNA within the various study subject groups, correlation analyses were also performed with the available parameters.
Methods
Study subjects
Blood samples were collected prospectively, according to their availability, from the vaccinated study participants after they expressed their willingness to participate and signed the informed consent form. All participants were over the age of 18. The study received ethical approval from the Regional Committee for the Research Ethics of the University of Pécs (Approval No. 8839-PTE 2021). This study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024.
The initial immunized group participants received their first immunization in January 2021 with two doses of the Pfizer-BioNTech mRNA vaccine, according to the vaccination schedule recommended by the producing company. Blood samples were collected at the first, second, and sixth months post-vaccination. Booster vaccinations were administered in November 2021, with additional blood samples collected one month before the booster, in the first 10–14 days after the booster, and at the third-month post-booster.
The control group consisted of subjects from 2019, before the emergence of COVID-19 in the country. Inclusion criteria required CRP levels below 5 mg/L, normal routine blood counts, and the absence of anemia, severe acute, or known chronic diseases. The COVID-19 group comprised patients with PCR-confirmed SARS-CoV-2 infection, with severe COVID-19 condition requiring intensive care treatment. The sepsis group data were also retrospectively collected from intensive care unit (ICU)-treated patients diagnosed with sepsis, with high procalcitonin (PCT) levels (>0.5 ng/mL).
Blood sample collection and analyses
All laboratory analyses were conducted at the Department of Laboratory Medicine, University of Pécs, using the Sysmex XN9000 analyzer (Sysmex Co., Kobe, Japan) in conjunction with CellaVision® for routine EDTA-whole blood analysis. This system enables complete blood count (CBC) analysis, generation of May-Grünwald-Giemsa-stained peripheral blood smears, digital image acquisition for white blood cell classification, and manual differential counting. CellaVision® identified various cell categories, including classic degenerated lymphocytes and cellular structures, including PNAs. PNA count was determined using the Sysmex XN9000 hematology analyzer and CellaVision® Peripheral Blood Application.
The Sysmex® XN-9000 analyzer prepares blood smears with a predefined angle and speed in relation to the measured patient hematocrit. The prepared blood smears’ quality is automatically assessed by the analyzer with a built-in cell localization test, checking the ability of the equipment in distinguishing between leukocytes, erythrocytes, and artifacts. The threshold of the quality assessment is a localization greater than 97% of the slides and an artifact ratio of less than 30%. In the present study, inter-rater reliability was optimized by the analysis of the peripheral blood smears by two specialists. Discrepancies were then solved with a collaborative reassessment of the smears. The process helped minimize individual biases and resulted in a more reliable data acquisition. Each neutrophil presenting at least one membrane-bound platelet was classified as a PNA. The PNA rate was calculated as the number of PNAs divided by the total neutrophil count detected on the peripheral blood smear by the software.
Additional laboratory tests were also performed. High-sensitivity C-Reactive Protein (hs-CRP) was measured using CRPHS Cardiac C-Reactive Protein (Latex) High Sensitivity reagent kit on the Roche Cobas c503 and Cobas c702 analyzers. Anti-S-protein antibody was measured using the Elecsys Anti-SARS-CoV-2 S assay on the Roche Cobas e801 analyzer (Roche, Basel, Switzerland).
The results of the subject groups with COVID-19 and sepsis laboratory analyses were collected retrospectively, using the first CBC data after admission to the ICU, including peripheral blood smear analysis.
Statistical analysis
Statistical analyses were performed using R (version 4.4.0) and RStudio (2024.09.1 Build 394). The data distribution was non-Gaussian, non-parametric tests were performed for statistical analysis. Paired sample comparisons were conducted using the Wilcoxon signed-rank test, while unpaired comparisons were performed using the Wilcoxon rank-sum test. To assess correlations, Spearman's rho correlation analysis was used.
Confidence intervals were determined with bootstrapping procedure, generating 5000 bootstrap samples by resampling the original data with replacement. Spearman's rho was calculated for each bootstrap sample. 95% Confidence interval has been then obtained from the distribution of the bootstrapped rho values, using the percentile method, by selecting the 2.5th and 97.5th percentiles of the bootstrap distribution.
The threshold of significance was set at p < 0.05, and results were visualized using appropriate graphical representations in R, with “ggplot2” and “ggpubr” libraries. The reporting of this study conforms to STROBE guidelines. 27
Results
This study included subjects stratified into groups based on their immunization process: The initial immunization group (n = 48) and the booster group (n = 30). To evaluate and compare the findings of the immunized cohorts, retrospectively collected laboratory data were analyzed from patients with severe COVID-19 (n = 80) and sepsis (n = 26), along with a control group (n = 19). Descriptive analyses by age and sex are presented in Table 1.
Study groups’ size, age, and sex distributions.
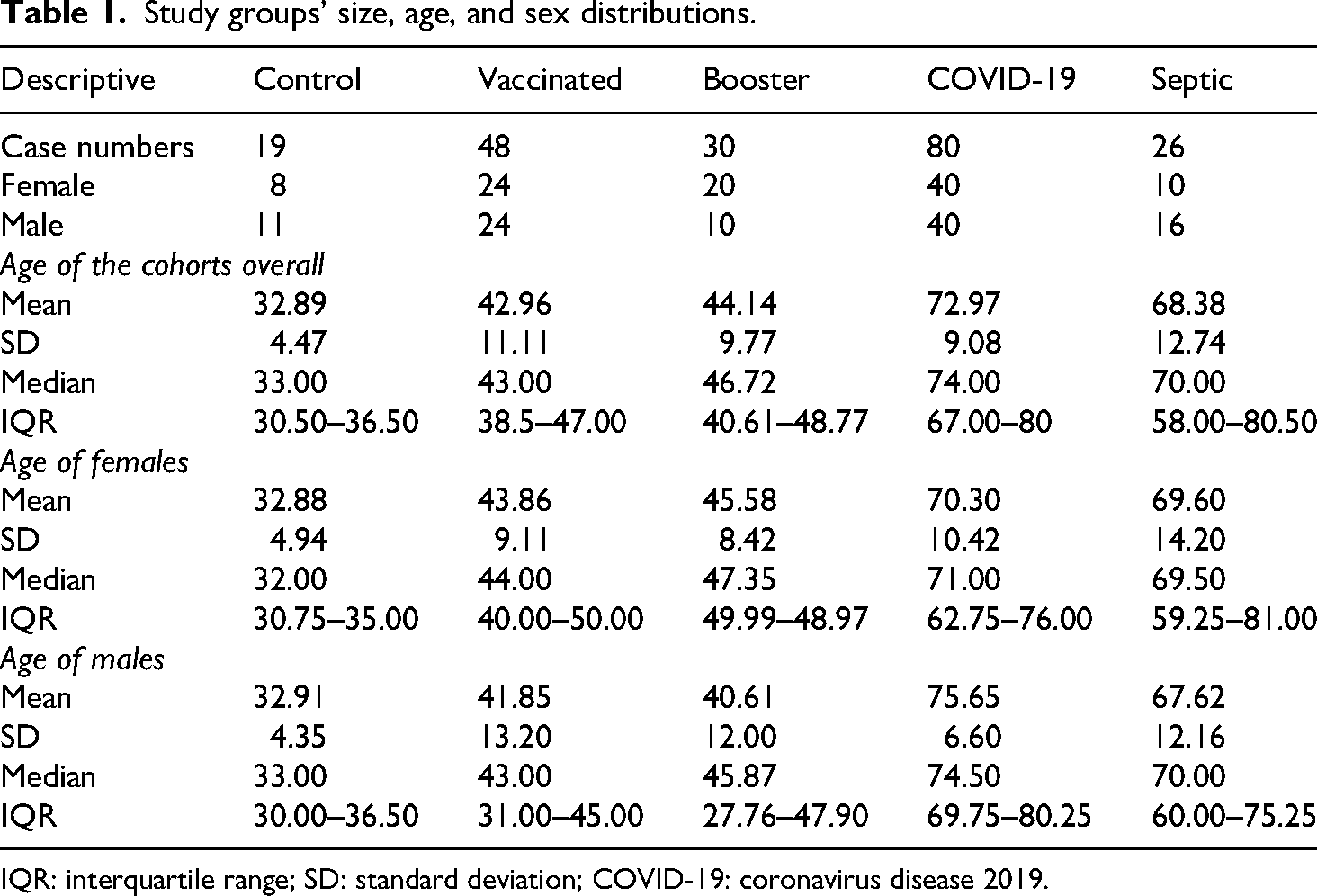
IQR: interquartile range; SD: standard deviation; COVID-19: coronavirus disease 2019.
The median PNA ratio in the control group was 3.60% [1.80–11.45]. Platelet counts were lower in the group of septic patients compared to the other groups. Leukocyte counts and neutrophil granulocyte counts were different in the septic and COVID-19 groups (Supplemental Table 1).
The comparative analysis of PNA values across the examined groups is represented in Figure 1. Since most of the data was not normally distributed, nonparametric tests were used for comparisons. PNA values in the septic patient group were comparable to those in the control group. In the group of COVID-19 patients, PNA values were higher than in the control group and showed the greatest variability among the examined groups.

Platelet–neutrophil aggregate (PNA) ratio across the study groups compared to the control group.
Within one month following the first immunization, PNA values were significantly higher compared to the control group and gradually decreased during the first six months but remained higher than the control values. The values before the booster vaccination were similar to those in the groups following the first immunization but were still higher than the control values. No significant PNA increase was observed after the third vaccination, compared to pre-booster measurements. Three months after the booster vaccination, PNA values showed a decrease and were not different from the control group values.
The PNA values and the parallel S-protein-neutralizing antibody levels in vaccinated and booster-vaccinated individuals were also analyzed (Figure 2). S-protein levels declined significantly after the fourth month following the first immunization, while PNA values showed a moderate decrease. Post-booster antibody levels significantly increased during both measurement periods despite comparable PNA values.

Within-group comparisons of the vaccinated and booster study subjects.
The correlation between PNA values and log-transformed anti-S-protein antibody levels after the first two doses of the primary immunization was moderate (rho = 0.31, p = 0.0071) and significant. In the booster-vaccinated group, no correlation was found between anti-S-protein and PNA values (rho = −0.11, p = 0.380). A positive moderate correlation (rho = 0.354 [0.032–0.607], p = 0.034) was observed between the PNA rate and log-transformed anti-S-protein antibodies in the samples drawn 150–180 days following the first two doses of primary immunization In the case of the samples acquired in the first 14–30 days and 30–60 days following primary immunizations, no significant correlation could be observed. In the case of the booster group, no significant correlation could be observed in any of the obtained sample groups (Figure 3).

Spearman-correlation between platelet–neutrophil aggregate (PNA) and log-transformed anti-S-protein antibody levels in the two immunized groups.
Subsequently, we examined the relationship between PNA and platelet count, white blood cell count, and neutrophil granulocyte count across our groups (Figure 4). Strong, positive linear correlations were found between PNA and platelet count in the COVID-19 and septic groups. A weak correlation was observed between PNA and platelet count following the first immunization. No correlation was identified between PNA and either leukocyte or neutrophil granulocyte counts in any group.
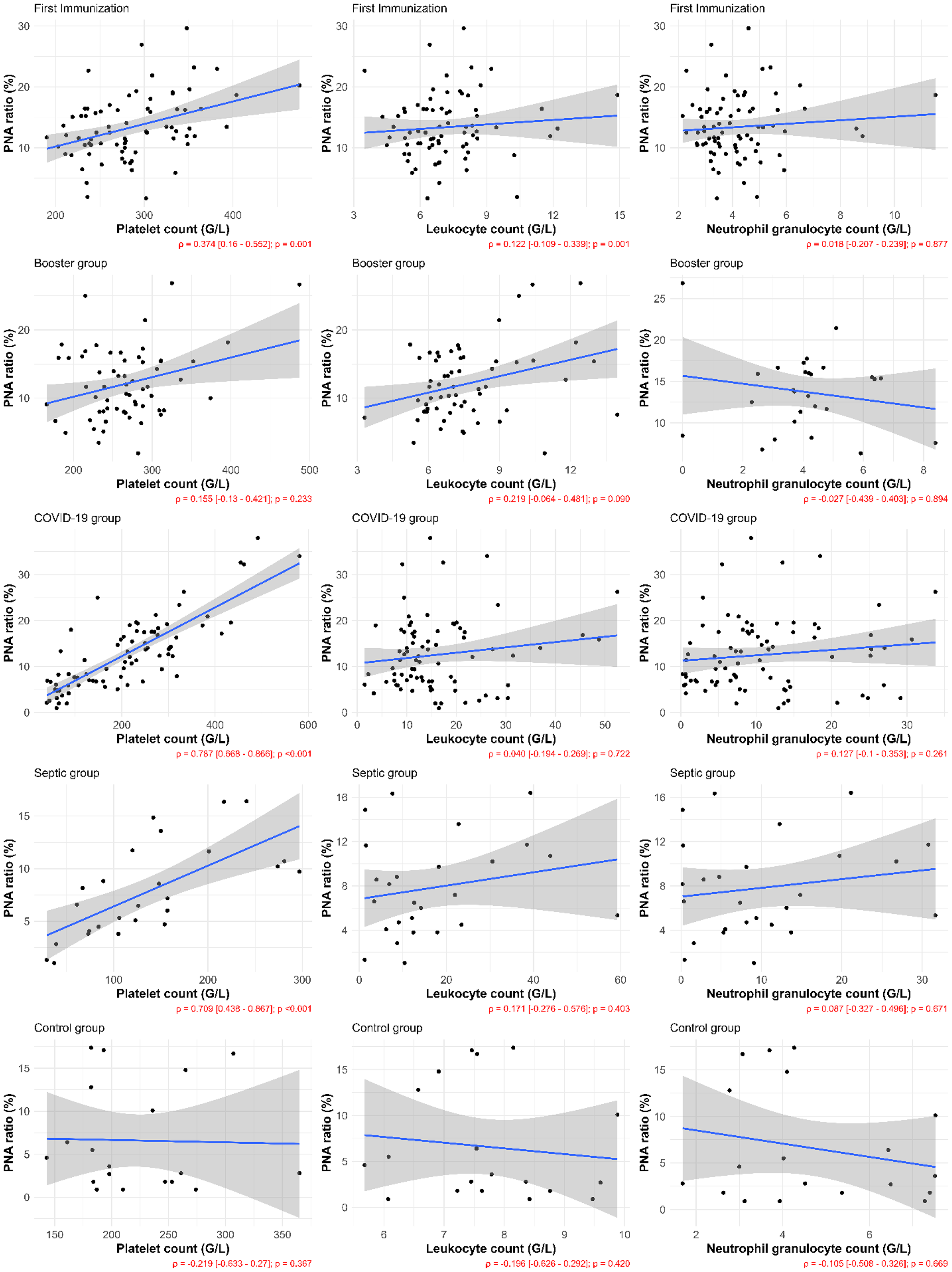
Spearman-correlation analyses of platelet–neutrophil aggregate (PNA) with platelet count, leukocyte count, and neutrophil granulocyte count, respectively in each study group (Spearman's rho—ρ).
Discussion
Following initial immunization, PNA values transiently increased, returning to control levels by approximately six months, though median values remained elevated. Concurrently, anti-S-protein antibody concentrations declined. The prolonged PNA elevation during the first six months post-initial immunization may be linked to the extended presence of mRNA and recombinant Spike protein, which Boros et al. 28 detected for over a month and six months postvaccination, respectively, potentially causing sustained platelet and neutrophil activation.
Booster vaccination increased anti-S-protein antibody levels, but PNA values did not significantly change from the already elevated pre-booster levels, which were higher than controls. The elevated pre-booster PNA values might be attributed to the booster administration coinciding with Hungary's fourth pandemic wave, potentially increasing population infection rates and prolonged platelet activation from asymptomatic viral infections. 29 The differing immune response to a booster, acting as a reminder dose, could explain the lack of further PNA elevation. This aligns with Zhou et al., 30 who reported similarly elevated pre-booster PNA levels in a Japanese cohort that did not increase post-third vaccination, suggesting reproducibility across populations. Furthermore, the stable PNA ratio post-booster mirrors observations that vaccinated COVID-19 patients exhibit lower platelet and neutrophil activation than unvaccinated patients, correlating with better clinical outcomes. 24
In septic and COVID-19 patients, strong positive correlations between PNA ratio and platelet count suggest that elevated PNA is linked to increased platelet numbers, which can also indicate inflammation, 31 although severe sepsis may present with leukocytopenia and thrombocytopenia. In a study investigating the role of PNA among patients with deep venous thrombosis (DVT), Zhou et al. 32 found no correlation between PNA ratio and platelet count. However, according to the results of the aforementioned study, PNA level correlated with total platelet activation, highlighting the underlying mechanism of PNA formation, requiring the activation of both, platelets and neutrophil granulocytes.
Post-immunization, weaker PNA-platelet correlations imply that PNA elevation results from platelet and neutrophil activation even at normal cell counts, not solely from increased platelet numbers. Moreover, a study conducted by Mediu et al. 33 found no inflammatory processes between 21 and 31 days after the administration of the second dose of the vaccine in subjects immunized against SARS-CoV-2.
Study limitations include incomplete follow-up for all participants after the first immunization (though complete for the booster, allowing paired tests) and the absence of pre-vaccination data for the initial immunization group. The control group data were retrospectively collected from a pre-pandemic period to exclude asymptomatic SARS-CoV-2 infections. During the timeframe of the analysis, it cannot be ruled out that asymptomatic, unrecognized COVID-19 infection occurred in some of the cases even before the first immunization. Precise data on previous SARS-CoV-2 immunization or infection in the septic patient group could not be obtained, with the exception of the pre-pandemic control group, which data was collected in the pre-pandemic period.
These findings suggest that the PNA ratio may indicate immune activation after vaccination against SARS-CoV-2 and similar cellular interactions might occur after other vaccines, indicating immune activation and proposing PNA assessment as a potential marker. Post-vaccination PNA elevation for other pathogens could correlate with vaccine efficacy, offering a cost-effective method to monitor immune responses, especially in resource-limited healthcare settings, as PNA determination can use routine laboratory equipment or even manual smears.
Conclusions
The results of this study may suggest the importance of PNA ratio evaluation after immunization against SARS-CoV-2, and also in COVID-19 disease and sepsis. PNA ratio can indicate the activation of the immune and hemostatic system. PNA evaluation may be an emerging promising, cost-effective tool for assessing the immune response after vaccinations.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251352079 - Supplemental material for Increased platelet–neutrophil aggregate ratio after severe acute respiratory syndrome coronavirus 2 vaccination and severe infection
Supplemental material, sj-docx-1-sci-10.1177_00368504251352079 for Increased platelet–neutrophil aggregate ratio after severe acute respiratory syndrome coronavirus 2 vaccination and severe infection by Barbara Réger, Margit Solymár, Sándor Pál, Viktória Temesfői, Attila Miseta and Zsuzsanna Faust in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504251352079 - Supplemental material for Increased platelet–neutrophil aggregate ratio after severe acute respiratory syndrome coronavirus 2 vaccination and severe infection
Supplemental material, sj-docx-2-sci-10.1177_00368504251352079 for Increased platelet–neutrophil aggregate ratio after severe acute respiratory syndrome coronavirus 2 vaccination and severe infection by Barbara Réger, Margit Solymár, Sándor Pál, Viktória Temesfői, Attila Miseta and Zsuzsanna Faust in Science Progress
Footnotes
Ethical considerations
This study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024. The study is reported according to STROBE guidelines. This study was approved by the Regional Committee for the Research Ethics of the University of Pécs (Approval No. 8839-PTE 2021).
Informed consent
Participants were provided information about the study procedure, risks and benefits, and their rights. Written informed consent was obtained, including consent for publication by the participants.
Author contributions
Conceptualization: R.B., F.Zs., and P.S. Methodology: R.B., F.Zs., T.V., and P.S. Validation: R.B. and P.S. Formal analysis: P.S. Investigation: R.B, F.Zs, P.S., and T.V. Data curation: P.S. and S.M. Writing—original draft: P.S., T.V., and S.M. Writing—review & editing: M.A., T.V., and F.Zs. Visualization: P.S. Supervision: F.Zs. and M.A.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The raw data supporting the conclusions of this article will be made available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
