Abstract
Abdominal cocoon syndrome is a rare cause of small-bowel obstruction characterized by a fibro-collagenous membrane that surrounds the intestine. Here, we report a documented case in which extensive fibrous membrane stripping was combined with intraoperative placement of a transnasal intestinal decompression tube, achieving rapid recovery and durable patency. We describe a patient in his 60s with intermittent abdominal distension, colicky pain, and complete arrest of flatus and stool. Contrast-enhanced computed tomography revealed clustered jejuno-ileal loops enveloped by soft tissue density, raising suspicion of encapsulating peritonitis. Diagnostic laparoscopy identified a thick, whitish membrane that covered the stomach, whole small intestine, colon, and mesentery; after extensive adhesiolysis, the operation was converted to laparotomy for complete membrane stripping and placement of a transnasal intestinal decompression tube. Histology demonstrated proliferative fibrous tissue without IgG4-positive plasma cell infiltration, excluding IgG4-related disease. The patient resumed oral intake on postoperative day 2, was discharged on day 22, and remained asymptomatic with patent bowel loops on 3-month imaging. This case underscores the importance of cross-sectional imaging for early suspicion, supports a strategy of laparoscopic exploration followed by open decortication when necessary, and suggests that intraoperative nasointestinal tube placement may mitigate early recurrent obstruction.
Keywords
Introduction
Abdominal cocoon (AC), also known as idiopathic sclerosing encapsulating peritonitis (SEP), is an uncommon disorder in which the small intestine—and occasionally other abdominal viscera—are wrapped by a thick, grayish-white fibrous membrane that resembles a cocoon.1–3 Because the initial clinical picture is usually limited to recurrent episodes of abdominal distension, pain and mechanical obstruction, preoperative diagnosis is difficult and mismanagement is frequent. Therefore, increasing awareness among clinicians is a prerequisite for timely surgical intervention.
IgG4-related disease (IgG4-RD) is a systemic fibro-inflammatory condition characterized by dense lymphoplasmacytic infiltrates rich in IgG4 positive plasma cells, storiform fibrosis and, often, obliterative phlebitis. 4 Although its classical targets are the pancreas, salivary glands, and biliary tree, sporadic reports have demonstrated gastrointestinal involvement presenting with chronic abdominal pain, intractable vomiting, or even bowel perforation. 5 These manifestations substantially overlap with those of AC and can result in diagnostic confusion, particularly in centers where intraoperative frozen sections or specialized immunohistochemistry are not routinely available.
The two entities, however, diverge fundamentally in both pathogenesis and therapeutic paradigm. IgG4-RD responds well to glucocorticoids and B-cell-directed immunosuppression, while AC requires meticulous surgical adhesionolysis with or without membrane stripping; failure to distinguish them may expose patients to unnecessary immunosuppression or delayed definitive surgery. 4 Therefore, the surgical team must maintain a high index of suspicion and combine imaging, intraoperative findings and histopathology—including IgG4 immunostaining—to secure an accurate diagnosis. The latest consensus statement of the International IgG4-RD Working Group explicitly recommends tissue confirmation before corticosteroid therapy in any suspected gastrointestinal case.6,7
We report a patient in his 60s with extensive encapsulation of the stomach, small intestine, colon, and mesentery. Comprehensive immunohistochemical analysis—performed on samples obtained after conversion from laparoscopy to laparotomy—showed negligible IgG4-positive plasma-cell infiltration, thereby excluding IgG4-RD. We detail diagnostic reasoning, surgical decision making, and short-term outcome, and we review the contemporary literature on membrane stripping combined with placement of the nasointestinal ileus tube. By contrasting this case with the IgG4-RD spectrum, our objective is to highlight a practical diagnostic pathway that can guide surgeons faced with similar presentations.
Clinical data
Ethical Statement: Ethical approval for this case report was obtained from the Ethics Committee of Shanghai Siylyl Hospital (Approval No. SYYEC2025-002). During data collection, all potential identifiers were removed to protect patient privacy.
Patient Consent: Written informed consent was obtained from the patient for surgical and postoperative management and publication of this case report, including any accompanying images.
Medical history
Since April 2024, a Han Chinese patient in his sixties has experienced recurrent episodes of abdominal distention with pain, accompanied by flatulence and cessation of defecation. He was previously diagnosed with small intestinal obstruction at another institution through abdominal computed tomography (CT) scan and gastroscopy; conservative treatment offered temporary relief. However, a few months ago, symptoms returned and worsened, which resulted in his admission to Shanghai No. 411 Hospital (Shanghai, China) in August 2024. He denied any relevant family history and had no history of long-term smoking or alcohol consumption.
Physical examination
Vital signs were stable. Physical examination revealed general abdominal distension, with no obvious intestinal or gastric peristaltic waves. Auscultation revealed absence of bowel noises. On percussion, there was a tympanic sound with negative shifting dullness. On palpation, there was moderate muscle tension and the abdomen was somewhat tender, but there was no rebound tenderness. There was no mass to sense. Digital rectal examination was not exceptional.
Preoperative imaging
Abdominal CT centered on the right mid-abdomen revealed a tightly clustered conglomerate of small intestine loops arranged in multilayered concentric coils. The bowel walls were uniformly thickened (4–6 mm) with homogeneous enhancement, while the interloop mesenteric fat was compressed and streaked by engorged vessels. CT showed a smooth crescent-shaped rim, with a density marginally higher than the surrounding fat, circling the bowel cluster, a feature highly suggestive of a fibrous capsule. No free intraperitoneal gas was present (Figure 1). The combination of loop clustering, fibrous-appearing envelope, and mesenteric compression is highly suggestive of encapsulating peritoneal sclerosis (abdominal cocoon).
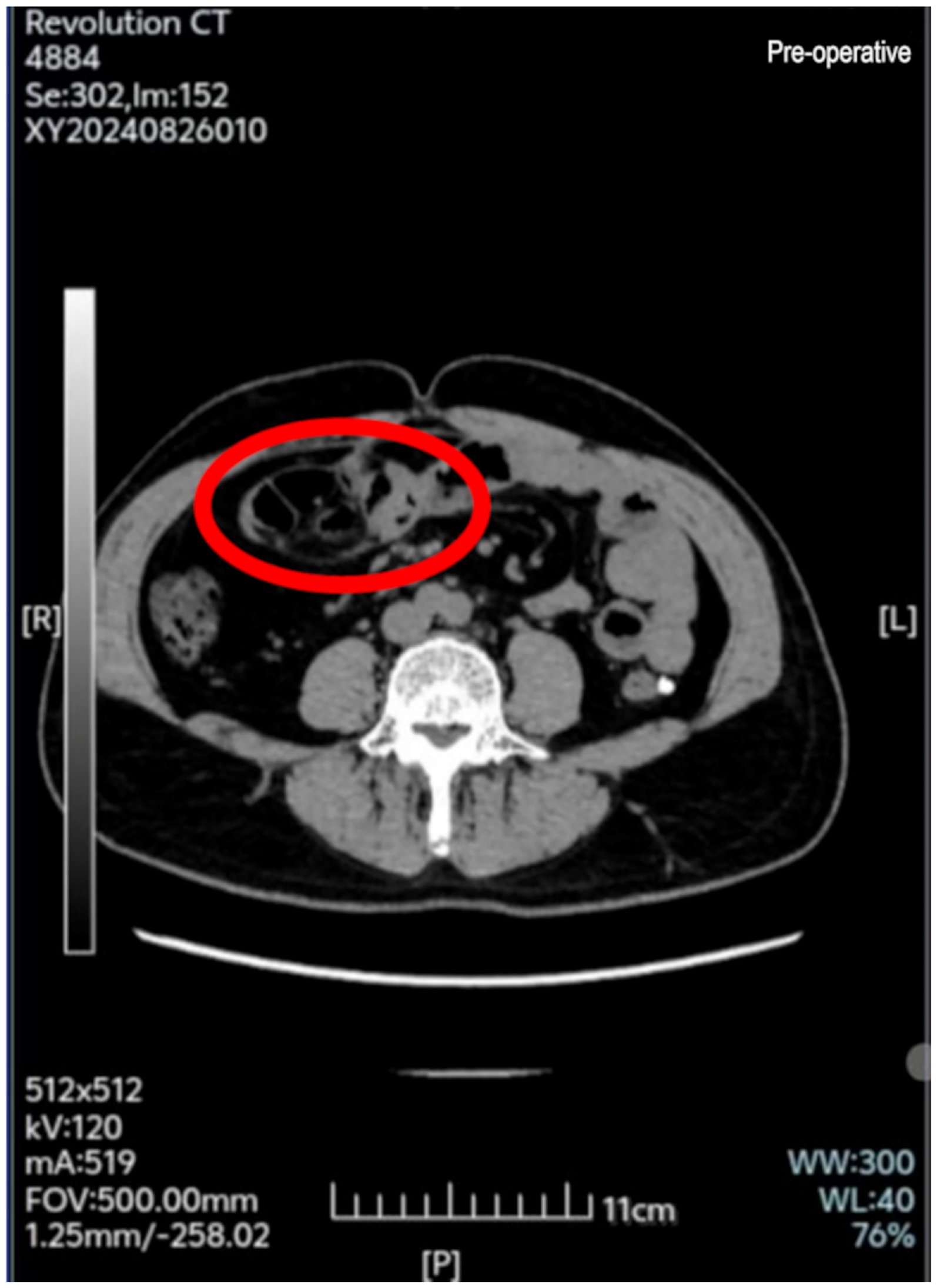
Abdominal CT with contrast optimization (axial view, right mid-abdomen). The red ellipse outlines a tightly clustered mass of small-bowel loops arranged in multilayered, concentric coils, and encased by a smooth soft tissue rim that is slightly denser than the surrounding fat, characteristics consistent with a fibrous capsule. Bowel walls are uniformly thickened with homogeneous enhancement, and the interloop mesenteric fat is compressed and streaked by engorged vessels. These findings are characteristic of encapsulating peritoneal sclerosis (abdominal cocoon). CT: computed tomography.
Colonoscopy, hindered by inadequate preparation, revealed multiple diverticula and abundant fecal material but no obstructing lesion. Ancillary studies, including abdominal ultrasound, transthoracic echocardiography, and electrocardiography, were not remarkable.
Preoperative laboratory tests
Complete blood count, coagulation profile, liver and kidney function tests, multiple tumor markers, cardiac enzymes, infectious disease screening, urinalysis, and routine stool examination showed no significant abnormalities.
Treatment course
After admission in August 2024, the patient first underwent the requisite laboratory and imaging tests. Conservative therapy was then initiated, consisting of fasting, nasogastric decompression, intravenous fluids, gastric mucosal protection, hepatoprotective agents, and nutritional support (details provided in the Postoperative Recovery section). Patient symptoms did not change much. Laparoscopic exploration was performed under general anesthesia in August 2024. Instrument mobility became severely restricted because the fibrous capsule covered almost the entire small intestine. After finishing the safest possible laparoscopic adhesiolysis, the surgeons therefore switched to laparotomy to obtain a wider field for complete membrane stripping.
Intraoperative inspection revealed: (1) absence of ascites; (2) mall-bowel adhesions forming a conglomerate mass; (3) a fibrinous membrane covering both intestinal serosa and mesentery; (4) scant purulent exudate on the bowel surface; (5) a mildly edematous, hyperemic appendix with peri-appendiceal adhesions; and (6) retracted omentum encasing the gastric greater curvature and transverse colon (Figure 2).

Laparoscopic exploration revealing extensive grayish-white fibrous membrane in the abdominal cavity. (a) Partial small bowel; (b) encapsulated small bowel mass; (c) retracted greater omentum encapsulating stomach and transverse colon; and (d) ileocecal region after partial fibrous membrane removal.
Intraoperative diagnoses were: (1) adhesive bowel obstruction (abdominal cocoon); (2) multiple colonic diverticula; (3) hypertension; and (4) diabetes mellitus. Extensive adhesiolysis, nasointestinal ileus tube placement, and appendectomy were subsequently performed.
Postoperative tests and pathology
To determine whether this patient's condition was related to IgG4-RD affecting the gastrointestinal tract, we performed immunohistochemical testing in addition to routine Hematoxylin and Eosin (HE) staining. Blood samples were also specifically tested for IgG4-related indicators. Both blood tests and postoperative pathology results did not support the diagnosis of IgG4-RD.
Postoperative pathological HE staining revealed: small intestinal adhesions showed extensive proliferation of peritoneal fibrous tissue with collagenization, partially involving adipose tissue, containing capillary and small vessel proliferation, with minimal inflammatory cell infiltration, and no significant lymphocyte or plasma cell infiltration. The appendix showed chronic inflammation with proximal lumen obliteration and minimal pus accumulation in the distal lumen. The appendiceal mesenteric adipose tissue showed proliferative fibrous tissue involvement with focal lymphocytic infiltration. Immunohistochemistry showed: CD38 (occasional +), CD138(−), CD31 (vessels +), CD34 (vessels +), D2-40 (mesothelium +), IgG (occasional +), IgG4(−), SMA (minimal +), CD20 (minimal +), CD3 (minimal +), B-catenin (membrane +). Immunohistochemistry showed pathogenic diagnosis: without any indication of peritoneal fibrous tissue development on the small intestine surface or IgG4-related illness, chronic appendicitis with suppurative inflammation is present. Comprehensive study was advised to involve further correlation with serum IgG4 levels and clinical history.
IgG4, complement C3, C4, IgA, IgG, IgM, IgE, anticyclic citrullinated peptide antibody, antistreptolysin O, rheumatoid factor, total complement activity, erythrocyte sedimentation rate, and complete blood count revealed no significant deviations.
Postoperative recovery
We have developed a thorough treatment timeline (Figure 3) that graphically shows the main milestones and therapeutic progress from initial onset to postoperative follow up so that the patient's whole treatment course may be presented completely.

Chronological timeline of abdominal cocoon management. Diagram summarizing the key milestones—symptom onset, hospital admission, surgical intervention, postoperative recovery, and follow-up evaluations. POD: postoperative day.
Routine fluid replacement, anti-infection precautions, dietary support, and fasting constituted part of postoperative therapy. On surgical day 2, the patient had his first bowel movement; until postoperative day 14, he intermittently missed defecation. Nine hours after oral treatment, follow-up gastrointestinal iodine contrast studies revealed delayed small bowel transit, upper abdominal jejunal dilatation with stepladder air-fluid levels, and no contrast in the colon. The colon showed minimal gas and contents. The nasointestinal tube was placed in the pelvic small intestine (Figure 4).

Postoperative day 12 gastrointestinal iodine contrast study.
The patient was discharged on postoperative day 22, during which time the nasointestinal tube was gradually withdrawn partially but not completely removed. Abdominal examination at postoperative day 45 follow-up revealed no significant anomalies; normal flatus and stool resulted in gradual removal of the nasointestinal tube. Follow up by November 2024 (postoperative day 81) showed that the patient had returned to regular food and lifestyle routines many days earlier, without recurrence of abdominal distension or cessation of flatus and feces. Follow-up gastrointestinal iodine contrast study revealed that the patent digestive tract was free from notable blockage (Figure 5).
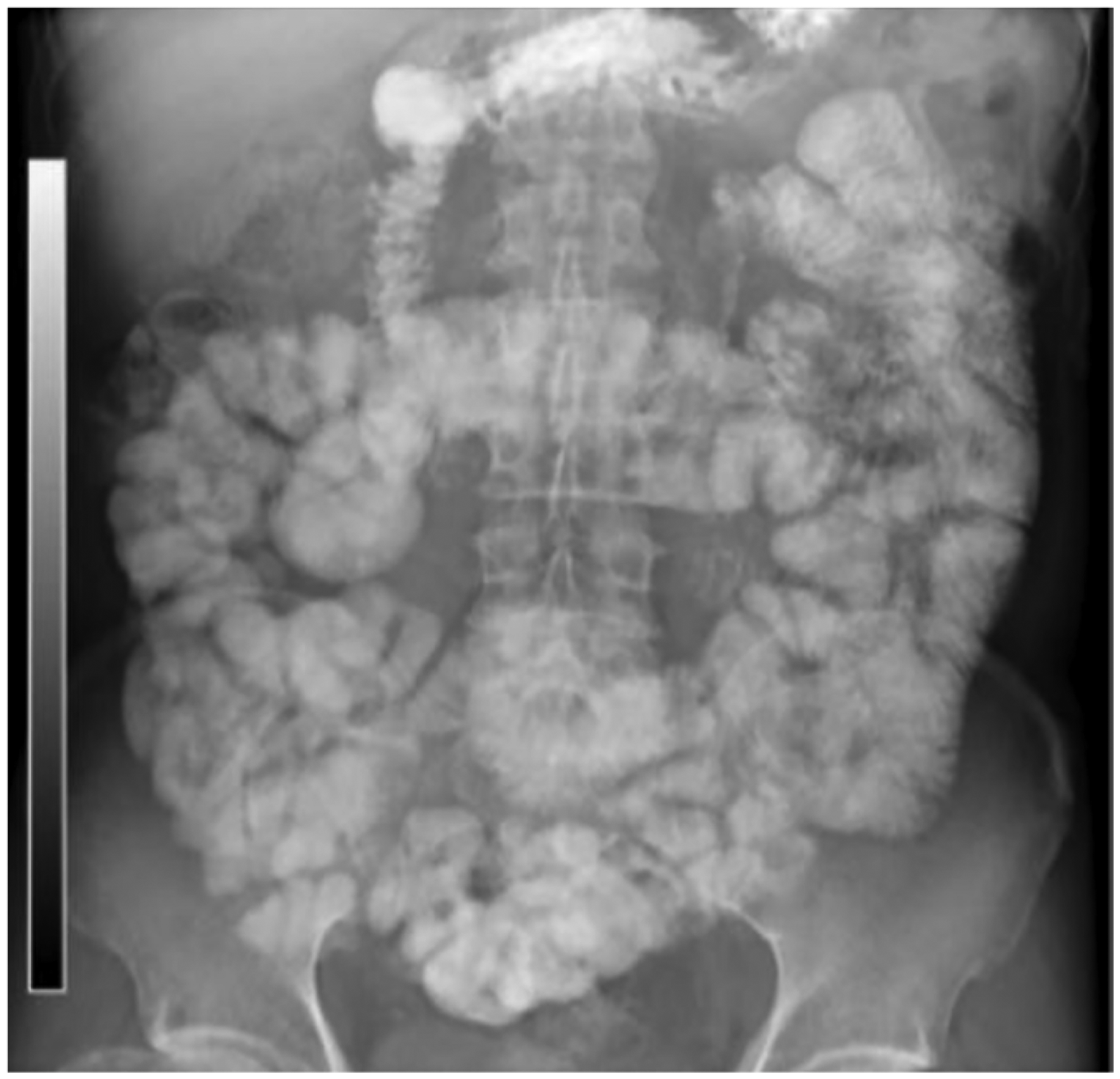
Postoperative day 81 gastrointestinal iodine contrast study.
Follow-up outcomes: At the 3-month outpatient review, the patient remained asymptomatic; contrast CT confirmed patent, nondilated small-bowel loops with no recurrent adhesions.
Patient-reported outcome: Visual-analogue-scale pain score was 0/10 from postoperative day 5 onward, and the patient had resumed unrestricted daily activities at week 8.
Intervention adherence and tolerability: Nursing logs recorded 100% compliance with daily nasointestinal-tube flushing; only mild, transient throat discomfort was noted.
Adverse events: No postoperative complications, readmissions, or unexpected events occurred during the 3-month surveillance period.
Pharmacotherapeutic Summary: When signs of postoperative obstruction emerged on postoperative day 6, the patient was started on a short-course, multimodal regimen: (1) mosapride citrate 5 mg orally twice daily (postoperative days 6–15) and domperidone 10 mg orally 3 times daily (postoperative day s7–15) to enhance gastrointestinal motility; (2) octreotide acetate 0.1 mg subcutaneously every 8 hours (postoperative days 6–15) to suppress intestinal secretions; (3) cefoperazone 3.0 g intravenous infusion twice daily (postoperative days 7–16) for surgical-site infection prophylaxis; (4) glycerin enema 20 mL per rectum twice daily (postoperative days 8–10) to facilitate defecation; (5) polyene phosphatidylcholine 20 mL intravenous drip once daily (postoperative days 7–16 ) as a hepatoprotective agent; and (6) vitamin B₁ 100 mg intramuscularly once daily (postoperative day 10 until discharge) to prevent vitamin depletion after prolonged fasting. No drug-related adverse events were recorded. Bowel sounds normalized by postoperative day 15, oral intake was gradually resumed, and the patient was discharged uneventfully on postoperative day 22.
Discussion
Etiology and epidemiological characteristics
In our situation, intraoperative finding of appendicitis most certainly played a major role in the development and aggravation of the patient's illness; yet, the presence of primary etiologies cannot be absolutely ignored. Abdominal cocoon can be classified into primary and secondary types. Usually ascribed to congenital peritoneal or vascular developmental abnormalities, 8 the pathogenesis of primary abdominal cocoon is still unknown and may be associated with asymptomatic peritoneal infection from retrograde menstruation. 9
Secondary abdominal cocoon has been associated with peritoneal dialysis, tuberculosis, prior abdominal surgery, and systemic lupus erythematosus. Each condition can trigger chronic peritoneal inflammation, leading to fibroblast proliferation and fibrin deposition, and ultimately to fibrous encapsulation.10–14
The epidemiological features of abdominal cocoon remain not entirely known. Worldwide cases have been recorded with variances depending on location, age group, sex, and lifestyle patterns. For those on peritoneal dialysis, for example, Chinese studies show an incidence rate ranging from 0.7% to 3.3%. 15
Young women patients are more often affected in India, perhaps due to lifestyle and geographical elements. 16 In terms of gender distribution, although early studies claimed female preponderance, current studies show that male cases are not unusual. 17
Clinical manifestations and imaging features
Usually involving fibrous membrane encapsulation of the small intestine, this disorder rarely affects several abdominal organs. 18 Our case is noteworthy for its extensive involvement, with fibrous membrane encapsulating the appendix and nearly all of the small intestine, while the greater gastric curvature and transverse colon were enclosed by retracted omentum. The patient primarily presented with recurrent episodes of paroxysmal abdominal distension, pain, and obstructive symptoms including cessation of defecation and flatus, showing no pathognomonic clinical features. Abdominal CT demonstrated small bowel obstruction with air–fluid levels. While some patients present with palpable abdominal masses, none was detected in our patient, likely due to intestinal fixation to the retroperitoneum and abundant abdominal adipose tissue. Based on clinical analysis, we suggest that AC should be considered when patients present with recurrent acute or subacute small bowel obstruction without other apparent causes, especially when supported by imaging findings.
Diagnostic approach
The initial diagnosis of abdominal cocoon primarily relies on imaging tests. CT scan is the preferred technique, typically revealing a characteristic clustering of small intestinal loops with membrane encapsulation, known as the “cauliflower sign.” 19 While gastrointestinal contrast tests offer limited assistance in early diagnosis, they can indicate small bowel aggregation and distortion. Magnetic resonance imaging is less frequently employed in diagnosing abdominal cocoon, despite its advantages in revealing peritoneal disease. Additionally, while endoscopic studies have limited diagnostic value, they can help rule out other gastrointestinal disorders.
Although CT scan is the initial diagnostic tool of choice, surgical exploration remains the most accurate diagnostic approach. The decision between direct laparotomy and laparoscopic exploration should consider several criteria. Even if conversion to open surgery may be necessary after confirmation, laparoscopic exploration is recommended as the first approach in highly suspected cases. This is due to the uncertainties surrounding preoperative diagnosis and the benefits of laparoscopy, which include a minimally invasive method, enhanced visibility, and multiple viewing angles. 1
To systematically summarize the diagnostic and treatment decision-making process for abdominal cocoon, we have developed a detailed surgical decision flowchart (Figure 6) based on our case experience and literature review. This flowchart demonstrates the complete therapeutic approach from patient admission and conservative treatment to surgical plan selection, which may serve as a reference for clinicians in their practice.
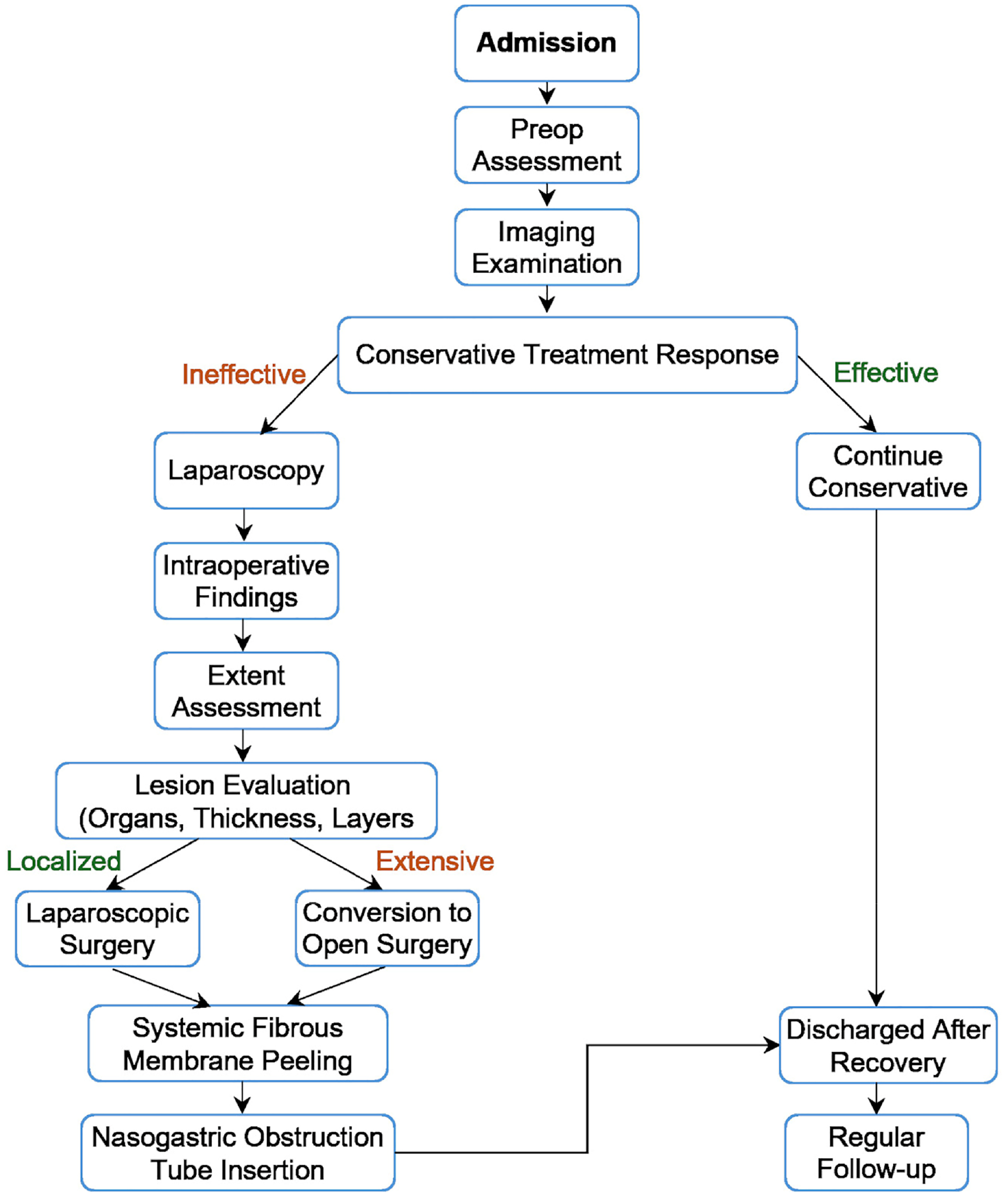
Detailed surgical decision flowchart for abdominal cocoon.
Regarding the timing of conversion to open surgery, we recommend starting laparoscopically to release adhesions between the small intestine and surrounding structures (abdominal wall, colon, stomach) until the intestine is adequately mobilized. If a diffusely encasing fibrous membrane or limited instrument mobility compromises safe dissection, the surgeon should promptly convert to laparotomy. The open approach provides a broader, unobstructed operative field, eliminates the limitations of intracorporeal vision and instruments, and allows comprehensive membrane stripping and adhesiolysis.
Differential diagnosis
Since both conditions present with recurrent abdominal pain, vomiting, and intestinal obstruction, gastrointestinal involvement of IgG4-RD must be taken into consideration. 5 Differential diagnosis heavily relies on postoperative histological and immunohistochemical results.
With abundant IgG4-positive plasma cells in afflicted tissues, the main pathological characteristics of IgG4-RD are storiform fibrosis, obliterative phlebitis, and plasma cell invasion. With lymphocytes positive for CD20, CD38, or plasma cells positive for CD138, immunohistochemical staining reveals IgG4-positive cell infiltration. 4 Furthermore, a noteworthy feature is raised serum IgG4 levels. Although most patients have a high recurrence rate, extended corticosteroid treatment is usually required to keep remission since IgG4-RD responds well to glucocorticoid therapy. On the other hand, abdominal cocoon mostly calls for surgical treatment, thus precise differentiation is essential for the suitable therapeutic intervention.
Treatment strategy selection
The severity and the patient's general condition determine the approach to treating abdominal cocoon. Patients with mild symptoms might find conservative management, including fasting, gastrointestinal decompression, and nutritional support, appropriate. For patients showing intestinal obstruction or other severe complications, however, surgical intervention becomes necessary. Maximizing cocoon membrane removal, adhesiolysis, complete peritoneal lavage, and occasionally bowel resections and anastomosis when needed constitute traditional surgical concepts. 20
Compared to traditional surgery that involves only fibrous membrane removal and adhesiolysis, intraoperative placement of a nasointestinal ileus tube significantly reduces the risk of postoperative bowel obstruction recurrence. 21 Furthermore, prophylactic long intestinal-tube splinting has been reported to mitigate recurrence, particularly in high-risk patients.22,23
With better intestinal function recovery than simple gastrointestinal decompression, patients undergoing tube placement show noticeably higher daily gastrointestinal decompression volumes than those in conventional surgery groups, so facilitating reduction of intestinal inflammation and burden. Whereas the conventional surgery group mostly experienced grades 2 and 3 complications, the Clavien–Dindo complication classification 24 shows that most complications in the tube placement group were grades 0 and 1. Six-month follow up revealed higher readmission rates for bowel obstruction in the traditional surgery group.
Regarding postoperative nasointestinal tube care, based on our case experience, attention should be paid to tube-related respiratory infections. Prior to removal, adequate hydration helps reduce friction between the tube and intestinal wall, and multiple small-amplitude withdrawals are recommended to prevent intussusception. Tube removal should be considered only after confirmed patency on postoperative imaging tests and continuous good gastrointestinal function.
Prognosis and follow-up management
Timely diagnosis and treatment will determine the course of an abdominal cocoon. Patients who manage conservatively should be closely monitored since they are prone to recurrence. Although surgical patients may be at risk for postoperative complications, generally the prognosis is good. Improving patients’ quality of life depends on suitable postoperative care and long-term follow up.
Future research prospects
Rare but severe, abdominal cocoon is a condition needing great clinical suspicion and accurate imaging diagnosis; definitive diagnosis usually calls for surgical exploration. Although intraoperative nasointestinal ileus tube placement is a relatively novel approach to treating, current research suggests its effectiveness in improving postoperative adhesive bowel obstruction, preventing recurrence, and enhancing prognosis. Future studies should focus on developing more efficient diagnosis and treatment strategies, as well as investigating the epidemiological features of the abdominal cocoon.
Limitations
This case report is inherently limited by single patient, single center nature and by the specialized resources required for intraoperative nasointestinal ileus tube placement. In low-resource settings, acquisition and routine use of such devices may not be feasible, restricting wider adoption of our approach. Additional studies conducted in hospitals or institutions with different economic resources are needed to develop alternative, resource-appropriate strategies and to confirm the generalizability of our findings.
Conclusions
This report demonstrates the successful management of extensive abdominal cocoon through combined fibrous membrane stripping and intraoperative nasointestinal ileus tube placement. Key lessons include: (1) CT scan showing the characteristic “cauliflower sign,” is crucial for early suspicion of AC; (2) immunohistochemical analysis, including IgG4 staining, is essential to exclude IgG4-RD, which requires completely different therapeutic approaches; (3) initial laparoscopic exploration followed by conversion to open surgery when necessary provides optimal visualization while maintaining a minimally invasive approach; (4) intraoperative placement of a nasointestinal ileus tube appears to facilitate recovery and reduce the risk of recurrent obstruction, though larger studies are needed to confirm this benefit. Our patient's excellent 3-month outcome supports this multimodal surgical strategy for managing this rare but challenging condition.
Guidelines
The reporting of this case conforms to the CARE guidelines. 25
Footnotes
Acknowledgements
The authors thank Dr Changqing Xu, Director of Radiology at Shidong Hospital Shanghai, for his guidance on image interpretation, and appreciate the assistance of all supporting staff and institutions. ChatGPT was used for language polishing and the content was reviewed and verified.
Ethical considerations
The study was approved by the Ethics Review Committee of Shanghai Siylyl Hospital (approval number: SYYEC2025-002) and was conducted in accordance with the principles of the current Helsinki Declaration of 1975 and its 2024 revisions, as well as local, regional, and international regulations pertaining to clinical research.
Author contributions
LZ was involved in conceptualization and methodology; FZ in data collection; WL in literature review; AB in data analysis; GW in surgical intervention; YZ in patient management; CD in data interpretation; FG in pathological analysis; JH in supervision; BG in resources and project support; LZ in writing—original draft; and LZ, FZ, WL, AB, GW, YZ, CD, FG, JH, and BG in writing—review and editing. All authors have read and approved the final manuscript and accept full responsibility for its content.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Military Key Discipline Construction Projects of China, (grant number HL21JD1206).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data supporting this study are contained in the article; additional de-identified files are available on request.
