Abstract
Objectives
This systematic review and meta-analysis aims to evaluate whether alendronate therapy improves survival rates in patients with osteoporosis, in addition to its known benefits in reducing fracture risk.
Methods
A comprehensive literature search was conducted across Embase, Web of Science, Medline, Cochrane Library, and ClinicalTrials.gov to identify randomized, placebo-controlled clinical trials involving alendronate therapy in osteoporosis patients. The primary outcome was overall survival. Data were extracted and analyzed using fixed-effects or random-effects models based on heterogeneity. The quality of evidence was assessed using the GRADE approach, and publication bias was evaluated using funnel plots.
Results
A total of 13 randomized controlled trials, involving 15,560 participants, were included in the meta-analysis. Alendronate treatment did not significantly improve survival rates in osteoporosis patients (RR, 1.00; 95% CI, 1.00–1.01). Subgroup analyses, including studies focusing on postmenopausal women and trials lasting three years or more, also showed no significant association between alendronate treatment and survival rates. The studies included in this analysis exhibited low heterogeneity, and no significant publication bias was detected.
Conclusions
The findings of this meta-analysis suggest that alendronate therapy does not enhance survival rates in osteoporosis patients, despite its effectiveness in reducing fracture risk. Therefore, the primary focus of osteoporosis treatment should remain on fracture prevention.
PROSPERO registration number: CRD420251034567.
Introduction
Osteoporosis impacts nearly 200 million women globally, with roughly one-third of women and one-fifth of men over 50 being affected. The frequency of osteoporosis-related fractures is expected to increase notably, especially in developing nations where aging populations coincide with lifestyle shifts.1,2 Additionally, fractures caused by osteoporosis are linked to a 20% decrease in survival rates. 2
The 30-day mortality rate following fractures is generally reported to range from 5% to 8%, with the 1-year mortality rate for hip fractures reaching up to 22%. 3 Age significantly influences the 30-day post-fracture mortality rate, being less than 1% for patients under 65, but increasing to nearly 7% for those aged 80 and above. 4 Additionally, research shows that mortality rates remain high for several years after a fracture. For example, the 5-year absolute mortality rate is particularly notable for hip and clinical vertebral fractures, with age-adjusted standardized mortality ratios (SMRs) reaching 6.68 for women with hip fractures and 8.64 for vertebral fractures. 5
Alendronate is a commonly prescribed bisphosphonate that effectively lowers the risk of fractures in individuals with osteoporosis. It functions by inhibiting osteoclast activity, which strengthens bones and decreases bone turnover. 6 Clinical trials consistently demonstrate that alendronate significantly reduces the occurrence of both vertebral and non-vertebral fractures in postmenopausal women with osteoporosis.6,7 For example, one study found a 61% decrease in symptomatic vertebral fractures in patients treated with alendronate compared to those receiving a placebo. 6 However, it remains debated whether alendronate improves overall survival rates in osteoporosis patients. Some observational studies have hinted at a possible association between bisphosphonate use and lower mortality rates—suggesting reductions of 25%–60%—but these results are affected by confounding variables such as the patients’ pre-existing health conditions and the types of fractures they have experienced.8,9
Currently, there are no meta-analyses explicitly defining the relationship between alendronate treatment and survival rates. If alendronate could improve survival rates in osteoporosis patients, it might not only reduce fracture risk but also warrant further clinical application. Therefore, we conducted a meta-analysis of randomized, placebo-controlled clinical trials involving alendronate therapy to evaluate its impact on survival rates.
Methods
The study has been conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 10 and AMSTAR (Assessing the Methodological Quality of Systematic Reviews) guidelines. 11 This study was registered post-study at PROSPERO (https://www.crd.york.ac.uk/prospero/#guidancenotes): registration number CRD420251034567.
Data sources and searches
We conducted a comprehensive literature search across Embase, Web of Science, MEDLINE (via PubMed), Cochrane Library, and ClinicalTrials.gov, covering publications up to August 8, 2024. The specifics of our search criteria are provided in Supplementary Table 1. Additionally, we discovered further references by thoroughly examining the bibliographies of relevant studies and reviews included in our analysis.
Selection of studies
The eligibility criteria for this study included: (1) randomized, placebo-controlled clinical trials; (2) participants diagnosed with osteoporosis based on bone mineral density criteria without requiring the presence of fragility fractures; (3) use of alendronate in the experimental group; (4) clear reporting of mortality data at the end of the trial; and (5) a minimum trial duration of six months. The exclusion criteria were: (1) study designs such as review articles, case series, case reports, letters, conference abstracts, or reviews; (2) data that could not be extracted; (3) duplicate publications that only reported the final, comprehensive trial results; and (4) inappropriate selection or lack of a control group. For clinical trials that investigated multiple drug treatments, each treatment arm was individually compared to the placebo group. The search was limited to articles published in English.
After a thorough literature search, one researcher (SR) independently reviewed the title and abstract of each record, with another researcher (JQH) providing validation. To ensure maximum data collection, articles were excluded during the title and abstract screening phase only if they clearly met the exclusion criteria. The full text of the remaining records was then carefully examined, and articles that met the inclusion criteria were included in the analysis. Any disagreements between the two researchers were resolved through discussion or by consulting a third researcher (XD).
Data extraction
Data were extracted into a standardized spreadsheet developed using Microsoft Excel (version 2019, Microsoft Corporation, Redmond, WA, USA). The data extraction process encompassed the following key components: (1) fundamental study information, including the title, publication year, and corresponding author; (2) demographic characteristics, such as age and nationality; and (3) detailed trial-specific parameters, including alendronate dosage, trial duration, and mortality outcomes, among other relevant variables. The extraction procedure was conducted independently by one reviewer (SR) and subsequently validated by a second (LZB). Any discrepancies or uncertainties were resolved through consensus discussion or by consulting a third investigator (XD).
Evaluation of article quality
The assessment of randomized trials for potential bias was carried out using the revised Cochrane Risk of Bias tool. 12 This evaluation included aspects such as sequence generation, allocation concealment, blinding of participants, blinding of outcome assessors, handling of incomplete outcome data, and reporting bias. Two researchers (SR and JQH) independently rated each aspect as having either a low risk, high risk, or unclear risk of bias. Any disagreements in ratings were resolved through discussion or by consulting a third investigator (XD).
To evaluate the quality of evidence for all outcomes, two researchers (LZB and WG) used the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) method (GRADE Pro, version 3.6). This assessment considered five factors—risk of bias, inconsistency, indirectness, imprecision, and other potential sources of bias—to judge each outcome. The evidence was categorized as high, moderate, low, or very low, depending on the probability that additional research could influence confidence in the effect estimates.
Statistical analysis
Heterogeneity was assessed using the I² statistic, with values of 50% or less indicating low to moderate variability between studies, leading to the use of a fixed-effects model to combine effect measures. For I² values exceeding 50%, signaling significant between-study heterogeneity, a random-effects model was utilized. Additionally, subgroup analyses were performed for the indicators. Publication bias was evaluated through funnel plots, along with Egger's and Begg's statistical tests. Risk ratios (RRs) comparing treatment to placebo were expressed as 95% confidence intervals (CIs). Data analysis was conducted using Review Manager (RevMan) version 5.4, developed by the Nordic Cochrane Center in partnership with the Cochrane Collaboration.
Results
Characteristics of the included studies
Supplementary Figure 1 illustrates the study selection process. We gathered 343 records from various sources including Medline (via PubMed), Web of Science, Embase, Cochrane Library, and ClinicalTrials.gov. After removing duplicate and irrelevant studies, we reviewed 39 published reports to evaluate their suitability for full-text analysis. Ultimately, our meta-analysis included 13 randomized placebo-controlled trials13–25 with a total of 15,560 participants, as detailed in Table 1. Of these participants, 7769 were assigned to the placebo group and 7791 to the treatment group.
Summary of the characteristics of the studies included.

NR: not reported.
The studies were carried out in a diverse range of countries across Asia, Europe, and the Americas, notably including the United States, China, Japan, Hong Kong, and Italy. Most participants in the included studies were over the age of 50, including postmenopausal women and older adults.
Risk of bias
Supplementary Table 2 shows the results of the bias risk assessment for the studies included. Some of the studies14,15,20,24 did not specify the details of random sequence generation, leading to potential selection bias. Additionally, a few studies14,17,18,20 were limited by their open-label design, which prevented the double-blinding of both participants and researchers. Despite these issues, the overall risk of bias is considered very low due to the predominance of large-scale randomized clinical trials.
Outcomes of meta-analysis
The alendronate treatment did not significantly improve survival rates in osteoporosis patients (RR, 1.00; 95% CI, 1.00–1.01; Figure 1).13–25 The results of the subgroup analysis for postmenopausal women (Figure 2)20–25 similarly indicated no significant association between alendronate treatment and survival rates (RR, 1.00; 95% CI, 0.99–1.01). Additionally, clinical trials of alendronate treatment lasting 3 years13,16,21 or more also showed no significant correlation with survival rates (RR, 1.00; 95% CI, 0.99–1.01; Figure 3). All subgroup analyses exhibited low heterogeneity (Table 2).
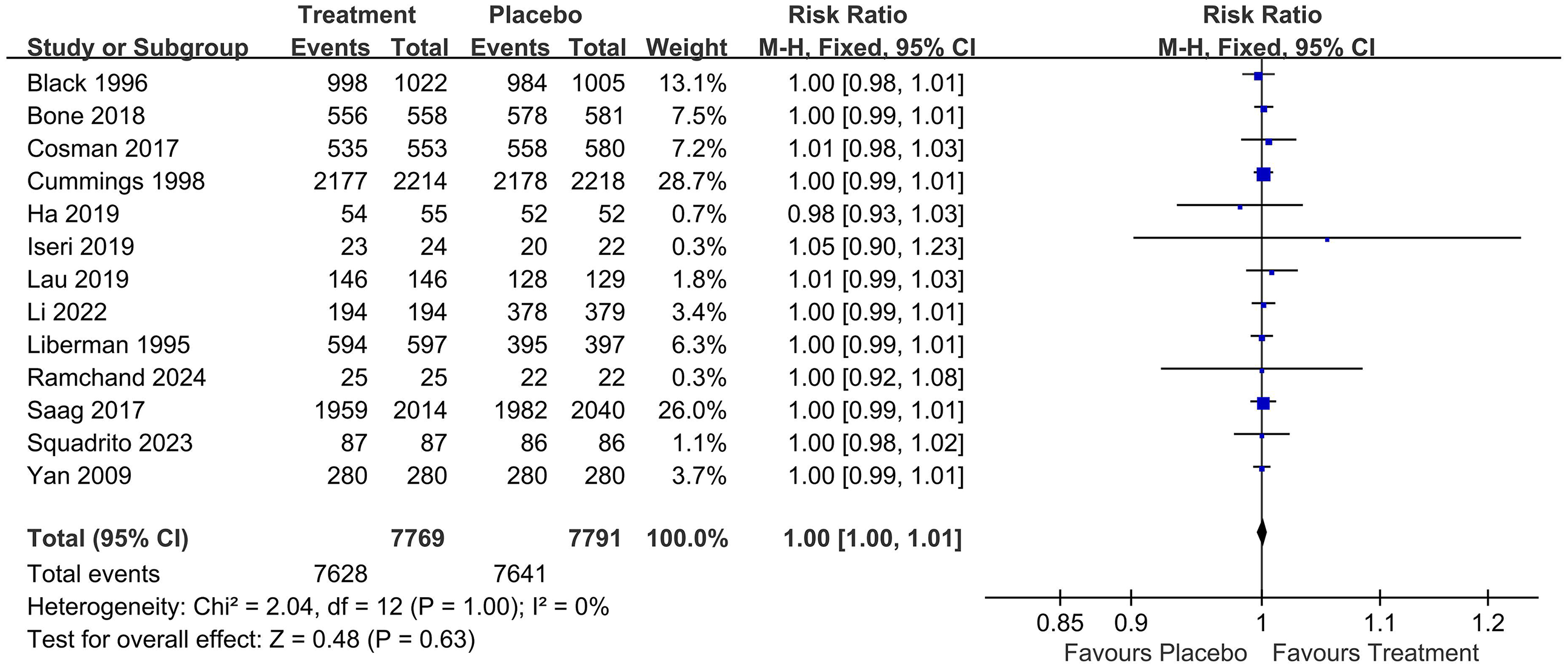
Forest plot for alendronate treatments and overall survival rates.

Forest plot of alendronate treatment and overall survival rates in postmenopausal women.

Forest plot of alendronate treatment with at least 3 years of study for osteoporosis and overall survival rates.
Overview of results and evaluation of evidence quality.
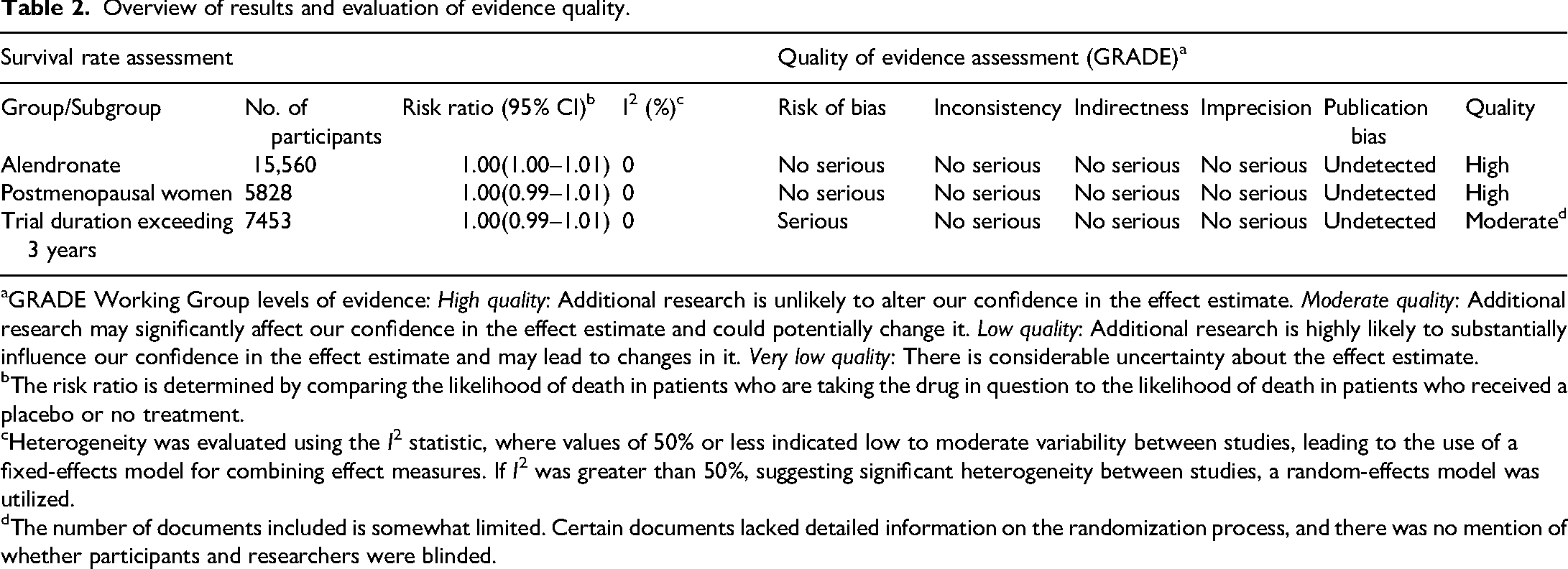
GRADE Working Group levels of evidence: High quality: Additional research is unlikely to alter our confidence in the effect estimate. Moderate quality: Additional research may significantly affect our confidence in the effect estimate and could potentially change it. Low quality: Additional research is highly likely to substantially influence our confidence in the effect estimate and may lead to changes in it. Very low quality: There is considerable uncertainty about the effect estimate.
The risk ratio is determined by comparing the likelihood of death in patients who are taking the drug in question to the likelihood of death in patients who received a placebo or no treatment.
Heterogeneity was evaluated using the I 2 statistic, where values of 50% or less indicated low to moderate variability between studies, leading to the use of a fixed-effects model for combining effect measures. If I2 was greater than 50%, suggesting significant heterogeneity between studies, a random-effects model was utilized.
The number of documents included is somewhat limited. Certain documents lacked detailed information on the randomization process, and there was no mention of whether participants and researchers were blinded.
Funnel plot, evidence quality, and recommendation strengths
Figure 4, which serves as a funnel plot, displays symmetry on both sides, suggesting a high level of homogeneity among the studies and indicating no significant publication bias. Table 2 shows the GRADE evidence ratings, with all studies rated as Moderate to High quality. Given that all studies included in this meta-analysis are RCTs with large sample sizes, and despite some minor issues with blinding procedures, the overall quality of evidence can be regarded as high.

The funnel plot.
Discussion
Statement of principal findings
This meta-analysis indicates that alendronate therapy may not improve survival rates in osteoporosis patients. Both subgroup analyses (postmenopausal women and trials lasting more than 3 years) also support this conclusion. The heterogeneity of this meta-analysis was very low (I² = 0), and no publication bias was detected. The GRADE assessment indicates that the evidence quality of this study is high.
Comparison with previous studies
An observational study showed that patients with osteoporosis who were treated with alendronate had a 25%–60% lower overall mortality rate than those who did not receive the treatment. This significant reduction suggests that the drug may have a direct impact beyond merely preventing fractures. 9 A 5-year prospective study similarly showed a 27% reduction in the risk of death with oral bisphosphonate therapy. 26 Furthermore, another study verified that integrating fracture liaison services with bisphosphonate treatment lowered the mortality risk.27,28
Nonetheless, the previously mentioned studies might have overlooked potential confounding factors that could influence the observed reduction in mortality. The decrease in mortality rates could possibly be explained by the “healthy adherer effect,” a phenomenon recognized in research where individuals who consistently follow placebo treatments in clinical trials tend to show lower mortality rates. 29 For example, a study within the Women's Health Initiative showed that women in the placebo group who adhered to at least 80% of the placebo treatment had a 36% lower overall mortality rate compared to those who adhered less than 80%. Notably, this reduction in mortality remained significant even after adjusting for various potential confounding factors. This suggests that individuals who consistently follow their treatment regimens may naturally have better health than those who do not. This effect is particularly important to consider in observational studies of osteoporosis treatments, as it is estimated that only about half of women prescribed oral osteoporosis medications adhere to the treatment for a year, with even fewer continuing long-term.30,31 Conversely, the decrease in mortality rates observed in the previously mentioned studies might be due to periodic increases in the population size.
Research targeting specific groups, such as postmenopausal women, has demonstrated that bisphosphonates like alendronate can substantially decrease the risk of vertebral and non-vertebral fractures, both of which are linked to higher mortality. Some studies propose that the decrease in fracture risk might indirectly help reduce mortality rates, but this has not been conclusively established.6,32
Alendronate functions by suppressing osteoclast activity, thereby reducing bone resorption. This results in higher bone mineral density (BMD) and a lower risk of fractures. The pharmacological actions of bisphosphonate drugs in treating osteoporosis mainly involve the following aspects: inhibiting bone resorption, enhancing osteoblast activity, boosting the formation and deposition of new bone tissue, which in turn increases bone density, and improving bone quality in osteoporosis patients; 33 and reducing the release of mineralization precursors, thereby slowing the bone tissue mineralization process and further decreasing the risk of osteoporosis. 17 Given that fractures, especially those of the hip and spine, are linked to higher mortality, preventing these fractures may help enhance survival rates.6,9 Certain clinical trials have not monitored patients for a sufficient duration to accurately evaluate long-term mortality outcomes. The impact of bisphosphonates on mortality might take years to become evident, making it challenging to draw conclusions from shorter studies.
It is important to note that all participants in the included studies were diagnosed with osteoporosis based solely on BMD criteria, and none of the trials specifically enrolled patients with prior fragility fractures. This distinction is clinically relevant, as fragility fractures are potentially associated with significantly higher short-term and long-term mortality rates. Consequently, the results of our analysis may not be generalizable to populations with established fragility fractures. Future studies focusing on this high-risk subgroup are warranted to determine whether alendronate may confer additional survival benefits in such populations.
Although alendronate does not confer a survival benefit, it has been shown to positively impact the quality of life in patients with osteoporosis. A randomized study involving 44 postmenopausal Japanese women with osteoporosis demonstrated that alendronate significantly improved pain-related quality of life. 34 Moreover, alendronate treatment resulted in a significant increase in lumbar spine bone mineral density after just four months of therapy. 34 Although our meta-analysis did not demonstrate a survival benefit associated with alendronate, it remains a cornerstone in reducing fracture risk. However, improving patient outcomes in osteoporosis management requires a more comprehensive approach. Fracture Liaison Services (FLS), which provide systematic post-fracture care, have been shown to reduce secondary fracture rates and improve long-term outcomes through coordinated assessment, treatment initiation, and follow-up. Incorporating such multidisciplinary care strategies can complement pharmacologic interventions and help address the broader impact of osteoporotic fractures on morbidity and mortality.
Limitations
Our meta-analysis has several limitations. It's crucial to acknowledge that the connection between alendronate treatment and survival rates might take several years to emerge. Although we did not find a significant association between alendronate treatments lasting three years or more and survival rates, this time frame could still be considered relatively brief. Post-study registration may have introduced bias to our results and is a limitation of this study.
Conclusion
This meta-analysis of randomized placebo-controlled clinical trials suggests that alendronate use in treating osteoporosis does not seem to be associated with an increase in survival rates, even though it clearly reduces fracture risk. As a result, osteoporosis treatments should primarily focus on lowering fracture risk, as recommended by clinical guidelines.
Supplemental Material
sj-docx-2-sci-10.1177_00368504251348587 - Supplemental material for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-sci-10.1177_00368504251348587 for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials by Rui Sun, Zhibin Lan, Gang Wu, Qi Ma, Di Xue, Xue Lin and Qunhua Jin in Science Progress
Supplemental Material
sj-doc-3-sci-10.1177_00368504251348587 - Supplemental material for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials
Supplemental material, sj-doc-3-sci-10.1177_00368504251348587 for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials by Rui Sun, Zhibin Lan, Gang Wu, Qi Ma, Di Xue, Xue Lin and Qunhua Jin in Science Progress
Supplemental Material
sj-doc-4-sci-10.1177_00368504251348587 - Supplemental material for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials
Supplemental material, sj-doc-4-sci-10.1177_00368504251348587 for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials by Rui Sun, Zhibin Lan, Gang Wu, Qi Ma, Di Xue, Xue Lin and Qunhua Jin in Science Progress
Supplemental Material
sj-doc-5-sci-10.1177_00368504251348587 - Supplemental material for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials
Supplemental material, sj-doc-5-sci-10.1177_00368504251348587 for Does alendronate enhance survival rates in osteoporosis patients? A meta-analysis of randomized controlled trials by Rui Sun, Zhibin Lan, Gang Wu, Qi Ma, Di Xue, Xue Lin and Qunhua Jin in Science Progress
Footnotes
Acknowledgements
Thanks to all authors for their contributions to this article.
Author contributions
Rui Sun and Zhibin Lan was involved in the study conception and design; the data acquisition and interpretation; the drafting of the article; and the provision of final approval of the version to be published. Gang Wu, Qi Ma, Di Xue, and Xue Lin were involved in the study conception and design; the data acquisition and interpretation; the drafting of the article; and the provision of final approval of the version to be published. Qunhua Jin was involved in the study conception and design; the acquisition and interpretation of the data; the writing and critical revision of the manuscript for important intellectual content; and the provision of final approval of the version to be published.
Funding
This study was supported by the National Natural Science Foundation of China (No.U22A20285); National Natural Science Foundation of China (No. 82160433); Key R&D Project of Autonomous Region (No. 2023BEG02018); Autonomous Region Major Scientific and Technological; Achievements Transformation Project (No. 2023CJE09037); Ningxia Medical University General Hospital “Medical Engineering Special” (No. NYZYYG-001); Key R&D Project of Autonomous Region (No. 2024AAC03601) and Ningxia Medical University Research Grant Project (XT2023035).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
