Abstract
Objective
There is crosstalk between the thyroid hormone and the renin-angiotensin-aldosterone system, but the clinical application is lacking. Our study aimed to verify the correlation between renin and other thyroid-related biomarkers and their association with treatment prognosis.
Methods
This retrospective study recruited eighty consecutive patients treated for hyperthyroidism in a medical center between 2021 and 2023. Among participants, 53 were newly diagnosed, and 27 were recurrent cases. Laboratory measurements, including renin, aldosterone, and other thyroid-related biomarkers, were assessed, while the associations of biomarkers and clinical symptoms were also analyzed. The clinical variables and biomarkers were entered into the Cox regression model to identify the independent risks associated with time to achieve euthyroidism.
Results
The correlation matrix showed that free T4 was positively correlated either with T3 (r = 0.755, P < 0.001) or renin (r = 0.341, P = 0.002) but not with aldosterone (r = 0.055, P = 0.631). In ROC analysis for biomarkers and clinical symptoms, either free T4 (AUC = 0.754, P < 0.001) or thyroid-stimulating hormone receptor antibody (TSHRAb) (AUC = 0.645, P = 0.026) demonstrated effective prediction of tachycardia; however, renin was slightly positive for tachycardia without significance (AUC = 0.580, P = 0.233). Regarding thyrotoxic periodic paralysis, the renin showed significant prediction power (AUC = 0.727. P = 0.021) rather than free T4 (AUC = 0.543, P = 0.748) or TSHRAb (AUC = 0.688, P = 0.063). The median time to achieve euthyroidism was 166 days for newly onset patients and 216 days for recurrence, but the time-to-event curves showed no difference (log rank P = 0.728). Under Cox regression modeling, TSHRAb was found to independently predict late euthyroidism (Hazard ratio 0.946, 95% confidence interval 0.901–0.993, P = 0.026), and the renin had no impact (HR 0.995, 95% CI 0.987–1.003, P = 0.215).
Conclusions
Renin but not aldosterone was positively correlated with free T4. Certain biomarkers measured at diagnosis were associated with hyperthyroid symptoms. Free T4 and TSHRAb might predict tachycardia, while renin might predict thyrotoxic periodic paralysis; nevertheless, only higher TSHRAb was associated with delayed euthyroid state achievement.
Keywords
Introduction
Hyperthyroidism is a common thyroid disorder with various etiologies, the most prevalent being Graves’ disease, which typically affects individuals aged 30–50 1 with a prevalence rate of approximately 2% in females and 0.5% in males. 2 Autoantibodies are often present and involved in pathogenesis in patients with Graves’ disease, including the thyroid-stimulating hormone receptor antibody (TSHRAb) and anti-thyroid peroxidase antibody (anti-TPO Ab). 3 Beyond common symptoms like anxiety, hand tremors, tachycardia, and diarrhea, the cardiovascular impact of hyperthyroidism warrants particular attention, as it can be life-threatening. In hyperthyroidism, elevated thyroid hormones bind to receptors on cardiac myocytes, leading to increased cardiac contractility and cardiac output4–7; additionally, thyroid hormones can stimulate the sympathetic nervous system. Previous studies have shown an increase in the number of β-adrenergic receptors in the renal cortex in hyperthyroidism.8,9 These often result in an elevated heart rate and increased cardiac output; in cases of poorly controlled hyperthyroidism, it can progress to arrhythmias, congestive heart failure, or even death. 10
The renin-angiotensin-aldosterone system (RAAS) is also essential for regulating cardiac and vascular physiology and is involved in numerous pathological conditions, such as hypertension, heart failure, and renal disease. 11 The cascade of RAAS primarily functions through renin, angiotensin II, and aldosterone to regulate blood volume, electrolyte balance, systemic vascular resistance, and blood pressure. 12 When the RAAS is activated persistently, the heart, kidneys, and other organs will be affected by inappropriate expansion of the intravascular and extravascular volumes, ultimately contributing to the progression of heart failure. 13
Moreover, researchers have explored the interaction between thyroid hormones and the RAAS. 14 Several studies have reported that thyroid hormones might modulate the RAAS; however, most evidence has been derived from animal studies15–19; furthermore, due to the limited number of studies, the clinical manifestations and potential applications of thyroid hormone's influence on the RAAS remain unclear.
Accordingly, our study aimed to verify the association between RAAS parameters, with a particular focus on renin and thyroid-related markers to explore potential clinical applications, while a further aim intended to elucidate whether the initial biomarkers measured at the diagnosis of hyperthyroidism were associated with further treatment outcomes.
Materials and methods
Participants
This retrospective study included a cohort of 116 consecutive patients treated for hyperthyroidism in the Endocrinology Outpatient Department at Chang Gung Memorial Hospital between January 2021 and December 2023. Inclusion criteria comprised patients aged over 18 years, presenting with either newly diagnosed or recurrent hyperthyroidism, with blood samples collected within one month of disease onset. Patients with incomplete data were excluded from the study. Ultimately, 80 patients met the study criteria, of whom 53 were newly diagnosed cases and 27 were recurrent cases. All participants received regular follow-up during the enrolled time and were managed with antithyroid drugs (ATDs). The study endpoint was defined as achieving a euthyroid state. None of the patients were pregnant, experienced thyroid storm, or had known adrenal disease. The Institutional Review Board of Chang Gung Memorial Hospital approved this retrospective study (No. 202500047B0), while the reporting of this study conformed to STROBE guidelines. 20 All data were de-identified, and no personally identifiable information was reported. We conducted our study in accordance with the Helsinki Declaration of 1975 as revised in 2024.
Clinical characteristics and data
Clinical characteristics recorded for each patient included age, gender, body mass index (BMI), smoking status, and concurrent conditions including chronic kidney disease (CKD), cerebrovascular accident (CVA), heart failure (HF), or atrial fibrillation (Af). Concurrent or subsequent symptoms were documented, including goiter, hypertension (HTN), tachycardia, and thyrotoxic periodic paralysis (TPP). Additionally, the types of antithyroid drugs (ATDs) administered during the treatment period (propylthiouracil (PTU), methimazole (MMI), carbimazole (CBZ)) and the use of β-blockers were recorded.
Laboratory measurements were collected within one month of disease onset, including thyroid function tests (TSH, free T4, T3), thyroid autoantibodies (antithyroid peroxidase antibody (anti-TPO Ab) and thyroid-stimulating hormone receptor antibody (TSHRAb). RAAS-related measurements included plasma renin concentration and plasma aldosterone concentration with additional laboratory parameters including white blood cell count (WBC), hemoglobin (Hb), platelet count (PLT), alanine aminotransferase (ALT), potassium (K), and total bilirubin (Bil-T) also being measured.
Statistical analysis
Comparisons between first-diagnosed and recurrent hyperthyroid patients were conducted using Pearson's chi-square test for categorical variables and the Mann–Whiney U-test for continuous variables, where appropriate. The predictive power of thyroid-related biomarkers for hyperthyroid symptoms was assessed using ROC curve analysis, with area under the curve (AUC) values and statistical significance determined, while Kaplan–Meier curves were used to visualize the time to euthyroid state. The ability of each variable to predict euthyroid (hazard ratio) was calculated using a multivariate Cox regression model with adjustments for covariates to identify independent risk factors. The hazard ratios and 95% confidence interval of variables correlated with time to euthyroid are presented by forest plot. All statistical analyses were performed using the Statistical Package for the Social Sciences (IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp.) software.
Results
Characteristics of patients with hyperthyroidism
Among the 80 consecutive patients with hyperthyroidism included in the study, 53 were in the new onset group and 27 in the recurrence group. Patient demographic data is presented in Table 1. The median age was 38.84 years in the new onset group and 42.61 years in the recurrence group (P = 0.094) with females constituting the majority in both groups, accounting for 62.26% and 74.07% respectively (P = 0.291). There were no significant differences in concurrent diseases and subsequent symptoms between the two groups, and none of the patients had CKD. For HF, the new onset and recurrence groups included n = 2 (3.77%) and n = 1 (3.70%) patients, respectively (P = 1.000). Af or pAf was present in n = 4 (7.55%) in the new onset group and n = 4 (14.81%) in the recurrence group (P = 0.432); CVA occurred in n = 2 (3.77%) in the new onset group and none in the recurrence group (P = 0.547); while the new onset group had a higher proportion of patients with HTN (35.85% vs 18.52%, P = 0.110) and tachycardia (47.17% vs 33.33%, P = 0.236) compared to the recurrence group, although neither reached statistical significance. Additionally, there were n = 3 (5.66%) patients with thyrotoxic periodic paralysis (TPP) in the new onset group and n = 2 (7.41%) in the recurrence group (P = 1.000). There were no differences between the two groups in the types of ATDs used (P = 0.367) or in β-blocker usage (P = 0.379).
Clinical characteristics and anti-thyroid medications of participants.
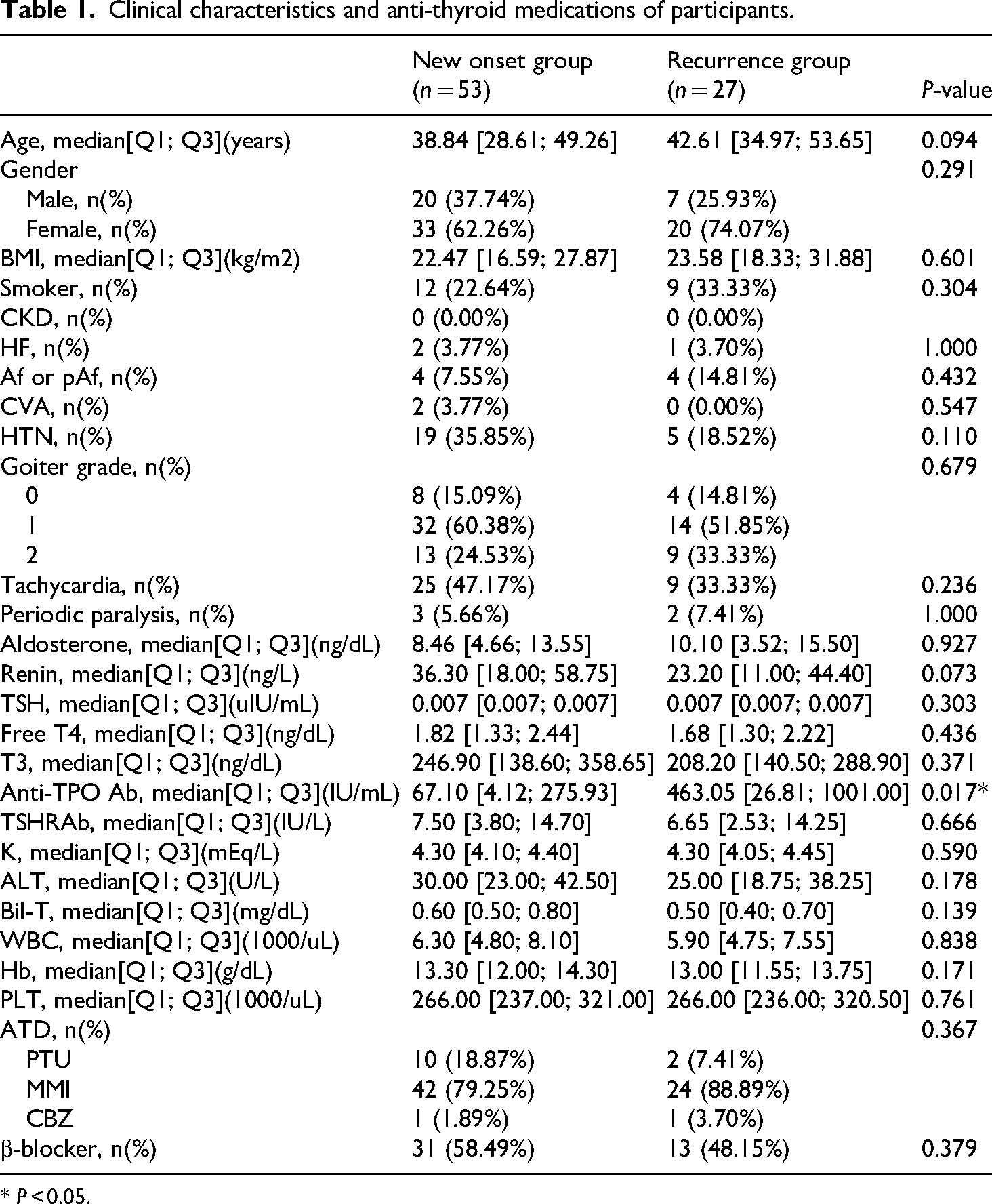
* P < 0.05.
Biochemistry
The biochemical profiles of the new onset and recurrence groups are presented in Table 1. Thyroid hormones showed no significant differences between the two groups, while both free T4 and T3 were slightly higher in the new onset group. Regarding thyroid autoantibodies, the median level of anti-TPO Ab was significantly higher in the recurrence group (67.10 IU/mL vs 463.05 IU/mL, P = 0.017), while the median TSHRAb level was slightly higher in the new onset group (7.50 IU/L vs 6.65 IU/L, P = 0.666). RAAS-related biomarkers, renin, and aldosterone, did not differ significantly between the groups, although renin levels were slightly higher in the new onset group compared to the recurrence group (36.30 ng/L vs 23.20 ng/L, P = 0.073). Other blood biochemical parameters, including WBC, Hb, PLT, K, ALT, and Bil-T showed no differences between the groups.
Association and predictive value among renin, thyroid biomarkers, and clinical outcomes
The correlations between various biochemical markers are presented in Figure 1. As expected, there was a significant positive correlation between the thyroid hormone free T4 and T3 (r = 0.755, P < 0.001), as well as between the RAAS biomarker renin and aldosterone (r = 0.446, P < 0.001). Notably, a statistically significant positive correlation was also observed between free T4 and renin (r = 0.341, P = 0.002), but not between free T4 and aldosterone (r = 0.055, P = 0.631).

Correlation matrix between biochemical markers measured at the time of hyperthyroidism diagnosis. There were significant positive correlations between free T4 and T3 (r = 0.755, P < 0.001) and between renin and aldosterone (r = 0.446, P < 0.001). Free T4 also correlated positively with renin (r = 0.341, P = 0.002) but not aldosterone (r = 0.055, P = 0.631).
Figure 2 presents the ROC analysis for biomarkers and clinical symptoms. Free T4 (AUC = 0.754, P < 0.001) and TSHRAb (AUC = 0.645, P = 0.026) demonstrated effective predictive ability for tachycardia, and while renin showed a slightly positive predictive value for tachycardia, it did not reach statistical significance (AUC = 0.58, P = 0.233). Additionally, renin exhibited significant predictive power for TPP (AUC = 0.727, P = 0.021), while no predictive capability for TPP was observed in free T4 (AUC = 0.543, P = 0.748) or TSHRAb (AUC = 0.688, P = 0.063). Regarding hypertension, no predictive capability was found in free T4, renin, or TSHRAb.

The ROC analyses for biomarkers to predict clinical manifestations. Free T4 (AUC = 0.754, P < 0.001) and TSHRAb (AUC = 0.645, P = 0.026) effectively predicted tachycardia, while renin showed a weak and non-significant predictive value (AUC = 0.58, P = 0.233). Renin significantly predicted TPP (AUC = 0.727, P = 0.021), whereas free T4 (AUC = 0.543, P = 0.748) and TSHRAb (AUC = 0.688, P = 0.063) did not. None of the biomarkers demonstrated predictive value for hypertension.
Factors and impacts on time to euthyroid
Figure 3 displays the Kaplan–Meier plot showing the cumulative probability curves of the euthyroid state, with a median time to achieve euthyroidism of 166 days for new-onset patients and 216 days for recurrent cases, and no significant difference was noted (log-rank P = 0.728). Figure 4 shows that Cox regression modeling identified TSHRAb as an independent predictor of late euthyroidism (Hazard ratio (HR): 0.946, 95% Confidence Interval (CI): 0.901–0.993, P = 0.026), and age was associated with early euthyroidism (HR: 1.033, 95% CI: 1.007–1.061, P = 0.014). However, renin (HR: 0.995, 95% CI: 0.987–1.003, P = 0.215), free T4 (HR: 1.168, 95% CI: 0.694–1.968, P = 0.559), anti-TPO Ab (HR: 1.000, 95% CI: 0.999–1.001, P = 0.703) and gender (HR: 1.649, 95% CI: 0.480–5.672, P = 0.427) showed no impact on time to achieve euthyroidism.

Kaplan–Meier plots for the cumulative probability curves of the euthyroid state. The median time to achieve euthyroidism was 166 days for new-onset patients and 216 days for recurrent cases, with no significant difference observed (log-rank P = 0.728).
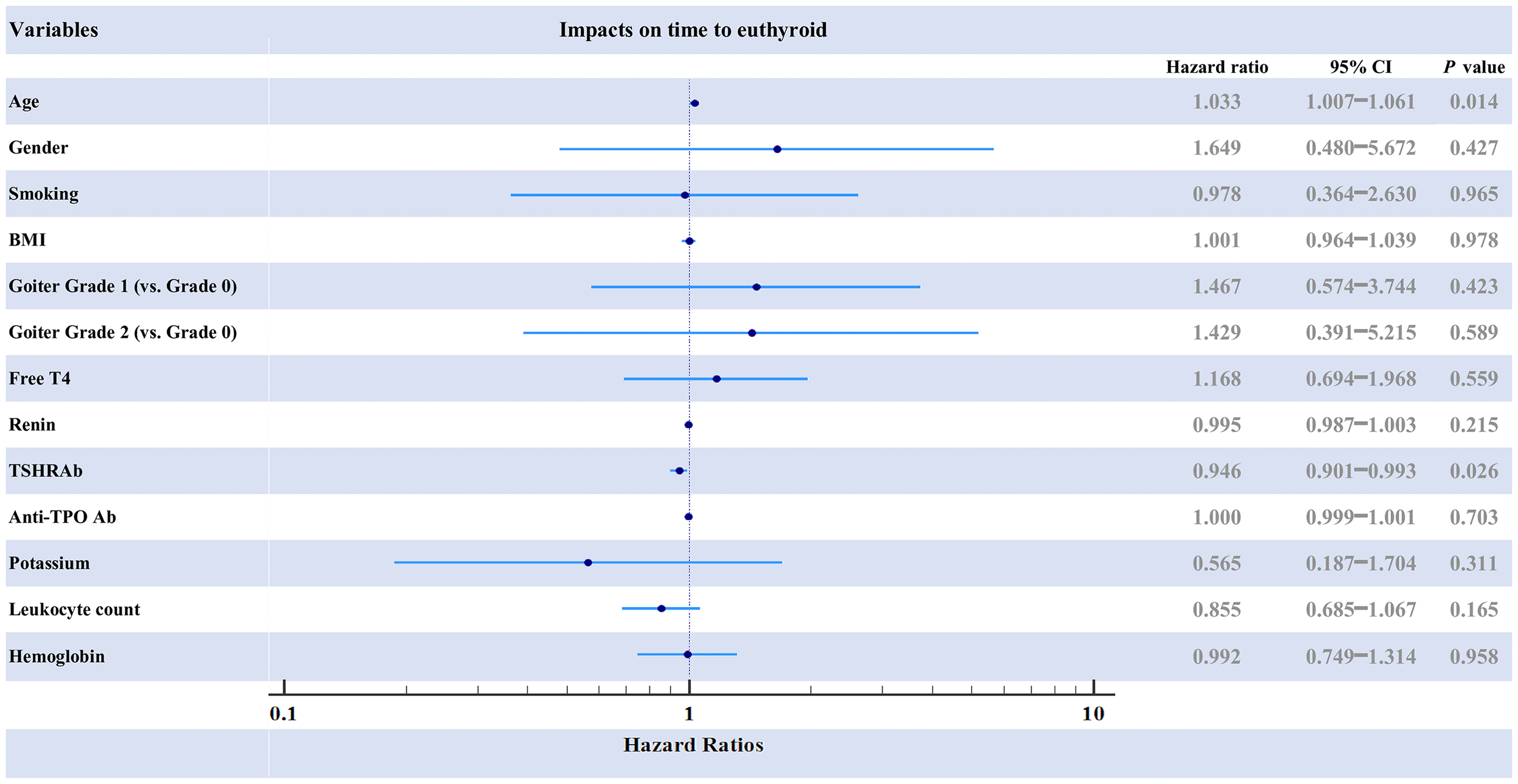
Forest plot of hazard ratios for clinical variables and biomarkers associated with time to achieve euthyroidism, analyzed using the Cox regression model. TSHRAb was an independent predictor of delayed euthyroidism (HR: 0.946, 95% CI: 0.901–0.993, P = 0.026), while age was associated with earlier euthyroidism (HR: 1.033, 95% CI: 1.007–1.061, P = 0.014). In contrast, renin (HR: 0.995, 95% CI: 0.987–1.003, P = 0.215), free T4 (HR: 1.168, 95% CI: 0.694–1.968, P = 0.559), and anti-TPO Ab (HR: 1.000, 95% CI: 0.999–1.001, P = 0.703) had no significant impact on the time to achieve euthyroidism.
Discussion
Our study demonstrated a positive correlation between plasma renin concentration and free T4, consistent with some prior investigations in vitro or in vivo. Marchant et al. found that hyperthyroidism activated the plasma RAAS, increasing plasma renin activity (PRA) by 47% and concentration by 52% after 7 days of thyroid hormone supplement in rats. 17 Another animal research study revealed that PRA and renal expression of renin mRNA were elevated by 152 to 155% in hyperthyroid rats. 18 In human studies, one investigation revealed that individuals with Graves’ disease exhibited significantly elevated PRA compared to control subjects; conversely, following thyrostatic treatment, the PRA levels in these patients were comparable to those of the control group. 21 These studies consistently support our findings on the correlation between renin and thyroid hormones in patients with hyperthyroidism.
The previous literature has proposed several mechanisms to explain how thyroid hormone increases renin levels and activates the RAAS in hyperthyroidism. Since most renin in the blood is synthesized and secreted by the kidneys, thyroid hormone appears to stimulate renal renin synthesis by enhancing renin mRNA expression with a subsequent increase of renal renin levels and plasma angiotensin II levels. Through experiments in vivo and in vitro, Kobori and Ichihara et al. demonstrated that thyroid hormone directly stimulates the transcription of renin mRNA without involving the sympathetic nervous system; furthermore, thyroid hormone enhances renin secretion from juxtaglomerular cells by activating a calcium-dependent signaling mechanism.18,22 They also indicated that thyroid hormones stimulate the renin gene by binding to specific thyroid hormone receptors, which then interact with the thyroid hormone response element (THRE) in the promoter region of the renin gene. This interaction enhances the transcriptional activity of the renin gene, resulting in greater production of renin mRNA and, ultimately, increased renin synthesis. 23 In addition to directly enhancing renin mRNA, other indirect mechanisms including increased sympathetic stimulation and renal perfusion changes might contribute to the elevated renin level. Since renin levels can be elevated through β-adrenergic stimulation, 24 and Haro et al. demonstrated an increase in β-adrenergic receptor density in the renal cortex during experimental hyperthyroidism, 8 it is suggested that thyroid hormones could indirectly promote renin release by enhancing the body's sensitivity to catecholamines through interactions with the sympathetic nervous system, particularly in hyperthyroidism.7,9 The theory of renal perfusion change suggests that thyroid hormones, by reducing systemic vascular resistance through smooth muscle relaxation or endothelial nitric oxide production, decrease renal perfusion. This reduction triggers juxtaglomerular cells to secrete more renin, leading to the activation of the renin-angiotensin-aldosterone system (RAAS).25–27 However, it is important to note certain pharmacological factors in our patient population that could have influenced the observed correlations. Most patients in our study were undergoing ATD therapy, which directly reduces thyroid hormone production. Approximately half the patients were also treated with β-blockers, which were supposed to suppress renin secretion. 28 As a result, the true physiological correlation between thyroid hormone levels and RAAS activation may have been underestimated in our study population.
The positive correlation between thyroid hormones and the RAAS suggests that monitoring renin levels in hyperthyroid patients might aid in managing associated cardiovascular risks. Thyroid hormone-stimulated renin elevation could serve as a potential marker for the severity of hyperthyroid-induced cardiovascular alterations, providing valuable insights for targeted therapeutic strategies. We further investigated the correlation of these biomarkers with clinical symptoms of hyperthyroidism. The analysis using ROC curves revealed a positive predictive relationship between free T4 and TSHRAb levels for tachycardia.
Thyroid hormones increase the sensitivity of the heart to catecholamines, which are stimulants of heart rate. They achieve this by upregulating β-adrenergic receptors in the heart, making cardiac cells more responsive to sympathetic nervous stimulation, which results in a faster heart rate.29,30 Although evidence on the direct effects of TSHRAb on the heart is limited, 31 the positive correlation between TSHRAb and tachycardia in hyperthyroid patients is unsurprising, as these autoantibodies are the primary cause of hyperthyroidism. 32 By contrast, renin has some predictive power in the symptom of tachycardia but is less effective than Free T4 and lacks statistical significance. Interestingly, though not strongly associated with tachycardia, our result showed that renin exhibited significant predictive power for thyrotoxic periodic paralysis (TPP). TPP is a rare condition observed in hyperthyroid patients, characterized by sudden episodes of muscle weakness, primarily in the lower limbs, caused by hypokalemia. The primary mechanism involves hyperthyroidism enhancing the activity of Na+/K + ATPase pumps in cell membranes, which leads to a substantial intracellular shift of potassium, resulting in reduced serum potassium levels. 33 Additionally, RAAS activation, particularly through aldosterone, promotes potassium excretion via the kidneys. Together, these effects may further decrease serum potassium levels, heightening the risk of TPP in hyperthyroid patients, although currently, very few studies have addressed this relationship.34,35 Furthermore, activation of tissue renin-angiotensin system (RAS) probably contributes to the pathogenesis of hypokalemia in hyperthyroid patients. In addition to the circulating RAS, the emerging concept of tissue RAS has gained clinical significance, highlighting its expression across various tissues, where changes in its activity have been linked to numerous diseases.36,37 Hyperthyroidism has been documented to promote an increase in cardiac renin levels and stimulate the generation of angiotensin II 19 ; it also raises the levels of angiotensin type 1 (AT1) and type 2 (AT2) receptors in the heart,17,38 contributing to structural and functional alterations. 14 Researchers have also explored how thyroid hormones influence the vascular RAS by modulating its components and affecting vascular tone.14,39 Nevertheless, the precision relationship between tissue RAS and TTP still requires further investigation for validation.
Regarding treatment outcomes and associated risk factors, we observed that the time to euthyroid was not significantly different between patients with new-onset and recurrent disease. The initial level of TSHRAb was the only factor influencing the time to reach a euthyroid state, with higher values predicting delayed remission. The finding suggested high TSHRAb levels are associated with a more difficult course to sustained euthyroid in hyperthyroid patients, consistent with prior investigations disclosing that high TSHRAb levels are predictive of disease relapse after therapy.40–42 In contrast, although associated with initial clinical presentation, neither free T4 nor renin levels were associated with the time to achieve euthyroidism. Since plasma renin levels would fluctuate due to factors like dietary sodium loading, posture, activity, and sex steroids, clinical use of renin measurement for predicting euthyroidism will be difficult.
The present study had limitations because of its retrospective design, which inherently relied on existing data and might therefore introduce biases. In our study, no comparison of continuous change of biomarkers was made post-treatment, requiring further prospective studies for verification. Moreover, only a limited number of patients have TPP; therefore, the predictive power of renin might be overestimated. Another limitation of our study is related to the measurement of renin levels according to plasma renin concentrations rather than plasma renin activity; thus, caution should be exercised when interpreting or applying these results. The absence of plasma angiotensin II data precluded a more comprehensive analysis of the RAAS. Notwithstanding, we still consider accession to and analysis of these biomarkers in hyperthyroid patients to be valuable for clinical application and research inspiration.
Conclusions
In conclusion, our study demonstrates a positive correlation between renin and free T4, with potential clinical relevance for symptom prediction in hyperthyroidism including the association of free T4 with tachycardia and renin with TPP. A significant association was found between TSHRAb and time to achieve euthyroidism, whereas renin and free T4 showed no such relationship. These findings suggest that measuring plasma renin level, in addition to standard thyroid biomarkers, may provide complementary insight into disease severity and symptom risk stratification, though it does not appear to be associated with disease progression. We hope this study serves as a reference for the potential clinical application of thyroid-related biomarkers in guiding personalized therapeutic strategies for hyperthyroid patients.
Footnotes
List of abbreviations
Af: atrial fibrillation; ALT: alanine aminotransferase; Anti-TPO Ab: anti-thyroid peroxidase antibody; AT1: angiotensin type 1; AT2: angiotensin type 2; ATDs: antithyroid drugs; ATPase: adenosine triphosphatases; AUC: area under curve; Bil-T: total bilirubin; BMI: body mass index; CBZ: carbimazole; CI: confidence interval; CKD: chronic kidney disease; CVA: cerebrovascular accident; Hb: hemoglobin; HF: heart failure; HR: hazard ratio; HTN: hypertension; K: potassium; MMI: methimazole; mRNA: messenger ribonucleic acid; PRA: plasma renin activity; PLT: platelet; PTU: propylthiouracil; RAAS: renin-angiotensin-aldosterone system; RAS: renin-angiotensin system; ROC: receiver operating characteristic; STROBE: strengthening the reporting of observational studies in epidemiology; THRE: thyroid hormone response element; TPP: thyrotoxic periodic paralysis; TSH: thyroid-stimulating hormone; TSHRAb: thyroid-stimulating hormone receptor antibody; WBC: white blood cell; pAf: paroxysmal atrial fibrillation.
Acknowledgements
The authors thank the staff and participants of these studies for their important contributions.
Ethical considerations
The studies involving human participants were reviewed and approved by the Institutional Review Board of Chang Gung Memorial Hospital, Taiwan (No. 202500047B0). The IRB allowed for the exemption of the informed consent of subjects since the study was retrospectively analyzed on anonymized data (National Department of Health Medical Affairs No.1010265083).
Author contributions
C.-W.L. derived the concept. H.-Y.W. did the data curation. I.-W.C. did the formal analysis. H.-Y.W. wrote the original draft. All authors assisted with reviewing the manuscript, as well as approving the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and analyzed during the current study are not publicly available due to IRB regulation but are available from the corresponding author upon reasonable request.
