Abstract
This study investigated the impact of sulfate-resistant mineral admixture (SP2) and untreated sugarcane bagasse ash (SCBA) as a partial substitute for cement on the physico-mechanical properties and microstructure of Portland cement concrete. Four concrete mixtures were prepared for the investigation including a control mixture, SP2 mixture (4.7% SP2), SCBA mixture (5% SCBA), and SP2-SCBA mixture (4.7% SP2 and 5% SCBA). The SP2 mixture exhibited superior performance across all assessed engineering properties in comparison to the other mixtures. Moreover, a comparative analysis was conducted to evaluate the performance of untreated SCBA used in this study versus processed SCBA from recent research. Remarkably, untreated SCBA, despite having minimal impact on compressive strength, it demonstrated notable improvements in tensile strength. When combined with 4.7% SP2, the untreated SCBA mixture achieved a remarkable 37.9% increase in splitting tensile strength compared to the control mixture, outperforming similar mixtures using processed SCBA and silica fume in previous studies. Microstructure analysis revealed a large amount of spongy, fibrous hydrated structures in both the SCBA and SP2-SCBA mixtures. These structures provide a rational explanation to the increase in splitting tensile strength of the concrete containing SCBA. The findings highlight the tradeoff between using untreated SCBA and treated SCBA as sustainable cement replacements. Although untreated SCBA can only be used at a low replacement level, it offers unique engineering benefits alongside significant energy savings.
Keywords
Introduction
Sulfate attack in concrete is a complex chemical phenomenon characterized by the reaction of soluble sulfate ions with various compounds present in the hydrated cement matrix. This complex chemical reaction creates new compounds, causing the concrete to expand and eventually deteriorate. In specific, the presence of sulfate in seawater can induce the formation of gypsum (CaSO4·2H2O) through a reaction with Portlandite (Ca(OH)2) or result in the generation of ettringite (3CaO·Al2O3·3CaSO4·32H2O) by reacting with aluminate phases (C3A).1–4 Knowing that Ca(OH)2 and aluminate phases are the sources of the problem, longstanding solutions have been devised to mitigate sulfate attacks in concrete. Cement characterized by enhanced sulfate resistance has traditionally been produced in two approaches: by reducing the C3A content within the cement composition, or by substituting a high proportion of conventional Portland cement with an appropriate granulated blast furnace slag or other supplementary cementitious materials (SCMs).5,6 While low C3A cement mentioned in the former approach is rarely seen in the contemporary market, the latter approach proves to be more practical and feasible for use in construction. As previously mentioned, the latter approach is more widely adopted, and for a long history, slag and fly ash are among the most commonly used SCMs, either individually or in combination, for making concrete. The combined use of slag and fly ash offers significant advantages over using either material alone, particularly in terms of drying shrinkage, chloride resistance, and mechanical properties. 7 This phenomenon is often referred to as the synergistic effect, which has been demonstrated in previous studies on both cement concrete and geopolymer concrete. In such systems, slag contributes to early-age strength due to its latent hydraulic properties, while fly ash enhances long-term durability through pozzolanic reactions. Ultimately, sulfate resistance is significantly improved when geopolymer or cementitious systems with lower calcium content are used.
Presently, there are numerous commercial products for sulfate-resistant mineral additives, offering more options for making sulfate-resistant concrete instead of relying solely on fly ash or granulated blast furnace slag. These sulfate-resistant mineral admixtures are typically produced from silica fume, granulated blast furnace slag, or fly ash through an effective grinding technology. This makes them have ultra-fine particles, high amorphous silica oxide content, and very high reactivity at an early age. The mechanism of sulfate-resistant mineral admixtures in enhancing the sulfate-resistant capacity of concrete is indeed quite similar to that of silica fume or granulated blast furnace slag, as both contribute to improving the impermeability of the concrete surface. However, the dosage of sulfate-resistant mineral admixtures in concrete mixture proportioning is significantly smaller compared to the substantial amount of granulated blast furnace slag needed to achieve a similar effect. Figure 1(a) and (b) presents the wave-dissipating concrete blocks that have been used in the Tuy Hoa beach, situated within Phu Yen Province, a central region of Vietnam. These blocks are fabricated using a 4.7% sulfate-resistant mineral admixture, named SP2, from the Lotus Chemical Joint Stock Company, to replace cement. Remarkably, these concrete blocks have withstood the harsh marine environment for over four years, maintaining commendable surface conditions as shown in Figure 1(a). Consequently, the formulation of this mixture continues to be relied upon for the fabrication of concrete blocks intended for marine applications in that specific location, as depicted in Figure 1(b). To thoroughly understand how the SP2 admixture affects concrete, we cast concrete specimens in a controlled lab setting using the same mixture proportion used to make the wave-dissipating concrete blocks. This allowed us to closely examine the physical, mechanical, and microstructure of these concrete materials.

Wave-dissipating concrete blocks located in Phu Yen Province: (a) after over 4 years in service; (b) new concrete blocks using SP2 admixture have been cast.
Besides that, the compatibility of using sugarcane bagasse ash (SCBA) in conjunction with the sulfate-resistant mineral admixture to produce concrete was also investigated in this study. Sugarcane bagasse ash refers to the residual ash collected through the process of burning sugarcane bagasse to produce heat for generating electricity. The substantial annual production of this by-product material necessitates responsible handling to mitigate its environmental impacts. The suitability of SCBA in the context of concrete construction has been the subject of extensive investigation by numerous researchers. These studies can be categorized into two primary approaches: using SCBA as a substitute for cement,8–20 or using it as a substitute for fine aggregate.21–25
Cordeiro 9 studied the SCBA that was burnt in a boiler at temperatures ranging from 700 to 900 °C. The analysis revealed that the SCBA had a significant silica content, accounting for 78.3% of the ash. However, it was observed that nearly 60% of the silica existed in the form of quartz, while 16% was present as cristobalite, and 24% existed in an amorphous state. The authors explained that the quart content came from sand sticking to the sugarcane during harvesting. They also found that the high temperatures in the boiler caused the formation of cristobalite. The substantial presence of quartz in SCBA offers a plausible explanation for its favorable potential as a substitution for fine aggregate with a pretty high level of replacement. Sales 21 conducted a study using low-reactivity SCBA as a substitute for sand in concrete production, replacing it with up to 30%. The findings showed that the concrete produced with SCBA with up to 30% exhibited higher strength compared to the control concrete. Modani 22 observed a similar outcome and reported that concrete with 10% and 20% SCBA replacement for sand demonstrated higher compressive strength than the control concrete after 28 days. However, it is important to note that the aggregate type significantly affects the sorption characteristics and sulfate resistance of concrete. 26 Using raw SCBA as a fine aggregate replacement can possibly increase the sorption characteristics as it is typically more porous than natural sand. Dense and non-reactive aggregates such as basalt or quartzite can enhance magnesium sulfate resistance by minimizing sulfate ingress and limiting the extent of chemical degradation in the cement paste.
Meanwhile, when considering SCBA as SCMs, a considerable number of studies, most of which were conducted a relatively long time ago, reported that the optimal content of SCBA should be limited to 10%.8–10,12,13,19 One of the main challenges of using SCBA instead of cement in concrete is that most types of SCBA have poor reactivity. Recent studies tried to increase the reactivity of SCBA before using it in concrete. For instance, El-said et al. 27 investigated the effects of raw and processed SCBA in concrete and found that while raw SCBA did not improve the mechanical properties of concrete, the processed SCBA, achieved through one hour of regrinding and reburning at 600°C for two hours, increased the concrete compressive strength and flexural strength up to 12% and 8%, respectively. However, their research also indicated the optimal amount of processed SCBA to partial substitute for cement weight remains at 10%. Exceeding this percentage leads to a decline in concrete density, compressive strength, and flexural strength. Another study by Hussien and Oan, 28 which followed a similar procedure to process SCBA, also achieved significant improvements in compressive strength and splitting tensile strength, increasing by 20% and 33%, respectively. However, their findings indicated that the optimal SCBA replacement level was only 5% when used as a partial substitute for cement. Similarly, Daniel et al. 29 incorporated 5% processed SCBA as a cement replacement and 10% recycled polyethylene terephthalate (PET) as a sand replacement. Their results showed a slight decrease in compressive strength, with reductions of 3.58% at 28 days and 2.54% at 56 days compared to the control concrete.
To enhance the reactivity of SCBA, the carbon-rich fibrous unburnt particles, which have no pozzolanic properties, must be removed by sieving through a 300 μm sieve and then the SCBA needs to be ground to cement fineness (300 m2/kg). 19 However, while grinding is acknowledged as an effective method for enhancing the reactivity of SCBA, it is important to consider the significant energy consumption associated with this process. Based on a study, 9 it is estimated that about 250 kWh/t of energy is needed to grind SCBA in an industrial ball mill so that the SCBA is fine enough to be partially used as cement replacement in concrete without compromising compressive strength properties. It is worth noting that the total electrical energy consumption for cement production is just about 100 kWh/t. 30 Let's compare that to the 250 kWh/t needed just to grind SCBA, it is clear that grinding SCBA requires much more energy. Therefore, energy efficiency should be carefully considered when using this technique.
This study investigates the potential of incorporating SP2 and low-reactivity SCBA in concrete, particularly for applications in Vietnam's coastal regions. While recent research has focused on enhancing the pozzolanic reactivity of SCBA through processing methods like grinding and reburning, the maximum feasible replacement level remains constrained to 5–10% of cement content, despite the added energy and cost. In contrast, this study demonstrates that even raw SCBA, without any additional processing, can replace 5% of cement without compromising mechanical properties, offering a more energy-efficient and practical alternative for sustainable concrete production. To do that, a comparative analysis was also conducted to highlight the tradeoffs between engineering performance and energy efficiency when using untreated versus processed SCBA. Furthermore, to the best of the authors’ knowledge, no internationally published research has examined the characteristics and applicability of SCBA produced in Vietnam for use in the construction sector, despite its potential as a viable SCM. Given that SCBA composition varies based on factors such as sugarcane variety, growth conditions, and combustion processes, 31 it is crucial to conduct dedicated studies to assess its suitability in Vietnam and explore methods for minimizing its environmental impact. Additionally, the combination of SCBA with SP2 presents an opportunity to enhance the performance of concrete while enabling the direct utilization of untreated SCBA, eliminating the need for energy-intensive treatment methods and broadening the potential for sustainable material use in construction.
Methodology
Materials and mixture proportions
The binding materials employed in this research (see Figure 2) included 40-grade Portland cement (OPC) sourced from Vissai Cement Company, SCBA obtained from a sugar production plant in Phu Yen Province, Vietnam, and SP2 provided by Lotus Chemical Joint Stock Company. The cumulative percentage passing sieve of 45 μm and the activity index at 7 days of SP2 are 4.5% and 122.6%, respectively. The chemical compositions and relevant properties of these binding materials can be found in Table 1 and Figure 3. According to Table 1, the main components of OPC were CaO (66.34%), followed by SiO2 (21.86%), which contributed to formation of OPC hydration products (i.e. C–S–H gels and CH). Meanwhile, the major components of SCBA were SiO2 (65.44%), followed by Al2O3 (4.46%), while SP2 contained SiO2 (88.40%) and Al2O3 (3.51%). These compositions indicate that both SP2 and SCBA have the potential to react with CH produced during OPC hydration, resluting in the formation of secondary C–S–H gel.

Binding materials: (a) OPC; (b) SCBA; (c) SP2.
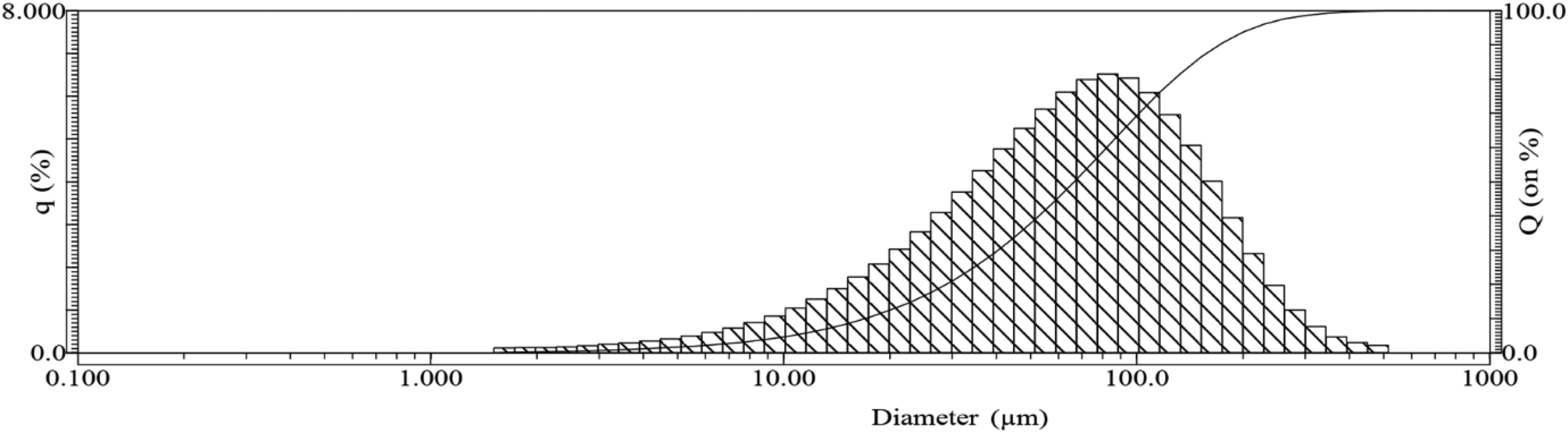
Particle size distribution of SCBA.
Chemical compositions and other properties.

For the composition of the aggregate, river sand was used as the fine aggregate, while crushed stone with a maximum particle size of 20 mm was used as the coarse aggregate. Their particle size distribution is shown in Figure 4. To maintain consistent workability with an equal slump of 10 cm, tap water was used in combination with a superplasticizer. The superplasticizer dosage ranged from 0.8% to 1% of the cement content. There are four concrete mixture proportions as presented in Table 2. The control mixture exclusively employed OPC as the binding material. In mixture SP2, 4.7% of total binding materials were SP2, while in mixture SCBA, 5% of total binding materials were SCBA. The final mixture, named SP2-SCBA, stands for the combination of 4.7% SP2 and 5% SCBA as the total binding material. All mixtures had an equal water-to-binder (W/B) ratio of 0.37. The rationale for selecting the replacement ratios of SCBA and SP2 was to ensure that the 28-day compressive strength of the concrete incorporating SCBA and SP2 remained at least equal to that of the control mixture and met the target strength of 50 MPa.
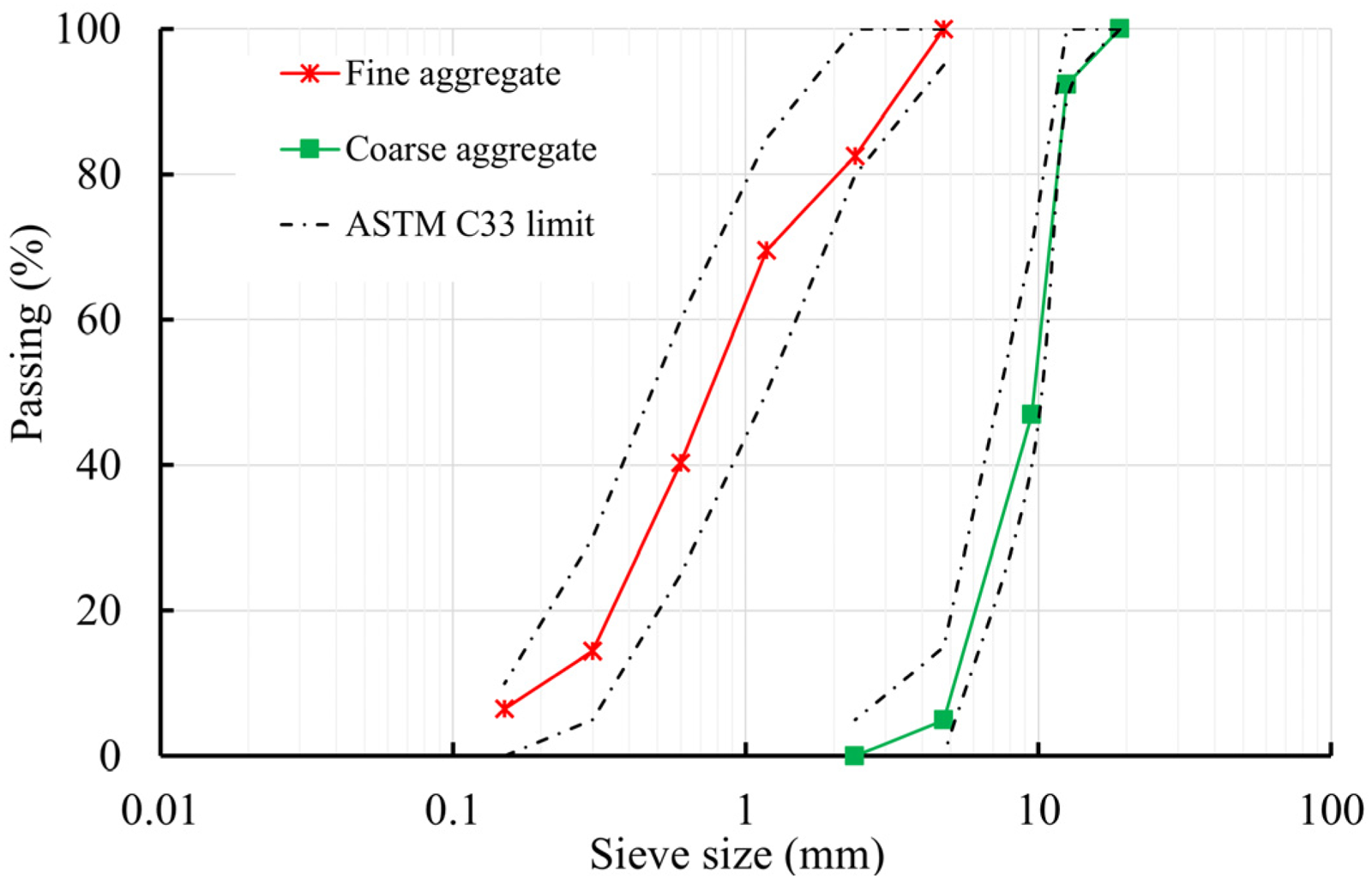
Particle size distribution of aggregates.
Concrete mixture proportions.
Mixing, casting, and curing
The mixing procedure began by dry blending the binding materials and river sand in a pan mixer for a duration of 3 minutes. Subsequently, the crushed stone was added to the mixer, followed by an additional 3 minutes of mixing. Lastly, tap water, along with the appropriate dosage of superplasticizer was introduced into the mixture. The entire mixture underwent a further 4 minutes of mixing. Fresh concrete after mixing was cast into the steel molds. The concrete surfaces were then covered by plastic films. All the samples were placed in the laboratory and cured at a room temperature of 25 °C. After 24 h, the samples were demolded and then cured in either freshwater, ambient air, or seawater until the designated testing day. It is important to note that most of the data reported in this research were based on freshwater curing. Data for ambient air and seawater curing were partially reported to support the trends observed in the freshwater curing results.
Measurements
Compressive strength and splitting tensile strength
The compressive strength test, conducted in accordance with TCVN 3118, 32 was performed on cubic samples with dimensions of 150 × 150 × 150 mm. Meanwhile, the splitting tensile strength was conducted based on the ASTM C496. 33 This test was carried out on cylindrical samples with dimensions of 150 mm in diameter by 300 mm in height. The compressive strength test was performed on days 7, 28, and 56, while the splitting tensile strength test was performed on day 28.
Sorption characteristics
Water absorption, apparent porosity, and sorptivity were performed on cubic samples with sizes of 100 × 100 × 100 mm at 28 and 56 days. The results of testing represent the average of two replicate samples. Water absorption and apparent porosity were tested based on guidance from ASTM C642-13.
34
At the time of testing, the concrete samples were placed in the drying oven at 100 ± 5 °C for 24 h. Afterward, the samples were naturally cooled in the laboratory, and the dry mass (A) was determined (Figure 5(a)). Next, the specimens were submerged in water for 48 h. Subsequently, they were placed in a suitable receptacle filled with tap water and boiled at a temperature of 90°C for 5 h (Figure 5(b)). After the boiling process, the specimens were left to cool at room temperature for 14 h, and their saturated surface-dry mass (C) were determined. Finally, the specimens were suspended by wires, and their apparent mass in water (D) was determined (Figure 5(c)). The absorption after immersion and boiling of the concrete, and apparent porosity were determined according to the following formulas.
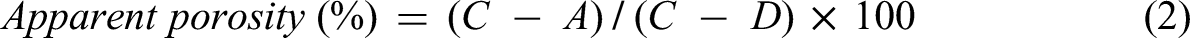

Water absorption and apparent porosity tests: (a) weighting dry mass of sample; (b) immersing and boiling samples in water; (c) weighting sample in water.
The procedure for conducting the sorptivity test follows the guidelines outlined in ASTM C1585-13.
35
Prior to the test, the four lateral surfaces of the specimens were cleaned and then coated with waterproof epoxy adhesive. Subsequently, the specimens underwent a drying period at 60 °C for 48 h (Figure 6(a)), then they were placed in a sealed plastic box at room temperature for an additional 24 h (Figure 6(b)). The top surfaces were covered with plastic films to prevent possible water evaporation (Figure 6(c)). To initiate the test, the specimens were weighted to determine the initial mass m0 (g). A stray with absorbent paper was prepared, and the test specimens were placed on it. Water was introduced into the tray, maintaining a water surface approximately 1.0 cm from the bottom of the tray throughout the test. The mass mt (g) of the sample was then recorded at specified time intervals: 1, 5, 10, 20, 30, 60, 120, 180, 240, 300, and 360 min. The cumulative water absorption (I) was calculated using equation (3). Meanwhile, the sorptivity index (S) was correlated with I and the square root of elapse time (t1/2) according to equation (4).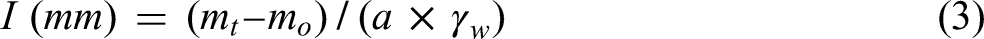


Sorptivity test: (a) dry the samples after covering lateral surfaces with epoxy adhesive; (b) place the samples in a sealed plastic box for 24 h; (c) submerge the bottom of samples in a tray containing water and absorbent paper.
Scanning electron microscopy
Scanning electron microscopy (SEM) analysis (JSM-IT200, USA) was used to identify the specific morphological characteristics of the samples. The SEM analysis was performed on one sample of each mixture after 56 days of ageing. Samples from the core of the cylindrical specimens were selected. The samples were then crushed into the size of about 5 mm and immersed in acetone to stop hydration and carbonation (Figure 7(a)). Before testing, the samples were dried in a vacuum desiccator for 24 h. The samples were then coated with platinum. An acceleration voltage of 15 kV and a magnification from 500 × to 2000 × were used to observe the microstructural differences between the samples. Additionally, the Energy Dispersive X-ray Spectroscopy (EDS) was employed to identify the elemental composition presented in the samples (Figure 7(b)).

Scanning electron microscopy (SEM) analysis; (a) samples for the test; (b) scanning electron microscopy equipment.
The experimental test method in this study can be generally found in Figure 8.

Flowchart of experiment.
Results and discussion
The results in the following subsections present data for four mixtures: Control, SP2 (4.7% SP2 as a cement replacement), SCBA (5% SCBA as a cement replacement), and SP2-SCBA (4.7% SP2 and 5% SCBA as a cement replacement). The fifth mixture, T-SP2-SCBA, represents the theoretical value of the synergistic effect between SP2 and SCBA and is calculated using equation (5). This equation is a modification of the theoretical synergistic effect equation between fly ash and silica fume as presented by Radlinski and Olek.
36
A synergistic effect refers to a phenomenon in which the combined action or interaction of multiple elements produces a result that is greater than the sum of their individual effects. In other words, if a synergistic effect between SP2 and SCBA exists, their combined impact is more significant or potent than what would be expected based on their individual contributions.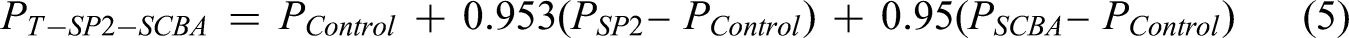
Mechanical properties
The data depicted in Figure 9 illustrates the compressive strength and splitting tensile strength of various concrete mixtures cured in fresh water. At 7 days, the compressive strength of the SP2 mixture surpassed that of the control mixture. However, both the SCBA and SP2-SCBA mixtures exhibited lower compressive strength values compared to the control mixture. Overall, the changes in concrete compressive strength observed during this testing age were relatively modest, ranging from 1.2 to 1.8 MPa. As the concrete aged, the SP2 mixture consistently outperformed the other mixtures in terms of enhancing compressive strength. In comparison to the control mixture, the SP2 mixture demonstrated significant improvements, with compressive strengths that were notably higher. Specifically, at 28 and 56 days, the SP2 mixture showed enhancements of 8.9 MPa and 9.8 MPa, respectively. It can be seen from Table 1 that the specific surface area and SiO2 content of SP2 are much higher than those of OPC. Consequently, the compressive strength at both early and later ages of the SP2 mixture is higher than that of the control mixture due to the filler effect and pozzolanic reaction of SP2. In contrast, there were no substantial improvements in compressive strength observed with the mixtures containing SCBA at 28 days. In fact, the SCBA mixture even exhibited lower compressive strength (52.4 MPa) than the control mixture (53.0 MPa). At 56 days, both the SCBA and SP2-SCBA mixtures displayed slightly higher compressive strength values than the control mixture. The late-stage development in compressive strength indicates that the untreated SCBA from Vietnam used in this study has the potential to perform a pozzolanic reaction to improve the concrete compressive strength at later ages. This late-stage development aligns well with the results found in studies of untreated SCBA from Mexico.37,38 In these studies, using 10% untreated SCBA as cement replacement in mortar demonstrated higher compressive strengths than the control mixture at 28, 56, 90, 450, and 600 ages. 37 Similarly, using 20% untreated SCBA as cement replacement in concrete decreased the compressive strength at early ages but increased it after 90 days compared to the control mixture. 38

Results of compressive strength and splitting tensile strength.
The results for splitting tensile strength at the 28-day showed a similar pattern to what was observed in the 28-day compressive strength, as shown in Figure 9. Notably, the SCBA mixture exhibited a slightly higher splitting tensile strength at 28 days compared to the control mixture, despite having a slightly lower compressive strength at the same test day. Furthermore, there was a discernible enhancement in splitting tensile strength from the SP2-SCBA mixture to the control mixture at 28 days. While the SP2-SCBA mixture showed only a marginal 0.38% increase in compressive strength at 28 days compared to the control mixture (rising from 53.0 MPa to 53.2 MPa), the corresponding figure for splitting tensile strength demonstrated a significant 37.9% increase (climbing from 3.06 MPa to 4.22 MPa). This observation holds particular interest, given that established building codes39,40 and researchers41,42 commonly propose formulas predicting splitting tensile strength as functions or square-root functions of concrete compressive strength. This implies a close relationship between splitting tensile strength and compressive strength, where an increase in compressive strength is anticipated to result in a corresponding increase in splitting tensile strength, although at a comparatively smaller rate. 43 Therefore, the unexpected 31% increase in splitting tensile strength with only a 1.5% increase in compressive strength for the SP2-SCBA mixture over the SCBA mixture prompts further investigation in the subsequent sub-section to identify whether this observation can be attributed to physical or chemical effects. In fact, the effect of untreated SCBA on the splitting tensile strength remains inconclusive. 44 Some studies have reported a decrease in splitting tensile strength,45,46 while others indicate a modest increase when used as a 10% replacement for cement.47–50 The potential reduction in tensile strength due to partial cement replacement with SCBA has been attributed to higher drying shrinkage, resulting from the loss of capillary water. This occurs because untreated SCBA has lower porosity compared to Portland cement, which increases the concrete's water demand. Consequently, SCBA concrete may develop wider cracks than the control mix due to greater capillary water loss. This reduction in tensile strength is more pronounced in mixtures with a high water-to-binder (W/B) ratio. In contrast, improvements in tensile strength were reported when replacing cement with SCBA at levels up to 10% in mixtures with a lower W/B ratio. To support this observation, Figure 10 presents a summary of recent studies illustrating the effect of W/B ratio on tensile strength improvements. Since the concrete mixture investigated in this study had a very low W/B ratio (0.37) and was carefully cured in freshwater until the testing day, the risk of drying shrinkage was minimized. Consequently, the observed increase in tensile strength in this study can be attributed to these favorable conditions.

Summary of the effect of SCBA on concrete tensile strength from recent studies.
The results for both the compressive and splitting tensile strengths of the SP2-SCBA mixture consistently fall lower than those of the theoretical value of synergistic effect (T-SP2-SCBA). This observation clearly indicates the absence of a synergistic interaction in OPC, SP2, and SCBA systems, at least within the dosage range investigated in this study. In alternative ternary cementitious systems, such as those involving Portland cement, fly ash, and silica fume, noteworthy observations have been made by researchers, indicating a synergistic effect of fly ash and silica fume that positively influences the compressive strength properties of concrete.36,51 This synergistic effect was attributed to plausible chemical interactions between fly ash and silica fume, wherein the finely dispersed fly ash particles were covered by the silica fume grains, thereby augmenting cement hydration and refining the resultant hydrated matrix. 52 Additionally, the observed synergistic effect may come from physical interactions, where the particle sizes of cement, fly ash, and silica fume exhibit a well-graded distribution, leading to a superior packing density of concrete compared to instances where fly ash or silica fume is employed individually. 53 Hence, it can be inferred from the outcomes of this investigation that the SP2 and SCBA may lack compatibility to create sufficient chemical or physical interactions needed to enhance the concrete's strength indices.
Sorption characteristics
The assessment of water absorption, apparent porosity, and sorptivity properties holds considerable significance in the evaluation of the durability and performance of concrete structures, particularly in environments characterized by exposure to water and moisture.54,55 Water absorption serves as a direct metric for quantifying the concrete's capacity to absorb water, providing valuable insights into the material's susceptibility to water ingress. Apparent porosity, on the other hand, functions as an evaluative parameter for the volume of void spaces within the concrete structure that are readily accessible from the surface. Finally, sorptivity is instrumental in gauging the rate at which water permeates the concrete surface through capillary suction. Lower values in these properties usually indicate a concrete structure with enhanced resistance against harmful environmental factors like seawater.
Figure 11 presents the water absorption (Figure 11(a)), apparent porosity (Figure 11(b)), and sorptivity data (Figure 11(c)) for four distinct mixtures cured in fresh water, alongside the theoretical value denoting the synergistic effect of SP2-SCBA. The trend in the dataset shows that the SP2 mixture consistently performs better than the other three mixtures. This is evident in its ability to achieve the lowest values across all three sorption indices at different testing ages. SP2 showed notable mitigation in water absorption, with a reduction of approximately 15% at 28 days and 24.5% at 56 days in comparison to the control mixture. Corresponding reductions in apparent porosity were observed at 15.6% and 23.8% for the same durations, while reductions in sorptivity stood at 24.6% and 26%, respectively. In contrast, the inclusion of SCBA in the mixtures showed negligible impacts on these indices compared to the control mixture. However, an exception is discerned in the SP2-SCBA mixture, where a distinct reduction in sorptivity at a later age is observed when compared to the control mixture (Figure 11(c)). The SP2-SCBA mixture also exhibited the most substantial enhancement in the sorptivity index from 28 days to 56 days, with a significant decrease of 15.3%. This reduction exceeds threefold the improvement observed in the apparent porosity index of the same mixture. The sorptivity index is influenced not only by porosity but also by factors such as pore diameter and distribution, 56 and porous materials characterized by smaller pores will likely have a decelerated water uptake during sorptivity tests. This suggests although the porosity of the SP2-SCBA mixture did not undergo significant changes between 28 days and 56 days, there was a significant enhancement in its pore structures via the pozzolanic reaction of SCBA, so that the sorptivity index was very much improved. It is also obvious that no synergistic effects are evident in the results of the three sorption properties within this ternary system.

Sorption properties of investigated mixtures: (a) water absorption; (b) apparent porosity; (c) sorptivity.
In Figure 12, the graph shows the sorption characteristics of the examined concrete mixtures with lines, while the mechanical properties described in section “Mechanical properties” are shown by shaded areas. The SP2 mixture demonstrated superior performance among the concrete mixtures, displaying minimized values in sorption characteristics. Simultaneously, it exhibited the highest values in compressive strength and splitting tensile strength indices. This outcome can be attributed to the composition of SP2, primarily comprising finely dispersed amorphous SiO2, which demonstrates high reactivity with Ca(OH)2 during cement hydration. Silica from SP2 can react with Ca(OH)2 as early as 12 h after mixing and provide a large amount of hydrates at the early ages. 57 Moreover, silica particles from SP2 can provide nucleation sites, facilitating the precipitation of cement hydration products from the solution. 58

Plots of sorption properties versus mechanical properties: (a) sorption properties versus compressive strength (28 days); (b) sorption properties versus compressive strength (56 days); (c) sorption properties versus splitting tensile strength (28 days).
In contrast, SCBA mixture did not show improvements in the sorption and mechanical properties of concrete at early ages, specifically at 28 days. Although the sorptivity and splitting tensile strength indices of the SCBA mixture exhibited better performance than the control mixture, the disparities in these values between the SCBA and control mixtures were marginal. However, at a later age (56 days), the SCBA mixture had lower values for water absorption, apparent porosity, and sorptivity indices compared to those of the control mixture. Additionally, its compressive strength was also higher than that of the control mixture at 56 days. This observation can be explained by the presence of SiO2 components in SCBA, which react with Ca(OH)2 generated during cement hydration. Consequently, the concrete structure is refined through the substantial formation of calcium silicate hydrate compound (C-S-H), resulting in increased densification that impedes water ingress. The improvements were not noticeable at 28 days, likely because of the low reactivity of the SCBA employed in this study. In contrast to the amorphous SiO2 particles derived from SP2, where 95% of the particles exhibit dimensions below 45 μm, Figure 3 shows that the particle sizes of SCBA are much larger, with many around 100 μm and some as large as 300 μm. Given this size disparity, the reactivity of SiO2 in SCBA is less reactive than in SP2.
When incorporating 4.7% SP2 with 5% SCBA together as cement replacement, properties like sorptivity, water absorption, apparent porosity, and compressive strength showed slight improvements compared with the SCBA mixture. The lack of effectiveness in producing a synergistic effect in this ternary cementitious matrix can be attributed not only to the large fineness of raw SCBA mentioned earlier, but also to the reduction in Ca(OH)2 availability for pozzolanic reaction. The effect of silica from SP2 on promoting the rate of hydration depends on the Ca(OH)2 availability, 58 but the incorporation of 5% SCBA as cement replacement may reduce the Ca(OH)2 content at early ages due to the decrease in cement content. As a result, there was not enough Ca(OH)2 availability for SP2 to react with. Similar results were observed when silica fume was present with fly ash. Weng et al. 57 found that the reaction between silica fume with Ca(OH)2 was delayed from 12 h to 1 day when 30% of fly ash replaced cement. Mien and Chau 59 also observed that when 40% fly ash replaced cement, the incorporation of 4% silica fume in the mixture showed a higher temperature rise due to heat of hydration compared to using 8% silica fume. Although SCBA in this study was just used as a level of 5% of cement replacement, which is considerably lower than the utilization of fly ash mentioned above, it still reduced the reactivity of silica from SP2. However, it is very interesting to note that, while some sorption properties such as sorptivity, water absorption, or apparent porosity did not exhibit significant improvement, the splitting tensile strength of SP2-SCBA at 28 days witnessed a significant improvement when compared to this value with those of control and SCBA mixtures. The value of 4.22 MPa of splitting tensile strength of SP2-SCBA is just slightly smaller than that of the SP2 mixture.
Comparative analysis of raw SCBA and processed SCBA in concrete performance
This section presents a comparative analysis of the performance of the concrete mixture incorporating raw SCBA used in this study versus those employing processed SCBA. The objective is to evaluate the trade-off between using untreated and treated SCBA. To ensure a fair comparison, only studies with mixture proportions closely resembling those used in this study are considered. For instance, Amin et al. 60 incorporated processed SCBA (25%) and silica fume (SF) (5%) in concrete with a water-to-binder ratio (W/B) of 0.35; Abdalla et al. 61 incorporated processed SCBA at levels of 10% and 20%, along with SF (5%), in concrete with a W/B of 0.35; Farrant et al. 62 incorporated processed SCBA (30%) and SF (10%) in concrete with a W/B of 0.5. Studies incorporating both SCBA and SF were selected because the SP2 used in this study closely resembles the SF material in terms of properties and performance.
Compressive strength comparison
Figure 13 illustrates a comparative analysis of compressive strength performance between untreated SCBA (this study) and treated SCBA mixtures, highlighting significant differences in both early-age and long-term performance. For 28-day compressive strength, untreated SCBA exhibited minimal impact on compressive strength. In contrast, although treated SCBA can be used at significantly higher replacement levels, most mixtures incorporating treated SCBA showed a notable reduction in early-age strength. However, treated SCBA used at 10% or 20% replacement combined with 5% silica fume (SF) demonstrated a moderate improvement of approximately 6% in compressive strength. At later ages, untreated SCBA combined with 4.7% SP2 achieved a 3.47% increase in compressive strength at 56 days. Meanwhile, treated SCBA mixtures such as 10% treated SCBA or 25% treated SCBA with 5% SF showed a considerable reduction in compressive strength at 91 days. The most significant strength improvement at later ages was observed with 10% treated SCBA combined with 5% SF. From this comparison, it can be concluded that treated SCBA only provides a clear advantage over untreated SCBA at later ages when used at a 10% replacement level in combination with 5% SF.

Comparative analysis of compressive strength.
Splitting tensile strength comparison
The comparison of splitting tensile strength (Figure 14) reveals that the untreated SCBA (5%) combined with SP2 (4.7%) significantly improved the splitting tensile strength by 37.91% at 28 days. This remarkable result was not observed in any of the selected studies involving treated SCBA. The best performance achieved using treated SCBA was with a 20% replacement level; however, the improvements in splitting tensile strength at both 28 and 91 days were considerably lower compared to the untreated SCBA combined with SP2 in this study. It is suspected that the treatment process, which involves burning and sieving to enhance SCBA reactivity, may result in the loss of a substantial amount of fibrous particles. These fibrous particles are believed to contribute significantly to the enhancement of splitting tensile strength, potentially explaining the superior performance observed with untreated SCBA and SP2.

Comparative analysis of splitting tensile strength.
Water absorption comparison
For water absorption, it can be seen in Figure 15 that the treated SCBA significantly reduced the water absorption of concrete. The highest water absorption value was observed when treated SCBA (25%) was incorporated with SF (5%). In contrast, the untreated SCBA had minimal effect on reducing concrete's water absorption, regardless of the presence of 5% SP2. Although the curing period for the untreated SCBA data was shorter than that of the treated SCBA data, it is evident that untreated SCBA cannot achieve comparable performance in reducing water absorption, even if cured for up to 91 days. This is because untreated SCBA tends to be more porous and less reactive than treated SCBA, limiting its ability to promote the pozzolanic reaction.

Comparative analysis of water absorption.
Trade-off between treated and untreated SCBA considering energy consumption
The performance of untreated and treated SCBA in concrete mixtures reveals a notable trade-off between the benefits gained through treatment processes and the associated energy costs. As shown in Table 3, untreated SCBA demonstrates the potential for enhancing concrete properties, particularly at lower replacement levels, without requiring additional energy-intensive treatment. Untreated SCBA, when used at a 5% replacement level, showed improvements in both compressive and tensile strength at later ages, indicating its potential as a sustainable SCM. Although untreated SCBA may not be suitable for higher replacement levels, its ability to enhance performance without additional processing energy makes it a viable option in applications where energy conservation is a priority.
Performance comparison of treated and untreated SCBA in concrete considering energy consumption.

Note: (↑): increase; (↓): decrease; (-): neutral; NA: not available.
Conversely, treated SCBA allows for higher replacement levels; however, this comes with trade-offs in both energy demand and performance consistency. Treatments such as grinding, drying, and calcination involve significant energy consumption, yet the resulting material does not consistently outperform untreated SCBA. For instance, Amin et al. reported reduced compressive strength when SCBA was finely ground and incorporated at 25% replacement, despite the additional 5% SF. Similarly, Farrant et al. observed substantial strength reductions at 30% and 50% replacement levels, despite employing calcination to improve SCBA reactivity. Notably, Abdalla et al. found that although treated SCBA was used, the most effective mix was limited to 10% SCBA combined with 5% SF, suggesting that even with additional treatment, the optimal replacement level remains relatively moderate.
These findings emphasize that while treatment can enhance SCBA's performance in some contexts, untreated SCBA presents a promising alternative for improving concrete properties in an energy-efficient manner, particularly at low replacement levels. Thus, the selection of untreated or treated SCBA should carefully consider the balance between energy consumption, performance gains, and practical application constraints.
Scanning electron microscopy and energy dispersive spectroscopy analysis
SEM results
The SEM analysis depicted in Figures 16 and 17 highlights the distinct characteristics of the hydrated product in the control and SP2 mixtures at various magnifications, respectively. The images reveal a dense and rigid structural microstructure of the SP2 mixture in comparison with that of the control mixture, indicating a more developed hydration process. In contrast, Figure 18 showed SEM images of the SCBA mixture, which has a spongier structure with a substantial presence of unreacted cementitious materials. Notably, Figure 18(b) highlights numerous unreacted particles with diameters exceeding 10 μm, a phenomenon less pronounced in the SP2 mixture (see Figure 17). Evidently, the structure of the SCBA mixture exhibited increased void content and diminished rigidity compared to the SP2 mixture. The increased porosity and unreacted cementitious particles observed in the SCBA mixture (Figure 18) may likely contribute to its lower compressive strength (see Mechanical properties section) because voids may reduce the effective load-bearing capacity, while unreacted particles limit further hydration. Moreover, the presence of unreacted particles in SCBA-containing mixtures could influence long-term durability (see Sorption characteristics section). Of particular interest are the abundant spongy-fibrous structures observed in the SEM images of the SCBA mixture. Many previous studies agree that raw SCBA consists of particles of various shapes and sizes, including unburned fibrous particles, prismatic particles, and mostly irregular particles.19,44,63 The spongy-fibrous structures that we observed in this study are likely the unburned fibrous particles that result from incomplete combustion in the production of raw SCBA. These structures, characterized by their elongated nature and tendency to interconnect, are notably absent in both the SEM images of the control mixture and the SP2 mixture. Many previous studies agree that raw SCBA consists of particles of various shapes and sizes, including unburned fibrous particles, prismatic particles, and mostly irregular particles. As a result, despite minimal gains in compressive strength, we assume that this unique fibrous structure in the SCBA mixture (Figure 18) and SP2–SCBA mixture (Figure 19) provides a plausible explanation for the observed enhancement in splitting tensile strength compared to the control mixture because it could enhance the interfacial bonding between cement matrix and aggregates and prevent the crack propagation. Additionally, these elongated particles may act as micro-reinforcements, improving tensile stress distribution (see Mechanical properties section).

SEM images of mortar extracted from the control sample at 56 days.

SEM images of mortar extracted from SP2 sample at 56 days.

SEM images of mortar extracted from concrete (SCBA mixture) at 56 days: (a) and (b) spongy particles; (c) and (d) fibrous product structures.

SEM images of mortar extracted from SP2-SCBA sample at 56 days: (a) and (b) magnification from a fibrous structure position; (c) and (d) magnification from a non-fibrous structure position.
To further support the assumption that the fibrous structures in SCBA could lead to an increase in splitting tensile strength, additional data are presented in Figure 20. These additional data are collected from mixtures that are similar to those previously presented, except they have different curing conditions (seawater and open-air) compared to the original data, which had freshwater curing condition. It is clearly observed that while the compressive strength decreases from the control mixture to the SCBA mixture, the splitting tensile strength increases. It is consistent with the results in Mechanical properties section regarding SCBA mixture. SEM analysis also revealed a significant presence of fibrous structures in the SCBA samples cured in seawater and open-air conditions, as shown in Figure 21.

Compressive strength and splitting tensile strength of additional data underwent open-air and sea water curing conditions at 28 days.

Fibrous structures from SCBA mixtures captured from additional data.
EDS results
Figure 22 illustrates the EDS spectra corresponding to the analyzed regions of control, SP2, SCBA, and SP2–SCBA mixtures after a testing period of 56 days. The EDS results reveal the presence of primary elements, including Carbon (C), Oxygen (O), Magnesium (Mg), Sulfate (S), Aluminum (Al), Silicon (Si), and Calcium (Ca). These elements intricately combine to form quartz (as observed in samples containing sand) and hydration products such as C-S-H, ettringite, and Ca(OH)2. The EDS spectra also provide quantitative information, presenting the percentages of each element detected in the samples.

EDS spectra correspond to SEM image: (a) control mixture; (b) SP2 mixture; (c) SCBA mixture; (d) SP2-SCBA mixture.
The carbon content in SCBA (12.24 wt%) and SP2-SCBA (9.98 wt%) mixtures was markedly higher than that in the control (5.67 wt%) and SP2 (5.60 wt%) mixtures. This observation aligns with expectations, given the notably elevated loss on Ignition (LOI) of SCBA as provided in Table 1. The substantial presence of unburnt carbon in SCBA can detrimentally impact cement hydration. The previous studies64–66 incorporated unburnt carbon at varying proportions (2% to 24%) into the OPC-Fly ash system with a composition of 80% OPC and 20% fly ash. Their findings indicated a discernible impact on the alkalinity of the cement with an increase in carbon content. This phenomenon can be attributed to the carbonation process, wherein carbon particles serve as a surface for the deposition of calcium carbonate (CaCO3), facilitating the reaction between carbon dioxide (CO2) and calcium hydroxide (Ca(OH)2). Consequently, the alkalinity of the concrete experiences a reduction. This process is shown through the following reactions (equations (6) to (8)).


If the observed reduction in alkalinity attributed to unburnt carbon holds true in the ternary system under investigation, it offers a plausible explanation for the underperformance of the SP2-SCBA mixture in various investigated aspects when compared to the SP2 mixture. SP2, known for its high reactivity, contributes to early-age strength development in concrete. However, the elevated presence of unburnt carbon in SCBA reduced alkalinity and impeded the pozzolanic reaction. This could explain the higher porosity (see Sorption characteristics section) of the SCBA mixture compared to the SP2 mixture.
Additionally, the inclusion of SP2 and SCBA would make the structure of concrete even more complex through the formation of secondary C-S-H with different Ca/Si ratios. According to Parada et al., 38 the Ca/Si atomic ratio of primary C-S-H (P) generated from cement hydration ranges from 1.57 to 2.1. Meanwhile, the secondary C-S-H (S), which is formed by the pozzolanic reaction is less than 1.6. It can be found from Figure 22 that the average Ca/Si atomic ratio of the spectral area in the control specimen is 1.94 (with a percentage of Ca and Si atomic at 17.98 and 9.29%, respectively), making it a representative example of C-S-H(P). In contrast, the average Ca/Si atomic ratio of spectral area for the other specimens ranges from 0.88 to 1.09 (these values can be calculated from the percentage of Ca and Si atomic in the table). Therefore, the results suggest that these products are C-S-H(S), which are formed by the pozzolanic reaction of SP2 or SCBA. Consequently, the formation of C-S-H(S) could be an explanation for the improvement of the mechanical properties and sorption characteristics of specimens incorporating SP2 and SCBA, as discussed in Mechanical properties and Sorption characteristics sections.
Conclusion
By investigating the mechanical and sorption properties alongside microstructure via SEM and EDS, the feasibility of using waste materials (SCBA) along with the SP2 in concrete was comprehensively investigated. The following conclusions were drawn.
The untreated SCBA from Vietnam used in this study has the potential to be used as SCMs in concrete production. However, due to the significant presence of unburned particles and high LOI, it has low pozzolanic reactivity and can only be considered for a limited replacement level of 5% of OPC, compared to the optimal replacement level of 10% cement reported in previous studies using other untreated SCBA. A synergistic effect within the ternary system comprising Portland cement, SP2, and SCBA on concrete engineering properties was not observed. This absence of synergy can be attributed to the low pozzolanic reactivity inherent in the raw SCBA, as well as the inadequate particle size distribution of the ternary cementitious material employed. Concrete with a 5% replacement of cement by SCBA exhibited negligible effects on compressive strength, water absorption, apparent porosity, and sorptivity at the early age of 28 days. Nevertheless, by day 56, this particular mixture demonstrated performance comparable to that of the control mixture. Another noteworthy benefit of the SCBA mixture is its enhanced 28-day splitting tensile strength in comparison to that of the control mixture. This enhancement in splitting strength in SCBA mixture compared to the control mixture is observed in all three different curing conditions. Statistically, previous research indicates that the improvement in splitting tensile strength with SCBA concrete tends to be observed in concrete with a low W/B (below 0.5). The microstructure of mixtures containing SCBA revealed a substantial presence of spongy-fibrous hydrated structures. This discovery provides a rational explanation for the notable improvement in the splitting tensile strength of mixtures containing SCBA compared to the control mixture. The superior properties of the SP2 mixture were confirmed through laboratory experiments. In comparison to the control mixture, the incorporation of 4.7% of the SP2 resulted in concrete exhibiting an 18% increase in 56-day compressive strength, a 42% enhancement in 28-day splitting tensile strength, and substantial densification of the microstructure, leading to reduced water absorption and sorptivity This explains why wave-dissipating concrete blocks made with this mixture have performed well in the tough marine environment for four years while maintaining a commendable esthetic appearance. While treated SCBA can provide better compressive strength performance at higher replacement levels, untreated SCBA can offer notable improvements in tensile strength and significant energy savings. This distinction emphasizes the importance of selecting the appropriate SCBA type based on project requirements and sustainability goals. Despite the low pozzolanic reactivity of raw SCBA, it can be incorporated with SP2 to replace approximately 10% of cement to produce concrete with equivalent or better properties compared to the control mixture. The direct use of raw SCBA in concrete can save a lot of energy by eliminating the need for additional SCBA treatment processes.
These findings highlight the potential of using untreated SCBA with SP2 to produce sustainable concrete that exhibits enhanced tensile strength with minimal reduction in compressive strength. However, additional experiments such as X-ray diffraction (XRD), thermogravimetric analysis (TGA), and Fourier transform infrared spectroscopy (FTIR) should be carried out to further validate these results. Moreover, investigating the long-term performance of SCBA and SP2 concrete (e.g. at 182 or 365 days) and considering aggressive environmental conditions, such as chloride ingress and sulfate exposure, is crucial. Lastly, a comprehensive cost analysis comparing untreated and treated SCBA would provide valuable insights for material selection in sustainable concrete production. The findings in this study can serve as a foundation for further exploring optimized concrete mixtures incorporating sulfate-resistant mineral admixtures and sugarcane bagasse ash, as well as investigating the long-term performance of these materials in various environmental conditions. Moreover, all relevant experimental details and data in this study can contribute to developing predictive models for the mechanical and durability properties of similar concrete mixtures.
Footnotes
Acknowledgements
This study has received funding from Mientrung University of Civil Engineering (MUCE), Vietnam. We express our gratitude for the time and resources provided by MUCE. Additionally, our sincere appreciation goes to the students of classes D17 and D18 in the faculty of Infrastructural Engineering-Technology, MUCE, for their assistance in the preparation and casting of samples.
Author contributions
Vinh Ngoc Chau: methodology, formal analysis, visualization, writing—original draft. Viet Quoc Dang: project administration, conceptualization, supervision, methodology, writing—review & editing. Kenji Kawai: writing—review & editing, validation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data can be shared upon reasonable request.
