Abstract
In this study, Cypress fruit-based-activated carbon (ACCF) was employed to investigate the removal of Zn
Introduction
Water quality is crucial for life and resources. Heavy metals pollute water, are toxic, bioaccumulate, and degrade slowly. 1 Heavy metals can bind to cellular proteins, leading to harmful effects. This affects the survival, growth, and reproduction of aquatic life. Chronic exposure to sublethal contamination can be ecologically devastating. Heavy metals enter the water through industrial discharge, urban runoff, and agricultural drainage. 2 Human activities such as mining, releasing industrial waste and sewage, and using agricultural and household products that contain heavy metals have increased environmental levels of these pollutants. 3 This study concerns the removal of zinc, copper, and nickel ions from water. These heavy metals are at moderate to low pollution levels and can have toxic effects on the aquatic environment. For zinc (Zn), the threshold hardness theory suggests that its toxicity is due to interference with calcium uptake and/or toxicity resulting from Zn's high affinity for soft ligands such as sulfide, especially at low water hardness. However, at moderate water hardness, the inhibition of Ca uptake is protected by competitive water effects and the formation of less toxic carbonate complexes. The toxic response to Zn exposure is typical of an essential metal, where disruption of homeostasis occurs before mortality. 4 Copper metal is unique compared to other metals because its toxicity can be critical in soft water because of the high bioavailability of the free ion. However, the activity of the copper-free ion decreases as water hardness increases because of the competition from other cations, which leads to reduced toxicity. 5 Nickel is considered toxic to aquatic organisms encompasses algae, crustaceans, and fish. 6 Several studies have proved that nickel can obstruct the growth and photosynthesis of aquatic plants, as well as disrupt the reproduction and development of aquatic invertebrates. 7 Moreover, the biomagnification of nickel elements through the food chain can influence human health by consuming contaminated seafood, as it can accumulate in the tissues of aquatic organisms. 8 Different treatment methods and technologies have been employed to remove hazardous elements from water. These methods and technologies consist of flotation, electrochemical methods, coagulation, filtration, precipitation, adsorption, ion exchanges, reverse osmosis, and membrane technologies. 9 One of the most widely recognized and effective methods for the removal of heavy metals from water is adsorption. This process is generally easy to design and operate, and they are proficient in removing a diverse array of contaminants from polluted water, including heavy metals, even at low concentrations. 10 The availability and affordability of adsorbents, such as activated carbon, make adsorption a feasible solution for water treatment. Furthermore, the reversibility of adsorption enables the potential recovery and reuse of the adsorbent, rendering it a more environmentally sustainable approach than other techniques used for similar purposes. 11 Numerous researchers provided great efforts to produce more effective, cheaper, and environmentally friendly activated carbons. Therefore, various precursors, such as agricultural and agroindustrial by-product materials, have been utilized as precursor materials for activated carbon production by using various physical and chemical activation processes. 12 In this study, the cypress fruit-derived activated carbon (ACCF) was selected as an adsorbent for removing Zn, Cu, and Ni ions from water due to its abundance, renewability, and minimal competing uses, making it a cost-effective and environmentally sustainable material. Cypress fruit contains natural precursors for carbonization, enabling the development of porous structures and functional groups ideal for adsorption (Gan, 2021). 13
The research emphasizes optimizing adsorption parameters to maximize the removal efficiency of heavy metals both individually and simultaneously, offering a cost-effective and sustainable solution to mitigate water contamination.
Materials and methods
Chemicals
All chemicals used in this study; Zinc (II) nitrate, Zn(NO3)2 (99.5%), copper (II) nitrate dihydrate, Cu(NO3)2.2H2O (95%), nickel (II) nitrate, Ni(NO3)2 (98%), Sodium hydroxide, NaOH, (97%), and hydrochloric acid (HCl) were purchased from Sigma Aldrich. All solutions were prepared using double-distilled water.
Preparation of solutions
A 500 ppm stock solutions of Zn(NO3)2, Cu(NO3)2.2H2O, and Ni(NO3)2. Each solution was separately prepared in a 500 mL volumetric flask using double-distilled water to confirm purity. Afterward, these stock solutions were diluted further with doubled distilled water to attain the desired final concentrations for each metal ion according to the conducted experiments. For the simultaneous experiments, a mixture encompassing all three metal ions was prepared by combining calculated aliquots of the diluted stock solution. The pH of solutions was adjusted using solutions of 0.1 M NaOH and HCl to attain the desired experimental conditions.
Adsorbent preparation and characterization
The cypress fruit was obtained from the Isra University campus in Jordan. Afterward, the cypress fruits underwent washing and were then dried at a temperature of 100 °C for 48 h. They were subsequently ground, impregnated with 98% phosphoric acid, heated to a temperature of 450 °C, and finally washed with 1.0 M NaOH solution to attain neutralization. 14 The Prepared cypress fruit-based activated carbon (ACCF) underwent a process that encompassed crushing and sieving, resulting in a particle size of 180 μm (Figure 1). Fourier transform infrared (FTIR, TENSOR 27 model from BRUKER) was used to assign the functional groups at the ACCF surface. The scanning electron microscope (SEM; Apreo 2 S LoVac, USA) was used to confirm the adsorption process.
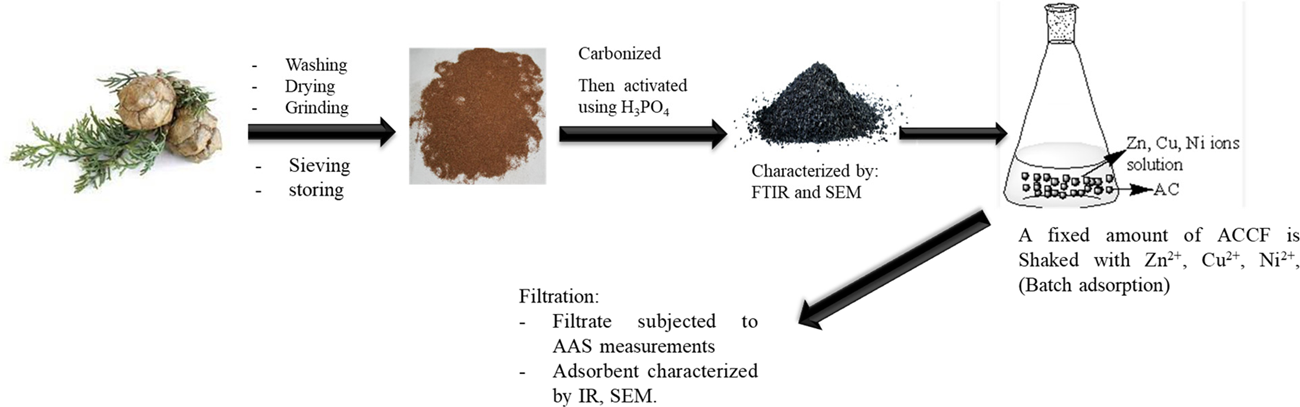
Preparation of ACCF and batch adsorption.
Adsorption batch experiment
Solutions of each metal ion with different concentrations were shaken with a stated mass of ACCF for a set time. The study inspected adsorption parameters such as ACCF dosage (20–10 mg), initial metal ion concentration (20–120 mg L−1), pH, shaking time (3–11), and temperature (298, 308, and 318 K). Further experiments were performed using a solution of the three metal ions to investigate the simultaneous adsorption process. The concentrations of the metal ions were determined using an atomic absorption spectrometer (ContrAA 800 from Analytik Jenna, Germany). The amount of adsorbed metal ions (qe in mg g−1) was calculated based on Equation 1 in which Ci and Ceq (mg L−1)are the initial and equilibrium concentrations, respectively:
where v and w are the volume (L) and weight of ACCF adsorbent (g), respectively.
The percentage of removal was also calculated based on Equation (2):
Regeneration of ACCF adsorbent
A chemical regeneration was employed. The absorbent of ACCF was washed with 0.1 M HCl solution to remove residual adsorbates, followed by drying at 60 °C for 6 h. The regeneration efficiency was assessed by comparing the adsorption capacity of regenerated ACCF with fresh ACCF over 5 cycles.
Results and discussion
Characterization of adsorbent
Fourier transform infrared
The FTIR spectroscopy results (Table 1) offered valuable insights into the functional groups present in the ACCF adsorbent and their role in the adsorption of metal ions. The presence of a variety of functional groups on the ACCF surface is verified by the distinct peaks observed in the spectra. It is essential to comprehend these functional groups to elucidate the adsorption properties of ACCF and its potential applications in the removal of metal ions. It is important to note that the ACCF surface has a successful interaction with Zn2+, Cu2+, and Ni2+ ions, as evidenced by the observed variations in absorbance and intensity during metal ion adsorption. The emergence of new peaks in the 400–600 cm−1 region is particularly noteworthy. This spectral range frequently corresponds to metal–oxygen bond vibrations, which implies the formation of coordination bonds between the oxygen atoms present on the ACCF surface and the adsorbed metal ions (Zn2+, Cu2+, and Ni2+). The effective adsorption of the metal ions onto the ACCF material is strongly supported by these findings. The FTIR spectra are depicted in Figure 2 both before and after adsorption.

Fourier transform infrared (FTIR) spectra for the adsorbent (ACCF) before and after adsorption.
Fourier transform infrared (FTIR) results of ACCF, ACCF-zn, ACCF-cu, and ACCF-Ni, ACCF-mix.

Scanning electron microscope and energy-dispersive X-ray spectroscopy
SEM analysis (Figure 3) provides compelling evidence for the successful adsorption of Zn

Scanning electron microscopy (SEM) analysis of (a) ACCF, (b) ACCF-Zn2+, (c) ACCF-Cu2+, (d) ACCF-Ni2+, (e) ACCF-mix.
Energy-dispersive X-ray spectroscopy (EDS) analysis (Figure 4) identified various elements in the synthesized ACCF adsorbent, including carbon, oxygen, phosphorus, sodium, and potassium. EDS confirmed the presence of adsorbed metal ions. As shown in Table 2, the weight percentages of Zn

Energy-dispersive X-ray spectroscopy (EDS) analysis of (a) ACCF, (b) ACCF-Zn2+, (c) ACCF-Cu2+, (d) ACCF-Ni2+, and (e) ACCF-mix.
Energy-dispersive X-ray spectroscopy (EDS) analysis of ACCF before and after adsorption.

Batch adsorption
Influence of adsorbent amount
This study inspected the influence of ACCF dosage on the adsorption of Zn
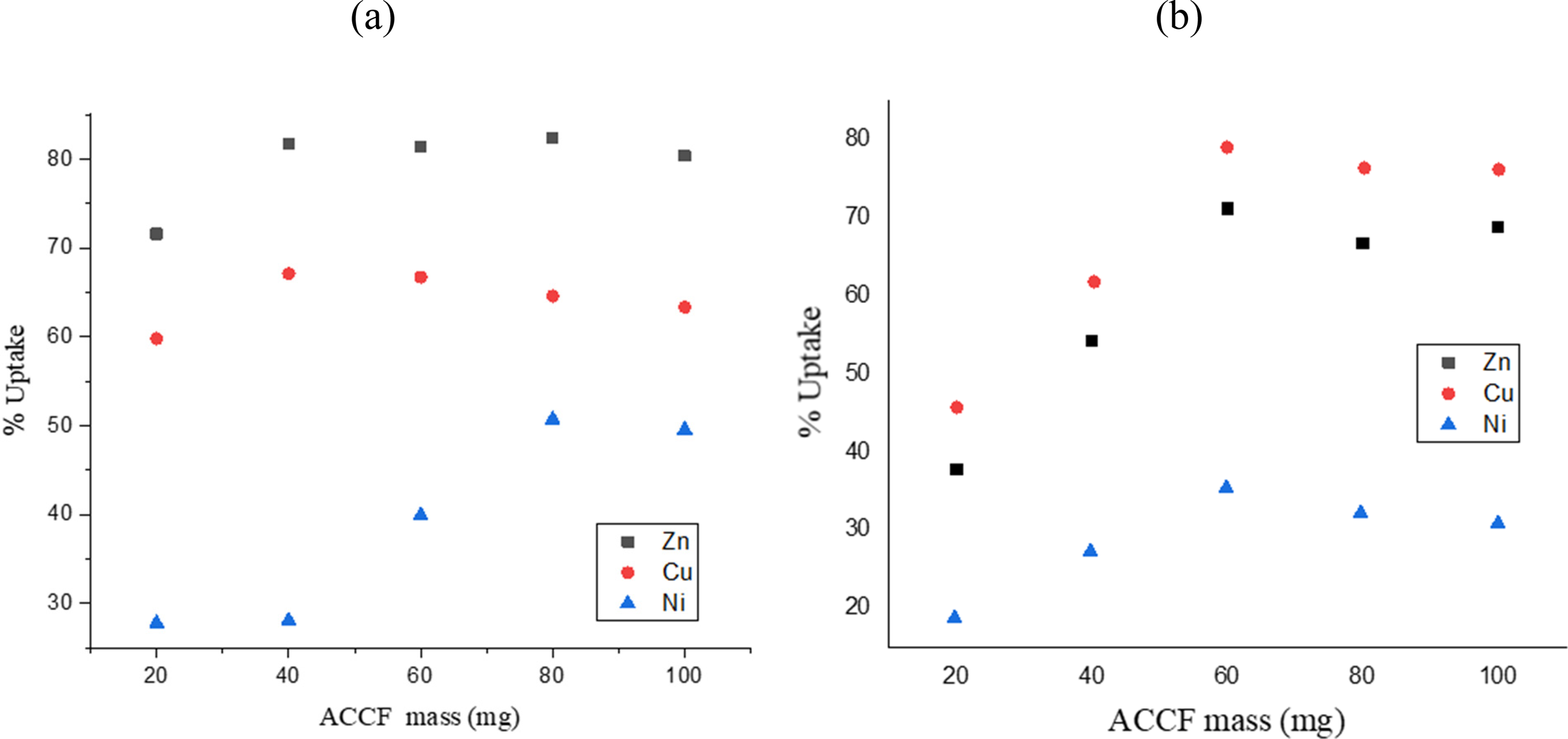
Influence of ACCF mass on the adsorption of (a) Zn2+, Cu2+, & Ni2+ and (b) ion mixture.
Influence of ions initial concentration
This study explored the influence of initial metal ion concentration (30–120 mg/L) on their adsorption onto ACCF at a constant temperature (298 ± 1 K). Figure 6 reveals a clear correlation between initial concentration and adsorption for each metal ion and the mixture. Notably, exceptional uptake percentages of 81.36% (Zn
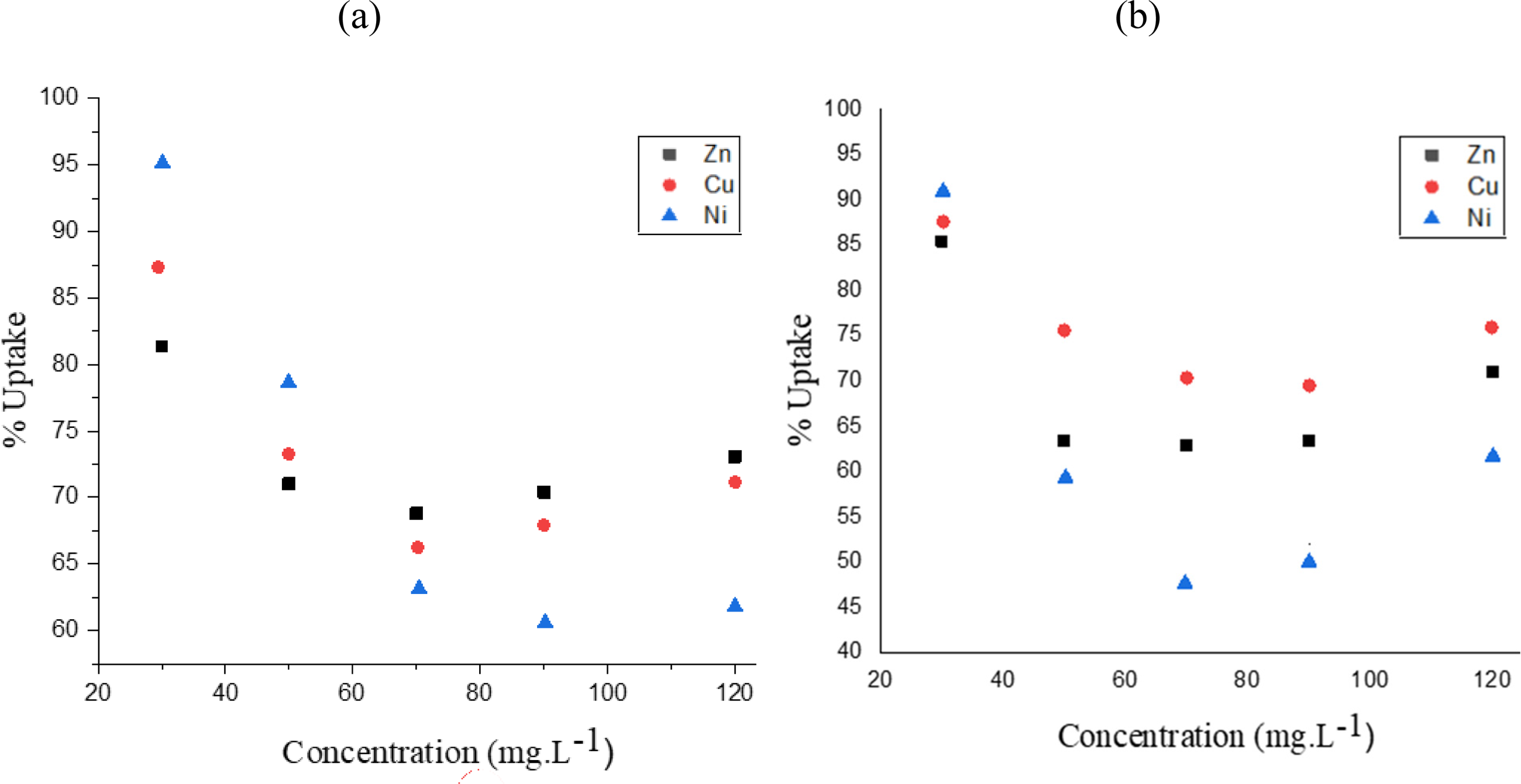
Influence of the initial concentration on the adsorption for (a) individual ion and (b) mixture ions.
Influence of adsorption contact time
To determine the perfect contact time required for maximum adsorption onto ACCF, triplicate batch adsorption experiments were conducted for each metal ion. The experiments involved shaking 40 mg ACCF with separate solutions of 30 mg L−1 Zn

Influence of adsorption contact time for (a) individual metal ion and (b) simultaneous uptake of metal ions.
Influence of PH
The pHpzc of the ACCF surface was reported as 7.0 by.
15
This study investigated the influence of solution pH (3–11) on the adsorption of Zn
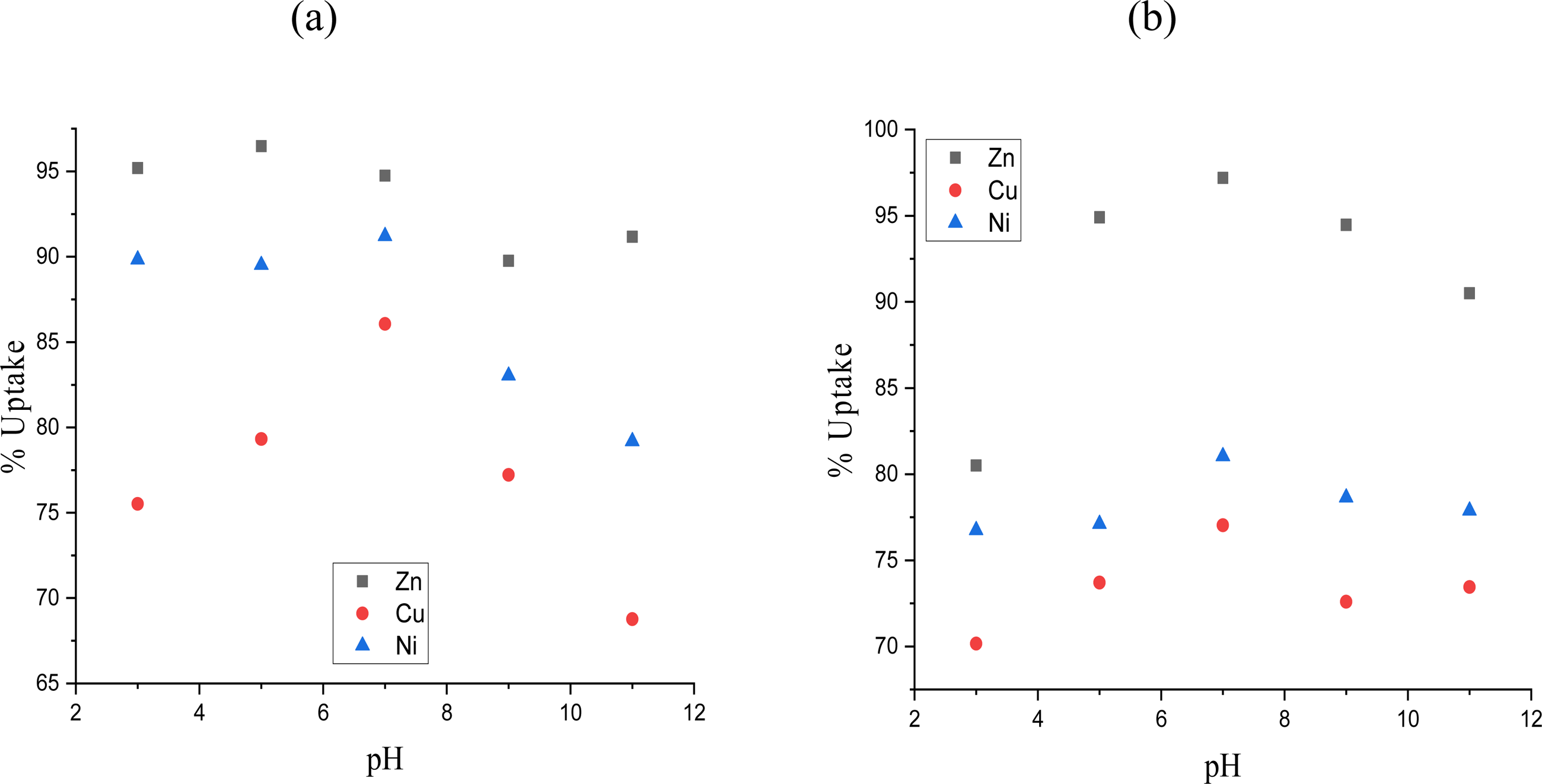
Impact of pH on the uptake of (a) individual metal ions and (b) mixture by ACCF.
Kinetics and mechanism
To understand the mechanisms controlling the adsorption of Zn
Kinetics models equations.

PFO: pseudo-first order; PSO: pseudo-second order; IPD: intra-particle diffusion.
Figures 9 and 10 provide compelling evidence that the PSO kinetic model best describes the adsorption behavior of Zn

Linear kinetics studies of the adsorption of the metal ions on ACCF (a) PFO and (b) PSO. PFO: pseudo-first order; PSO: pseudo-second order.

Non-linear kinetics studies of the adsorption of metal ions on ACCF for (a) Zn2+, (b) Cu2+, and (c) Ni2+.
Results of kinetics studies for the adsorption of Zn2+, Cu2+, Ni2+, and mixture of metal ions onto ACCF.

PFO: pseudo-first order; PSO: pseudo-second order; IPD: intra-particle diffusion.
The contribution of IPD to the adsorption mechanism of heavy metal ions onto ACCF was evaluated using the equations presented in Table 3.
The IPD model suggests that the rate-limiting step depends on the value of the intercept (C) obtained from the model fitting (refer to Table 3 for the equations). When C is zero, IPD is the sole factor controlling the adsorption rate. However, a non-zero C indicates that surface adsorption also plays a role alongside IPD in determining the overall rate. Based on Figure 11, surface adsorption appears to be the rate-limiting step for Zn

(A) Linear and (b) Nonlinear intra-particle model for the adsorption of the Zn2+, Cu2+, & Ni2+ on ACCF.
The values of R2 and constants for all kinetics models were summarized in Table 4.
Adsorption isotherms
To understand the nature and characteristics of the adsorption process for Zn2+, Cu2+, and Ni2+ onto ACCF, various adsorption isotherm models were employed. These models, including Langmuir, Freundlich, and Temkin (refer to Table 5 for details), helped assess the interaction between the adsorbent (ACCF) and the adsorbates (metal ions). By analyzing the fit of these models, insights could be gained into whether the adsorption process is primarily physical or chemical in nature.
Linear and non-linear isotherms model equations.

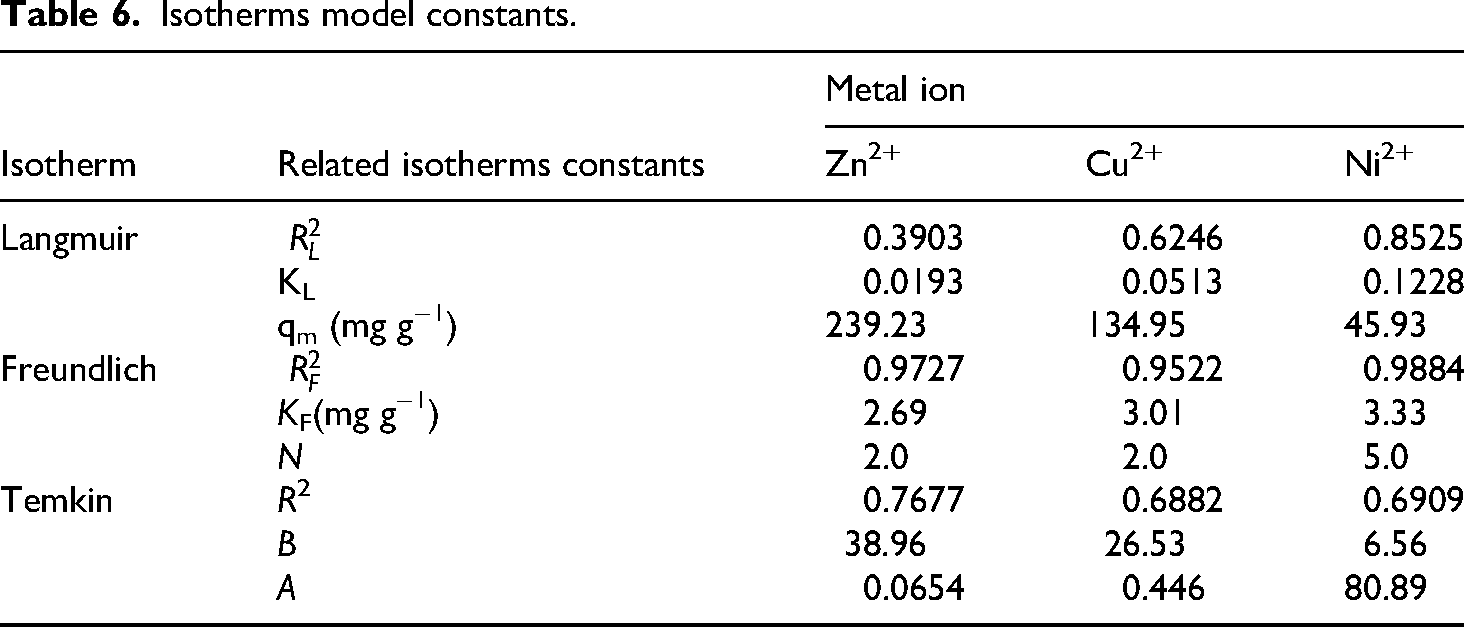
The linear forms of the three isotherm models (Langmuir, Freundlich, and Temkin) are presented in Figure 12(a) to (c) for visual analysis. Table 6 summarizes the key parameters obtained from these plots, including correlation coefficients (R²) and relevant constants. Notably, the Freundlich isotherm exhibited the highest R² values for Zn2+ (0.9727), Cu2+ (0.9522), and Ni2+ (0.9884) adsorption onto ACCF. These high R² values suggest a good fit for the Freundlich model, indicating a non-uniform ACCF surface with heterogeneous adsorption sites for these metal ions. All adsorption processes are favorable at high metal ion concentrations (chemical interactions occur), as shown by the Freundlich model n parameter values greater than unit (Table 6). Table 6 shows that these data are valid regardless of the metal ion or biosorbent, indicating that all biosorption processes use comparable principles (Ciobanu et al., 2024Click or tap here to enter text.). The values of Temkin constant values, B (38.96, 26.53, & 6.56 for Zn2+, Cu2+, and Ni2+, respectively) indicate a significant interaction between the metal ion and the ACCF adsorbent. 17

The linear isotherms of (a) Langmuir, (b) Freundlich, and (c) Temkin of Zn2+, Cu2+, and Ni2+ removal by ACCF.
Isotherms model constants.
Thermodynamics
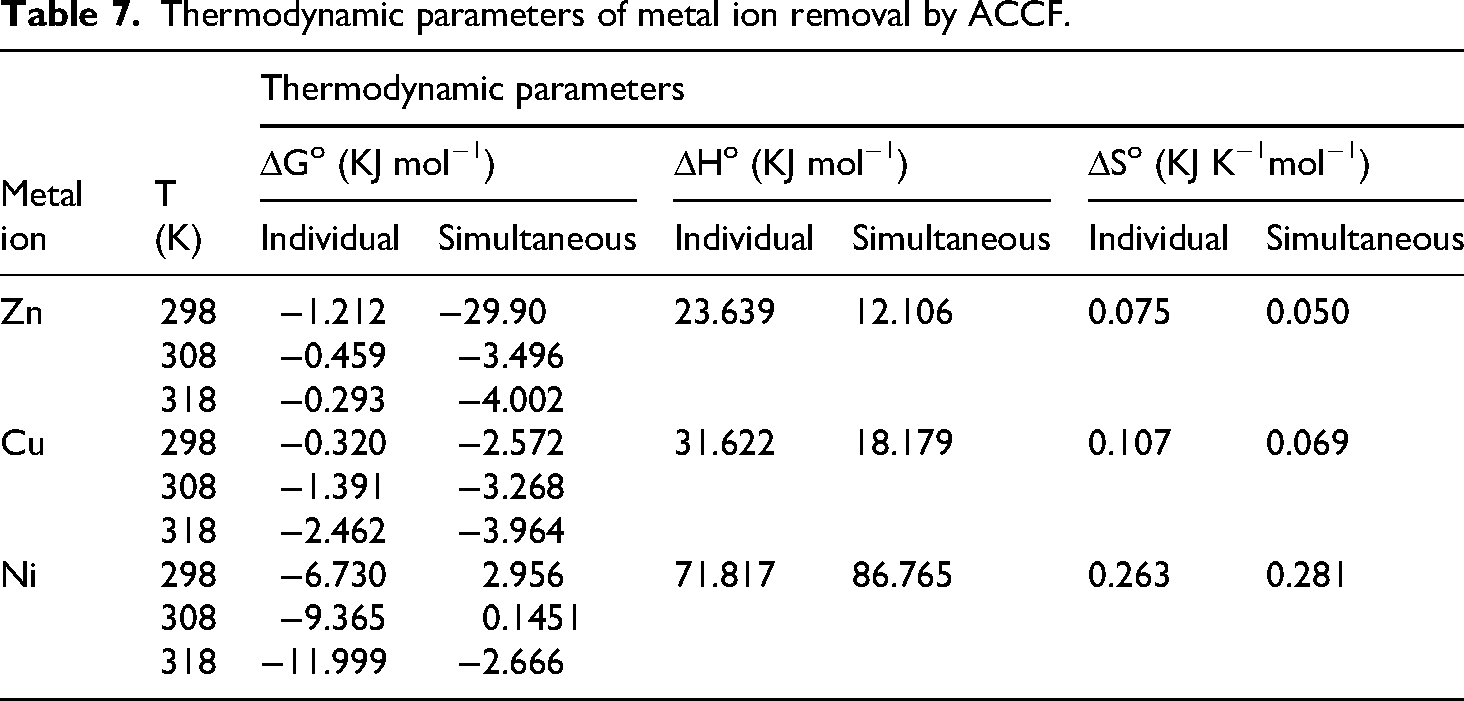
To further understand the effectiveness of metal ion removal by ACCF, thermodynamic parameters were measured under controlled conditions. Experiments were carried out at three constant temperatures (298 ± 1.0 K, 308 ± 1.0 K, and 318 ± 1.0 K) with continuous agitation (1.0 h). Key thermodynamic parameters (ΔG⁰, ΔH⁰, and ΔS⁰) were calculated to reveal the nature of the adsorption process. The results are presented in Table 5. The standard Gibbs free energy change (ΔG⁰) was calculated using ΔG° = ΔH°-TΔS°. Vant Hoff equation was used to determine the enthalpy change (ΔH⁰) and entropy change (ΔS⁰) from the slope and intercept of a plot of ln(K) vs. 1/T:
K corresponds to the adsorption equilibrium constant according to the best-fitted model, and R is the universal constant of ideal gases, 8.314 J K−1 mol−1. The negative ΔG° value (Table 7) indicates that ACCF adsorbs metal ions spontaneously for both individual and simultaneous removal. The positive ΔHo value (Table 7) indicates the endothermic nature of the ions. 18 Adsorbent affinity and irregularities in the adsorbate/adsorbent interface are implied by the positive value of ΔSo. 19
Thermodynamic parameters of metal ion removal by ACCF.
Regeneration of ACCF adsorbent
After conducting numerous experiments, it was determined that 0.1 M HCl acid is an appropriate candidate for the recovery investigation. The adsorption process was carried out under the effectiveness parameters, after which the adsorbed heavy metal ions were desorbed by shaking the ACCF in a 0.1 M HCl solution. The ACCF was then dried and the adsorption/desorption process was repeated multiple times to evaluate its reusability. Atomic absorption analysis was conducted on metal ions of Zn2+, Cu2+, and Ni2+ that were eluted from ACCF. The subsequent equation was employed to determine the percentage of recovery:
20

Cad represents the adsorbed concentration of metal ions and Cde represents the desorbed concentration. The adsorption/desorption performance of Zn2+, Cu2+, and Ni2+ ions using ACCF was evaluated over five cycles, as shown in Figure 13. The results demonstrate a decreasing trend in recovery efficiency with each successive cycle for all three metal ions. Among the metals, Cu2+ consistently exhibited the highest recovery efficiency across all cycles, which suggests a stronger affinity of ACCF for copper ions. Conversely, Ni2+ displayed the lowest recovery percentages, indicating comparatively weaker interaction with the adsorbent. Zn2+ recovery efficiency remained intermediate but showed a similar decreasing trend with repeated cycles. Figure 13(a) demonstrates that the recovery of metal ions decreased, going from 90.83.29% to 77.25% for Zn2+, from 82.80% to 49.19% for Cu2+, and from 65.98 to 42.50 for Ni2+. This suggests that ACCF can be utilized again and again. For the combination of the three metal ions, similar outcomes were attained (Figure 13(b)).

Regeneration of ACCF adsorbent.
Comparison with other adsorbents
The maximum uptake of heavy metal ions by ACCF was compared to other activated carbon adsorbent. (Table 8). The comparison shows that ACCF has satisfied adsorption capacity, which is better than other adsorbents.
Comparison of ACCF Uptake capacity with other adsorbents.
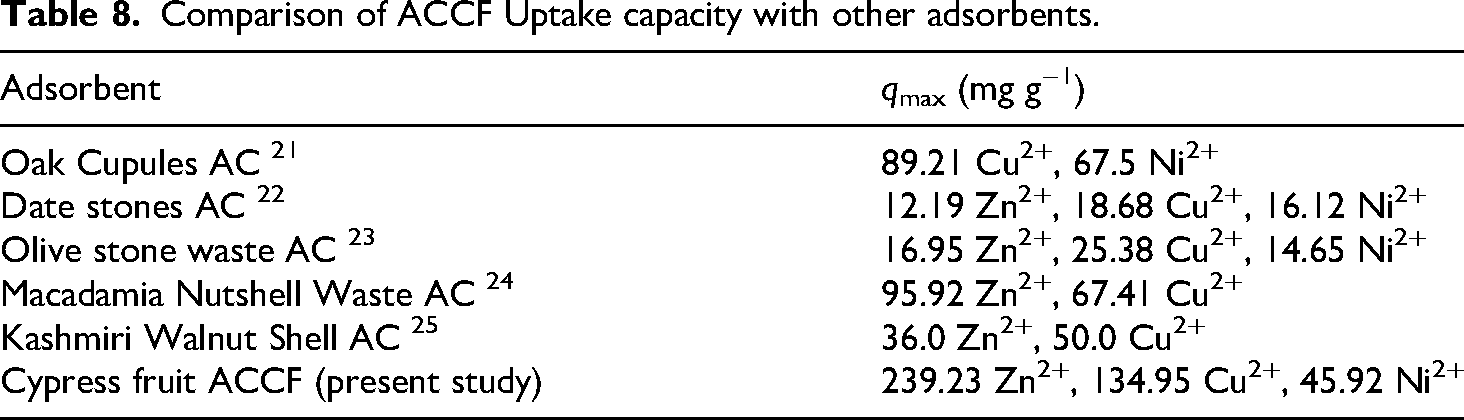
Conclusion
This study demonstrates the efficacy of cypress fruit-based activated carbon (ACCF) for removing Zn2+, Cu2+, and Ni2+ from aqueous solutions using a batch adsorption process. The experiments definitively identified the effectiveness parameters for maximizing the removal efficiency of these metal ions using ACCF. The experimental results indicate that the best conditions for Zn2+, Cu2+, and Ni2+ individual removal using ACCF are as follows: 60 min of agitation for Zn2+, 100 min for Cu2+ and Ni2+, 0.04 g for Zn2+ and Cu2+, and 0.08 for Ni2+. Furthermore, it was found that acidic solutions are more effective in removing Zn2+ and Ni2+ than neutral solutions. However, for Cu2+, the neutral solution demonstrated superior performance. It was discovered that the following were ideal for the simultaneous removal of Zn2+, Cu2+, and Ni2+: Agitated for 40 min, with 0.06 g of adsorbent. In addition, the adsorption process works better with neutral solutions. ACCF exhibited a stronger attraction for Zn2+ ions compared to Cu2+ and Ni2+. Kinetic and isothermal analyses revealed a strong fit between the adsorption of Zn2+, Cu2+, and Ni2+ by ACCF and both the Freundlich isothermal model and the PSO kinetic model. ACCF removes heavy metal ions from the environment using an endothermic and presumably spontaneous process, according to thermodynamic research. Results revealed 239.23 mg g−1 uptake capacity of Zn2+, 134.95 mg g−1 for Cu2+, and 45.93 mg g−1 for Ni2+. After five regeneration cycles, ACCF may lose effectiveness yet remain functioning. Future studies will investigate surface modifications of ACCF to improve selectivity, alongside exploring strategies for scaling up its utilization.
Footnotes
Acknowledgements
The authors would like to express their appreciation to the Isra University Innovation Center (IUIC) for their vital role in testing the samples. The state-of-the-art infrastructure at IUIC significantly contributed to the accuracy of our findings, and we are grateful for their commitment and professionalism.
Ethical considerations
We confirm that this work is original and has not been published elsewhere nor is it currently under consideration for publication elsewhere.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. The first draft of the manuscript was written by Alaa Mahmoud Al-Ma’abreh and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This research was funded by Isra University with grant number of [8-38/2020/2021].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The article contains the data that substantiates the results of this investigation.


