Abstract
Objective
Intranasal analgesic sprays represent a safe, efficacious method for pain relief, with a shorter working time compared to oral painkillers. This study aimed to develop nasal sprays using commonly available over-the-counter analgesics, providing an alternative treatment option that is more convenient and potentially more effective in managing pain, particularly in pediatric patients.
Methods
Three different nasal spray formulations with the contents of diclofenac sodium, ibuprofen, and paracetamol were created, and characterization studies were completed. The possible cytotoxic, genotoxic, and apoptotic effects of nasal sprays were studied on human normal skin fibroblastic cells (CCD-1079Sk) using WST cell viability test, alkaline single-cell gel electrophoresis analysis, and acridine orange/ethidium bromide staining, respectively.
Results
The formulations’ physical appearance and drug content were assessed, yielding nonsignificant results (p > 0.05). All formulations were determined at pH 5.5–6.2 so that the pH values of the prepared formulations were compatible with the pH value of the nasal mucosa. Selected nasal spray formulations were stable for 90 days, and the safe doses were chosen as 0.0625, 0.375, and 1.25 mg/mL for diclofenac, ibuprofen, and paracetamol, respectively, by not showing toxicity even at 24 h.
Conclusion
This study demonstrated that nasal sprays containing paracetamol, ibuprofen, and diclofenac sodium can be successfully formulated. These new formulations may provide alternative treatment and easier application for patients unable to swallow or refuse to take oral analgesics.
Introduction
Intranasal (IN) drug administration represents a noninvasive method for delivering medications. The historical practice of applying moisture to the nasal passages as a soothing treatment dates back to ancient times. The utilization of medicinal substances through IN sprays gained prominence primarily in the 20th century. 1 In modern times, IN sprays have gained significant traction and are particularly recommended as targeted therapeutic options, especially for conditions like allergic rhinitis. This approach enables precise delivery of medications directly to the nasal mucosa, enhancing patients’ overall quality of life. This method also prevents delays in treatment when compared to oral administration.2–4 Many studies in the literature show the effectiveness of IN drug application. Ketamine, fentanyl, oxytocin, and calcitonin are a few examples that have been studied as IN applications.3,5–7
Paracetamol (also known as acetaminophen in the USA), ibuprofen, and diclofenac are over-the-counter analgesic drugs commonly used all over the world. Paracetamol is one of the most popular analgesic drugs used around the world, and it is a popular choice for children under 12 years of age and pregnant or breastfeeding women. 8 Ibuprofen is another commonly used analgesic and antipyretic drug for relieving acute pain, inflammation, and fever. Ibuprofen is widely considered to be the best-tolerated drug of its class, has been described as the mildest nonsteroidal anti-inflammatory drug with the fewest side effects, and has been in clinical use for a long time.9,10 Diclofenac, available worldwide in various formulations, including oral, intravenous, suppository, transdermal patches, and gel, is a widely recognized nonsteroidal anti-inflammatory drug demonstrating its efficacy in managing acute and chronic pains. 11
The aim of the study was to formulate nasal sprays containing paracetamol, ibuprofen, and diclofenac sodium since these drugs are known for their minimal side effects and are available over the counter without a prescription. By creating these sprays, it was aimed to provide alternative treatment and easier application for patients unable to swallow oral analgesics due to underlying neurological deficits, the pediatric age group who may refuse to take oral analgesics and have painful deglutition after tonsillectomy. The new formulations were analyzed for their physicochemical properties besides their possible cytotoxic, apoptotic, and genotoxic effects in vitro.
Materials and methods
Chemicals and materials
Diclofenac sodium, ibuprofen, paracetamol, glycerin, carboxymethyl cellulose (CMC), Tween 80, sodium chloride, benzalkonium chloride, sodium citrate, and citric acid were of analytical standard and obtained from Sigma-Aldrich, Inc. (St Louis, MO, USA).
Formulation preparation
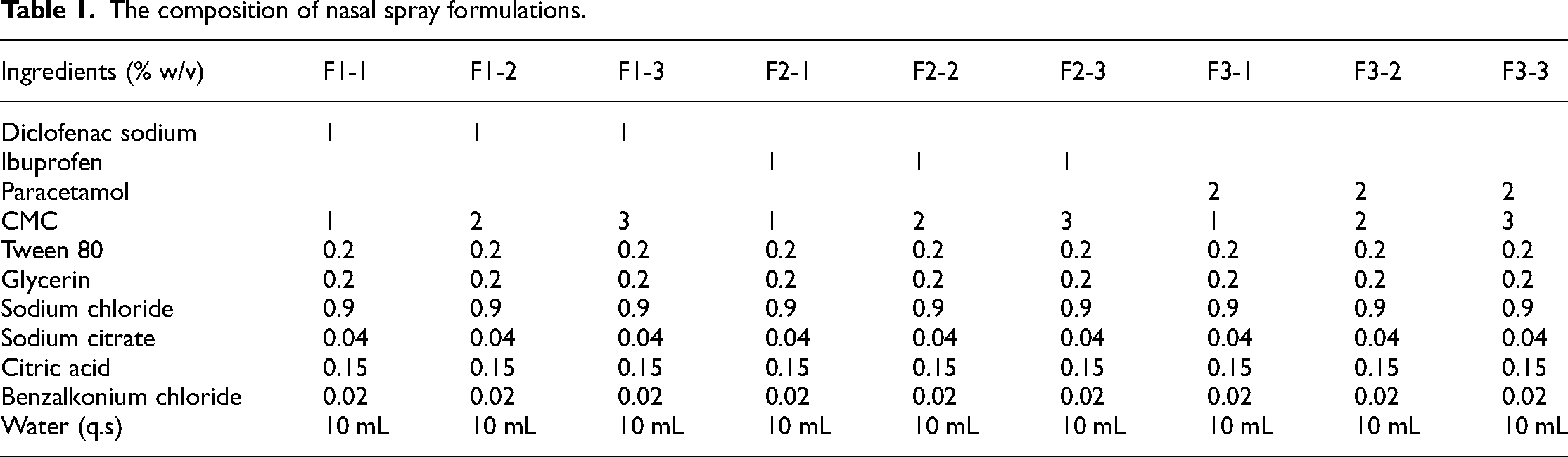
Three different nasal spray formulations containing diclofenac sodium, ibuprofen, and paracetamol were prepared separately according to the conventional production technique. 12 CMC was dispersed in bidistilled water under magnetic stirring, which is used as a mucoadhesive polymer in the concentration range of 1%–3%. After homogenizing the CMC dispersion, active ingredients at constant concentrations were added to CMC dispersion (phase I). Sodium citrate and citric acid were dissolved in bidistilled water, which is used to maintain the pH of the formulations (phase II). Glycerin, Tween 80, sodium chloride, and benzalkonium chloride were dissolved in bidistilled water and used as a viscosity enhancer, surfactant, isotonic agent, and preservative, respectively (phase III). Phase II and phase III were added to phase 1 under the magnetic stirrer (phase IV), and the volume was made of bidistilled water. Then, the nasal spray formulations were placed in a clean and dry nasal spray bottle. All the formulations were prepared in a sterile aseptic condition in the laminar flow chamber. The ingredients of all the formulations are indicated in Table 1.
The composition of nasal spray formulations.
Physicochemical property evaluation
Clarity test
Visual inspection was conducted on the formulations to detect the presence of any foreign particles, and formulations containing such particles were discarded.
pH determination
pH meter (Jenway, Britain) was used to determine the pH values of newly formulated nasal sprays.
In vitro release studies
The in vitro drug release of new formulations was analyzed with the Franz diffusion cell (The Hanson Vertical Diffusion Cell). 13 A nylon membrane (MW cutoff 250 kDa, pore size 0.45 mm) was placed between the donor and the receiver compartment. The receiver compartment was filled with water containing 0.5% sodium dodecyl sulfate to ensure sink condition. The recipient medium was stirred continuously at 400 rpm. Three puffs of the nasal spray formulations were applied to the membrane in the donor compartment. At 0, 0.25, 0.5, 1, 2, 3, 4, 5, and 6 h, 0.5 mL samples were taken from the receptor phase by replacing with an equal volume of fresh medium. The amount of drug released was analyzed by reversed-phase high-pressure liquid chromatography (RP-HPLC).
Stability evaluation of the formulations
The optimized nasal spray formulations were monitored for 3 months to assess their stability. During this time, they were stored at a controlled temperature of 25 ± 2°C and maintained under conditions of 60% ± 5% relative humidity. At predefined intervals throughout the 3 months, samples were taken from the formulations for analysis. These samples were then subjected to various tests, including examination of physical appearance, pH levels, and drug content. The physical appearance of the formulations was visually inspected to detect any changes such as color alterations, phase separation, or particulate matter formation. pH measurements were taken to monitor potential variations in the formulations’ acidity or alkalinity, which could impact their stability. Additionally, the drug content of the samples was quantitatively assessed to determine whether the formulations retained their intended drug concentrations over time.
Analytical determination of active ingredients
The amounts of diclofenac sodium, ibuprofen, and paracetamol ingredients were determined using RP-HPLC system equipped with a UV-Vis detector (Shimadzu, Japan). Filtered and degassed phosphate buffer (pH 3.0):acetonitrile (40:60, v/v) was used for diclofenac sodium, while phosphate buffer (pH 7.5):methanol (70:30, v/v) was used for ibuprofen and paracetamol as a mobile phase. The mobile phase was delivered at a flow rate of 1 mL/min through a GL Sciences InertSustain C18 column (250 × 4.6 mm, 5 μm) maintained at 25 °C. The UV detector was set at 210 nm for diclofenac sodium and 220 nm for ibuprofen and paracetamol detection.
The HPLC method for active ingredients was performed following the International Conference of Harmonization (ICH) Q2 (R1) guidelines. Linearity studies were conducted on standard curves ranging from 0.05 to 10 µg/mL, which were prepared using a stock solution of diclofenac sodium, ibuprofen, and paracetamol. The peak area showed a linear correlation with diclofenac sodium (r2 = 0.9993), ibuprofen (r2 = 0.9990), and paracetamol (r2 = 0.9994). The drug-free spray formulations did not interfere with the diclofenac sodium, ibuprofen, and paracetamol peaks, indicating the selectivity of the method. Additionally, the HPLC method was validated for accuracy, precision, and stability.
A content uniformity study was conducted to determine the formulations’ drug content. One hundred microliters of the formulations was dissolved and diluted using acetonitrile and filtered through a 0.45 µm membrane filter. RP-HPLC (Shimadzu, Japan) was used to quantify the total contents of diclofenac sodium, ibuprofen, and paracetamol in the formulations.
In vitro toxicity evaluation
Culture of cells
The human normal skin fibroblastic cells (CCD-1079Sk, CRL-2097, ATCC, USA) were used for toxicity and genotoxicity studies of the formulations. The cells were cultured in Eagle's minimum essential medium (Biochrome, Germany) complemented with 10% fetal bovine serum (Biochrome, Germany), 100 U/mL penicillin, and 100 μg/mL streptomycin (Biochrome, Germany). The cells were cultured at 37 °C in humidified atmosphere containing 5% CO2.
Cell viability assay
The possible cytotoxic effect of diclofenac sodium, ibuprofen, and paracetamol formulations was analyzed by the WST cell viability test (Roche, Germany). The WST test is favored in cytotoxicity studies due to its safety, simplicity, and sensitivity. The assay relies on the reduction of the WST tetrazolium salt by mitochondrial dehydrogenases enzyme in viable cells to produce a colored formazan product. The amount of formazan produced is directly proportional to the number of metabolically active cells, allowing for precise quantification of cell viability. Briefly, 1 × 104 fibroblast cells were seeded to each well of 96-well plates and treated with different concentrations of nasal sprays for 1, 6, and 24 h at 37°C. After the incubation period, 10 μL WST working solution was added to each well and incubated for 3 h at 37 °C. Viability was calculated as a percentage relative to the nontreated control group by assessing the absorbance at 440 nm using the Varioskan Flash Microplate Reader (Thermo Scientific, USA). The acquired data were averaged to determine a safe dosage. According to previous literature, >80% cell viability was accepted as low cytotoxicity. 14 Therefore, the concentration resulting in viability higher than 80% was accepted as nontoxic in our study. A statistical analysis was also conducted to identify a nontoxic dose that did not result in statistically significant changes in viability (p > 0.05).
Acridine orange/ethidium bromide double staining
The apoptotic effect of formulations was assessed using the acridine orange/ethidium bromide (AO/EB) dual stain technique, which is a simple and inexpensive method used to differentiate apoptotic cells from living cells, as described by McGahon et al. 15 The AO/EB test is a qualitative method used to distinguish between live (viable) and dead (nonviable) cells based on the permeability of cell membranes. Live cells with intact membranes exclude EB and thus fluoresce green, while dead or dying cells with compromised membranes take up EB and fluoresce red. Three doses of formulations based on the 24-h cytotoxicity results were applied to the fibroblast cells for 24 h. The cells were then removed from the plates and stained with AO/EB (Sigma-Aldrich, USA). The stained cell samples were then observed using a fluorescent microscope system (Leica SFL4000, Germany). By analyzing a minimum of 200 cells in each sample, the proportions of viable cells (green staining) and apoptotic cells (red staining) were ascertained.
Alkaline single-cell gel electrophoresis analysis
Possible genotoxic effects of three doses that do not show cytotoxic effects on fibroblast cells within 24 h were evaluated by the alkaline single-cell gel electrophoresis analysis (Comet Analysis) method as described by Singh et al. 16 Cells were seeded on a six-well plate (approximately 2 × 105 cells per well) and incubated for 24 h with nontoxic doses. Cells were removed from the plates and mixed with low melting agarose (0.65%), then pipetted onto slides coated with agarose (1%). The slides were stored at 4 °C until the gel solidified. They were then placed in the lysis buffer, which consisted of 2.5 M NaCl, 100 mM Na2EDTA, 10 mM Tris-HCl (pH 10–10.5), 1% Triton X-100, and 10% DMSO (added just before use), for a minimum of 1 h. The slides were rinsed with cold PBS and placed in fresh alkaline electrophoresis solution (300 mM NaOH, 1 mM EDTA-Na2, pH 13) for 20 min at 4 °C to unwind the DNA. The cells were then electrophoresed for 25 min (25 V, 300 mA), and the gel was neutralized with 400 mM Tris-HCl buffer and then stained with EB (5 mg/mL) and examined using a fluorescence microscope system (Leica SFL4000, Germany). One hundred cells, on average, were counted and scored for each concentration, and tail density (% tail) was calculated as DNA damage by using Comet Assay IV, Perceptive software (Suffolk, UK).
Statistical analysis
Statistical analyses were conducted using one-way ANOVA with Tukey’s post hoc test. A p-value of less than 0.05 was considered statistically significant. The IBM SPSS package program for Windows (Version 20, Armonk, NY, IBM Corp.) was used for all statistical analyses. The experiments were performed in triplicate, and the data is presented as mean ± standard deviation.
Results
Physical and chemical characterizations of the formulations
Clarity of the formulations
The view of the content of the nasal spray formulations was analyzed for all formulations. There is no change in color and clarity of the formulation, as shown by the product integrity in all formulations, as indicated in Table 2.
Characterizations of nasal spray formulations.

T: transparent.
The pH of the formulations
The pH of the nasal spray formulation is very significant, primarily to prevent nasal mucosa irritation, to avoid pathogenic microorganisms’ growth, and to maintain normal physiological ciliary movement. 17 All formulations were determined at pH 5.5–6.2 so that the pH values of the prepared formulations were compatible with the pH value of the nasal mucosa.
Drug content
The diclofenac sodium, ibuprofen, and paracetamol content in the spray formulations were determined to analyze the potential loss of active ingredients during the preparation of nasal spray formulations. In conclusion of the studies, it was analyzed that the active ingredients were recovered from the nasal spray formulation at a high rate ranging from 95.3 ± 0.5% to 98.2 ± 0.6% (Table 2).
In vitro drug release of the nasal spray formulations
Drug release behaviors of three different nasal spray formulations containing diclofenac sodium, ibuprofen, and paracetamol, respectively, were determined by the diffusion method. In vitro release studies were implemented to choose appropriate formulations for cell culture studies. As shown by the in vitro release profiles given in Figure 1, 77.56% ± 2.32% for diclofenac sodium (F1-1), 73.12% ± 3.05% for ibuprofen (F2-1), and 85.07% ± 1.12% for paracetamol (F3-1) were released from the nasal spray formulations in the first 2 h. According to the in vitro release results, the increase of polymer concentration in spray formulations caused a slower release of active ingredients. Therefore, F1-1, F2-1, and F3-1 were selected from nasal spray formulations prepared for cell culture studies (Figure 1).
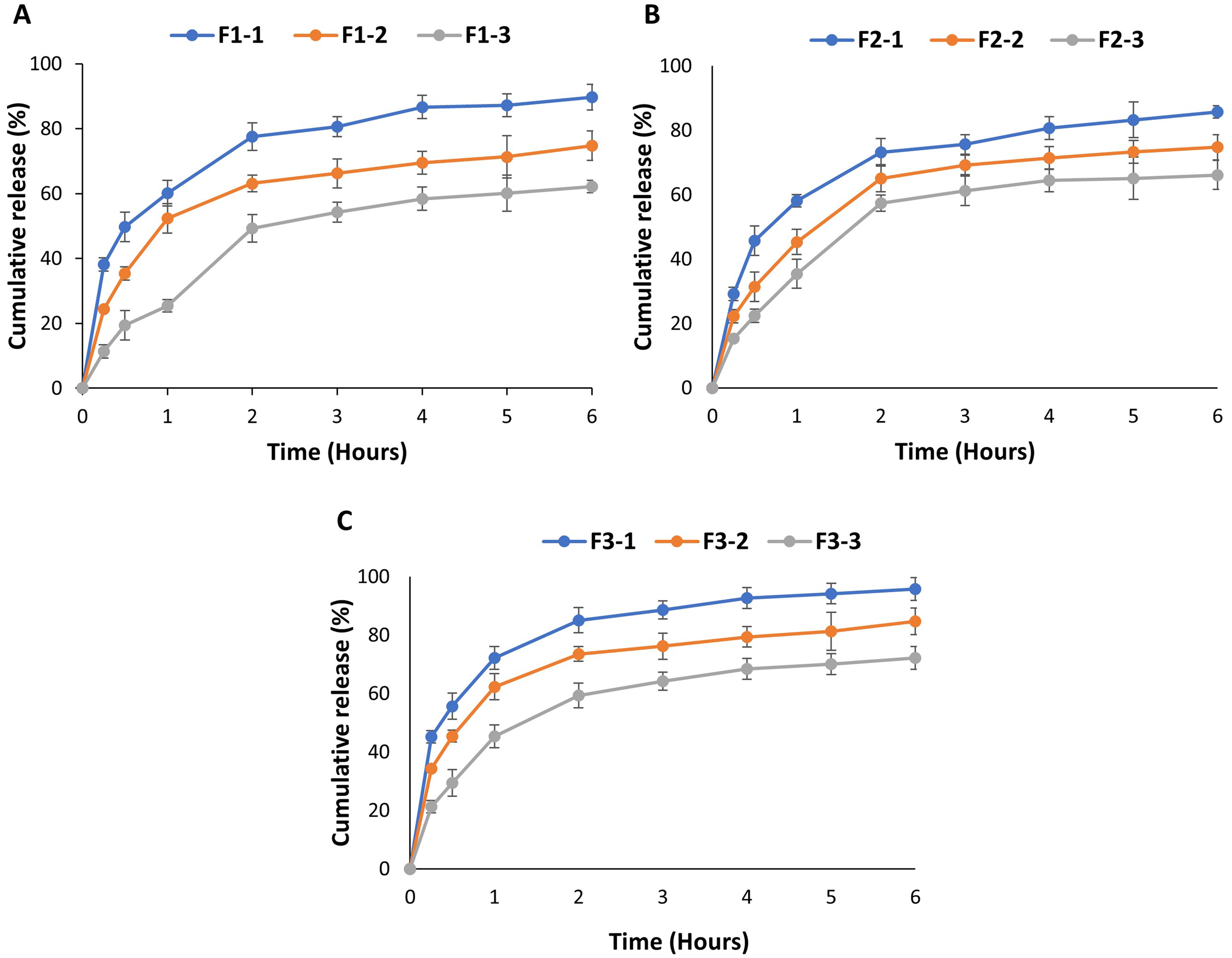
In vitro release profiles of nasal spray formulations containing (a) diclofenac sodium, (b) ibuprofen, and (c) paracetamol. The results were presented as mean ± standard deviation based on three independent experiments.
Stability studies
The stability analysis of the nasal spray formulations was conducted under controlled conditions, with the samples stored in a refrigerator at 25 ± 2°C and 60% ± 5% relative humidity for a span of 90 days. Throughout this period, several characterization parameters were monitored to assess the stability of the prepared spray formulations stored in appropriate conditions. Specifically, the physical appearance, pH, and drug content of selected formulations were analyzed at the outset and at various intervals. The results of these assessments were presented in Table 3. Notably, no statistically significant variations were observed in the outcomes (p ˃ 0.05). The results achieved showed that the selected nasal spray formulations maintained their stability for 90 days.
The results of stability studies of selected nasal spray formulations.

Nontoxic doses of diclofenac sodium, ibuprofen, and paracetamol formulations
In this study, CCD-1079Sk human normal skin fibroblastic cells were used as a model of the progressive formation of cellular toxicity. For cytotoxicity studies, CCD-1079 cells were treated with different concentrations of nasal sprays for 1, 6, and 24 h. The doses showing toxicity within 24 h were eliminated. The safe doses were chosen as 0.0625, 0.375, and 1.25 mg/mL for diclofenac, ibuprofen, and paracetamol, respectively, by not showing toxicity even at 24 h (Figure 2). For later studies, concentrations around nontoxic dose (0.0325 and 0.125 mg/mL for diclofenac; 0.25 and 0.5 mg/mL for ibuprofen; 0.625 and 2 mg/mL for paracetamol) were also studied for apoptosis and genotoxicity assays.

Viability of CCD-1079Sk cells that were incubated with various concentrations of diclofenac sodium, ibuprofen, and paracetamol for 1, 6, and 24 h. The results were presented as mean ± standard deviation based on three independent experiments (n = 3). Asterisk represents that the indicated concentration of the drug decreased the cell viability below the acceptable (>80%) range at 24 h and p < 0.05.
Elimination of potential apoptotic effect of formulations at nontoxic doses
Signs of apoptosis that may occur due to formulations investigated through analysis of cell morphology by light microscope. No change was seen in cell morphology at primary observation on any cells treated by all formulations at nontoxic doses for 24 h. Then, AO/EB staining, which is commonly used as a fluorometric method to analyze viable and apoptotic cell ratios, was used to observe any possible apoptotic effect of formulations. The cells were treated with diclofenac sodium, ibuprofen, and paracetamol formulations at nontoxic doses for 24 h and then analyzed by fluorescence microscope for viable and apoptotic cells. The concentrations that were chosen as safe doses (0.0625, 0.375, and 1.25 mg/mL for diclofenac, ibuprofen, and paracetamol, respectively) showed apoptotic ratios that were similar to nontreated control cells at 24 h (Figure 3).

After 24 h of treatment with diclofenac sodium, ibuprofen, and paracetamol, the cells were stained with AO/EB and then analyzed under a fluorescence microscope at 20× magnification, where viable cells appeared green and dead cells appeared red. The results were presented as mean ± standard deviation.
Elimination of potential genotoxic effect of formulations at nontoxic doses
Since drugs can also result in DNA damage, three doses around nontoxic doses of new formulations were analyzed for any genotoxicity. The alkaline comet assay is a very common method for measuring DNA damage via the migration of DNA under electrophoresis conditions. According to the results of the comet assay, DNA damage was not observed at determined nontoxic doses of diclofenac sodium, ibuprofen, and paracetamol formulations. The mean tail intensity values were similar to those of the nontreated control group, even though they showed an increased pattern with the increasing concentrations (Figure 4).

After 24 h of treatment with diclofenac sodium, ibuprofen, and paracetamol, cells were electrophoresed and stained with ethidium bromide and then analyzed under a fluorescence microscope at 40× magnification, and tail intensities were calculated. The results were presented as mean ± standard deviation.
Discussion
Pain is a public problem with a very important social and economic impact worldwide. 18 Pain has the most adverse effect on the quality of life among all chronic conditions. 19 Acute pain is one of the most frequent reasons why patients are seeking medical care. 20 Generally, chronic pains such as migraine or neuropathic and orthopedic pains are complex and multifaceted phenomena and difficult to treat. 21
IN drug delivery is an effective and noninvasive method for administering drugs systemically. After nasal administration, drugs are quickly cleared from the nasal cavity, leading to rapid systemic drug absorption. 22 IN route offers advantages such as ease of administration, rapid onset of action, and avoidance of first-pass metabolism. 23
IN analgesia serves as a method to ensure both safe and prompt pain relief without causing any discomfort. This eliminates the need for a delayed response often associated with the administration of oral analgesics. 24 The pharmacokinetics of IN drug administration offer the advantage of maintaining therapeutic drug concentrations, facilitating effective pain relief through controlled drug absorption, and mitigating the occurrence of minimal side effects. In children, IN drug administration is often used as an alternative to parenteral opioids for pain relief.3,4
There are many classes and examples of medications that are currently used or studied IN. Ketamine is one of these drugs that can be administered through IN, 25 and its effectiveness was shown by various literatures.26,27 In a study with 40 patients who had moderate or severe pain, IN ketamine reduced visual analog scale pain scores to a clinically significant degree in 88% of emergency department patients. 7 In a study comparing the effect of IN ketamine to intravenous paracetamol, it was shown that IN ketamine was more effective for analgesia in pediatric patients in all periods from 15 min to 24 h of adeno/tonsillectomy post operation. 28 Nasal ketamine bioavailability is 45%–50%, and it has been shown that a dose of 1.5 mg/kg (50 mg/mL) is an effective analgesic without dissociation or sedation.29–31 For IN ketamine spray, the maximum analgesic effect begins approximately 3–4 min before the maximum plasma concentration is reached. 32 IN fentanyl, recognized as the extensively investigated and frequently utilized form of IN analgesia, demonstrates a bioavailability of 77%.5,33 A total of 1.5 μg/kg (50 μg/mL) of IN fentanyl has been demonstrated to be an analgesic equivalent to 0.1 mg/kg of IV morphine and begins to take effect in as little as 7 min due to its high potency and lipophilicity. 3 IN fentanyl has been demonstrated to be a secure and well-tolerated approach to analgesia. This method has proven effective in both pre -and postoperative pain management among pediatric patients, 5 as well as in the immediate management of acute pain. 34 Additionally, it has exhibited efficacy in addressing exacerbated pain in individuals with cancer. 35 It has been shown that IN oxytocin can significantly reduce responses to craniofacial pain in a specific region of the brainstem. 36 The effect of intranasally administered oxytocin has been illustrated in its ability to alleviate migraine and other forms of pain. 36 The pain-relieving properties of intranasally administered oxytocin have been verified in various contexts, including mild traumatic brain injury-related pain, 37 wound pain, 38 chronic low back pain, 39 and chronic pelvic pain. 40 IN desmopressin has been used to reduce pain in patients with acute kidney injury. Constantinides et al. 41 reported a complete resolution of colic pain 30 min after IN administration of desmopressin.
The IN drugs used as analgesics, such as ketamine and fentanyl, are regulated under green prescription and have dose limitations. The use of ketamine may result in side effects such as sedation, nausea, vomiting, and respiratory depression 42 while fentanyl would cause dose-related addiction since it is a morphine-derived drug. Their uncontrolled use outside the hospital can cause undesirable side effects. Therefore, in this project, it was aimed to formulate nasal sprays containing over-the-counter analgesics that do not require a prescription. Diclofenac sodium, ibuprofen, and paracetamol are drugs that are known for their minimal side effects, and they are available over the counter without a prescription.
Although there are limited studies on the effectiveness of IN delivery of over-the-counter drugs, some have demonstrated promising results. For instance, Ogawa et al. 43 conducted a pharmacokinetic and pharmacodynamic evaluation of acetaminophen transport to the central nervous system and systemic circulation following IN administration. They compared this with other routes of administration and found that acetaminophen was rapidly transported to the brain, with higher concentrations observed in the olfactory bulb after IN delivery. This resulted in a significantly greater antipyretic effect than that seen with other administration routes. Similarly, Wen et al. 44 investigated brain uptake in rats following IN administration of ibuprofen in a microemulsion formulation. Their study revealed a 4-fold increase in brain uptake of ibuprofen compared to the reference formulation and nearly 4- and 10-fold increases compared to intravenous and oral administration, respectively.
Evaluation of nasal sprays and nasal absorption depends on many parameters of the formulations. The absorption of drugs is influenced by various physicochemical properties, such as size, molecular weight, solubility, lipophilicity, charge, pKa, polymorph, chemical state, and physical status. 13 Additionally, formulation parameters play a pivotal role, with pH being a crucial aspect. pH adjustment serves the dual purpose of preventing nasal irritation and promoting effective drug absorption. The pH range of 4–7.5 is suitable for nasal sprays as the human nasal mucosa has a slightly acidic pH range of 5.5–6.5. 45 For this reason, the newly formulated nasal drugs containing diclofenac sodium, ibuprofen, and paracetamol were primarily investigated for their physical and chemical characterizations. All formulations were determined at pH 5.5–6.2 so that the pH values of the prepared formulations were compatible with the pH value of the nasal mucosa. Moreover, it was found that the active ingredients were extracted from the nasal spray formulation at a high rate. Also, the data has shown that the newly formulated nasal spray formulations maintained their stability for 90 days at 25 ± 2°C and 60% ± 5% relative humidity.
Then, these formulations were tested for toxicity in vitro. Nontoxic doses of the formulations were determined by performing cytotoxicity tests. In a nasal spray, the drug concentration should not exceed 5 mg. 13 The safe doses in this study were chosen as 0.0625, 0.375, and 1.25 mg/mL for diclofenac, ibuprofen, and paracetamol, respectively, by showing nontoxicity even at 24 h. Genotoxicity tests also showed that these doses are safe even at 24 h. The in vitro nontoxic concentrations serve as a guide for calculating a safe and effective volume for patient use. By translating these concentrations to appropriate therapeutic doses in future studies, we can determine a clinically relevant volume that stays within standard nasal administration limits, ensuring safety and accounting for factors like mucosal absorption and metabolic differences. The formulations were specifically designed for nasal administration, allowing the drugs to bypass the first-pass metabolism that happens with oral dosing. This approach achieves therapeutic effects at lower concentrations.
IN drug administration offers several advantages, such as ease of use and rapid onset of action. However, the limited volume capacity of the nasal cavity restricts the amount of drug that can be delivered at once, potentially hindering the therapeutic efficacy of medications that require higher doses. Additionally, the nasal cavity's mucociliary clearance mechanism may result in the premature elimination of the drug before complete absorption, which could compromise its effectiveness, particularly for drugs needing prolonged contact time. Furthermore, the daily dosage of nasal sprays necessary to achieve a therapeutic effect equivalent to oral administration can vary widely depending on the drug's bioavailability and the specific condition being treated. Therefore, further research is essential to determine the optimal daily dosage and efficacy of these nasal sprays, with oral administration serving as a control for comparison. Also, in the current manuscript, we used a fibroblast cell line to observe possible cytotoxic and genotoxic effects of the newly formulated nasal sprays. By focusing on one cell line, we were able to closely monitor specific biological responses. However, the use of a single-cell line presents a limitation, as it may not fully capture the biological diversity seen across different cell types or tissues, and future studies should consider validating the results in multiple cell lines.
Conclusion
This study demonstrates that over-the-counter painkillers paracetamol, ibuprofen, and diclofenac sodium can be effectively formulated as nasal sprays with stable physical and chemical properties for 90 days at 25 ± 2°C. In vitro toxicity tests confirmed that the selected doses are nontoxic even at 24 h. These newly formulated nasal sprays may offer an alternative treatment modality and a more convenient route of administration for patients who are unable to swallow oral analgesics due to underlying neurological deficits, the pediatric age group who may refuse to take oral analgesics and have painful deglutition after tonsillectomy, and may reduce the length of hospitalization for children.
Footnotes
Author contributions
Alper Yenigun contributed to the conception and design of the study. Ali Asram Sagiroglu formulated the nasal sprays. Vildan Betul Yenigun performed toxicity evaluations. Vildan Betul Yenigun drafted the manuscript. All authors critically revised the manuscript. Abdurrahim Kocyigit and Orhan Ozturan gave the manuscript final approval.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bezmialem Vakif University Scientific Research Projects Units [grant number 12.2018/6].
Previous presentation at conferences
This manuscript was presented as an oral presentation at the 43rd Turkish National Congress of Otorhinolaryngology and Head and Neck Surgery on 16–20 November, 2022, in Antalya, Turkey.
